Abstract
Introduction
Colorectal cancer is the third most common cancer after lung and breast cancer and the second leading cause of mortality worldwide.1,2 Survival rates have increased in recent years because of earlier diagnosis, introduction of adjuvant chemotherapy, and advances in the treatment of metastatic disease. 3 In patients with colorectal cancer stage III and in certain situations in stage II, adjuvant chemotherapy is administered after R0 resection. A 5-fluorouracil-based (5-FU) chemotherapy reduces the risk of recurrence by 30% to 40%, and the 5-year survival can be increased by 20% to 30%. The addition of oxaliplatin to 5-FU/leucovorin or capecitabine significantly improves the disease-free survival (DFS) and the overall survival (OS) compared with 5-FU/leucovorin.4-6 With this treatment strategy, survival improves but the quality of life (QOL) could be impaired. Besides DFS and OS time, QOL is an important outcome measure for cancer patients.
Colorectal cancer and/or the treatment toxicities of chemotherapy can have an adverse effect on physical, emotional, and social functioning and may affect QOL in short-term and long-term periods.7,8 Treatment strategies for better QOL in cancer patients are challenging and are still an unresolved issue. 9 Further research is needed to enable more precise, targeted treatment options. Initiating or maintaining exercise activity in cancer patients is a promising treatment strategy that still has to be fully investigated. 10 We still do not know a lot about how exercise and physical activity affect the recovery from cancer and/or the treatment toxicities. But relatively recent research showed that adequate levels of physical activity could reduce cancer-related fatigue, pain, and insomnia in posttreatment survivors.11-18 Furthermore, several observational studies showed a reduction in relapse rate, cancer-specific mortality, and overall mortality in patients with colon and breast cancer following exercise training.19-23 Data of these observational studies indicate a high therapeutic efficacy of physical activity after adjuvant therapy. Current recommendations from several reports14,24-26 provide exercise guidelines for cancer survivors, but randomized clinical trials are still needed to better define the impact of exercise on patient-reported outcomes (PROs), relapse rate as well as cancer-specific mortality.
The ongoing Colon Health and Life-Long Exercise Change trial (CHALLENGE trial) is designed to determine the effects of a 3-year structured physical activity intervention on outcomes for survivors of high-risk stage II or III colon cancer who completed adjuvant chemotherapy. 27 Besides the primary endpoint, 3-year DFS, secondary endpoints evaluate PROs assessing QOL, fatigue, sleep quality, and anxiety and depression as well as objective testing of physical functioning consisting of anthropometric measurements, cardiovascular fitness, and physical functioning.
This pilot study investigated the feasibility of a predefined 1-year structured exercise training after adjuvant chemotherapy in patients with locally advanced colorectal cancer. 28 Before initiating a large randomized trial with more than 700 patients, our study group investigated the feasibility, compliance, and logistics of a 1-year exercise training. Results concerning compliance of patients have been considered for sample size estimation and study planning for the following large randomized trial. Secondary endpoints in this pilot study included evaluation of PROs with regard to fatigue, emotional functioning, physical functioning, and global QOL. PROs were assessed using the European Organization for the Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ-C30) questionnaires. The EORTC QLQ-C30 has become the most widely used questionnaire for the assessment of QOL in cancer clinical trials. The EORTC QLQ-C30 covers cardinal symptoms of cancer and its treatment as well as patients’ functioning. Feasibility results were already published elsewhere. 28 In this article, results of PROs as well as compliance to the exercise program will be reported.
Methods
Eligibility and Study Design
The “ABCSG C07 EXERCISE” study was an academic phase II pilot study investigating feasibility, acceptability, and PROs of a 1-year structured exercise training, after adjuvant chemotherapy, in patients with locally advanced colorectal cancer. Eligibility criteria, design, and feasibility/acceptability results have been published previously. 28 Patients were eligible for exercise training if they had histologically confirmed diagnosis of locally advanced adenocarcinoma of the colon or rectum (stage II with risk factors and stage III) without distant metastases and who had completed adjuvant chemotherapy 4 to 16 weeks before inclusion. The recruitment window of 4 to 16 weeks after adjuvant chemotherapy allows recovery from the side effects of the chemotherapy and/or stoma reversal surgery in rectal cancer patients before inclusion in the exercise training. Further inclusion criteria were the following: Eastern Cooperative Oncology Group (ECOG) performance status 0 to 1, 29 age >18 years, adequate liver and kidney functions, and able to perform exercise training according to protocol. We excluded patients with (1) significant comorbid conditions precluding participation in an exercise training program, (2) local recurrences or distant metastases, (3) significant cardiovascular disease, (4) left bundle branch block, (5) past or current history of other malignant neoplasms other than colorectal cancer in the past 5 years (except basal cell carcinoma of the skin and/or in situ carcinoma of the cervix), (6) current treatment with chemotherapy or radiation, (7) current pregnancy, (8) patients unwilling to complete study, or (9) patients who are treated with β-blockers without the possibility to change therapy.
