Abstract
Chemotherapy is a major clinical treatment for managing patients with advanced and recurrent ovarian cancer. However, the clinical performance of chemotherapy is limited, and adverse effects have been observed. Integrating chemotherapy with current chemotherapeutic drugs and novel antitumor ingredients might improve the clinical performance of current chemotherapy for ovarian cancer. The aqueous extract of Solanum nigrum leaves (AE-SN), a key ingredient in many traditional Chinese medicine formulae, has exhibited tumor suppression efficacy in numerous human cancer cells but not in ovarian cancer cells. In this study, tumor suppression efficacy was determined using the ES-2, SKOV-3, and OVCAR-3 human ovarian cancer cell lines. The half-maximal inhibitory concentrations of the AE-SN in ES-2 and SKOV-3 cells were 1.052 and 1.779 mg/mL, respectively. AE-SN treatment increased the accumulation of mammalian microtubule-associated protein 1 light chain 3 A/B, an autophagic cell marker, in all the tested cell lines; however, it activated the cleavage of caspase-3, an apoptotic marker, only in SKOV-3 cells. Furthermore, the AE-SN also promoted tumor suppression efficiency of cisplatin, doxorubicin, and docetaxel in the tested ovarian cancer cells. In addition, AE-SN-enhanced cell death was associated with AE-SN-induced caspase-3 cleavage in SKOV-3 cells. In conclusion, the AE-SN exhibited tumor suppression efficacy and improved the tumor suppression efficiency of cisplatin, doxorubicin, and docetaxel in human ovarian cancer cells. Therefore, the AE-SN is a candidate antitumor ingredient that can be used in developing future integrated chemotherapy for managing ovarian cancer.
Introduction
Ovarian cancer is one of the most common types of gynecological cancer, with high mortality worldwide.1-3 Most ovarian cancer cases are diagnosed at late stages and have extremely low survival rates at advanced stages. 4 Surgery is the primary approach to treating ovarian cancer patients, with chemotherapy being an alternative approach to treating advanced and recurrent ovarian cancer when surgery is not feasible. However, the clinical performance of chemotherapeutic drugs, such as platinum analogs (eg, cisplatin and carboplatin), taxanes (eg, paclitaxel and docetaxel), and doxorubicin, is limited because of sensitivity, resistance, and adverse effects.5,6 Regarding increasing tumor suppression efficiency, combination chemotherapy based on platinum analogs and taxanes initially yielded unsatisfactory clinical outcomes.7-9 A recent strategy involves applying molecular-targeting drugs (eg, bevacizumab), which suppress tumor growth by blocking tumor-associated angiogenesis, and chemotherapeutic drugs (eg, carboplatin and paclitaxel); this strategy modestly prolonged the progression-free survival time by approximately 4 months.10-12 Therefore, new drugs or improved integrated chemotherapeutic strategies are still urgently required for improving current ovarian cancer therapy.
Complementary and alternative medicine (CAM) use is common among cancer patients worldwide. Although traditional medicinal herbs and nutritional supplements play a crucial role in CAM, sufficient scientific evidence of both their efficacy and efficiency is lacking. In the past few decades, studies have increasingly focused on traditional Chinese medicine (TCM) and particularly on its effectiveness in clinical cancer therapy. Two meta-analyses of adjunctive cancer therapy suggested that using TCM to treat colorectal cancer and non-small-cell lung cancer improves the survival rate and quality of life of cancer patients.13,14 It has also been suggested that TCM plays a protective role in liver function and alleviates hematotoxicity in cancer patients who receive chemotherapy.15,16 TCM is based on firmly established theoretical approaches, and most ingredients are natural herbal products. In the clinical practice of TCM, a therapeutic agent is typically prepared by mixing the aqueous extracts (AEs) of various medicinal herbs according to various formulae.17-19 Some common AEs of medicinal herbs present in TCM formulae used for cancer treatment (eg, Solanum nigrum [SN], Hedyotis diffusa, Curcuma longa, and Scutellaria barbata) exhibit anticancer efficacy in many human cancer cells in vitro.20-25 These findings indicate that appropriately using TCM ingredients, such as AEs of herbs, according to specific formulae might improve the clinical outcomes of current cancer therapies.
SN is a common elemental ingredient used in the clinical practice of TCM for treating cancer patients.21,25 Several recent studies have indicated that SN extracts exert antitumor effects on numerous cancer types, such as lung, breast, stomach, liver, colon, leukemia, prostate, bladder, and endometrial cancers.20-24 Some of these studies have examined the antitumor effects of SN on the basis of chemical compounds isolated from SN extracts, such as solamargines and degalactotigonin,20,25-27 and other studies have used various SN extracts, including aqueous, hydroalcoholic, and polyphenolic extracts.22-24,28-30 Because TCMs are typically prepared using AEs and administered orally, evaluation based on the AE of SN (AE-SN), which is similar to the clinical practice of TCM, may therefore provide useful preclinical data on both the tumor suppression efficacy and efficiency of SN. The AE-SN exhibits tumor suppressive capabilities in vitro and in vivo.22,31 In addition, it has recently been suggested that the AE-SN enhances the cytotoxicity of chemotherapeutic drugs, including cisplatin, doxorubicin, 5-fluorofacil, and docetaxel, in human colorectal cancer cells and that of docetaxel in human endometrial cancer cells.24,29 These findings lead to the hypothesis that the AE-SN could also enhance the tumor suppression efficiency of chemotherapy in human ovarian cancer cells.
This study evaluated the antitumor effects of the AE-SN on human ovarian cancer cells and the combinatorial effects of the AE-SN and 3 common ovarian chemotherapeutic drugs, cisplatin, doxorubicin, and docetaxel, in vitro. The collected data are informative regarding the clinical use of the AE-SN in chemotherapy with adjunctive TCM for treating ovarian cancer patients.
