Abstract
Coenzyme Q0 (CoQ0; 2,3-dimethoxy-5-methyl-1,4-benzoquinone), a major active constituent of Antrodia camphorata, has been shown to inhibit human triple-negative breast cancer (MDA-MB-231) cells through induction of apoptosis and cell-cycle arrest. Ecological studies have suggested a possible association between ultraviolet B (UVB) radiation and reduction in the risk of breast cancer. However, the underlying mechanism of the combination of CoQ0 and UVB in human estrogen receptor–positive breast cancer (MCF-7) remains unclear. In this study, the possible effect of CoQ0 on inducing apoptosis in MCF-7 cells under exposure to low-dose UVB (0.05 J/cm2) has been investigated. CoQ0 treatment (0-35 µM, for 24-72 hours) inhibits moderately the growth of breast cancer MCF-7 cells, and the cell viability was significantly decreased when the cells were pretreated with UVB irradiation. It was noted that there was a remarkable accumulation of subploid cells, the so-called sub-G1 peak, in CoQ0-treated cells by using flow cytometric analysis, which suggests that the viability reduction observed after treatment may result from apoptosis induction in MCF-7 cells. CoQ0 caused an elevation of reactive oxygen species, as indicated by dichlorofluorescein fluorescence, and UVB pretreatment significantly increased CoQ0-induced reactive oxygen species generation in MCF-7 cells. In addition, cells were exposed to CoQ0, and the induction of DNA damage was evaluated by single-cell gel electrophoresis (comet assay). CoQ0-induced DNA damage was remarkably enhanced by UVB pretreatment. Furthermore, CoQ0 induced apoptosis in MCF-7 cells, which was associated with PARP degradation, Bcl-2/Bax dysregulation, and p53 expression as shown by western blot. Collectively, these findings suggest that CoQ0 might be an important supplemental agent for treating patients with breast cancer.
Introduction
Breast cancer is the leading cause of cancer death for women in Taiwan. 1 The incidence rate of breast cancer among Chinese women has increased enormously with changes in lifestyle. Although incidence of breast cancer is increasing worldwide, the mortality rate has declined in developed countries as a result of a range of measures, including implementation of screening and introduction of adjuvant systemic treatments.2,3 Studies reveal that about two thirds of breast cancer patients have estrogen-receptor (ER)–dependent tumors and most patients are treated with tamoxifen (an ER antagonist) and aromatase inhibitors, which abates estrogen activity. 4 However, concerns about the safety and effectiveness of hormone therapy still exist due to side effects that substantially affect the patient’s quality of life. Therefore, it is necessary to explore more efficient potential therapeutic strategies to reduce the risk of breast cancer. 5
Ultraviolet (UV) radiation is a part of the sunlight spectrum, which can be categorized into 3 bands—UVA (320-400 nm), UVB (280-320 nm), and UVC (100-280 nm)—according to wavelength. 6 Among these, DNA is the best-known target of UVB. 7 Low doses of UVB cause DNA mutations, which eventually lead to tumor initiation, whereas high doses result in irreparable DNA damage, causing apoptosis and cell death. 8 The presence of singlet oxygen in apoptosis has also been reported in UVB-irradiated retinal pigment epithelial cells. 9 A combined use of UVB irradiation and arsenic treatment exhibits the antiproliferative and pro-apoptotic effects via activation of caspase-8 and -9 in keratinocytes. In addition, it has also been reported that UVB radiation efficiently increased the apoptosis in an ER-positive breast cancer cell line. 10
Antrodia camphorata, an indigenous medicinal mushroom, has been used in traditional Chinese medicine for protection of diverse health conditions such as muscle damage, fever, food poisoning, drug intoxication, diarrhea, hypertension, skin irritation, and cancer. 11 Accumulating evidence suggests that Antrodia camphorata possesses a broad range of biological activities, including antioxidant, anticancer, antihyperlipidemic, immunomodulatory, antimetastasis, hepatoprotective, antihypertensive, and anti-inflammatory properties.11,12 The mycelium and the fruiting bodies of Antrodia camphorata exerted strong anticancer activity against several cancers such as breast, liver, prostate, colon, bladder, oral, pancreatic, lung, and leukemic cells. The inhibitory effect against cancer cells by Antrodia camphorata could be mediated by different cellular mechanisms of actions, such as inhibition of metastasis and angiogenesis regulatory proteins; regulation of oncogene and tumor suppressor gene expression; downregulation of signal transduction cascades involving Nrf2, NF-κB, AP-1, and MAPK; induction of cell-cycle arrest; and apoptosis involving the Wnt/β-catenin, p53, death ligands, Bcl-2, and caspase families.11,13,14 According to our previous results of high-performance liquid chromatography analysis, the amount of CoQ0 in the fermented culture broth of Antrodia camphorata was 17.3% (254 nm). 15
Coenzyme Q0 (CoQ0 or Ubiquinone 0) is a redox-active ubiquinone compound that accumulates predominantly in mitochondria. Recently, we have reported the anti-angiogenic and anti-inflammatory properties of CoQ0 in vitro or in vivo.16,17 Several studies suggest that CoQ0 exhibits strong toxicity toward various cancer cell lines.18,19 CoQ0 treatment also was shown to decrease the cell proliferation in HepG2, A549, and SW480 cancer cell lines; 18 stimulate insulin secretion in pancreatic islets; 20 possess anti-angiogenic properties; 16 and inhibit oxidative damage in mouse blood and tissues. Despite its cytotoxicity, some in vivo studies exhibited no deleterious effects of a CoQ0 analog in combination with other nutrients. Notably, administration of CoQ0 mixture inhibited oxidative damage in blood, heart, liver, kidney, and spleen of rodents.21,22 Nevertheless, pharmacological activities of a single CoQ0 molecule against cancer and redox imbalance have not been fully studied, and precise signaling pathways involved are largely unknown.