All patients received a pulse-controlled training program based on the study by O’Donovan et al. 30 At baseline, a bicycle ergometry was performed for individual assessment of the basic performance status. The intensity of exercise training was personalized by the heart rate reserve according to the Karvonen Formula. 31 On the basis of size, weight, and body fat, the normal weight was calculated. The calculation of the normal weight is necessary for calculation of the metabolic equivalent of task (MET)-hours. The kilocalories, which correspond to MET-hours, were calculated after age and gender adjustment of the normal weight. The exercise training was performed 3 times per week for 1 year and was increased gradually in 3 phases (Table 1) until reaching 18 MET-hours. According to 3 cohort studies of Meyerhardt et al, the most benefit of improving survival was seen in patients doing at least 18 MET-hours of physical activity.19,20,23 Heart rate during training was controlled by a heart rate monitor. Table 1 shows the exercise training (prespecified training), the calculated heart rate, and MET-hours. Exercise training was performed using a bicycle ergometer or cross-trainer. In the participating centers, supervised physical activity sessions were available. Alternatively, patients were allowed to perform the exercise training program at home independently. All training sessions were documented in a diary by the patient and were controlled regularly during the study visits. In case of problems during the home-based training, further supervised sessions were done in the training centers. If patients performed other sports types (non-prespecified training), all training sessions had to be documented in a diary by the patient. Non-prespecified training was converted into MET-hours depending on type and duration of physical activity. 32 Physical performance status was evaluated at screening and after 3, 6, and 12 months of the exercise training by ergometry. After each ergometry, a new calculation was performed to update the training heart rate. The training structure and the intensity of training were similar for all patients and differed only by the individual pulse input. Patients were withdrawn prematurely from study training in the following circumstances: (1) recurrence of colorectal cancer, (2) noncompliance with study procedures (<75% of the planned complete MET-hours per week), (3) the patient requested to be withdrawn from the study, and (4) occurrence of a restrictive clinically significant cardiovascular disease.
Exercise Training Program (Prespecified Training) for 12 Months.

Abbreviations: MET, metabolic equivalent of task; HR, heart rate.
The trial was approved by the local institutional review board and was carried out in accordance with the Helsinki Declaration of 1975, as revised in 1983. Written informed consent was obtained from all patients before study inclusion. The evaluation of PROs was supported by a research grant of the “Oberösterreichische Krebshilfe.”
Assessment of Patient-Reported QOL and Symptom Burden
The patients’ QOL was assessed by the EORTC QLQ-C30 questionnaire (version 3.0) of the European Organization for Research at baseline, after 3, 6, 9, and 12 months of the structured exercise training electronically by a tablet computer. PRO data collection was done by using the Computer-based Heath Evaluation System (CHES), 33 which has already been successfully implemented on an international level both in clinical trials as well as for daily clinical routine. CHES is a software tool for the electronic collection, calculation, and presentation of PRO data as well as the web-based administration of case report forms. The EORTC QLQ-C30, 34 an internationally validated, brief, self-reporting, and widely used cancer-specific measure of health-related QOL, assesses various facets of functioning, symptoms common in cancer patients, and global QOL.35-37 The EORTC QLQ-C30 has become the most widely used questionnaire for the assessment of QOL in cancer clinical trials with more than 3000 PubMed entries. The 30-item questionnaire is composed of 5 multi-item functional scales that evaluate physical, role, emotional, cognitive, and social function, and 1 global health status/QOL scale. Three multi-item symptom scales measure fatigue, pain, and nausea/vomiting, and 6 single items assess symptoms such as dyspnea, insomnia, appetite loss, constipation, diarrhea, and financial difficulties. High functional scores represent better functioning/QOL, whereas a high symptom score indicates more severe symptoms. Differences of QOL scores of >20, 10 to 20, and 5 to 10 points were considered large, moderate, and small, respectively. 38
