Abstract
Introduction
Use of complementary and alternative medicines (CAMs) is widespread in patients with chronic diseases, particularly cancer. Although the rate of use varies depending on patient demographics and cancer type, it has been estimated that around half of all patients use CAM therapy in Western countries. 1 Patients with breast cancer are among the most likely users of CAMs, with a recent study reporting that 67% of women initiated some form of CAM following a diagnosis of breast cancer. 2
Despite the high prevalence of CAM use in patients with cancer, clinical evidence of their benefit is sparse, and although they are often assumed to be safe with regard to concurrent therapies, few studies have been carried out to demonstrate this.3,4 Furthermore, oncologists are often unaware that their patients are taking CAMs.5-7 This is particularly concerning considering the narrow therapeutic index of many anticancer agents, where small changes in pharmacokinetics could potentially lead to significant adverse or subtherapeutic effects.
Fucoidans are a group of sulfated carbohydrates derived from marine brown algae (eg, Fucus vesiculosus, Cladosiphon okamuranus, Laminaria japonica, and Undaria pinnatifida) and have long been used as dietary supplements in Asia for medicinal purposes. The reported benefits of fucoidan extracts include anticancer, anticoagulant, anti-inflammatory, antiviral, and immunomodulating activities. 8 The anticancer activity of fucoidans is well documented, with the first reports appearing in the 1980s.9,10 Since then, there have been numerous studies to show fucoidans can both directly and indirectly induce apoptosis in vitro in a number of different cancer cells lines.11-17 In recent years, there has been a growing body of evidence to suggest that the in vitro anticancer activity of fucoidans can be translated in vivo in animal models.17-22 One study investigated orally administered fucoidan (derived from Cladosiphon sp) in patients with colorectal cancer and found significantly reduced fatigue and increased tolerability to chemotherapy. 23 Patients taking fucoidan also had prolonged survival; however, this was not statistically significant in the small sample studied. Fucoidan is included in various dietary supplements that are sometimes taken by cancer patients. The possibility for developing fucoidan as an adjunct or sole anticancer agent has been discussed.24,25
There are only a few studies regarding the potential systemic availability of fucoidan after oral administration. Clinical studies indicate only very low absorption,26,27 in line with that of other carbohydrates, such as chondroitin sulfate. Animal studies show that there is potential for some fucoidan uptake and localization in organs and tissues. 28 There are currently no validated methods for the measurement of serum levels of fucoidan, so they were not assessed in the study described here.
There appear to be no studies that have investigated the potential pharmacokinetic interactions between fucoidans and conventional cancer therapies. The aim of this study was to investigate the effect of Undaria fucoidan co-administration on the pharmacokinetics of 2 commonly used hormonal therapies, letrozole and tamoxifen, in patients with breast cancer. Plasma concentrations are reported for letrozole, tamoxifen, and 2 active metabolites of tamoxifen, 4-hydroxytamoxifen and endoxifen (4-hydroxy-N-desmethyl-tamoxifen), before and after administration with fucoidan. In addition, patients were monitored for potential adverse effects.
Methods
Patients
This clinical study was carried out at the Royal Hobart Hospital, Hobart, Australia. Participants had active breast cancer and had been taking a stable once-daily oral dose of either letrozole (2.5 mg) or tamoxifen (20 mg) for a minimum of 4 weeks prior to the study, to ensure steady state had been achieved, with at least 3 weeks of therapy remaining in their treatment course. The patients were not randomized but were already taking their medication as advised by their physician. Other inclusion criteria were as follows: aged ≥18 years; able to complete documentation of the treatment and adverse events, and attend follow-up; and able to swallow capsules whole. Patients were excluded if they met any of the following criteria: reluctance or inability to cease other CAM at least a week prior to trial commencement; ECOG (Eastern Cooperative Oncology Group) performance of ≥3; life expectancy of ≤12 weeks; impaired hematopoietic (white blood cells < 3.0 × 109/L, absolute neutrophil count < 1.5 × 109/L, platelet < 100 × 109/dL), renal (glomerular filtration rate < 50 mL/min), or hepatic function (either aspartate transaminase/alanine transaminase > 2.5 ULN, or > 5 × ULN in case of liver metastases, or bilirubin > 1.5 × ULN); pregnancy or lactation; cerebral or leptomeningeal metastases that were unstable in spite of appropriate therapy; serious intercurrent illness; major surgery within 2 weeks prior to study commencement; concurrent radiotherapy; bowel obstruction; documented allergy to fucoidan; concurrent warfarin therapy; and participation in trials of other pharmacological agents during the time of this study.