Accumulating evidence suggests that many natural compounds from food and plants have chemotherapeutic and chemopreventive effect in several human cancers.23,24 A number of natural products extracted from Chinese herbs has been found to enhance chemotherapy by inducing apoptosis and exhibiting anticancer potential both in vitro and in vivo.25-27 These studies indicate effects of CoQ0 on anticancer activity against human triple-negative breast (MDA-MB-231) cancer cells through induction of apoptosis and cell-cycle arrest. 19 In our previous study, we demonstrated that CoQ0, a major active constituent of AC, significantly inhibited melanoma cell growth through the induction of cell-cycle arrest and apoptosis via Wnt/β-catenin signaling pathways. 28 Studies have suggested a possible association between UVB radiation and reduction in the risk of breast cancer. 29 However, the regulatory mechanisms of CoQ0 that produces its pro-apoptosis effects in MCF-7 breast cancer are unknown. In the current study, the effect of CoQ0 treatment alone and in combination with UVB has been examined on the cellular growth of MCF-7 breast cancer cells.
Materials and Methods
Reagents and Antibodies
CoQ0 (2,3 dimethoxy-5-methyl-1,4 benzoquinone) was purchased from Sigma-Aldrich (St Louis, MO). Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS),
Cell Culture
The estrogen receptor–positive MCF-7 (human breast adenocarcinoma) cell line was obtained from the Bioresource Collection and Research Center (BCRC, Taiwan). MCF-7 cells were grown in DMEM supplemented with 10% heat-inactivated FBS, 2 mM glutamine, and 1% penicillin-streptomycin-neomycin in a humidified incubator (5% CO2 in air at 37°C). Cultures were harvested and monitored for cell number by counting cell suspensions with a hemocytometer. Cell morphology was examined using phase-contrast microscopy (200× magnification).
UVB Irradiation and Sample Treatment
Prior to UVB irradiation, MCF-7 cells were washed with phosphate-buffered saline (PBS) and resuspended in fresh phenol red–free DMEM containing 1% FBS. Then, cells were exposed to UVB radiation at dose 0.05 J/cm2 (λmax, 312 nm; no detectable emission below 280 nm) using UVllink CL-508M (UVItec, Cambridge, UK) for 30 seconds. After UVB irradiation, the cells were treated with CoQ0 (0-35 µM) for 72 hours in DMEM containing 10% FBS.
Assessment of Cell Viability by MTT Assay
Cell viability was determined by the MTT colorimetric assay. MCF-7 cells (5 × 104 cells/well in 24-well plates) were treated with various concentrations of CoQ0 (0-35 µM) for 24 to 72 hours, before 400 µL 0.5 mg/mL MTT in PBS was added to each well. After incubation at 37°C for 2 hours, an equal volume of DMSO (400 µL) was added to dissolve the MTT formazan crystals, and the absorbance was measured at 570 nm (A570) using an ELISA microplate reader (µ-Quant, Winooski, VT). The percentage of cell viability was calculated as follows: (A570 of treated cells/A570 of untreated cells) × 100.
Flow Cytometric Analysis
Cellular DNA content was determined by flow cytometry using the propidium iodide (PI)–labeling method as described previously. 14 Briefly, MCF-7 cells were seeded at a density of 1 × 106 cells/dish in 10-cm dishes. After UVB (0.05 J/cm2) irradiation, cells were treated with increasing concentrations of CoQ0 (0-35 µM) for 72 hours. Cells were harvested, fixed in ice-cold 70% ethanol, stored at 4°C, washed with PBS (pH 7.2), treated with 25 µg/mL RNase A at 37°C for 15 minutes, and stained with 50 µg/mL PI for 20 minutes. Flow cytometric profiles were analyzed with ModFit software (Verity Software House, Topsham, ME). Apoptotic nuclei were identified as a subploid DNA peak and were distinguished from cell debris on the basis of forward light scatter and PI fluorescence.