Statistical Analysis
The scoring of the EORTC QLQ-C30 items was performed according to the EORTC scoring manual. All scores were linearly transformed to a 0 (very bad QOL) to 100 scale (very good QOL). In case of missing items, multi-item scores were calculated as the mean of nonmissing items if at least half of the items from the corresponding scale had been completed. 39 The results were compared with published QLQ-C30 reference data from the German general population. 40 These reference data were derived from a representative sample of the German adult population comprising 2634 women and 2050 men (age range = 16-92 years). 40
Results
Thirty patients were included in the Exercise C07 pilot study. Of these, all 14 patients from 1 center (4 women and 10 men, median age 57 years) participated in this subprotocol and filled out the QOL questionnaire at baseline (100%), after 3 months (85,7%), 6 months (78.6%), 9 months (71.4%), and 12 months (71.4%) of the structured exercise training. Due to early end of treatment because of distant recurrences in 2 patients and a serious adverse event (atrial fibrillation) in 1 patient, not all patients filled out the QOL questionnaire at all time points. Nine patients (64.3%) had rectal cancer (1 stage II, 8 stage III), and 5 patients (35.7) had colon cancer (1 stage II, 4 stage III). Half of patients received the FOLFOX-4 regime (50%), 5 patients (35.7%) received CAPOX, 1 patient (7.1%) received capecitabine, and 1 patient (7.1%) received the De-Gramont regime as adjuvant chemotherapy. Baseline sociodemographic and clinical characteristics of the participating patients of this subprotocol are presented in Table 2. Half of patients were employed. Seven out of 14 patients had comorbidities in the medical history. The most common comorbidity was hypertension in 3 patients, chronic pulmonary disease in 2 patients, and myocardial infarction and autoimmune thyroiditis in 1 patient each. In the first phase, a gradual increase of exercise training from 3 to 15 MET-hours weekly was planned, and a median of 9.96 MET-hours weekly was done (median 6.87 MET-hours prespecified training; median 0.00 MET-hours non-prespecified training). In the second phase, 15 MET-hours weekly were planned, and a median of 15 MET-hours weekly was done (median 10 MET-hours prespecified training; median 0.00 MET-hours non-prespecified training). In the third phase, 18 MET-hours weekly were planned, and 18 MET-hours weekly was done (median 9 MET-hours prespecified training; median 8.18 MET-hours non-prespecified training; Figure 1). Mean MET-hours in the first phase were 7.97, 4.51, and 12.48 MET-hours of prespecified, non-prespecified, and total training. In the second phase, the mean MET-hours were 10.08, 6.73, and 16.80, and in the third phase, 9.71, 13.75, and 23.46 MET-hours for prespecified, non-prespecified, and total training. Three patients were noncompliant to the exercise program defined as doing the training less than 75% of the planned cumulative MET-hours per week (prespecified and non-prespecified training). Eight out of 14 patients completed ≥75% of the planned MET-hours (noncompliance: 3 patients; early end of treatment: 1 patient with SAE, 2 patients due to disease recurrences). Patients experienced a performance increase (bicycle ergometry) from median 130 W at screening to median 176.5 W after 12 months, a reduction in heart rate at rest from median 76/min at screening to 70/min after 12 months, and a reduction in body fat content from median 26% at screening to median 22.5% after 12 months.
Characteristics of Study Population at Baseline.
