Abstract
Danggui Buxue Decoction (DBD), a classical formula of traditional Chinese medicine (TCM), has an impact on promoting hematopoiesis. The aim of our study was to determine whether DBD can prevent myelosuppression in breast cancer patients treated with adjuvant chemotherapy. We conducted a phase II randomized prospective controlled clinical study. From December 2013 to February 2015, 106 patients were enrolled and randomly assigned (1:1) to the TCM group and control group. The primary end point was incidence of grade 3-4 neutropenia. The secondary end points included incidence of grade 3-4 neutropenia in each cycle, incidence of anemia, and incidence of thrombopenia during 4 cycles. Seventeen patients withdrew from this study, and 89 patients were included in the final analysis. Incidences of grade 3-4 neutropenia during 4 cycles were 57.1% in the TCM group and 59.6% in the control group, and there was no significant difference (P = .816). Similarly, no significant differences were observed between the 2 groups for incidence of grade 3-4 neutropenia in each cycle. While incidences of anemia were 54.8% and 66.6% for the TCM group and control group, respectively (P = .280), incidences of thrombopenia were 11.9% for the TCM group and 4.3% for the control group (P = .248). No significant differences were observed for the incidence of other nonhematological toxicities between the 2 groups. DBD failed to prevent myelosuppression in breast cancer patients treated with adjuvant chemotherapy. Further studies are warranted to validate the efficacy of DBD in selected patients.
Keywords
Introduction
Chemotherapy is one of the main effective systemic treatments for breast cancer, previous studies have shown that anthracycline-based polychemotherapy reduced the annual breast cancer death rate by about 20% to 38%. 1 Nevertheless, patients who receive chemotherapy also suffer from different grades of hematologic and nonhematologic toxicities, such as neutropenia, thrombopenia, anemia, nausea, vomiting, diarrhea, stomatitis, and neuropathy. 2 Among those, acute myelosuppression is the most common hematologic toxicity and often occurs within a very short time after chemotherapy, which is associated with increased risk of life-threatening infections as well as chemotherapy dose reductions and delays that may compromise treatment outcomes. 3 Studies have revealed that incidence of grade 3-4 neutropenia in breast cancer patients treated with conventional chemotherapy regimens is 43% to 65%.4-6
To reduce the risk of chemotherapy-induced neutropenia, especially febrile neutropenia, the routine method is prophylactic treatment with granulocyte-colony stimulating factors (G-CSFs), such as filgrastim, lenograstim, and pegfilgrastim. 7 However, these drugs are not only expensive but also have some side effects. Beyond that, traditional Chinese medicine (TCM) is another choice to prevent neutropenia that is less expensive and has fewer adverse events, although there is a lack of high-level evidence and efficacy of TCM is controversial. 8 There are many successful medicines that have been derived from TCM in history, such as artemisinin and arsenic trioxide.9,10 Previous reports indicated that TCM treatment could reduce toxic side effects during radiotherapy and chemotherapy, such as myelosuppression, gastrointestinal reactions, hepatic or renal impairment, and skin and mucosa radiation injuries.11,12 Danggui Buxue decoction (DBD) is a classical formula of TCM to raise the “Qi” and nourish the “Blood,” consisting of Radix Angelicae sinensis (Danggui; RAS) and Radix Astragali (Huangqi; RA) with a ratio of 1:5. 13 Chemical components of TCM are complex; astragaloside IV, calycosin, formononetin, ferulic acid, and ligustilide have been extracted and verified from DBD. 14 Fundamental studies have revealed that DBD treatment significantly promotes hematopoiesis and thrombopoiesis in myelosuppression mouse model. 15 Here, we report a phase II prospective study exploring the efficacy of DBD in preventing myelosuppression induced by chemotherapy in breast cancer patients (ClinicalTrial.gov Number NCT02005783).
Patients and Methods
Study Design
This was a phase II prospective randomized controlled and open-label clinical study. Postoperative female breast cancer patients who were suggested to receive adjuvant chemotherapy with EC (epirubicin plus cyclophosphamide) or TC (docetaxel plus cyclophosphamide) were included. A stratified block randomization method was used. Patients in the study were stratified by chemotherapy regimens (EC or TC) and randomly assigned (1:1) to the TCM group or control group. Patients in the TCM group received EC or TC chemotherapy and DBD treatment, while patients in the control group only received EC or TC chemotherapy. All patients in our study were not allowed to be treated with G-CSFs proactively.