Measurement of Reactive Oxygen Species Generation
Intracellular reactive oxygen species (ROS) accumulation was detected by fluorescence microscopy using the cell-permeable fluorogenic probe DCFH2-DA method. Cells (1 × 105 cells/12-well) were cultured in DMEM medium that had been supplemented with 10% FBS, and the culture medium was replaced when the cells had reached 80% confluence. After UVB (0.05 J/cm2) irradiation, cells were treated with CoQ0 (35 µM) for 20 minutes. Then culture supernatants were removed, and incubated with nonfluorescent DCFH2-DA (10 µM) in fresh medium at 37°C for 30 minutes. ROS production was measured by changes in fluorescence due to the intracellular accumulation of dichlorofluorescein (DCF), which is caused by the oxidation of DCFH2. The DCF fluorescence stained cells were captured using fluorescence microscope (200× magnification; Olympus, Center Valley, PA). The fluorescence intensity was quantified using analysis LS 5.0 soft image solution (Olympus Imaging America Inc). The fold-increase of ROS generation was compared with the vehicle-treated cells, which were arbitrarily considered as 1-fold.
Comet Assay
The comet assay (single-cell gel electrophoresis assay) is an uncomplicated and sensitive technique for the detection of DNA damage at the level of individual eukaryotic cells. 30 The protocol used for the comet assay followed previously published guidelines. 31 After UVB (0.05 J/cm2) irradiation, MCF-7 cells (1 × 106 cells/10-cm dish) were treated with CoQ0 (35 µM) for 72 h. Cells were then suspended in 1% low-melting-point agarose in PBS (pH 7.4) and pipetted onto superfrosted glass microscope slides that had been precoated with a layer of 1% normal-melting-point agarose (which was warmed to 37°C prior to use). The agarose was allowed to set at 4°C for 10 minutes; then, the slides were immersed in lysis solution (containing 2.5 M NaCl, 100 mM EDTA, 10 mM Tris, and 1% Triton X-100) at 4°C for 1 hour. The slides were then placed in single rows inside a 30-cm wide horizontal electrophoresis tank containing 0.3 M NaOH and 1 mM EDTA (pH 13.4) at 4°C for 40 minutes to allow the separation of the 2 DNA strands (alkaline unwinding). Electrophoresis was performed in the unwinding solution at 30 V (1 V/cm), 300 mA, for 30 minutes. The slides were then washed 3 times for 5 minutes each with 0.4 M Tris (pH 7.5) at 4°C before they were stained with DAPI (1 µg/mL). Observations were made at a final magnification of 200× using a UV microscope with a 435 nm excitation filter. Visual scoring of the cellular DNA on each slide was based on the characterization of 100 randomly selected nucleoids. Comet-like DNA formations were categorized into 5 classes (0, 1, 2, 3, or 4) representing increasing DNA damage in the form of a “tail.” Each comet was assigned a value according to its class. The overall score for 100 comets ranged from 0 (100% of comets in class 0) to 400 (100% of comets in class 4), and the overall DNA damage in the cell population was expressed in arbitrary units. 32 The observation and analysis of the results were always performed by the same experienced observer. The observer was blinded and had no knowledge of the slide identity. DNA damage in cells, which was specified as DNA strand breaks including double and single-strand variants at alkali-labile sites, was analyzed under alkaline conditions (pH 13.4). DNA migration from the nucleus of each cell was measured with a computer program using the comet moment parameters as follows: comet moment = ∑0→n [(amount of DNA at distance X) × (distance X)]/total DNA. The tail moments were quantified using the Cometscore software (Sumerduck, VA).
Protein Isolation and Western Blot Analysis
After UVB (0.05 J/cm2) irradiation, MCF-7 cells (1 × 106 cells/10-cm dish) were incubated with CoQ0 (35 µM) for 72 hours. After incubation, the cells were harvested, pooled, washed once with PBS, and suspended in 100 µL of lysis buffer (10 mM Tris-HCl, pH 8, 320 mM sucrose, 1% Triton X-100, 5 mM EDTA, 2 mM DTT, and 1 mM PMSF). The cell lysates were maintained on ice for 20 minutes and then centrifuged at 15 000 × g for 30 minutes at 4°C. Total protein content was determined using Bio-Rad protein assay reagent (Bio-Rad, Hercules, CA), BSA as a standard. The protein extracts were mixed with sample buffer (62 mM Tris-HCl, 2% SDS, 10% glycerol, and 5% β-mercaptoethanol), and the mixture was boiled at 97°C for 5 minutes. Equal amounts (50 µg) of denatured protein samples were separated by SDS-PAGE (sodium dodecyl sulfate polyacrylamide gel electrophoresis) and then transferred onto polyvinylidene difluoride membranes overnight. The membranes were blocked with 5% nonfat dried milk in PBS containing 1% Tween-20 for 1 hour at room temperature, followed by incubation with primary antibodies overnight. The membranes were then incubated with either a horseradish peroxidase–conjugated anti-rabbit or anti-mouse antibodies for 2 hours prior to development using a chemiluminescent substrate (Millipore, Billerica, MA). Densitometric analyses were performed using commercially available quantitative software (AlphaEase, Genetic Technology Inc, Miami, FL), with the control representing 1.0-fold.