Study Design and Procedures
Fucoidan was given in the form of Maritech extract (Marinova Pty Ltd, Hobart, Australia), which was derived from Undaria pinnatifida using the proprietary Maritech extraction process. The total daily dose of Maritech extract was 1000 mg, given as one 500 mg capsule in the morning and at night after food. The study medication was manufactured under the code of Good Manufacturing Practice. The Maritech extract was standardized to 88.9% fucoidan content.
At baseline, patients underwent a physical examination and demographics were collected. Blood samples were also collected for toxicity and pharmacokinetic analysis. In addition to their existing hormonal therapy, patients then commenced fucoidan administration for a 3-week period. At the end of the dosing interval, patients underwent the same physical examination, and blood samples were collected for toxicity and pharmacokinetic analysis.
Adverse drug reactions of letrozole and tamoxifen were graded using the NCI Common Terminology Criteria for Adverse Events Version 4.0 for hematological and nonhematological toxicities. Adherence to fucoidan was measured by telephone calls and inspection of returned bottles on completion of the study.
The patients were asked about any new symptoms or changes since the introduction of fucoidan. The research nurse noted comments by subjects. The study was approved by the Human Research Ethics Committee (Tasmania) Network, and all patients provided written informed consent prior to study entry.
Materials
Reference standards of letrozole, tamoxifen, 4-hydroxytamoxifen, and endoxifen were purchased from Sigma-Aldrich (Castle Hill, New South Wales, Australia). Solid-phase extraction cartridges were purchased from Waters (Waters Corporation, Milford, MA). Water was prepared in-house using a Milli-Q gradient water purification system (Millipore, Bedford, MA), and all other solvents and chemicals were of high-performance liquid chromatography (HPLC) or analytical grade.
Sample Collection
Pre and post extract blood was collected in 2 ethylenediaminetetraacetic acid tubes (18 mL of blood per participant). One sample was sent to a NATA (National Association of Testing Authorities, Australia) accredited laboratory (Hobart Pathology, Tasmania) to assess hematological and biochemical toxicity, which was determined using the following tests: urea, electrolytes, and creatinine; liver function tests (LFTs); and full blood count. All hematological toxicity tests were performed according to NATA guidelines using standardized methods. The second blood sample was centrifuged within 30 minutes of collection at 2000g for 10 minutes at room temperature. The plasma was transferred using a disposable pipette into cryovials and stored in a −20°C freezer for later analysis of letrozole, tamoxifen, and tamoxifen metabolites.
Sample Preparation for Letrozole, Tamoxifen, and Tamoxifen Metabolite Analysis
Plasma samples (5 mL) were first protein-precipitated using ethanol (45 mL). The samples were vortex-mixed and kept overnight at −20°C, followed by centrifugation at 12 000g for 15 minutes. The resultant supernatant was concentrated on a miVac DNA centrifugal concentrator (Genevac Ltd, Suffolk, UK) to 2 mL. Ammonia buffer solution (1 mL, 0.025 mM, pH 10) was then added and samples were further extracted through solid-phase extraction cartridges. Solid-phase extraction cartridges were conditioned with methanol (1 mL), followed by water (2 mL), and the samples were loaded onto the cartridges and eluted under vacuum. Cartridges were again washed with water (2 mL) followed by extraction using methanol (4 × 250 µL). The resultant eluate was centrifuged at 12 000g for 10 minutes and subjected to HPLC analysis. All plasma samples were prepared in triplicate and each triplicate was analyzed in duplicate.