Materials and Methods
Preparation of the AE-SN
In the clinical practice of TCM, SN is generally administered as a crude extract of its leaves, which is obtained by heating the leaves with water. The AE-SN was prepared by referring to a TCM method, which was described in previous studies.24,29 In this study, 50 g of dried leaves of SN were minced, soaked in 750 mL of distilled water, and boiled at 100°C for 50 minutes. The solution was maintained at 100°C for 1 hour and condensed to a final concentration of 1 g/mL at 100°C. This condensed AE-SN was stored at −80°C until further experiments were conducted.
Cell Lines and Cell Culture Conditions
Ovarian cancer cell lines ES-2, SKOV-3, and OVCAR-3 were purchased from the Bioresource Collection and Research Center (Hsinchu, Taiwan). ES-2 is a poorly differentiated clear-cell carcinoma, and SKOV-3 is a well-differentiated ovarian adenocarcinoma; both cell lines are estrogen receptor positive; however, only ES-2 is progesterone receptor positive.32-34 OVCAR-3 is an adenocarcinoma cell line expressing both estrogen and progesterone receptors.35,36 Human ovarian granulosa (hOG) cells were collected from women after approval was received from the Taipei Medical University Clinical Institutional Research Board (TMU-JIRB 201105007). Among all the tested cell lines, the hOG cells were cultured in Dulbecco’s modified Eagle’s medium with 4500 mg/mL of
Evaluation of Tumor Cell Suppression Efficiency
The hOG, WI-38, ES-2, and SKOV-3 cells were seeded at a density of 5 × 103 cells, and the OVCAR-3 cells were seeded at a density of 1 × 104 cells per well of 96-well microplates and incubated overnight before treatment. The cells were then treated with 0, 0.1, 0.2, 0.5, 1, or 2 mg/mL of the AE-SN and incubated for 24 or 48 hours to measure the cell viability. To evaluate the combinatorial effects on the AE-SN with cisplatin, doxorubicin, or docetaxel, ovarian cancer cells were incubated with cisplatin (0 to 100 µM), doxorubicin (0 to 50 µM), or docetaxel (0 to 100 nM) and 0, 0.5, or 1.0 mg/mL of the AE-SN for 48 hours. The cell viability was measured using a 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay. To each well, 30 µL of a 5-mg/mL MTT phosphate-buffered saline solution (Bio Basic Inc, Markham, Ontario, Canada) was added, and the cells were incubated at 37°C for 3 hours. The medium mixed with the MTT solution was then replaced with 100 µL of dimethyl sulfoxide (JT Baker, Phillipsburg, NJ) and shaken for 15 minutes at room temperature in a darkroom to completely dissolve the MTT formazan products. The optical density at 550-nm absorbance was measured for each well on an Emax microplate reader (Molecular Devices, Sunnyvale, CA). To evaluate the tumor suppression efficiency, the half-maximal inhibitory concentration (IC50) of the AE-SN was determined according to the MTT assay results by using the CalcuSyn software (Biosoft, Cambridge, UK). The morphology of the cells was examined using a Nikon Eclipse TS100 microscope (Nikon Instruments, Melville, NY), and photographs were taken at 100× magnification.
Immunoblotting
The ES-2, SKOV-3 (5 × 105 cells per 6-cm dish), and OVCAR-3 (1 × 106 cells per 6-cm dish) cells were seeded and incubated overnight. All cells were then incubated with cisplatin, doxorubicin, or docetaxel with 0.5 or 1.0 mg/mL of the AE-SN for 48 hours. The cells were then harvested using a cell lysis buffer containing150 mM NaCl, 50 mM pH 7.5 Tris-HCl, 1% NP-40, 0.5% deoxycholate, 0.1% sodium dodecyl sulfate, 1 mM phenylmethylsulfonyl fluoride (Life Technologies, Grand Island, NY), 10 µg/mL of leupeptin, and 100 µg/mL of aprotinin. Harvested cell extract samples were equalized to 30 µg of total protein concentration by using a Bio-Rad protein assay kit (Bio-Rad Laboratories, Hercules, CA); the proteins were separated on a 12% sodium dodecyl sulfate-polyacrylamide electrophoresis gel and then transferred to a polyvinylidene fluoride membrane (Pall Corp, Port Washington, NY). Primary antibodies used for the immunoblotting analysis included those against caspase-3 (1:1000; Cell Signaling Technology, Danvers, MA), mammalian microtubule-associated protein 1 light chain 3 A/B (LC3A/B, 1:1000; Cell Signaling Technology), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH, 1:10 000; Abfroniter, Seoul, South Korea); the secondary antibody used for the hybridization was donkey antirabbit horseradish peroxidase–conjugated secondary antibody (1:10 000; Santa Cruz Biotechnology, Santa Cruz, CA). Immunoreactivity was then determined using a WesternBright electrochemiluminescence Western blotting detection kit (Advabsta, Menlo Park, CA). Semiquantitation of protein expression levels was performed using ImageJ software (National Institutes of Health, Bethesda, MD).