Statistical Analyses
The obtained data were analyzed using analysis of variance (ANOVA), followed by Dunnett’s test for pairwise comparison. Mean values are presented with their standard deviation (mean ± SD). Statistical significance between the treatments was defined as P < .05 for all tests.
Results
CoQ0 Enhances UVB-Induced Cell Death in MCF-7 Cells
Prior to investigating the apoptotic effects of CoQ0, the cytotoxic effects of CoQ0 on human breast cancer MCF-7 cells were examined using an MTT colorimetric assay. The cells were exposed to 0 to 35 µM of CoQ0 for 24 to72 hours. As shown in Figure 1, treating MCF-7 cells with 35 µM CoQ0 for 24 and 48 hours resulted in no significant effect on cell viability. However, cell viability dramatically decreased when the MCF-7 cells were treated with CoQ0 (35 µM) for 72 hours (Figure 1A-C).

CoQ0 treatment significantly enhances UVB-mediated inhibition of MCF-7 cell growth. (A-C) Cells were treated with various concentrations of CoQ0 (0-35 µM) for 24 to 72 hours. (D) Cells were irradiated with UVB (0.05 J/cm2) and then treated with or without CoQ0 (0-35 µM) for 72 hours. The effects of CoQ0 alone and along with UVB were determined by MTT assay. The results are the mean ± SD of 3 assays. *P < .05; **P < .01; ***P < .001 compared with untreated control cells. #P < .05; ##P < .01; ###P < .001 compared with UVB-irradiated cells.
In addition, CoQ0-enhanced cell death was assessed by pretreating the MCF-7 cells with UVB (0.05 J/cm2) for 72 hours. We observed that exposure to UVB (0.05 J/cm2) dramatically induced MCF-7 cell death, which was significantly increased by CoQ0 (35 µM, Figure 1D). These data further confirm that UVB-induced cell damage was remarkably enhanced by CoQ0 (35 µM). The sublethal concentration of CoQ0 (35 µM) were used for subsequent experiments.
CoQ0 Increases Apoptosis in UVB-Irradiated MCF-7 Cells
Flow cytometric analyses were conducted to assess the apoptotic levels in CoQ0 (0-35 µM) and UVB (0.05 J/cm2)-irradiated MCF-7 cells by measuring the fluorescence of the PI-DNA complex. The results showed that treatment of MCF-7 cells with CoQ0 dose-dependently increased the sub-G1 peak as compared to control (Figure 2A and C). Furthermore, UVB pretreatment possessed a greater ability to induce sub-G1 peak in MCF-7 cells than CoQ0 and UVB alone (Figure 2B and D). These data further suggest that CoQ0 treatment may enhance UVB-induced apoptosis in MCF-7 cells.
