Abstract
Cisplatin induces anorexia, weight loss, loss of adipose tissue, skeletal muscle atrophy, and serious adverse effects that can cause premature termination of chemotherapy. The aim of this study was to use an animal model to assess cisplatin therapy (3 cycles) with and without
Introduction
Cancer-associated cachexia is characterized by anorexia, body weight decrease, and loss of adipose tissue and skeletal muscle. 1 Cancer cachexia prevalence ranges from 50% to 80% in patients with advanced cancer. 2 In addition to tumor-induced anorexia and cachexia, there is evidence of chemotherapy-induced anorexia and cachexia.3-5 Among chemotherapeutic agents, cisplatin is a first-line, standard care agent for lung cancer with strong emetic activity. There is evidence that cisplatin induces anorexia, loss of adipose tissue and skeletal muscle atrophy,3,5-7 indicating that cisplatin produces serious adverse effects that can lead patients to discontinuation of treatment. The incidence of anorexia is approximately 15% among patients with cancer receiving high-dose cisplatin treatment. 8 These adverse effects can markedly decrease the quality of patients’ lives, aggravate suffering and even lead to discontinue their treatments. The development of an adjuvant or chemopreventive agent without decreased antitumor action that ameliorates these adverse responses is vital for building patient confidence and continuing treatment.
A short-term animal study has shown that
Materials and Methods
Drugs and Chemicals
Cisplatin and
Animals
Twenty-four male Wistar rats (initial average body weight between 176 and 200 g) were purchased from BioLASCO Taiwan Co, Ltd. Rats were housed in cages with a maximum of 4 rats per cage with a 12-hour light/12-hour dark cycle and fed an autoclaved diet with ad libitum access to standard laboratory rodent diet 5001 during the study period.
Experimental Design
After 2 weeks of acclimatization, animals were randomly divided into 3 equal groups of 8 animals each. Based on our previous study, 16 anorexia was induced by intraperitoneal administration of cisplatin at dose of 5 mg/kg body weight once a week for 3 weeks. The experimental design was as follows:
Saline group: Phosphate buffered saline (PBS) was orally administered daily with a single intraperitoneal injection of 0.9% (w/v) NaCl solution on the 1st, 8th, and 15th days.
Cisplatin group: A single intraperitoneal injection of cisplatin (5 mg/kg) dissolved in normal saline was administered on the 1st, 8th, and 15th days.
C2C12 Cell Cultures
The C2C12 murine skeletal muscle cell line (BCRC number 60083) was purchased from the Bioresource Collection and Research Center (Hsinchu, Taiwan) and cultured in Dulbecco’s modified Eagle medium (DMEM) (GIBCO, Rockville, MD) with 10% fetal bovine serum (HyClone, Logan, UT), glutamine (2 mM), penicillin (100 units/mL), and streptomycin (100 μg/mL) (Invitrogen, Carlsbad, CA). To induce differentiation, at 80% to 90% confluence, the medium was changed to the differentiating medium, consisting of DMEM supplemented with 2% horse serum (HyClone, Logan, UT), to induce myotube formation. After 5 days of culture, differentiated C2C12 myotubes were incubated in DMEM or cisplatin (50 μM) or pretreated with 5 mM
Determinations of Hepatic Lipid Metabolism and Muscle Atrophy–Related Gene Expressions by RT-PCR
Muscles and liver were weighed to the nearest 0.1 g. Muscle and liver samples were flash-frozen in liquid nitrogen and ground to a powder in a stainless steel mortar and pestle homogenizer. One milliliter RareRNA (GenePure Technology Co, Taichung, Taiwan) was used to homogenize parts of the frozen crushed muscle and liver samples to extract RNA. Analysis of RT-PCR (reverse transcription polymerase chain reaction) was carried out as described previously with some modifications. 17 The concentration of each RNA sample and the 260/280-nm absorbance (A260/A280) ratio was measured using the Amersham-Ultrosrec 2100 Pro Spectrophotometer. Only samples with A260/A280 ratios between 1.8 and 2.0 were used for further analyses. cDNA was reverse-transcribed from 2 µg total cellular RNA and amplified in a reaction volume of 50 µL. High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA) was used for reverse transcription. PCR was performed with 1 µL RT products, 10 × PCR buffer 2.5 µL, 10 mM dNTPs (1 µL), 1 µL of each primer of β-actin, 1 µL of each primer of gene to be tested and ProTaq DNA polymerase, in a final volume of 25 µL. The primer sequences for PCR amplification are presented in Table 1. Samples were equally loaded onto 2% or 1.5% agarose gel, stained with ethidium bromide, and visualized with UV transilluminator. The PCR reactions were performed on programmable thermal controller instrument-thermal cycler model 2400. Sequences of primers are presented in Table1. The relative mRNA levels of stearoyl-CoA desaturase-1 (SCD-1), fatty acid synthase (FAS), carnitine palmitoyltransferase-1α (CPT-1α), insulin-like growth factor gene 1 (IGF-1), myostatin, forkhead box O1 transcription factor (FOXO-1), muscle atrophy F-box protein (MAFbx), muscle RING finger-containing protein 1 (MURF-1), Myogenin and MyoD in each sample were normalized to β-actin content using the ImageJ software.