Statistical Analysis
The Student t test was used for comparing the data of the 2 groups, and a 1-way ANOVA was used to examine the dose-dependent effect of the AE-SN on the tested ovarian cancer cells. Statistical analyses were performed using SPSS (SPSS Inc, Chicago, IL). Combinatorial effects of the AE-SN and selected chemotherapeutic drugs were analyzed using CalcuSyn software (Biosoft). The combinatorial drug effects were evaluated according to the combination index (CI) value, which was based on the median effect method: CI <1 indicated a synergistic effect; CI =1 indicated an additive effect; and CI >1 indicated an antagonistic effect. 37
Results
Cytotoxicity of the AE-SN in Ovarian Cancer Cells
To evaluate the cytotoxicity of the AE-SN in human ovarian cancer cells, ES-2, SKOV-3, and OVCAR-3 cells were treated with 0 to 2 mg/mL of the AE-SN for 48 hours. Cell viability was determined using an MTT assay, which indicated that the growth of all the tested human ovarian cancer cells was suppressed by the AE-SN treatment in a dose-dependent manner (Figure 1A). Compared with the tested human ovarian cancer cells, normal hOG cells did not exhibit any growth inhibition after AE-SN treatment (Figure 1B). An analysis of IC50s (Table 1) indicated that OVCAR-3 cells were more resistant (IC50 > 2 mg/mL of the AE-SN) to the AE-SN treatment than were ES-2 and SKOV-3 cells (IC50 = 1.052 and 1.779 mg/mL of the AE-SN, respectively). The AE-SN-treated ES-2 and SKOV-3 cells exhibited obvious morphological changes and apoptotic features, such as cell shrinkage and the presence of nuclear fragments (only ES-2 cells) as well as the formation of lipid-like droplets, which is considered a feature of autophagy (Figure 2). In contrast, the AE-SN-treated OVCAR-3 cells exhibited no morphological changes, suggesting that OVCAR-3 cells were more insensitive to the AE-SN treatment than were the ES-2 and SKOV-3 cells. Moreover, a morphological observation of hOG cells treated with 1 mg/mL of the AE-SN for 48 hours indicated no difference from the control hOG cells (Figures 2G and 2H). AE-SN-induced cell growth inhibition is likely specific to ovarian cancer cells and does not occur in normal cells. Furthermore, the AE-SN treatment induced the LC-3 A/B II accumulation in ES-2, SKOV-3, and OVCAR-3 cells and caspase-3 activation in SKOV-3 cells (Figure 2I).
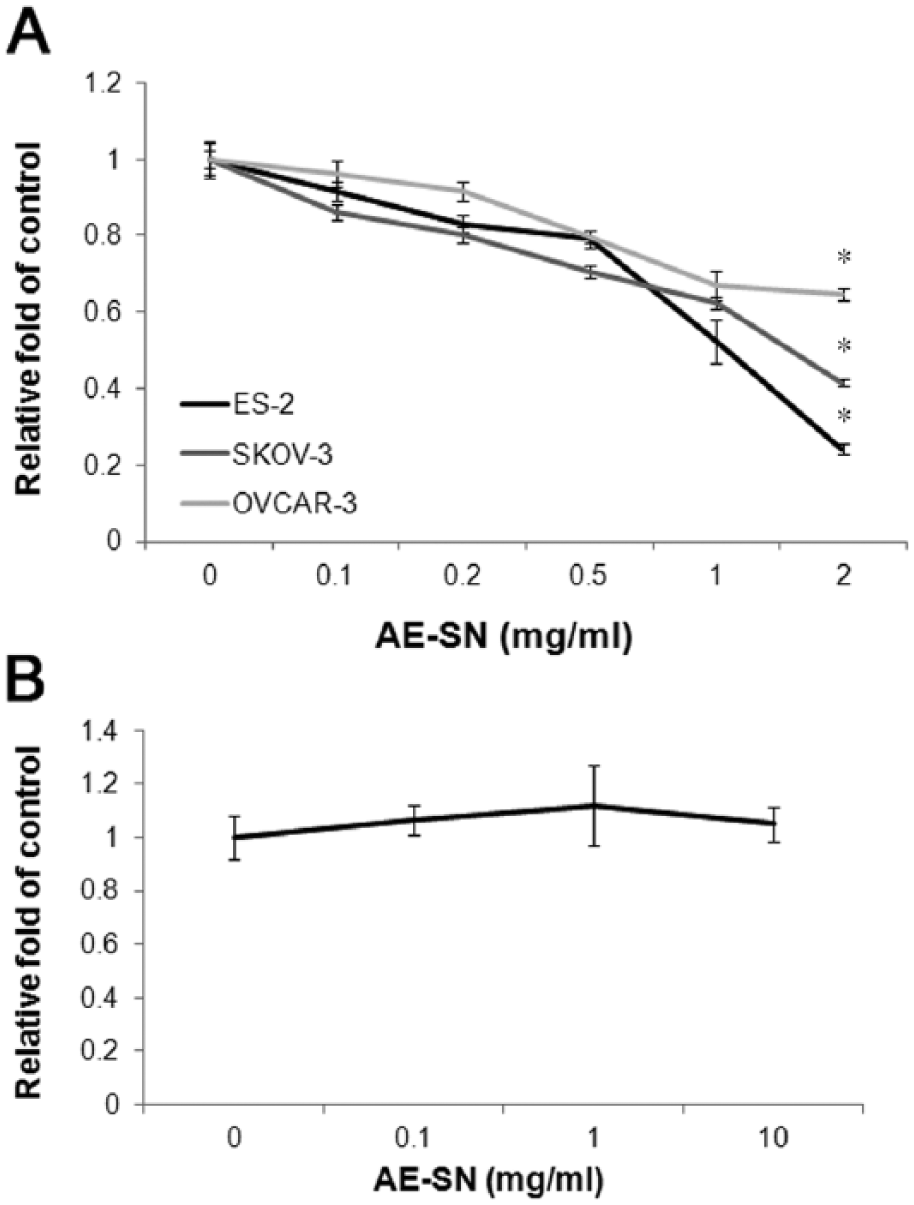
Cytotoxicity of the aqueous extract of Solanum nigrum (AE-SN) in ES-2, SKOV-3, and OVCAR-3 human ovarian carcinoma and normal human ovarian granulosa (hOG) cells: A. Ovarian carcinoma cells were treated with 0.1 to 2 mg/mL of the AE-SN for 48 hours; B. hOG Cells were treated with 0.1 to 10 mg/mL of the AE-SN for 48 hours. Cell viability was determined using a 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay. Experiments were repeated in triplicate. Data are presented as the mean ± SD. All ovarian carcinoma cell lines treated with the AE-SN showed dose-dependent effects in the MTT assay, whereas the hOG cells showed no difference when treated with the AE-SN.