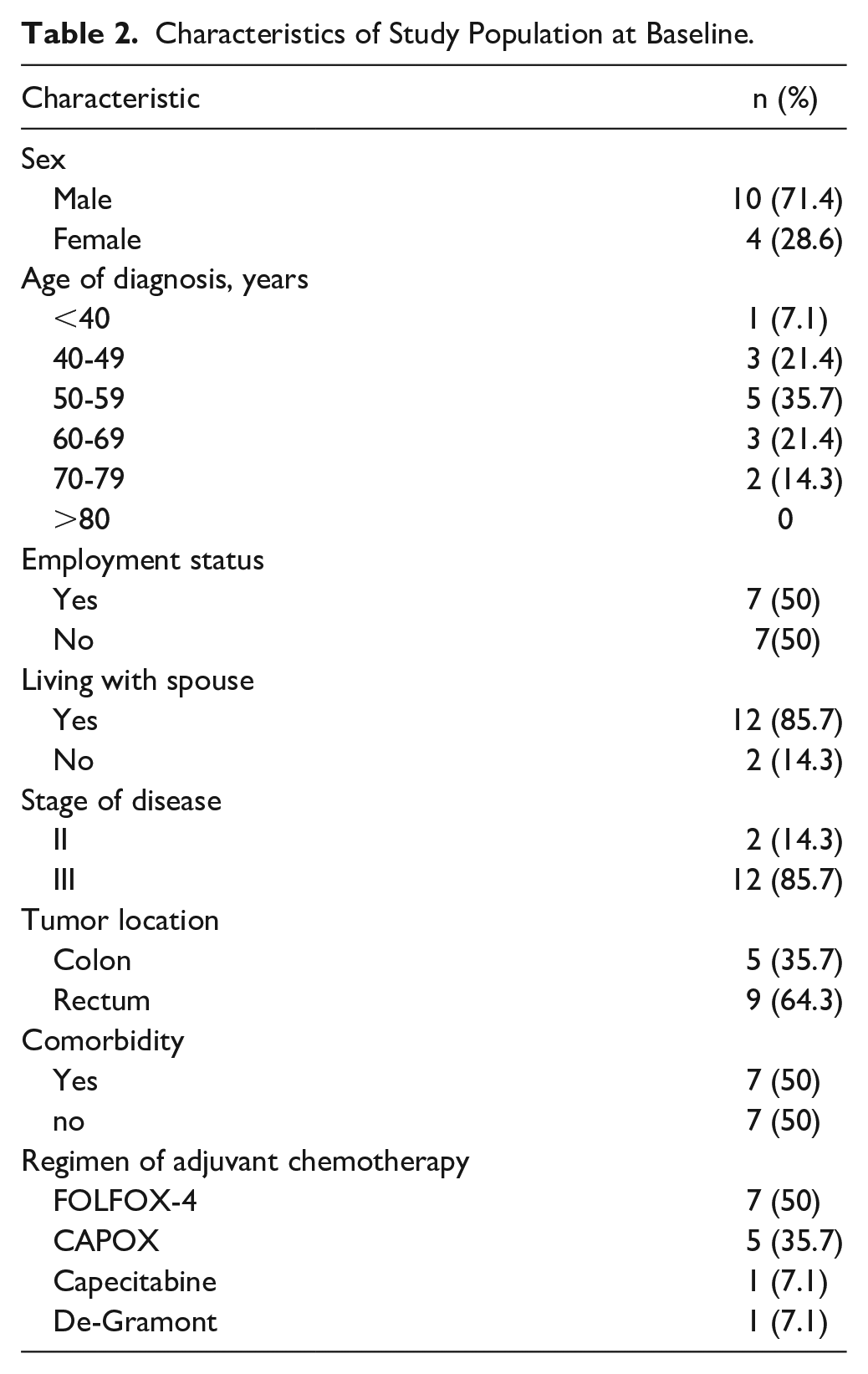

Overview of the planned and done exercise training. The line “planned” shows the prespecified exercise training as per protocol that should be performed during this 1-year exercise training program. The line “additional” shows the non-prespecified additional exercise training that was performed outside the protocol. The line “done” shows the part of the planned prespecified exercise training that was really done. The line “total” shows the performed prespecified and additional non-prespecified exercise training together.
Table 3 presents the mean scores for the QLQ-C30 subscales. Specific functional and symptom QOL scores compared baseline with 3, 6, 9, and 12 months during the structured exercise training and were compared with published reference data from a German general population. 40
Median and Mean scores for the QLQ-C30 subscales.

At baseline, colorectal cancer patients scored social functioning, emotional functioning, financial impact, and the symptoms insomnia and diarrhea much worse than the German general population. The biggest difference was seen in social functioning.
Between baseline and after 12 months of the structured exercise training, patients reported a “large” change (difference of scores greater than 20 points) for the social functioning, a small change (difference of scores greater than 5 points) for the physical and emotional functioning as well as for global QOL, and no change (difference less than 5 points) for role and cognitive functioning. Patients also reported a “moderate” change (difference of scores greater than 10 points) for pain, diarrhea, financial impact, and taste. The symptom scores including fatigue, nausea, dyspnea, insomnia, appetite loss, and constipation showed <5 points difference. At the end of the structured exercise training, social, emotional, and cognitive functioning had the same value as the German general population. Patients in our study had a better physical and role functioning as well as global QOL after 12 months of the structured exercise training than the German general population. With the exception of appetite loss and diarrhea, all other symptom scores were lower in the colorectal cancer group than in the German general population, indicating less frequent and/or less severe symptoms among colorectal cancer patients 1 year after the structured exercise training. The differences were most pronounced in the subscale of pain (mean 13.3 vs 28), fatigue (mean 21.1 vs 32), and insomnia (mean 20 vs 28). Due to the somewhat small sample size, a statistical analysis between the study groups (compliant vs noncompliant; colon cancer vs rectal cancer, employment status yes vs no) was not very meaningful. Nevertheless, an exploratory statistical analysis did not yield statistically significant results, neither for the compliant/noncompliant grouping nor for the 2 different diagnosis groups (colon cancer, rectal cancer) or for the employment status.