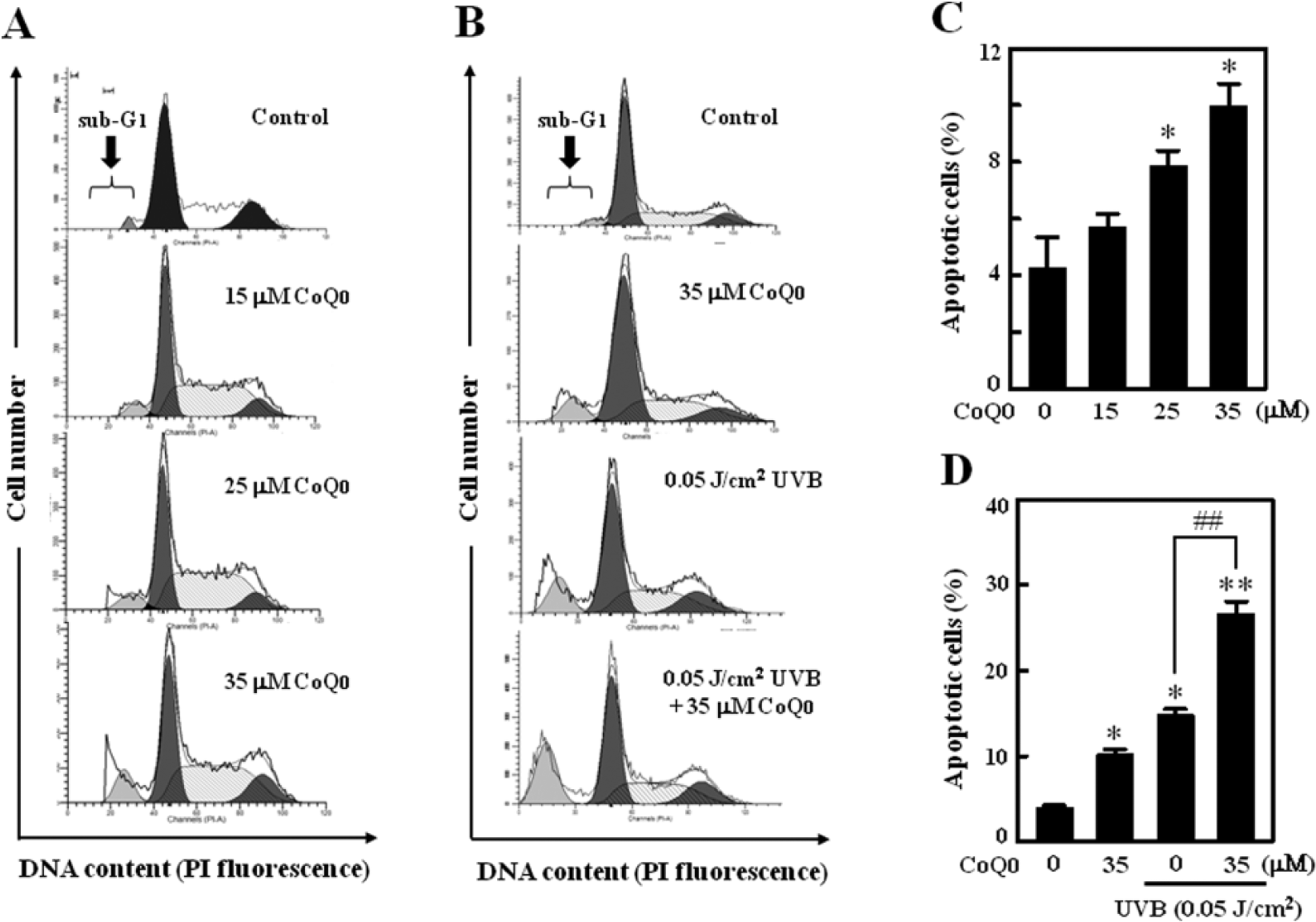
CoQ0 treatment significantly enhances sub-G1 peak in UVB-irradiated MCF-7 cells. (A) Cells were treated with various concentrations of CoQ0 (0-35 µM) for 72 hours. (B) Cells were irradiated with UVB (0.05 J/cm2) and then treated with or without CoQ0 (0-35 µM) for 72 hours. Cellular DNA content was determined by flow cytometry using the PI-labeling method as described in Materials and Methods. (C and D) The histograms indicate the percentage of apoptotic-positive cells induced by CoQ0 and UVB. The results are the mean ± SD of 3 assays. *P < .05; **P < .01; ***P < .001 compared with untreated control cells. #P < .05; ##P < .01; ###P < .001 compared with UVB-irradiated cells.
CoQ0 Induces Intracellular ROS Generation in UVB-Irradiated MCF-7 Cells
ROS generation has been implicated as an early event during apoptosis. To determine whether CoQ0-induced apoptosis is ROS-dependent, we examined the involvement of CoQ0 in ROS generation in UVB-irradiated MCF-7 cells. Fluorescence spectroscopy using DCFH2-DA as a fluorescent probe was performed to estimate intracellular ROS generation. The results indicated that MCF-7 cells exposed to CoQ0 (35 µM) for 20 minutes showed significant increase of fluorescence, which is proportionate to the amount of ROS generation. In addition, UVB irradiation markedly increased the intracellular ROS level as compared to control. Interestingly, CoQ0 treatment significantly enhanced the ROS generation in the UVB-irradiated cells (Figure 3). These data suggest that induction of excessive ROS formation by CoQ0 treatment may contribute to mitochondrial dysfunction and DNA damage in UVB-irradiated MCF-7 cells.

CoQ0 enhances UVB-induced ROS production in MCF-7 cells. Cells were irradiated with UVB (0.05 J/cm2) and then treated with CoQ0 (35 µM) for 20 minutes. (A) The intracellular ROS levels indicated by DCF fluorescence was measured by fluorescence microscopy (200× magnification). (B) Data are presented as fold-change along with fluorescent image. The results are the mean ± SD of 3 assays. *P < .05; **P < .01; ***P < .001 compared with untreated control cells. #P < .05; ##P < .01; ###P < .001 compared with UVB-irradiated cells.