IC50s of the AE-SN Calculated in Human Ovarian Carcinoma Cells. a
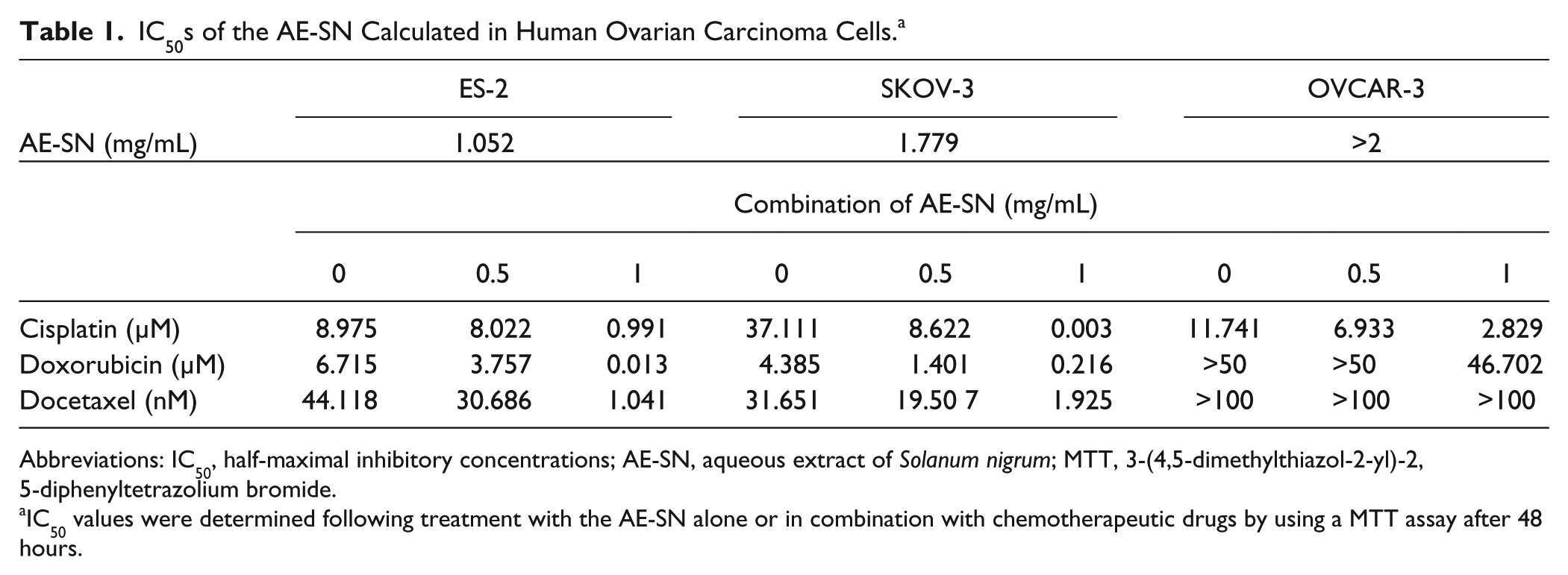
Abbreviations: IC50, half-maximal inhibitory concentrations; AE-SN, aqueous extract of Solanum nigrum; MTT, 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide.
IC50 values were determined following treatment with the AE-SN alone or in combination with chemotherapeutic drugs by using a MTT assay after 48 hours.