EC regimens were administrated with epirubicin 90 mg/m2 and cyclophosphamide 600 mg/m2 once every 21 days for 4 cycles. TC regimens were administrated with docetaxel 75 mg/m2 and cyclophosphamide 600 mg/m2 once every 21 days for 4 cycles. All patients were proactively treated with ondansetron hydrochloride 8 mg intravenously to decrease the risk of vomiting induced by chemotherapy. Patients receiving TC regimens also took dexamethasone tablets 7.5 mg orally twice a day for 3 days (the days before, on and after chemotherapy was administered). Patients in the TCM group received 1 dose of DBD (RAS 6 g and RS 30 g, concentrated traditional Chinese medicine granules; PuraPharm Corporation) twice daily during 4 cycles. Patients suffering from grade 3-4 neutropenia in the study were allowed to be treated with G-CSFs.
Eligibility Criteria
Inclusion criteria were as follows: women aged ≥18 years and <70 years; histologically confirmed invasive breast cancer and suggested to receive EC/TC regiments for adjuvant chemotherapy by the center’s multidisciplinary team; leukocyte ≥3 × 109/L; neutrophil ≥1.5 × 109/L; platelets ≥100 × 109/L; serum AST (aspartate aminotransferase)/SGOT or ALT (alanine aminotransferase)/AGPT ≤2.5 times of upper limit of normal range; serum creatinine/BUN equal to or less than the upper limit of normal range; no dysphagia and able to take the DBD; provided written informed consent according to the local ethics committee requirements; and an Eastern Cooperative Oncology Group (ECOG) Performance Score of 0 or 1. Patients were excluded if they had events as follows: metastatic breast cancer; family history of endometrial cancer or any other kind of gynecologic cancer; severe comorbidity that indicates intolerance to adjuvant chemotherapy; psychiatric disorder or other diseases leading to noncompliance to the therapy; known severe hypersensitivity to any drugs in this study; prior adjuvant chemotherapy of any kind of cancer; and hematologic disorders relative to aplasia. Patients could withdraw their consent if they had serious unacceptable adverse events or breached the study protocol, such as taking DBD irregularly or prophylactic treatment with G-CSFs.
All patients gave their written informed consent before inclusion in this study. The study was conducted in accordance with the Declaration of Helsinki. The protocol was reviewed and approved by the independent ethical committee/institutional review board.
Clinicopathological Information
Clinical data of patients enrolled in the trial were obtained from the electronic medical record system of Ruijin Hospital, including age, ECOG score, menstrual status, body mass index, parameters of routine blood tests, and biochemical parameters. Pathological sections were reviewed by 2 pathologists independently, including pathological type, histological grade, tumor size, number of lymph node metastases, estrogen receptor (ER) and progesterone receptor (PR), CerbB-2 and fluorescence in situ hybridization (FISH) test. ER and PR positivity was defined as more than 1% positive tumor cells with strong nuclear staining. Human epidermal growth factor receptor-2 (HER-2) positive status was confirmed by means of immunohistochemistry (with CerbB2 3+ indicating positive status) or FISH (with an amplification ratio ≥2.0 indicating positive status). Breast cancer staging was performed according to the seventh edition of American Joint Committee on Cancer (AJCC). 16 The treatment information of patients was obtained from the database of the center’s multidisciplinary team.
Assessment of Safety
Adverse events were recorded each cycle, including the severity of symptoms and frequency of adverse events. Data collected included leukocyte count, neutrophil count, hemoglobin, platelet count, fatigue, fever, infection, myalgia, arthralgia, stomatitis, diarrhea, nausea, vomiting, phlebitis, elevated liver enzymes, alopecia, rashes, and other reported adverse events. Every adverse event was graded according to the Common Terminology Criteria for Adverse Events, Version 3.0, from the US National Cancer Institute. Treatments and outcomes of the adverse events were also recorded.