Sequences of Primers Used in This Study.
Isolation of RNA and Real-Time RT-PCR
RNA from cultured C2C12 myotubes was isolated using RareRNA (GenePure Technology, Taiwan). Total RNA was reverse transcribed by High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, 4368813) to cDNA. The reverse transcription reaction conditions were 25°C for 10 minutes, 37°C for 120 minutes, and 85°C for 5 minutes. Real-time PCR was performed using ABI StepOnePlus real-time PCR system with gene-specific primers and Smart Quant Green Master Mix with dUTP and ROX (Protech, PT-GL-SQGLR-V3) using StepOne real-time PCR machine (Applied Biosystems, Foster City, USA). Ct values for both the target and internal control genes were calculated, and the relative changes in gene expression were analyzed by 2−ΔΔCt method. The mRNA of β-actin was used to normalize the total amount of cDNA on real-time PCR.
Primers were designed as follows:
Myogenin forward: 5′-ACAGCATCACGGTGGAGGATATGT-3′, reverse: 5′-CCCTGCTACAGAAGTGATGGCTTT-3′;
MyoD forward: 5′-ACGACTGCTTTCTTCACCACTCCT-3′, reverse: 5′-TCGTCTTAACTTTCTGCCACTCCG-3′;
MAFbx forward: 5′-CGACCTGCCTGTGTGCTTAC-3′, reverse: 5′-CTTGCGAATCTGCCTCTCTG-3′;
MuRF-1 forward: 5′-GGTGCCTACTTGCTCCTTGT-3′, reverse: 5′-CTGGTGGCTATTCTCCTTGG-3′.
Determination of Bone Microarchitecture Indices
For the assessment of bone microarchitecture indices in bone marrow, femur, and tibia were collected and fixed in 70% alcohol solution. Micro–computed tomography (micro-CT; Skyscan 1076) was used to conduct trabecular bone volume/total tissue volume (BV/TV; %), trabecular number (Tb.N; mm−1), trabecular thickness (Tb.Th; mm), and trabecular separation (Tb.Sp; mm) assays.