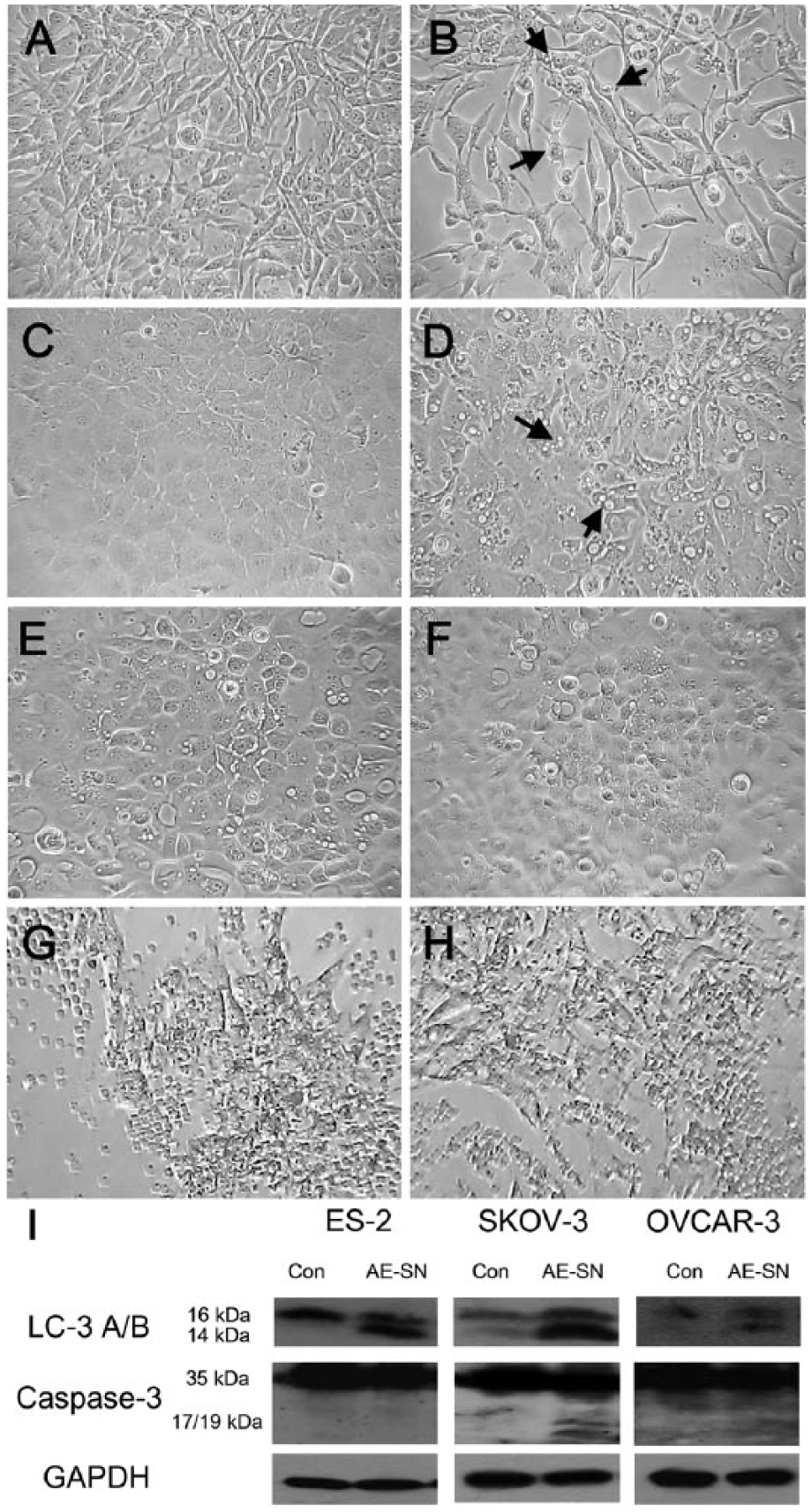
Morphological changes in human ovarian carcinoma and human ovarian granulosa (hOG) cells treated with the aqueous extract of Solanum nigrum (AE-SN). (A), (C), (E), and (G) ES-2, SKOV-3, OVCAR-3, and hOG cells were treated with a normal culture medium for 48 hours. (B), (D), (F), and (H) ES-2, SKOV-3, OVCAR-3, and hOG cells were treated with 1 mg/mL of the AE-SN for 48 hours. Arrows indicate cell shrinkage or nuclear fragments in ES-2 cells (B) and lipid droplet-like vesicles in ES-2 and SKOV-3 cells (B and D). Magnification = 100×. (I) Light chain 3 A/B (LC-3 A/B) II accumulation and caspase-3 cleavage in human ovarian carcinoma cells treated with 1 mg/mL of the AE-SN for 48 hours.
Combinatorial Effects of the AE-SN With Cisplatin, Doxorubicin, and Docetaxel on Human Ovarian Cancer Cells
According to previous results, AE-SN treatment could suppress tumor cell growth in the dose range of 0.5 to 2 mg/mL in the 3 tested ovarian cancer cell lines (Figure 1). Because treatment with 0.5 mg/mL of the AE-SN yielded 20.9%, 29.3%, and 20.2% tumor cell growth inhibition and treatment with 1 mg/mL of the AE-SN yielded 47.7%, 37.5%, and 32.9% tumor cell growth inhibition in ES-2, SKOV-3, and OVCAR-3 cells, respectively (Figure 1), these 2 doses were chosen as moderate and high doses for determining the combinatorial effects of the AE-SN with chemotherapeutic drugs. ES-2, SKOV-3, and OVCAR-3 cells were cultured with 0, 0.5, or 1 mg/mL of the AE-SN with cisplatin (2-100 µM in ES-2 and SKOV-3 cells and 1-50 µM in OVCAR-3 cells), doxorubicin (1 to 100 µM in ES-2 cells, 0.1 to 10 µM in SKOV-3 cells, and 1 to 50 µM in OVCAR-3 cells), or docetaxel (1 to 50 nM in ES-2 and SKOV-3 cells and 1 to 100 nM in OVCAR-3 cells) for 48 hours, and cell viability was analyzed using an MTT assay (Figure 3). The IC50 analysis indicated that both 0.5 and 1 mg/mL of the AE-SN enhanced the cytotoxicity of cisplatin, doxorubicin, and docetaxel in ES-2 and SKOV-3 cells but enhanced the cytotoxicity of only cisplatin in OVCAR-3 cells (Table 1).