HPLC-CAD (Charged Aerosol Detector) Conditions
Chromatographic analyses for letrozole, tamoxifen, 4-hydroxytamoxifen, and endoxifen were performed on a HPLC system (Thermo Fisher Scientific, North Ryde, New South Wales, Australia) consisting of a HPG-3400RS binary separation pump, WPS-3000TRS auto-sampler, and a TCC-3000RS column thermal compartment. The system was connected to Corona Ultra RS charged aerosol detector (Thermo Fisher Scientific), using a nitrogen flow of 35 psi and nebulizer temperature of 30°C. Instrument control and data acquisition were performed using Chromeleon software.
Separation of analytes was achieved using an Acquity UPLC HSS C18 SB column (100 × 2.1 mm internal diameter, 1.8 µm particle size; Waters, Milford, MA). The mobile phase consisted of 0.1% trifluoroacetic acid in water (mobile phase A) and 0.1% trifluoroacetic acid in acetonitrile (mobile phase B), with a flow rate of 1 mL/min and gradient elution of mobile phase B from 25% to 55% over 45 minutes. The temperature of the column and sample compartments was set at 40°C and 5°C, respectively, with an injection volume of 50 µL.
Calibration standards for letrozole, tamoxifen, 4-hydroxytamoxifen, and endoxifen were prepared in triplicate by spiking control plasma samples with standard analyte solutions and extracted in the same way as that detailed above for the test plasma samples. Calibration curves were linear over a concentration range of 15.6 to 500 ng/mL for letrozole and tamoxifen, and 1.56 to 50 ng/mL for 4-hydroxytamoxifen and endoxifen. Assay performance was assessed over 5 days (n = 6) at 3 concentrations for all analytes. Intra- and interday accuracy, precision, and reproducibility (% RSD) were found to be less than 9.1%, 10.7%, and 1.9%, respectively, for all analytes, and the extraction recovery of analytes from plasma samples was found to be at least 89.6% (range = 89.6% to 95.2%).
Data Analysis
Steady-state trough concentrations of letrozole, tamoxifen, 4-hydroxytamoxifen, and endoxifen were compared before and after fucoidan treatment, using a paired t test. A P value of less than .05 was considered significant.
Results
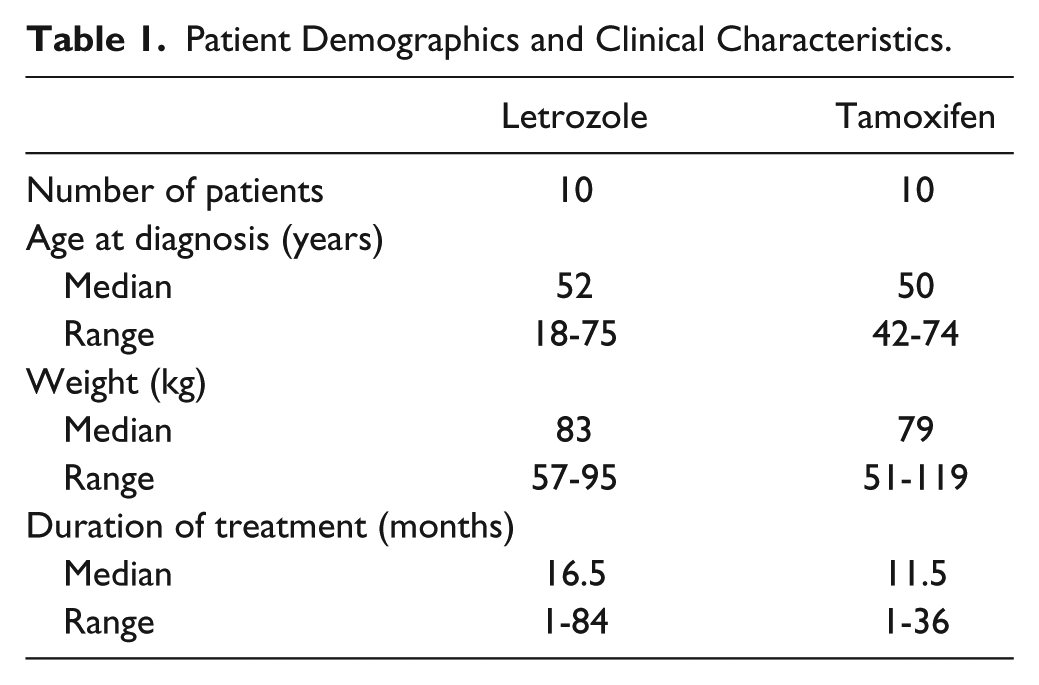
Twenty patients, all female, were included in this study; 10 were taking letrozole and 10 were taking tamoxifen. The median age of the participants was 59 years, with a range of 43 to 76 years. Patient demographics are summarized in Table 1. All participants successfully completed the study. There was a variety of self-reported subjective adverse events, including nausea, diarrhea, constipation, and headache. All were mild and transient. Conversely, 4 patients, 2 taking letrozole and 2 taking tamoxifen, reported a reduction in their bone and muscular pain during the 3-week fucoidan treatment period, as detailed in Table 2. There was no significant change in blood parameters (LFTs, renal function tests, and hematology) over the study period in all patients (data not shown), with one exception. Prior to fucoidan treatment, the LFTs for this patient were mildly abnormal (γ-glutamyl transferase [GGT] 140 units) and after 3 weeks of fucoidan, her GGT levels remained elevated (174 units). The cause of the raised GGT levels was not known; specifically, there was no evidence of hepatic metastases.