Histological Analysis
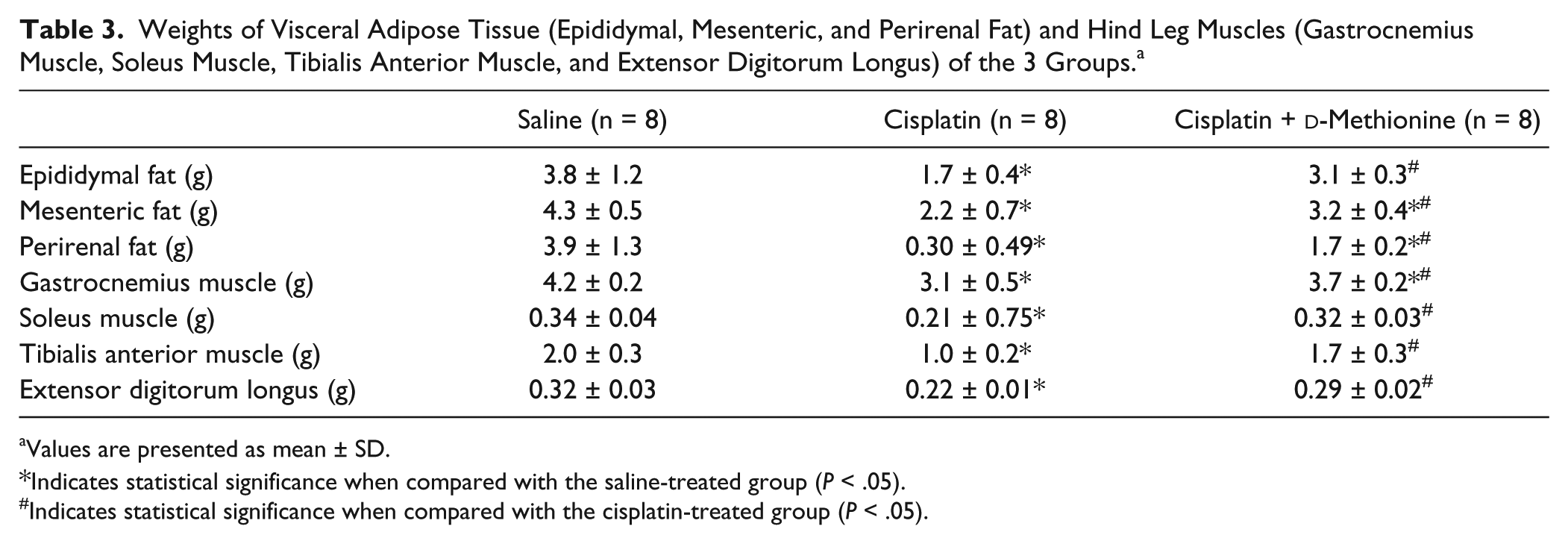
For the assessment of pathological changes of the stomach, stomach tissues were obtained and fixed in 10% formalin for 48 hours. Then, stomach samples embedded in paraffin were stained with hematoxylin and eosin (H&E) dye. After staining, slides were observed under Olympus BX60 microscope. In addition, gastrocnemius muscles were formalin fixed, embedded in paraffin, and cross-sectioned at the midpoint of the muscle. Sections were mounted on slides and stained with H&E. Digital images were captured using Zeiss Axio Imager A2 microscope and analyzed with ImageJ software for myofiber diameter (original magnification, 200) and cross-sectional area (CSA). Myofiber diameter was determined as described previously 5 by measuring the longest minor axis (the perpendicular bisector of the major axis) of at least 100 neighboring myofibers per animal. CSA of at least 50 muscle fibers/animal were measured.
Statistical Analysis
IBM SPSS Statistics 19 was used for all statistical analysis. All data are presented as mean ± standard deviation (SD). Statistical comparisons of the different treatment groups were carried out by 1-way analysis of variance followed by Tukey’s test adjustments for multiple comparisons. P < .05 was considered statistically significant.
Results
d -Methionine Alleviates Weight Loss, Promotes Food and Water Intake During Cisplatin Treatment
A previous study has shown that guinea pigs treated with

Changes in body weight, food intake, and water intake during the experimental period among the 3 groups. (A) Body weights, (B) food intake, and (C) water intake were recorded daily. Data are presented as mean ± SD, n = 8. Differences were analyzed by 1-way analysis of variance. *Indicates statistical significance when compared with the saline-treated group (P < .05). #Indicates statistical significance when compared with the cisplatin-treated group (P < .05).
d -Methionine Alleviates the Decreases in Spleen and Liver Weights and Increases Some Relative Organ Weights
Table 2 shows that cisplatin treatment resulted not only in significant reductions in body weight and spleen and liver weights but also an increase in kidney relative weight compared with the saline-treated group. In the group receiving cisplatin combined with
Changes in Organ Weights and Tissue Somatic Indices (TSI) After Cisplatin and

Values are presented as mean ± SD.
TSI means relative tissue weight (tissue weight/body weight, g/g).