Statistical Analysis
The study was designed to have a power of 80% to detect an absolute reduction of 20 percentage points for grade 3-4 neutropenia from 60% to 40% in the TCM group, at a 2-sided significance level of .05. The target enrollment was 53 eligible patients for each group based on the Simon 2-stage design, considering a withdrawal rate of 15%.
The primary end point of this study was incidence of grade 3-4 neutropenia, which was defined as the proportion of patients suffering from grade 3-4 neutropenia during 4 cycles of adjuvant EC or TC chemotherapy. The secondary end points were as follows: incidence of grade 3-4 neutropenia in each cycle; incidence of any grade anemia during 4 cycles of chemotherapy; incidence of any grade thrombopenia during 4 cycles of chemotherapy. Categorical variables between different groups were presented as frequencies and compared using χ2 tests (2-sided Pearson or Fisher exact test). Statistical analyses were performed by SPSS software (version 13.0, SPSS Inc, Chicago, IL). A P value <.05 was considered statistically significant.
Results
Study Population
A total of 106 postoperative early breast cancer patients who were suggested to receive EC or TC chemotherapy were enrolled and randomly assigned (1:1) to the TCM group and the control group in our prospective study from December 2013 to February 2015. Of these patients, 83 patients received EC regimens and 23 patients received TC regimens. Seventeen patients withdrew from this study due to different reasons, and the total rate of withdrawal was 16.0% (Figure 1). Therefore, with a follow-up of 4 chemotherapy cycles, 89 patients were included in the final analysis. No chemotherapy dose reduction was necessary for patients in our study, and 2 patients who received EC regimens had treatment delays because of severe neutropenia.

Enrollment and follow-up.
The clinicopathological characteristics of patients from the 2 groups are listed in Table 1. Among the 89 patients, 50 patients (56.2%) were ≤50 years and 39 patients were older than 50 years. A majority of tumors (94.4%) were invasive ductal carcinoma, and the remaining 5 tumors were invasive lobular carcinoma and mucinous carcinoma. Thirty-seven tumors were grade I to II, 47 tumors were