Patient Demographics and Clinical Characteristics.
Patients With Reduction in Pain.

Individual trough concentrations of letrozole, tamoxifen, 4-hyroxytamoxifen, and endoxifen before and after 3 weeks of treatment with fucoidan are shown in Figure 1. No significant differences in these concentrations were detected, and the results are summarized in Table 3. Normalizing for body weight, height, and age had no influence on drug or metabolite levels and did not alter the statistical outcome (data not shown).

Plasma trough concentrations (ng/mL) of (A) letrozole, (B) tamoxifen, (C) 4-hydroxytamoxifen, and (D) endoxifen before and after 3 weeks of fucoidan treatment in individual patients receiving either letrozole (n = 10) or tamoxifen (n = 10).
Mean Plasma Trough Concentrations (ng/mL) of Letrozole, Tamoxifen, 4-Hydroxytamoxifen, and Endoxifen, Before and After 3 Weeks of Fucoidan Administration a .

N = 10; data are presented as mean ± standard deviation.
Paired t test.
Discussion
The use of CAMs in conjunction with standard cancer therapies has become commonplace over the past 20 years. Despite this, clinical evidence of their safety when used with conventional therapies remains scarce. In addition, due to the “natural” origin of many CAMs, it is often assumed by patients that these products are safe and nontoxic, and therefore fail to mention CAM use to their oncologist.3,4,7,29 As evidenced by many potent conventional therapies, including anticancer agents, being derived from natural products (eg, digoxin, morphine, docetaxel, topotecan) and with the potential to produce serious toxicity if used inappropriately, the assumption of CAM safety can be erroneous. Well-conducted trials investigating the effect of CAMs on conventional drugs are warranted to generate evidence-based information on the safe combined use of these agents.
There is an abundance of in vitro studies, and an increasing number of in vivo studies in animal models, showing the anticancer activity of the commonly used CAM, fucoidan. 8 As a result, research interest and a wider public awareness of its use as a potential anticancer agent has grown in recent years. A number of studies have demonstrated that fucoidan, irrespective of source, does not appear to exert any toxic effects in animals or humans when given as monotherapy, after either acute or chronic exposure. For instance, doses of up to 2000 mg/kg/day have been administered to rats with no adverse effects. 30 Clinical trials in humans have reported no adverse effects at oral doses of 1 g daily for 12 weeks, 31 or 3 g daily for 12 days. 32 However, there have been no studies to date to investigate if fucoidan has any effect on the pharmacokinetics of conventional cancer therapies.