Indicates statistical significance when compared with the saline-treated group (P < .05).
Indicates statistical significance when compared with the cisplatin-treated group (P < .05).
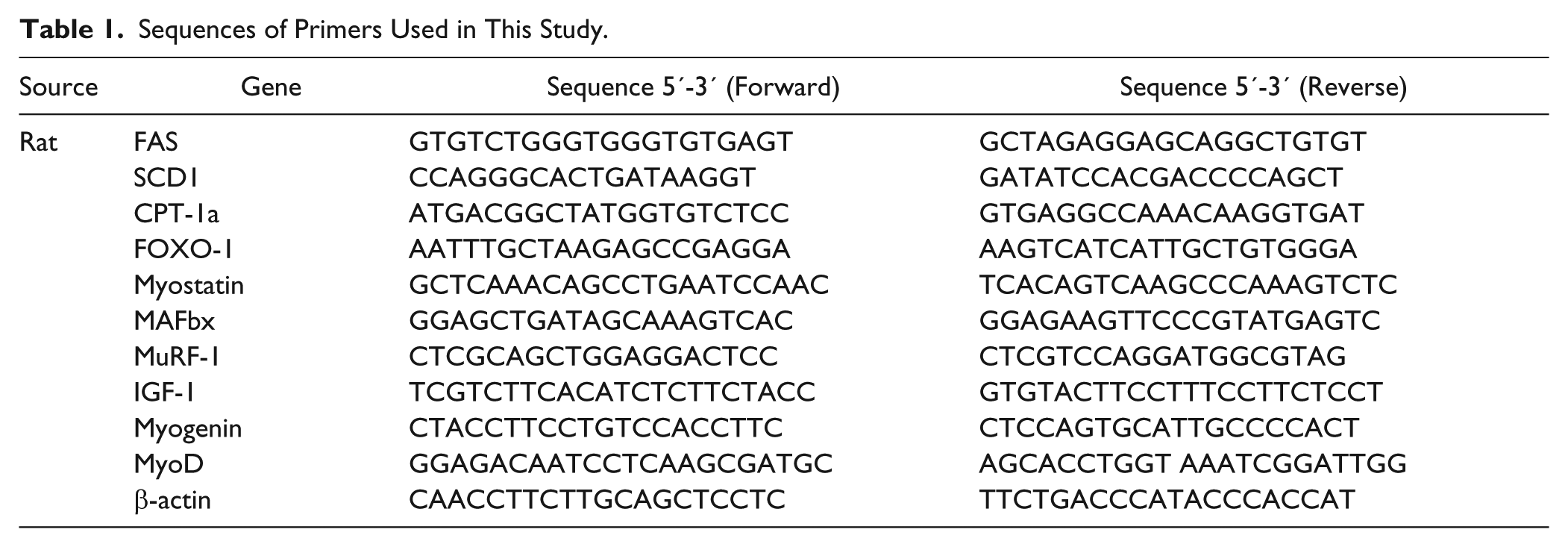
d -Methionine Administration Improves Gastrointestinal Dysfunction
Anorexia and early satiety are 2 main factors for decreased food intake after cisplatin treatment. It is widely accepted that cisplatin frequently induces gastrointestinal dysfunction.