grade III, and the other 5 tumors did not have grade information. Among these patients, 26 patients were AJCC stage I, 49 patients were stage II, and 14 patients were stage III. A total of 62 (69.7%) patients were ER positive, 48 (53.9%) patients were PR positive, and only 19 (21.3%) patients were HER-2 positive. There were no significant statistical differences between the TCM group and the control group for all of these clinicopathological variables.
Baseline Clinicopathological Characteristics.
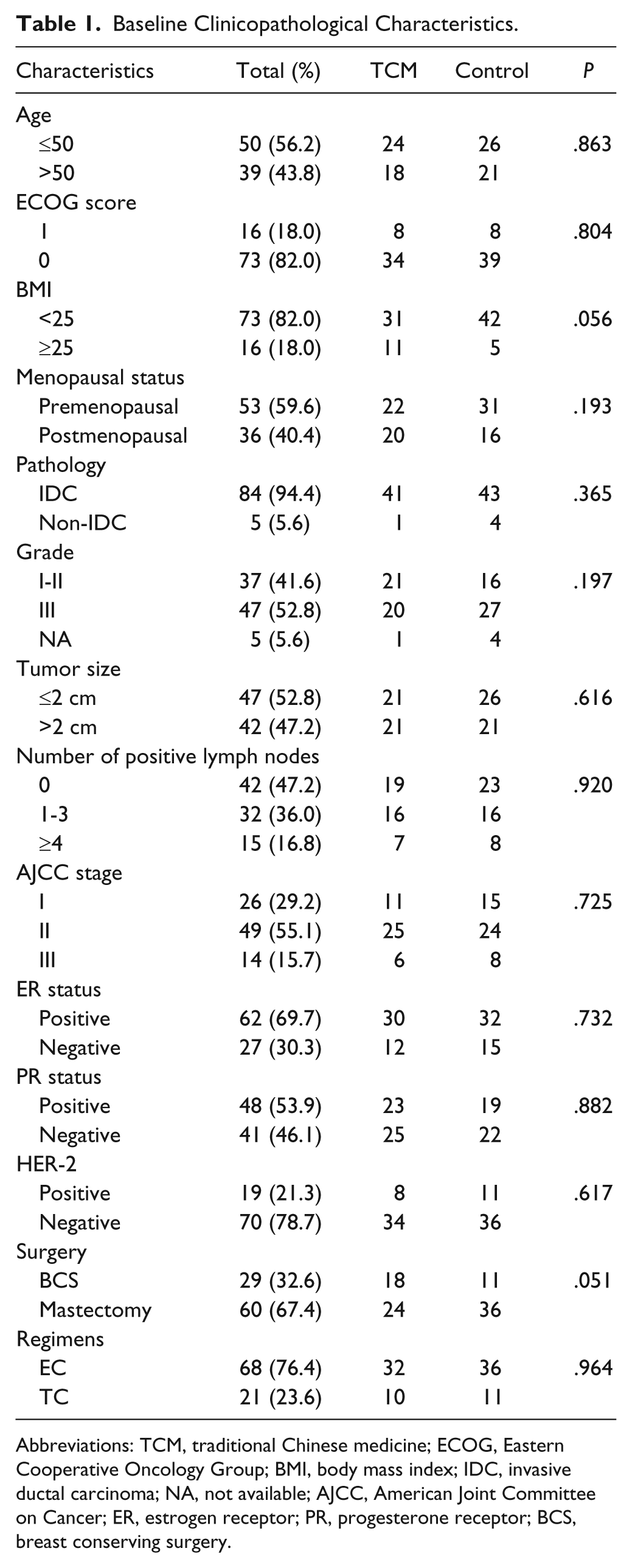
Abbreviations: TCM, traditional Chinese medicine; ECOG, Eastern Cooperative Oncology Group; BMI, body mass index; IDC, invasive ductal carcinoma; NA, not available; AJCC, American Joint Committee on Cancer; ER, estrogen receptor; PR, progesterone receptor; BCS, breast conserving surgery.
Incidence of Grade 3-4 Neutropenia
During 4 cycles, a total of 24 (57.1%) patients in the TCM group and 28 (59.6%) patients in the control group had the adverse event of grade 3-4 neutropenia. There was no significant difference between the 2 groups (P = .816; see Figure 2 and Table 2). Only one patient in the TCM group had the adverse event of febrile neutropenia.

Incidence of grade 3-4 neutropenia, anemia, and thrombopenia.
Incidence of Grade 3-4 Neutropenia, Anemia, and Thrombopenia.

Abbreviations: AE, adverse event; TCM, traditional Chinese medicine; RR, relative risk; CI, confidence interval.
In the first cycle, there were 19 (45.2%) patients in the TCM group and 22 (46.8%) patients in the control group having the adverse event of grade 3-4 neutropenia (P = .882). In the second cycle, there were only 7 and 11 patients suffering from grade 3-4 neutropenia, respectively, and no significant difference between 2 groups was observed (P = .430). In the third cycle, there were 9 and 13 patients experiencing grade 3-4 neutropenia, respectively (P = .496). And in the fourth cycle, 8 patients in the TCM group and 11 patients in the control group had the adverse event of grade 3-4 neutropenia. There was no significant difference between them (P = .617; see Figure 3 and Table 3). In addition, incidence of grade 3-4 neutropenia in the first cycle was significantly higher than that in the subsequent 3 cycles (P < .001).