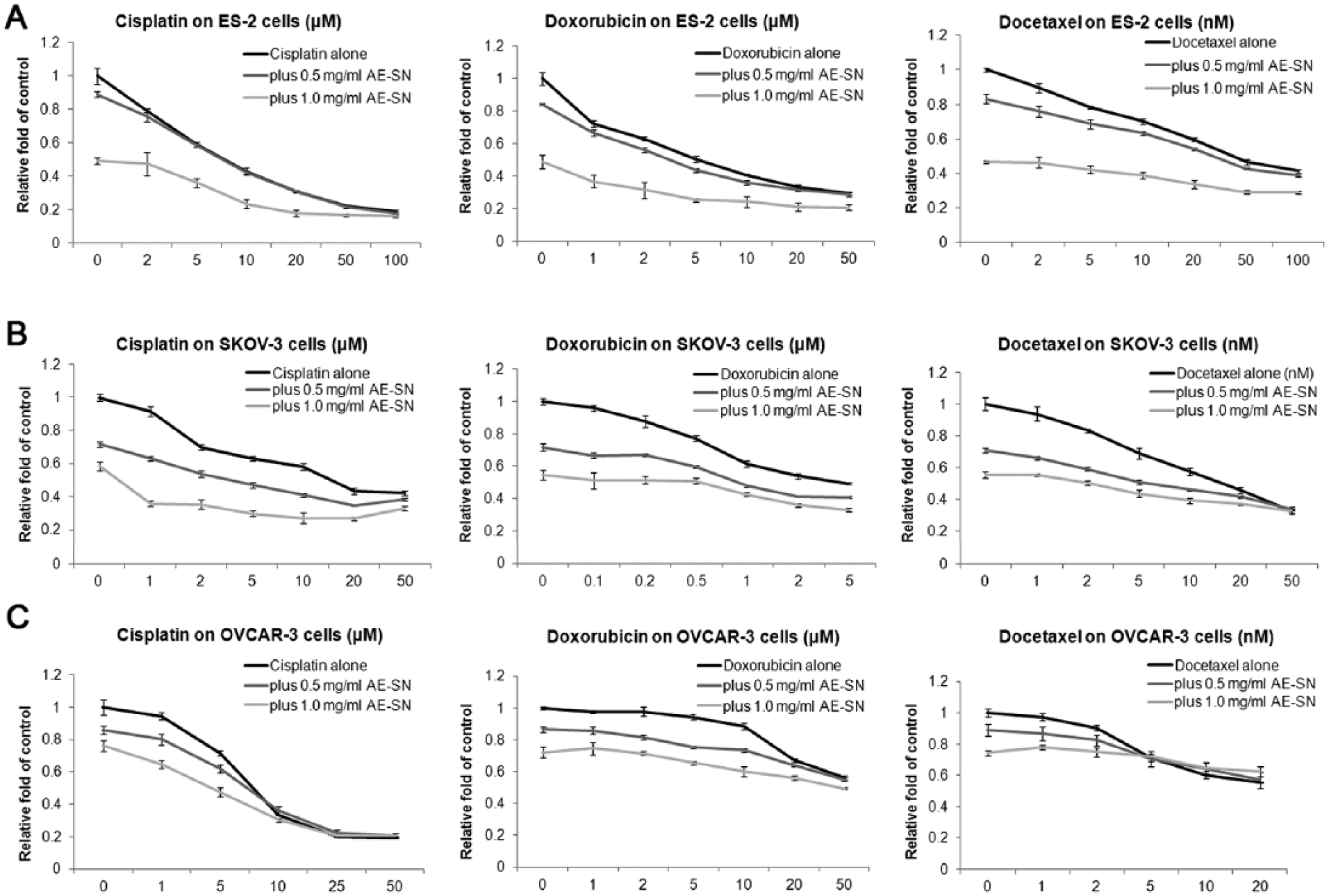
Combined cytotoxicity of the aqueous extract of Solanum nigrum (AE-SN) and selected drugs: A. ES-2 cells were treated with 0 to 100 µM cisplatin, 0 to 50 µM doxorubicin, or 0 to 100 nM docetaxel and 0, 0.5, or 1 mg/mL of the AE-SN for 48 hours. B. SKOV-3 cells were treated with 0 to 100 µM cisplatin, 0 to 5 µM doxorubicin, or 0 to 50 nM docetaxel and 0, 0.5, or 1 mg/mL of the AE-SN for 48 hours. C. OVCAR-3 cells were treated with 0 to 50 µM cisplatin, 0 to 50 µM doxorubicin, or 0 to 20 nM docetaxel and 0, 0.5, or 1 mg/mL of the AE-SN for 48 hours. Cell viability was determined using a 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide assay. Experiments were repeated in triplicate. Data are presented as means ± standard deviations.
To further identify the combinatorial effects of the AE-SN with the tested chemotherapeutic drugs, the CI was analyzed using Calcusyn software. CI values of <1, 1, and >1 indicated synergistic, additive, and antagonistic effects, respectively. The CI values for the middle dose range of cisplatin, doxorubicin, and docetaxel combined with 0.5 and 1 mg/mL of the AE-SN were mostly less than 1 (Table 2). All CI values for the ES-2 cells exposed to 2 to 50 µM doxorubicin and 0.5 mg/mL of the AE-SN were <1, suggesting that this combination produces a synergistic effect. CI values greater than 1 indicated an antagonistic effect and mostly occurred with the lowest and highest doses of the 3 chemotherapeutic drugs. These results might not indicate a true antagonistic effect because of the minimal and maximal cytotoxic effects observed with the start and end doses of the chemotherapeutic drugs, as indicated by CI values of 1.936 and 1.823 for 1 µM (the start dose) and 100 µM (the end dose) doxorubicin, respectively, combined with 0.5 mg/mL of the AE-SN. The most considerable antagonistic effect was observed only for the combination of 0.5 or 1 mg/mL of the AE-SN and 0.1 to 0.5 µM doxorubicin in the SKOV-3 cells. Overall, the results of both IC50 and CI analyses suggested that the AE-SN treatment enhances the tumor suppression efficiency of cisplatin, doxorubicin, and docetaxel in human ovarian cancer cells.
Analysis of the Combinatorial Effects on the Aqueous Extract of Solanum nigrum (AE-SN) and Selected Drugs. a
Cell viability of the cells cotreated with the aqueous extract of Solanum nigrum (AE-SN) in combination with cisplatin, doxorubicin, or docetaxel are from the previous cytotoxicity study (Figure 3). The combination index (CI) was determined using Calcusyn software. A CI <1 indicated a synergistic effect, a CI =1 indicated an additive effect, and a CI >1 indicated an antagonistic effect.