18
To determine whether cisplatin treatment affect feeding efficiency and gastric emptying, we recorded the gastric content load and calculated the GEI on the day of euthanasia. There were visible signs of stomach distension and gastric stasis in cisplatin-treated rats when compared with saline and cisplatin combined with
Effects of
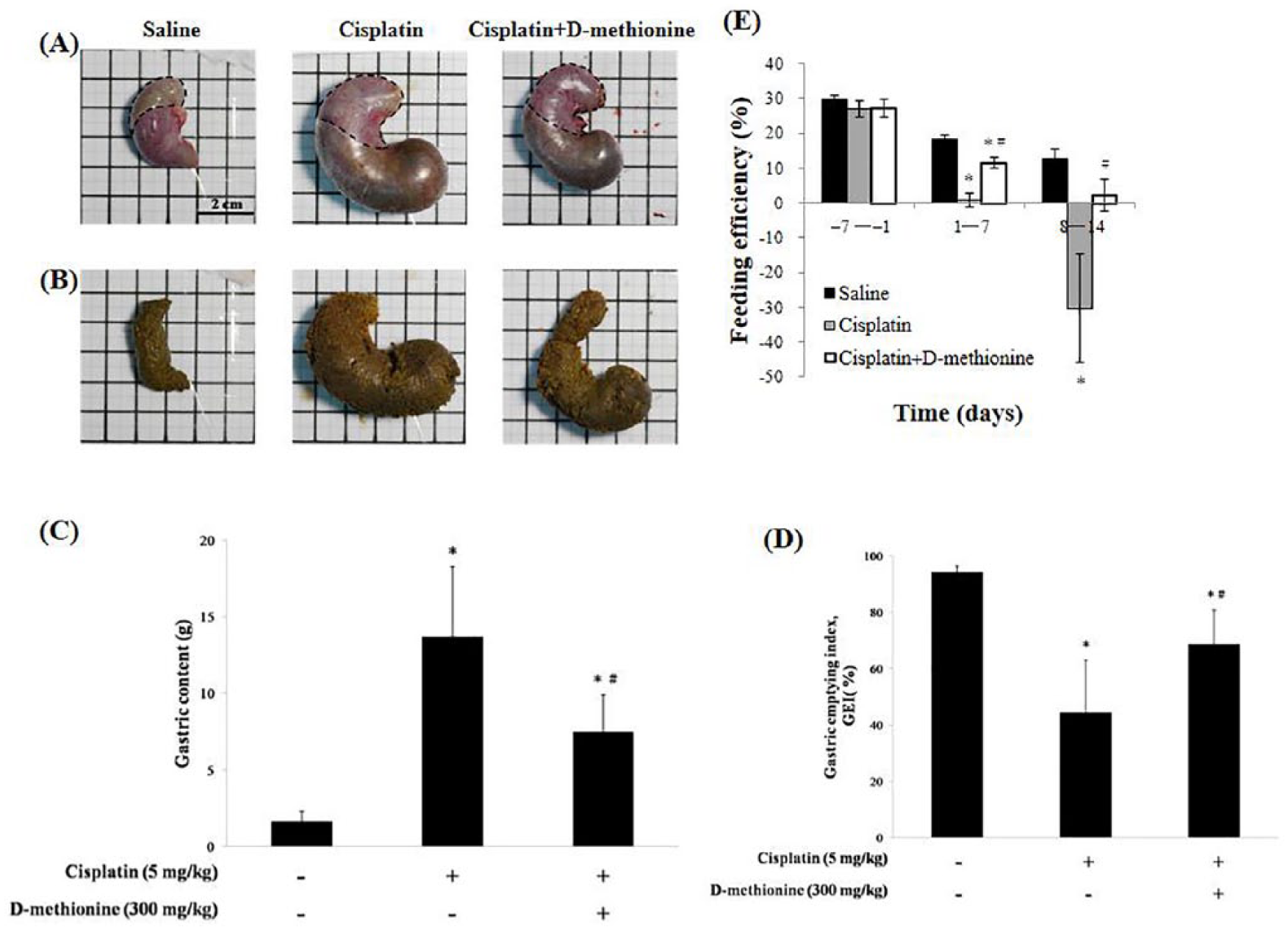
Effects of Cisplatin and d -Methionine on the Weights of Visceral Fat and Skeletal Muscle
We examined whether reduced body weight results in marked depletion of visceral fat and skeletal muscle, by measuring the weights of visceral adipose tissue (epididymal, mesenteric, and perirenal) and skeletal muscle (gastrocnemius muscle, soleus muscle, tibialis anterior muscle, and extensor digitorum longus). We found that the weights of visceral adipose tissue and skeletal muscle mass were significantly reduced in cisplatin-treated rats when compared with saline group (Table 3). In the cisplatin combined with
Weights of Visceral Adipose Tissue (Epididymal, Mesenteric, and Perirenal Fat) and Hind Leg Muscles (Gastrocnemius Muscle, Soleus Muscle, Tibialis Anterior Muscle, and Extensor Digitorum Longus) of the 3 Groups. a
Values are presented as mean ± SD.