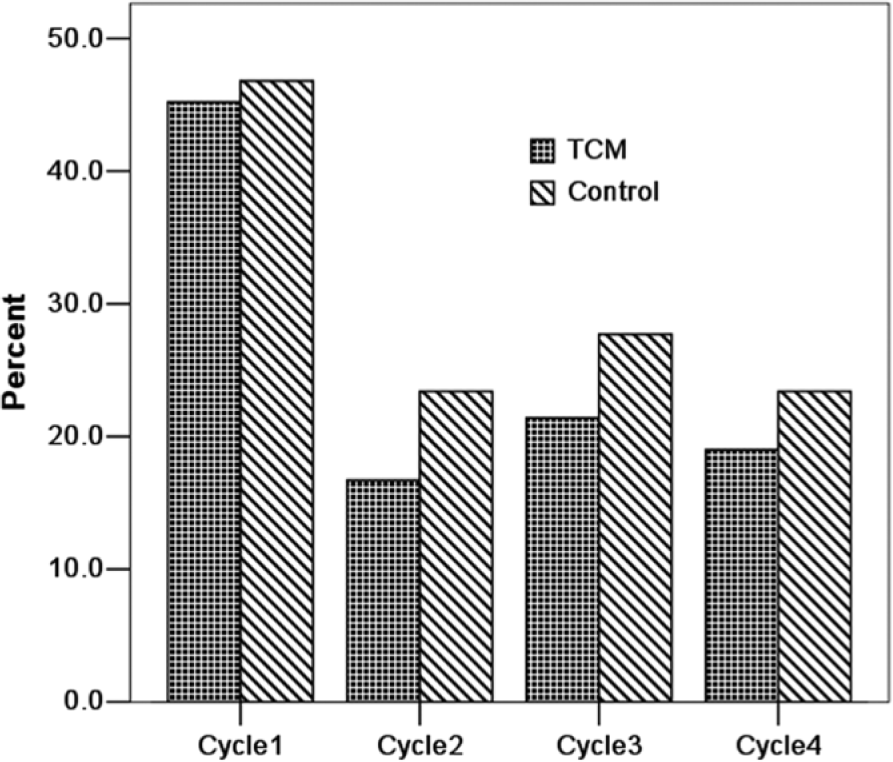
Incidence of grade 3-4 neutropenia in every cycle.
Incidence of Grade 3-4 Neutropenia in Every Cycle a .

Abbreviations: TCM, traditional Chinese medicine; RR, relative risk; CI, confidence interval; OR, odds ratio.
First cycle versus second to fourth cycles: OR = 2.085, 95% CI = 1.517-2.866, P < .001.
According to prior stratification, 68 patients received EC regimens and 21 patients received TC regimens (Table 1). Incidence of grade 3-4 neutropenia in patients receiving EC regimens were 65.6% for the TCM group and 66.7% for the control group, for which there was no significant difference (P = .928), while incidence of grade 3-4 neutropenia in patients receiving TC regimens were 30.0% for the TCM group and 36.4% for the control group. Similarly, no significant differences was observed between the 2 groups (Table 4).
Incidence of Grade 3-4 Neutropenia in Patients Treated With EC or TC Regimen.

Abbreviations: TCM, traditional Chinese medicine; RR, relative risk; CI, confidence interval; OR, odds ratio; EC, epirubicin plus cyclophosphamide; TC, docetaxel plus cyclophosphamide.
Incidence of Anemia and Thrombopenia
Incidences of any grade anemia and thrombopenia are shown in Table 2 and Figure 2. Among these patients, 23 (54.8%) patients in the TCM group and 31 (66.0%) patients in the control group suffered from anemia (P = .280). At the same time, the incidences of thrombopenia were 11.9% and 4.3% in the 2 groups, respectively; no significant difference was observed (P = .248). Only 2 patients in the control group suffered from grade 3 anemia and all the others were grade 1-2 anemia. No patients had the adverse event of grade 3-4 thrombopenia.
Nonhematological Toxicities
In addition to these hematological toxicities, many patients also suffered from different grades of nonhematological toxicities in the follow-up, most of which were grade 1 or 2 (Table 5). All the patients in the 2 groups had alopecia. The second most common were gastrointestinal reactions: incidences of nausea and vomiting in the TCM group were 81.0% and 66.7%, while incidences of these 2 adverse events in the control group were 85.1% and 74.5%, respectively. Fatigue induced by chemotherapy was very common: 31 (73.8%) patients in the TCM group and 32 (68.1%) patients in the control group had different grades of fatigue. A small number of patients in the 2 groups also had other adverse events, such as fever, infection, myalgia, arthralgia, stomatitis, diarrhea, phlebitis, elevated liver enzymes, and rash.
Incidence of Other Nonhematological Toxicities.