CoQ0 Enhances UVB-Induced DNA Damage in MCF-7 Cells
DNA damage, represented by DNA single-strand breaks, was indicated by an increase in tail lengths. DNA damage was evaluated with the comet assay, in which the tail length is an important quantitative parameter. Therefore, the effect of CoQ0 treatment (35 µM for 72 hours) on cellular DNA damage was evaluated in a single-cell gel electrophoresis comet assay. A total toxicity scale was generated while considering the comet lengths. Our results showed that a significant increase in comet length was observed after MCF-7 cells were treated with CoQ0 for 72 hours (Figure 4A and B). Furthermore, comet length and tail moment were dramatically increased when the cells were pretreated with UVB irradiation (Figure 4C and D), clearly indicating that CoQ0 treatment enhanced UVB-induced DNA damage.

CoQ0 increases UVB-induced DNA damage in MCF-7 cells as assessed by an alkaline comet assay. (A) The cellular DNA was stained with DAPI and photographed using a fluorescence photomicroscope. The comet-like DNA formations were categorized into 5 classes (0, 1, 2, 3, or 4) representing increasing DNA damage shown as a “tail.” Each comet was assigned a value according to its class. (B and C) Cells were irradiated with UVB (0.05 J/cm2) and then treated with CoQ0 (35 µM) for 72 hours. The overall score for 100 cells ranged from 0 (100% of comets in class 0) to 400 (100% of comets in class 4). The observation and the analysis of the results were always performed by the same experienced person. The analysis was blinded, with the observer having no knowledge of the slide identity. (D) The tail moment was quantified as described in Materials and Methods. The results are the mean ± SD of 3 assays. *P < .05; **P < .01; ***P < .001 compared with untreated control cells. #P < .05; ##P < .01; ###P < .001 compared with UVB-irradiated cells.
CoQ0 Upregulates p53 and PARP Cleavage in MCF-7 Cells
The p53 tumor-suppressor protein plays an important role in the induction of apoptosis. 33 Therefore, we examined p53 expression to determine the possible role of p53 in the induction of apoptosis by CoQ0 and UVB in MCF-7 cells. Western blot analyses revealed that pretreatment of MCF-7 cells with UVB significantly increased the p53 expression compared to CoQ0 and UVB alone (Figure 5A).

Effect of CoQ0 on apoptotic proteins in UVB-irradiated MCF-7 cells. Cells were irradiated with UVB (0.05 J/cm2) and then treated with CoQ0 (35 µM) for 72 hours. (A-C) The protein levels of p53, PARP, Bax, and Bcl-2 were determined by Western blot. Relative changes in the Bcl-2 and Bax protein bands were measured by commercially available quantitative software (AlphaEase, Genetic Technology Inc, Miami, FL), with the control representing 1.0-fold. The results are the mean ± SD of 3 assays. *P < .05; **P < .01; ***P < .001 compared with untreated control cells. #P < .05; ##P < .01; ###P < .001 compared with UVB-irradiated cells.
In addition, the cleavage of PARP, a nuclear enzyme that is associated with DNA repair, is dependent on the proteolytic cleavage of caspase-3 and is considered to be an important biological characteristic of apoptosis. As shown in Figure 5B, the PARP (115 kDa) protein was cleaved into an 85 kDa fragment in MCF-7 cells in response to CoQ0 treatment. Furthermore, UVB pretreatment significantly increased the PARP cleavage as compared to control. Taken together, these data suggest that CoQ0-induced apoptosis in UVB-irradiated MCF-7 cells was accompanied by p53 activation and PARP degradation.
CoQ0 Dysregulates Bcl-2/Bax Expression in UVB-Irradiated MCF-7 Cells
Bcl-2 family proteins play a pivotal role in (mitochondria-mediated) apoptosis, either as activators (Bok, Bad, and Bax) or as inhibitors (Bcl-2, Bcl-xL, and Bcl-w). 34 The effect of CoQ0 and UVB on Bcl-2 and Bax protein levels were examined in MCF-7 cells. As shown in Figure 5C, CoQ0 and UVB caused a dramatic reduction in Bcl-2, and significant increase of Bax protein levels was observed. Interestingly, the combination of UVB and CoQ0 significantly increased the Bcl-2 and Bax protein ratio as compared to control. These results indicate that CoQ0 may alter the Bcl-2 and Bax protein ratio and induce apoptosis in UVB-irradiated MCF-7 cells.