Treatment With the AE-SN-Activated Caspase-3 Cleavage in SKOV-3 Cells
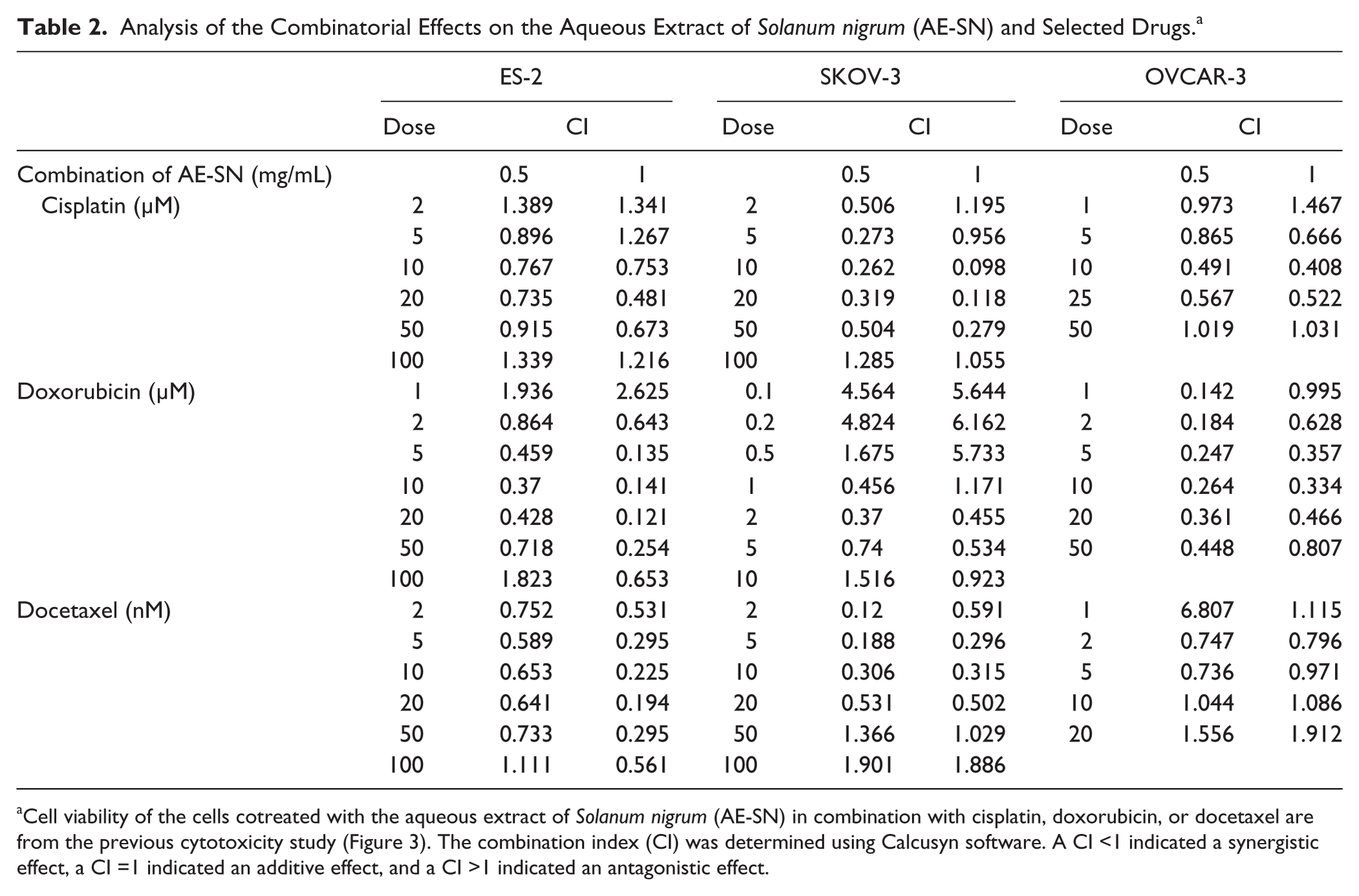
In previous results, AE-SN treatment suppressed ovarian cancer cell growth and enhanced the cytotoxicity of selected drugs, particularly activating the cleavage of caspase-3 in SKOV-3 cells. To determine whether the AE-SN treatment enhanced the cytotoxicity of the selected drugs through caspase-3 cleavage, SKOV-3 cells were treated with 1 mg/mL of the AE-SN alone or in combination with chemotherapeutic drugs. Caspase-3 was cleaved in SKOV-3 cells treated with both the AE-SN alone and in combination with drugs (Figure 4). The semiquantification results indicated that the AE-SN further enhanced cleaved caspase-3 accumulation when combined with the 3 drugs in SKOV-3 cells (Figure 4). These results suggest that the AE-SN enhanced the cytotoxicity of the selected drugs by promoting caspase-3 cleavage in ovarian cancer cells.