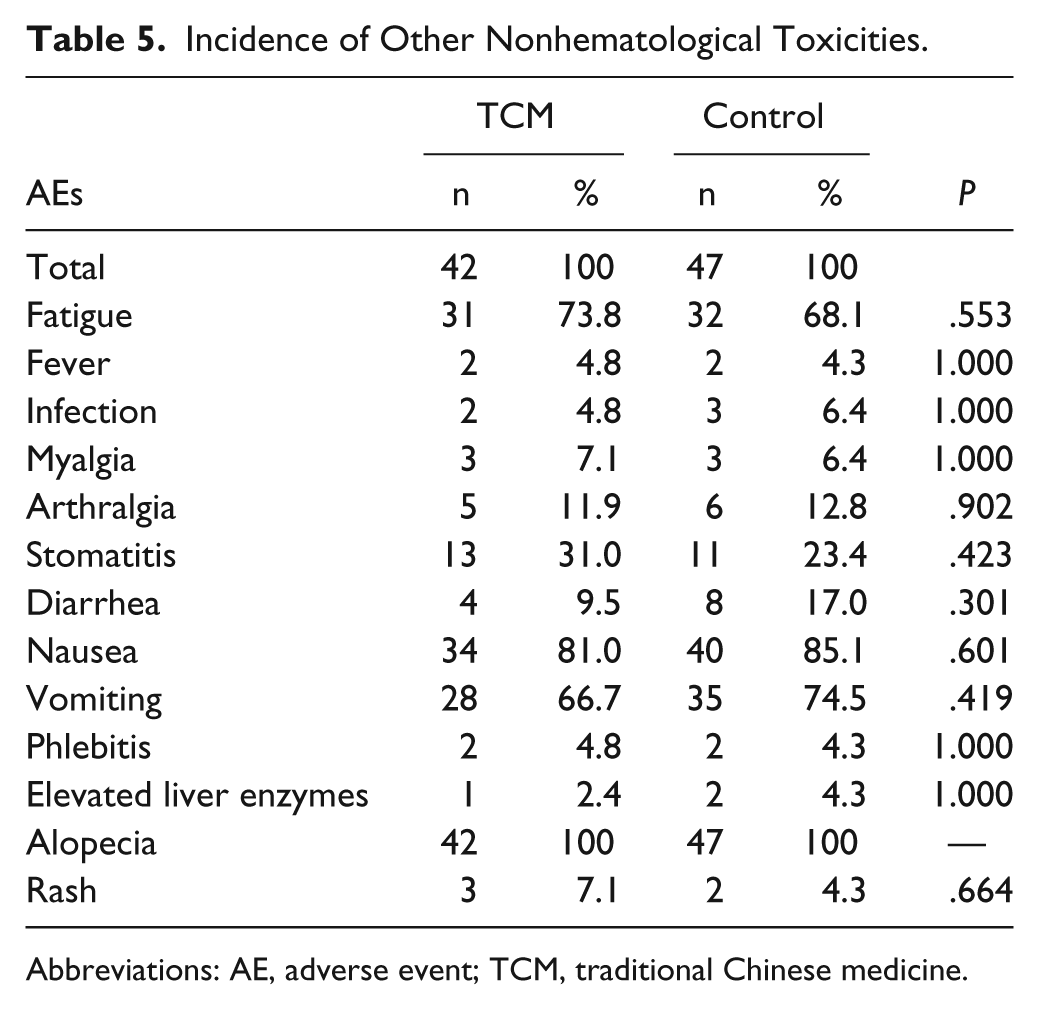
Abbreviations: AE, adverse event; TCM, traditional Chinese medicine.
Discussion
Our randomized prospective study tested the addition of DBD, a classical formula of TCM, as prevention for myelosuppression induced by chemotherapy in breast cancer patients and did not identify any meaningful improvement in the incidence of grade 3-4 neutropenia, anemia, or thrombopenia.
This result was not consistent with fundamental research. A previous in vivo study indicated that DBD treatment significantly increased the white blood cell counts and hematopoietic colony-forming unit (CFU) of myeloid cells in a myelosuppression mouse model. 15 However, in our study, the total incidence of grade 3-4 neutropenia was 57.1% in the TCM group and 59.6% in the control group; no significant improvement was observed after addition of DBD. Similarly, there were no significant differences for grade 3-4 neutropenia in each cycle. Nevertheless, incidence of grade 3-4 neutropenia in the first cycle was 45.2% and 46.8% for the 2 groups, respectively, but it decreased rapidly in the subsequent 3 cycles, ranging from 16.7% to 27.7%. This phenomenon is similar to previous reports that most neutropenia induced by chemotherapy occurs in the first cycle.3,17 In a study about patients with non–small cell lung cancer who were treated with CDE (cyclophosphamide, doxorubicin, and roxithromycin), incidence of febrile neutropenia was 24% in the first cycle and ranged from 3% to 11% in the subsequent cycles. 18 Beyond that, other studies also showed that 75% of febrile neutropenia occurred in the first cycle in breast cancer patients who were treated with docetaxel and doxorubicin. 3 This may be related to patients’ tolerance to chemotherapy and acceleration of bone marrow proliferation in the subsequent cycles, although the mechanism is still unclear.