Discussion
Breast cancer is the most common cancer among women and is the second leading cause of cancer-related death worldwide. 35 The inhibition of proliferation and the induction of apoptosis are currently being employed as criteria by which the efficacy of new therapeutic agents is evaluated in preclinical studies. 36 The results of the present study indicate that CoQ0 could be an anti–breast cancer agent in vitro. Our findings demonstrated that UVB pretreatment significantly induced apoptosis in MCF-7 cells, which was associated with the excessive production of ROS, DNA fragmentation, followed by a sequence of events, including PARP degradation, Bcl-2/Bax dysregulation, and p53 expression. Taken together, our findings are much more enlightening and revealed that CoQ0 is effective in the induction of apoptosis through accumulation of ROS in UVB-irradiated MCF-7cells.
Ubiquinone analogs have been shown to exhibit strong pro- or anti-oxidant properties. 37 They generate functional and structural damage through hypergeneration of ROS production. 38 On the contrary, they are also shown to reduce oxidative stress-induced cell death 39 by a mechanism that may involve their antioxidant properties. Ubiquinone analogs display noticeable tissue specificity when compared to other classes of PTP regulators. Studies have confirmed that the production of ROS and the regulation of PTP opening by ubiquinone analogs changes depending on the cell lines (which prevent any extrapolation from results obtained with liver mitochondria). Ubiquinone analogs represent a newly identified family of PTP regulators.37,40,41 Regardless of their partition coefficient or redox status, ubiquinone analogs are classified in 3 functional groups. According to whether they favor or inhibit PTP opening or are neutral but compete with active quinones, they are called PTP-inhibitory, PTP-activatory, or PTP-inactive quinones, respectively.
A large number of clinical and preclinical studies exhibit that coenzyme Q (CoQ) or ubiquinone analogs have therapeutic potential effects for inflammation, cancer, and several metabolic disorders.19,42,43 CoQ is a lipophilic molecule found in the inner side of the mitochondrial membrane of most aerobic organisms from bacteria to mammals. CoQ is composed of a redox active benzoquinone ring conjugated to an isoprenoid chain. The subscript in CoQ molecule refers to the number of isoprene repeat units, which include from CoQ0 to CoQ10. 42 Various analogs of CoQ have been shown to indicate either pro-oxidant or antioxidant properties. Antioxidant property has been represented by preventing oxidative stress-induced cells death.21,44 CoQ0, a redox-active compound without isoprenoid side chain that predominantly accumulates in mitochondria, has been shown to inhibit the activity of complex 1 of the mitochondrial respiratory chain and to prevent opening of the mitochondrial permeability transition pore. 45 We hypothesize that CoQ0, in the presence of UVB, dramatically changes mitochondrial function, which abundantly increases oxidative stress leading to subsequent mitochondrial permeability transition pore opening and induction of apoptosis in human breast cancer cells.
UVB is also a well-known genotoxic agent that can stimulate oxidative stress through the formation of ROS, such as superoxide anion radical, hydroxyl radical, and hydrogen peroxide, which leads to cell damage and DNA lesions. 46 UVB-induced apoptosis involves a highly complex mechanism. UVB activates death receptors, such as Fas, which eventually leads to the induction of apoptosis. However, nuclear DNA damage is considered as an important factor in UVB-induced apoptosis. 47 Furthermore, exposure to UVB alters the protein function by directly interacting with aromatic amino acids in proteins. However, it has been reported that in the presence of indole-3-carbinol, UVB induced apoptosis in G361 cells through DNA fragmentation, caspase-3 activation, and PARP cleavage. 48 UVB indirectly damages biomolecules through production of ROS and reactive nitrogen species by oxidizing lipids, proteins, and DNA. In addition, UVB generates ROS, which also play major roles in the execution of apoptotic cell death in an independent way. 49 In the present study, our results demonstrated that the combination of UVB and CoQ0 caused significant cell death compared to UVB and CoQ0 alone. We speculate that CoQ0 may be transformed to potent cytotoxic radicals under UVB irradiation, which subsequently leads to cell death.
Apoptosis-inducing agents are being examined as tools for the management of cancer treatment. Apoptosis is characterized by a number of well-defined features, including cellular morphological changes, internucleosomal DNA cleavage, chromatin condensation, and the activation of the caspase family. 50 Apoptosis activation occurs mainly through 2 well-characterized pathways in mammalian cells, death-receptor-dependent (extrinsic) and mitochondrial-dependent (intrinsic) apoptosis signaling cascades. 51 Recently, we also determined that the induction of apoptosis by AC treatment is mediated by the activation of both mitochondrial and death-receptor pathways. 52 In the present study, comet assays demonstrated that pretreatment of MCF-7 cells with UVB markedly induced apoptotic cell death associated with internucleosomal DNA fragmentation. Furthermore, UVB pretreatment significantly increased the sub-G1 accumulation in MCF-7 cells compared to control group. Cells undergoing apoptosis were found to be associated with elevated levels of cytochrome c in the cytosol with a corresponding decrease in the mitochondria. Cytosolic cytochrome c activates downstream effector caspases (including caspase-3), triggering apoptosis. 53 PARP, a nuclear protein, was shown to be required for apoptosis to proceed in various cell lines. Activated caspase-3 cleaves PARP (116 kDa), generating 89 kDa inactive fragment and cause apoptosis. 54 Our results demonstrated that the combination of CoQ0 and UVB treatment significantly reduced the pro-form of PARP, which demonstrated that CoQ0 could act as a chemopreventive agent with respect to inhibition of the growth of MCF-7 cells through the induction of apoptosis.