Discussion
To the best of our knowledge, this is the first study comparing QOL among stage II and stage III colorectal cancer patients after the end of adjuvant chemotherapy and during and after 1 year of a structured exercise training. The ongoing CHALLENGE trial 27 as well as the follow-up study of this trial—the ABCSG EXERCISE-C08 trial—analyze as secondary endpoints the effects of exercise training on QOL in stage II/III colorectal patients following chemotherapy. Due its ongoing status, no QOL results were published until now.
In the present trial, we compared the results with a German general reference population. 40 Decreased social and emotional functioning and specific symptoms like diarrhea, pain, and fatigue as well as financial difficulties represent main factors that impaired QOL among colorectal cancer patients after the end of adjuvant chemotherapy in our study. After 1 year of a structured exercise training, patients scored better on all function scales as well as all symptom scales with the exception of appetite loss than at the end of adjuvant chemotherapy. Interestingly, colorectal cancer patients’ scores at baseline for role functioning, fatigue, and insomnia were much better than the reference population, although they underwent a surgery and 6 months of adjuvant chemotherapy for their colorectal cancer. After 1 year of the structured exercise training, all function scales and all symptom scales were comparable or even better than in the German general population with the exception of diarrhea. Nausea gradually decreased over the year and was not present after end of the 1 year exercise training. A similar effect was seen with the symptom taste that was scored much worse at baseline than after 1 year. This effect is mainly attributed to the recovery from the side effects of cancer treatment rather than from the exercise training.
Arndt et al 41 measured the QOL in patients with colorectal cancer stages I to IV 1 year after diagnosis and compared the results with a German general population in a population-based study. Referring to these data, our patients showed better results in all functional scales after 1 year of a structured exercise training: physical functioning (Δ14.5), role functioning (Δ7.3), emotional functioning (Δ3.8), cognitive functioning (Δ6.5), social functioning (Δ7.2), and global QOL (Δ15.5) and in following symptom scales: fatigue (Δ14.4), nausea (Δ5.8), pain (Δ7.1), dyspnea (Δ9.8), insomnia (Δ12.1), constipation (Δ6.9), and financial difficulties (Δ7.6). Only diarrhea (Δ5.7) was scored worse and appetite loss was scored quite similar (Δ0.2). This population is more suitable for comparison with our study population as a reference population due to the same cancer diagnoses and the same evaluation time. It has to be mentioned that in the study from Arndt et al 41 patients with stage I to IV were included but the proportion of patients with stage IV was less than 10% (9.7%), and 49.2% of the patients received adjuvant chemotherapy and/or radiotherapy. Nothing is known about the amount of physical activity in this study. Nevertheless, it seems that our patients who are performing a structured exercise training for 1 year had a better QOL than comparable patients in the population-based study from Arndt et al 41 1 year after diagnosis of colorectal cancer.
Comparing the results with the EORTC-QLQ-C30 reference values for colorectal cancer is more difficult because there are only combined results for stage I to II and for stage III to IV patients available, and the evaluation time of the EORTC-QLQ-C30 questionnaire was at baseline (pretreatment). Data from patients currently receiving treatment or who are off treatment were excluded from the reference values.