According to prior stratification, subgroup analysis showed that there were no significant differences in the incidence of grade 3-4 neutropenia between 2 groups in patients treated with either EC or TC chemotherapy. However, we observed that incidence of grade 3-4 neutropenia was lower in patients treated with the TC regimen than patients treated with the EC regimen. By contrast, previous studies revealed that docetaxel had more hematological toxicity than doxorubicin. 19 In a phase III study, patients treated with TC suffered higher incidence of grade 3-4 neutropenia than AC (doxorubicin and cyclophosphamide; 61% vs 55%), 4 while the NATT trial at our center also showed that incidence of grade 3-4 neutropenia was only 24% in patients treated with TC, which was close to this study. 20 To analyze the reasons, we found that patients treated with TC in the NATT trial and our study both received dexamethasone prophylactically for anaphylactic reaction and body fluid retention. Meanwhile, dexamethasone could promote hematopoiesis and increase peripheral blood cell counts, which was the main treatment for neutropenia and thrombocytopenia caused by myelodysplastic syndrome and mononucleosis.21,22
As a consequence of myelosuppression, anemia and thrombocytopenia were also adverse events of chemotherapy. Previous studies revealed that DBD significantly enhanced the platelet recovery and CFU-megakaryocytic lineages in vivo.15,23 In addition, DBD also stimulated the erythropoietin expression through the hypoxia-inducible factor pathway in vitro.24,25 However, our study did not observe any improvement in the incidence of anemia and thrombocytopenia induced by chemotherapy after administration of DBD either. Beyond that, incidences of other nonhematological toxicities in the TCM group were similar to those in the control group, and safety of DBD was verified.
Our study did not give us an exciting result that DBD could prevent myelosuppression induced by chemotherapy with lower expenses and fewer adverse effects. Reasons were complicated as DBD was not a single chemical drug but a traditional Chinese formula. One reason may be that dosage of DBD in our study was not high enough to prevent myelosuppression induced by chemotherapy. Besides, chemical components of DBD are complex and the truly effective component is still unclear, dosage of which may be very low, just as the artemisinin derived from Artemisia annua for antimalaria treatment. 9 Beyond that, peripheral blood parameters were affected by various physiological, pathological, and physical factors. 26 In China, there are an abundance of traditional nutritional supplements and a variety of diets, which possibly affected our results. Another reason may be that we did not use DBD in selected populations dialectically as the original theory of TCM, which recommended us to treat patients with personalized medicine. 27 Of course, efficacy of DBD may not be as good as ancient books described, although some basic research revealed that DBD could promote hematopoiesis and thrombopoiesis in vivo and in vitro.15,24
Conclusion
In conclusion, our phase II clinical study demonstrated that DBD, a classical formula of TCM, failed to prevent myelosuppression in breast cancer patients treated with EC or TC chemotherapy. But the safety of DBD was verified in our study. Further studies about effective components extracted from DBD are warranted to find an ideal medicine that can prevent myelosuppression induced by chemotherapy in selected patients with fewer expenses and adverse effects.
Footnotes
Acknowledgements
We would like to thank all the patients who participated in our study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the grants from National Natural Science Foundation of China (Grant Numbers: 81172520, 81202087, 81202088, 81472462), Technology Innovation Act Plan of Shanghai Municipal Science and Technology Commission (Grant Numbers: 14411950200, 14411950201), Natural Science Foundation of Shanghai Municipal Science and Technology Commission (Grant Number: 12ZR1446400), and Joint Research Project of the Emerging Cutting-Edge Technology of Shanghai Shen-Kang Hospital Development Center (Grant Number: SHDC12014103).