The excessive generation of ROS can induce oxidative cellular stress, cell cycle arrest, loss of cell function, damage DNA, and apoptosis in cancer cells. 55 Moreover, ROS play a pivotal role in apoptosis by regulating the activity of certain enzymes involved in the cell death pathway.56,57 ROS triggered the opening of the mitochondrial permeability transition pore, depolarization of the mitochondrial membrane, and then the release of mitochondrial pro-apoptotic factors in the process of mitochondria-mediated apoptosis. 54 In the present study, CoQ0 and UVB pretreatment remarkably increased the production of intracellular ROS in MCF-7 cells, suggesting that CoQ0 and UVB might cause a synergistic effect that leads to increase in the intracellular ROS generation. Furthermore, increase in ROS level may act as upstream signaling molecules to induce mitochondria-mediated cell apoptosis in MCF cells.
The p53 tumor suppressor gene plays a critical role in controlling the cell cycle, genome integrity, and apoptosis in response to ionizing radiation, UV light, or certain genotoxic chemicals. 58 p53 activates the expression of target genes when it binds to regulatory DNA sequences, which is important for the inhibition of tumor formation as well as for mediating the cellular responses to many standard DNA damage-inducing cancer therapies by cycle inhibition and induction of apoptosis. 59 Previous studies suggest that p53-regulated apoptosis proceeds primarily through the intrinsic apoptotic program. 60 Accumulating evidence suggest that several natural chemopreventive and chemotherapeutic agents induce apoptosis by activating p53 and its target genes. 61 Research supports that p53 could be activated in response to ROS-dependent nuclear damage and could stimulate the induction of apoptosis. 62 Hence, in order to examine whether the ability of CoQ0 to induce apoptosis is due to the effect of p53, changes in the protein expression of p53 were examined by western blot. We found that CoQ0 treatment significantly increased the p53 expression in MCF-7 cells. In addition, UVB pretreatment dramatically increased the p53 level, which clearly indicates that in response to UVB radiation, however, CoQ0 enhances UVB-induced apoptosis in MCF-7 cells through a p53-dependent signaling pathway.
In mammalian cells, Bcl-2 gene family contains a number of anti-apoptotic proteins, such as Bcl-xL and Bcl-2, which are thought to be involved in resistance to conventional cancer treatment, while the pro-apoptotic proteins from the same gene family, including Bax, Bak, and Bad, may induce apoptotic cell death. 63 A hallmark of DNA damage-triggered apoptosis is reduced Bcl-2 expression and increased Bax expression. Hence, apoptosis largely depends on the balance between pro-apoptotic and anti-apoptotic protein levels. Activation of the p53 signaling pathway has been shown to play a pivotal role in apoptosis by altering the Bcl-2/Bax ratio. 64 Similarly, the present study indicates a significant inhibition of the anti-apoptotic protein Bcl-2 and a concomitant increase in the expression of the Bax, and p53 proteins by CoQ0 in UVB-irradiated MCF-7 cells. Taken together, our findings showed that UVB pretreatment enhanced its apoptotic effect possibly by upregulating p53, Bax expression, and downregulating Bcl-2 expression.
Conclusion
In conclusion, CoQ0 exhibits an antiproliferative effect by induction of apoptosis that is associated with p53 activation, PARP degradation, and alteration of Bcl-2 and Bax in MCF-7 cells. Furthermore, CoQ0 treatment significantly enhanced UVB-induced apoptosis in MCF-7 cells. As apoptosis has become a new therapeutic target in cancer research, these results confirm that CoQ0 can be used as a chemotherapeutic or chemopreventive agent that enhances UVB-induced apoptosis in human breast cancer cells. In addition, further investigation of its activity, in vivo, is necessary to elaborate and exploit this nascent promise.
Footnotes
Authors’ Note
Authors Hui-Min Wang and Hsin-Ling Yang contributed equally to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants MOST-104-2320-B-039-040-MY3, MOST-103-2320-B-039-038-MY3, NSC-103-2622-B-039-001-CC2, CMU103-ASIA-12, and CMU 103-ASIA-09 from the Ministry of Science and Technology, Asia University, and China Medical University, Taiwan.