We still do not know a lot about how exercise and physical activity affect the recovery from cancer or the side effects from the chemotherapy. But results of several reviews suggest that colorectal cancer patients may benefit from physical exercise both during and after cancer treatment. Colorectal cancer and its treatment strategies may result in psychophysical or functional impairment and has influence on social and family interactions. All these factors affect QOL. Fatigue, which compromises physical function and QOL, has been reported as the most frequent symptom of the underlying disease and the most distressing toxicity of the subsequent chemotherapy.13,14,42,43 A review by Mock 44 on the impact of various exercise programs on cancer-related fatigue found good evidence that exercise decreased levels of fatigue and increased QOL, mood, and functioning. This is concordant with other reviews on the impact of exercise on fatigue.10,13,14,45,46 Results of a randomized trial in postmenopausal breast cancer survivors showed a significant increase in peak oxygen consumption, global QOL, physical well-being, breast symptoms, and fatigue in the exercise group but not in the control group. 47 Similar results were found for prostate cancer patients. 48 In our patients, fatigue remained quite similar between baseline and 12 months after the structured exercise training, whereas global QOL scored better after 12 months. But fatigue was already scored very low at baseline. Fatigue is not the only part that negatively affects global QOL. Several physical and mental factors have an impact on global QOL. At baseline, more symptom items were scored higher than after 1 year, like diarrhea or pain, and physical/social/emotional functioning were scored worse at baseline. All these factors may have an impact on global QOL. After 1 year, these factors were scored better and this may influence the global QOL. Every 3 months, the EORTC QLQ C30 questionnaire was evaluated. Fatigue showed a fluctuation over time. After 3 months of exercise training, fatigue was scored lowest (at baseline 23.8 and after 3 months 16.7) and increased, respectively, to 25.3, 21.1, and 21.1 after 6, 9, and 12 months of exercise training. The intensity of the exercise training was increased gradually until reaching 18 MET-hours per week. Perhaps, there was a temporary worsening of fatigue due to the intensification of the exercise training and fatigue stabilized/decreased in the further course after adaption of the body to the exercise training.
About 21% of the patients were not compliant with the exercise training defined as doing less than 75% of the planned cumulative MET-hours per week. Patients who were doing the exercise training during the whole year reached the targeted MET-hours per week. Nevertheless, it must be mentioned that in the first 2 phases two thirds of the training were done with the prescribed training modalities (ergometer or cross-trainer; = prespecified exercise training), and one third was done with other sports facilities (non-prespecified exercise training). In the last phase, about half of the training was done as prespecified and the other half was done as non-prespecified. All training sessions were pulse-controlled and converted to MET-hours. One reasons for doing non-prespecified training was that ergometer training or cross-training was too boring over time. In the warm season, outdoor training was preferred. In the last phase, the training was done more independently at home, whereas in the first phase, the training was mainly done in the training center under supervision. This pilot study shows that intensive support is necessary to motivate patients for doing their training and that different sport facilities should be offered.
An important strength of this study is that our results were obtained from a homogeneous patient group. All patients received adjuvant chemotherapy due to colorectal cancer stage II/III and performed a pulse-controlled structured exercise training for 1 year after the end of adjuvant chemotherapy. All patients in whom the effects of exercise on QOL were evaluated came from one center and had the same training support. Other strengths are that the QOL was measured sequentially at baseline, after 3, 6, 9, and 12 months and that the data were collected electronically with a tablet computer. The small sample size and the lack of a control group might limit the interpretation of the result. Although we do not have a control group, the results were compared with existing data from Arndt et al 41 and a German general population. 40 The small sample size precluded the ability to conduct subgroup analysis to identify factors that may influence the relationship between exercise training and PROs. Due to the small sample size, a statistical analysis between study subgroups (compliant/noncompliant; colon cancer/rectal cancer) was not very meaningful. Nevertheless, an exploratory statistical analysis did not yield statistically significant results, neither for the compliance/noncompliance nor for the 2 different diagnosis groups (colon cancer vs rectal cancer). Furthermore, there were no statistically significant results on whether patients had an employment status or not regarding exercise training or compliance. Another limitation is that we only collected the data from one center participating in the main study. Nevertheless, it is our intention to verify the results in a follow-up study with a control group and an increased sample size in the upcoming ABCSG C08 EXERCISE II study with 788 patients. Results of the pilot study concerning compliance of patients have been considered for sample size estimation and study planning for the following large randomized trial.
Conclusion
In conclusion, this study has demonstrated improvements in social, physical, and emotional functioning as well as global QOL after 1 year of a structured, pulse-controlled exercise training after receiving adjuvant chemotherapy in patients with locally advanced colorectal cancer. To enhance compliance, sufficient support and different sport facilities should be offered. Further research is required to investigate the positive effect of exercise on PROs, DFS, and OS in cancer survivors.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the “Oberösterreichische Krebshilfe.”
Ethical Approval
All procedures performed in the study were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