Letrozole and tamoxifen were studied as they are among the most commonly utilized drugs in breast cancer patients. While there was no reason to think that fucoidan would interfere with hormonal therapy, there was a possibility that fucoidan would interfere with gastrointestinal absorption of the drugs, due to being a poorly absorbed and large molecular weight carbohydrate. However, this study showed that the co-administration of Undaria fucoidan did not significantly influence the steady-state trough plasma concentrations of the hormonal therapies, letrozole and tamoxifen, in patients with breast cancer. It also showed no significant differences in the plasma levels of the active metabolites of tamoxifen, 4-hydroxytamoxifen and endoxifen, before and after fucoidan administration, although a mean decrease of 21% in the level of 4-hydroxytamoxifen and a mean increase of 25% in the level of endoxifen was observed after fucoidan administration (see Figure 1 and Table 3). 4-Hydroxytamoxifen and endoxifen are considerably more potent than tamoxifen.33,34 In the case of 4-hydroxytamoxifen, plasma concentrations of this metabolite are not thought to correlate with treatment outcomes in breast cancer, 35 and so the reduction observed in this study is unlikely to be of clinical significance. On the other hand, the long-term benefit of tamoxifen therapy in breast cancer is thought to be directly related to maintaining endoxifen plasma concentrations above a threshold level.35,36 Although the change in endoxifen levels post-fucoidan was not statistically significant, further investigation is warranted to ascertain if there are any clinical consequences of the observed increase in plasma levels. The study was only a 3-week study, but that is quite sufficient for a drug interaction study. This study period is ample given the short half-lives of the anticancer drugs examined (ie, any changes in their steady-state plasma levels would be apparent by that time). Also, any potential effect of fucoidan on liver enzymes (whether inducing or inhibiting) would be fully apparent in that timeframe.
There have been a limited number of clinical studies investigating the effect of other CAMs on the pharmacokinetics of anticancer agents.37-42 One noteworthy study by Goey and coworkers showed that St John’s wort, commonly used as a “natural” antidepressant, significantly increased docetaxel clearance through induction of CYP3A4, giving rise to the potential for subtherapeutic dosing of this anticancer agent. 39 Garlic has also been shown to exert some inhibitory effects on docetaxel clearance; however, the changes reported were unlikely to result in any clinically significant consequences. 37 More recently, fish oil was shown to reduce the efficacy of cisplatin in tumor-bearing mice due to the presence of a chemoresistance-inducing fatty acid, raising concerns over the use of fish oil and consumption of certain fish by patients undergoing chemotherapy. 42 Other trials have demonstrated no clinically significant interactions between CAMs and anticancer agents; these include Echinacea with docetaxel, 38 ginkgo biloba with letrozole and tamoxifen, 40 and milk thistle with irinotecan. 41 Results from the current study can now be added to this small body of evidence to allow more informed patient-physician discussions and decisions regarding the use of fucoidan with hormonal cancer therapy.
Although biological activity has been reported after oral ingestion, 8 little research has been carried out to examine the absorption and fate of fucoidan and, consequently, the mechanisms of any potential interactions remain unknown. Fucoidans are large molecular weight polysaccharides, ranging from 10 kDa to 500 kDa, and therefore oral absorption and systemic exposure is thought to be low. 8 Nevertheless, using ELISA techniques, fucoidan has been detected in the blood and urine of humans after oral ingestion.26,27 Nagamine and co-workers recently demonstrated a dose-dependent increase in transport of Cladosiphon okamuranus-derived fucoidan across Caco-2 cells, and detected fucoidan in rat jejunal and liver cells after oral ingestion. 28 These findings give rise to the possibility of distribution, metabolism, and elimination based drug interactions.
Unlike the docetaxel-St John’s wort interaction, where cytochrome P450 enzymes are well known to play a key role,43-46 there is no evidence to suggest fucoidan affects the drug metabolizing enzymes responsible for the metabolism of letrozole or tamoxifen. Letrozole and tamoxifen are metabolized by CYP2A6 and CYP2D6, respectively, and genetic polymorphisms in these genes result in interindividual variation in the metabolism of these compounds.47,48 We did observe a large interpatient variability in drug and metabolite levels, which is consistent with other pharmacokinetic studies on letrozole47,49 and tamoxifen.50,51 Fucoidan has not been systematically tested against CYP enzymes, and the limited evidence from the small sample size in this study did not indicate any effect on the CYP enzymes.
In conclusion, administration of fucoidan showed no effect on the steady-state plasma concentrations of letrozole or tamoxifen and was well tolerated, with patients reporting no adverse effects that could be attributed to this dietary supplement.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr J. Helen Fitton is an employee of Marinova Pty Ltd, which supplied the fucoidan for the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this project was provided by the Royal Hobart Hospital Research Foundation.