Indicates statistical significance when compared with the saline-treated group (P < .05).
Indicates statistical significance when compared with the cisplatin-treated group (P < .05).
Effects of d -Methionine on Cisplatin-Induced Muscle Atrophy
The C2C12 cell line differentiates rapidly, forming contractile myotubes and producing characteristic muscle proteins. Hence, C2C12 myoblasts derived from satellite cells are commonly used as an in vitro system to study muscle development and differentiation. 3 The differentiation of C2C12 myoblasts into multinucleated myotubes is through cell fusion process. Terminally differentiated myotubes will form multinucleated myofibers which fill with myofibrils. Thus, skeletal muscles generally consist of bundles of multinucleated myofibers (fibroblast-like; fusiform). 19
Hind limbs in cisplatin-induced group were smaller than in saline-treated group and cisplatin combined with
Effects of d -Methionine on Hepatic Lipid Metabolism–Related gene Expressions Following Cisplatin Administration
Garcia et al
6
reported that cisplatin suppresses lipogenesis in liver. To understand the effects of

Effects of
Effects of d -Methionine on Muscle Atrophy–Related Gene Expressions Following Cisplatin Administration
To further investigate the possible anti-muscle atrophy mechanisms of
The Effect of Cisplatin and d -Methionine on Gene Expression of Myogenesis (Myogenin and MyoD) and Specific E3-Ubiquitin Ligases (MAFbx and MuRF-1) in C2C12 Myocytes
To further investigate whether there is a direct preventive effect of

Effects of
Trabecular Bone Microarchitecture Analysis
Micro-CT images demonstrated no differences in the microarchitecture of trabecular bone of tibia among the 3 groups of rats (Figure 6). Bone structural integrity and compactness were evident in the saline group. There were no differences in the degree of bone density in cisplatin and cisplatin combined with

Micro–computed tomography 3-dimensional image analysis of the tibia.
Morphological Parameters of Trabecular Bone Microarchitecture. a

Values are presented as mean ± SD.
Indicates statistical significance when compared with the saline-treated group (P < .05).
Indicates statistical significance when compared with the cisplatin-treated group (P < .05).
Histopathological Examination
As depicted in Figure 2, cisplatin inhibits food ingestion. Analytical results of H&E staining of mouse gastric fundus mucosa have revealed decreases in mucosal thickness and diffuse edema of submucosa after cisplatin administration. 21 We sought to see if cisplatin can lead to stomach injury, H&E staining was performed to evaluate histopathological changes to stomach tissues. No marked histopathological morphology was detected in the stomach tissues of the 3 groups (data not shown). Functional dyspepsia is a common functional gastrointestinal disorder without identifiable cause on conventional diagnostic evaluation. The symptoms are usually associated with food ingestion and include early satiety, feeling of fullness, bloating, belching, nausea, vomiting, and loss of appetite. 22 Thus, we speculated that cisplatin would cause functional dyspepsia rather than stomach injury.
Discussion
In the present study, we evaluated the efficacy of
Anorexia is usually defined as loss of desire to eat. Numerous studies have shown that cisplatin adversely affects food intake,3,5,7,23 which is significantly associated with declining body weight. In the present study, the toxic effects of cisplatin on appetite, body weight and feeding efficiency increased with accumulated treatment times and doses (Figure 2 E). This finding confirms that cisplatin-induced toxicity was dose dependent.
7
Changes in body weight and food intake after each cisplatin injection were reported by Malik et al,
24
who showed that administration of cisplatin significantly reduces food intake on the first day following cisplatin, reaching a nadir at 2 days and progressively recovering thereafter. We also observed the same phenomenon, as shown in Figure 1A and B. Our results revealed that the animals treated with
In addition, cisplatin has been shown to decrease feeding efficiency3,6 and to induce gastric stasis and stomach distension defined as an abnormal accumulation of gases and food in the stomach.
26
The relation of stomach distension to gastric emptying is shown in Figure 2A and D and reveals that gastric emptying is delayed by cisplatin, which is consistent with a previous report by Abalo et al.
27
Their radiographic study demonstrated that cisplatin induces notable gastric distension and this response is strongly visible for the first 2 hours after cisplatin injection.
27
Cisplatin may induce early satiety (the perceived feeling that the stomach is full soon after starting to eat) which is associated with reduced gastric emptying, and leads to appetite loss afterward. Our results revealed that
The complex multifactorial etiology of cisplatin-induced anorexia has yet to be fully elucidated. The results of in situ hybridization histochemistry and RT-PCR have shown that many feeding-regulating peptides in the hypothalamus, such as neuropeptide (NPY) and proopiomelanocortin (POMC), are involved in cisplatin-induced anorexia.24,29 In addition, cisplatin-induced anorexia is mediated by decreases in plasma ghrelin level and serotonin (5-HT).
30
The mechanisms of the protective effect of
Increase in kidney/body weight ratio has been suggested as a parameter of nephrotoxicity induced by cisplatin.
34
As shown in Table 2, the weights of spleen and liver decreased, and the ratios of heart/body weight, kidney/body weight and stomach/body weight increased, in cisplatin-treated rats. Orally administered
Loss of skeletal muscle mass is an important characteristic of cachexia.
3
FOXO-1, a member of the Foxo forkhead type transcription factors, plays a critical role in skeletal muscle atrophy signaling pathways and is markedly upregulated in skeletal muscle in energy-deprived conditions such as cisplatin-induced anorexia cachexia.
5
MAFbx and MuRF-1, ubiquitin-ligating enzymes, are involved in the proteolysis of myofibrillar proteins.
1
FOXO-1 can bind to the promoter regions of either the MuRF-1 or MAFbx gene, leading to increases in their expression levels within muscle. Increased expressions of MuRF-1 and MAFbx following atrophy stimulant are thought to be responsible for the shift in protein balance from net synthesis to net degradation, inducing a loss of muscle mass.
37
The cisplatin-induced wasting of muscle mass is mediated by the elevation of mRNA levels of MAFbx and MuRF-1, and by the decline in expressions of MyoD and myogenin.3,5 Consistent with these studies, we observed that the gene expressions of FOXO-1 and MAFbx in skeletal muscles are upregulated by cisplatin, resulting in muscle protein degradation. Long-term oral administration of
Cisplatin was found to induce C2C12 myotube atrophy either through the downregulation of Akt and myogenic differentiation (MyoD and myogenin) or the activation of autophagy system, myostatin pathways and ubiquitin ligases.3,20 A previous study demonstrated that the induction of C2C12 myotube atrophy is due to the activation of the NF-κB signaling pathway, not the ubiquitin-proteasome system.
39
The mechanisms of muscle wasting by cisplatin are complex and multifactorial. In agreement with the findings of previous reports,
3
cisplatin caused direct atrophy of C2C12 myotubes, characterized by increases in the levels of ubiquitin ligase (MAFbx and MuRF-1) mRNA expressions
36
and decreases in the mRNA levels of myogenic regulatory factors (MyoD and myogenin). The effects of cisplatin treatment on C2C12 cells are considered predictive of impairment of the process of muscle differentiation.
From Table 3, the rats treated with cisplatin combined with
It is also well known that chemotherapy can disrupt the bone microenvironment, leading to changes in marrow cellular composition.
40
Our results indicated that cisplatin treatment with/without
In conclusion, the chemopreventive efficacy of
Footnotes
Acknowledgements
The authors would like to thank Dr Kai-Li Liu (Department of Nutrition, Chung Shan Medical University) for her kind instructions and advice in maintaining the muscle cell culture and muscle fiber analysis. We also thank the Taiwan Mouse Clinic for technical support in micro-CT experiment.
Ethical Approval and Consent to Participate
The experimental protocol for this study was approved by the Instituted Animal Care and Use Committee, Chung Shan Medical University Experimental Animal Center, Taichung, Taiwan (Approval No: 1439). The treatment of the rats was according to the guidelines of the Instituted Animal Care and Use Committee of Chung Shan Medical University for the care and use of laboratory animals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Chung Shan Medical University Grant (CSMU-SHOW-105-02 and CSMU-JAH-106-03).