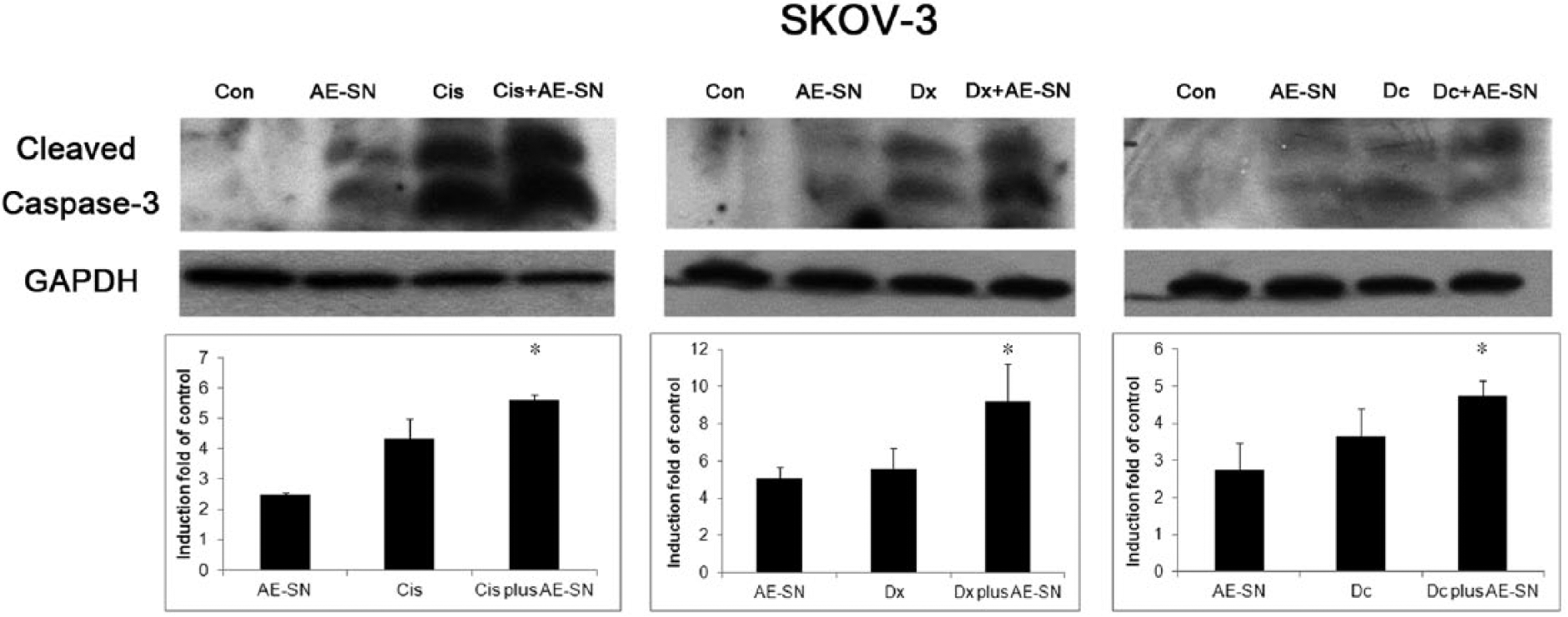
Treatment with the aqueous extract of Solanum nigrum (AE-SN) induced caspase-3 cleavage in SKOV-3 cells. SKOV-3 cells were treated with 0 or 1 mg/mL of the AE-SN (Con or AE-SN), 5 µM cisplatin (Cis), 1 µM doxorubicin (Dx), or 5 nM docetaxel (Dc) alone or in combination with 1 mg/mL of the AE-SN (Cis + AE-SN, Dx + AE-SN, and Dc + AS-SN) for 48 hours. Western blotting analysis was performed to detect caspase-3 cleavage (upper images). Semiquantification of the caspase-3 cleavage is shown below. Experiments were repeated in triplicate. Data are presented as the mean ± standard deviation.
Discussion
The AE-SN is a common ingredient in numerous TCM formulae used for managing various cancers; however, both the tumor suppression efficacy and efficiency of this extract have not been fully clarified. Recent studies on the AE-SN have demonstrated that the AE-SN alone activated cell death in several human cancer cells, including breast, lung, liver, colorectal, and endometrial cancer cells.22,24,29-31 Although AE-SN treatment induced both caspase-3 cleavage and LC-3 A/B II induction in human breast adenocarcinoma cells, AU565, and hepatocellular carcinoma cells (HepG2),22,31 AE-SN-treated human colorectal adenocarcinoma cells, HT-29 and DLD-1, as well as human endometrial adenocarcinoma cells, HEC-1A, HEC-1B, and KLE, only exhibited LC3 A/B II induction.24,29 In this study, the AE-SN induced LC-3 A/B II in ES-2, SKOV-3, and OVCAR-3 cells but activated caspase-3 cleavage only in SKOV-3 cells. These in vitro studies have indicated that the AE-SN treatment induced programmed cell death by activating autophagy through LC3 A/B II accumulation with or without capsase-3-related apoptosis depending on individual cancer cell types. In recent studies, the IC50 of 48-hour AE-SN treatment has ranged from 0.5 to 2 mg/mL in breast, liver, gastric, and endometrial cancer cells.24,29,31 In contrast to human cancer cells, human normal cells are more resistant to AE-SN treatment. As observed in this study, AE-SN treatment did not inhibit cell growth in normal hOG cells. These results suggest that AE-SN-induced cytotoxicity is specific to tumor cells. Moreover, the AE-SN-induced caspase-3 cleavage was observed only in SKOV-3 cells but not in ES-2 and OVCAR-3 cells, indicating that the AE-SN-mediated caspase-3 cleavage may vary among cell types. The exact signaling pathways mediated by the AE-SN that lead to the induction of tumor cell death, such as autophagy and apoptosis, in tumor cells remain unclear. These mechanisms must be studied further to improve the understanding of AE-SN-mediated tumor cell death.
The specific compounds present in the AE-SN that induced programmed cell death are not fully understood. Although Huang et al 16 suggested that flavonoids in the AE-SN are the major compounds inducing tumor cell death in human breast adenocarcinoma AU565 cells, 22 flavonoids were not confirmed to activate apoptosis or autophagic cell death in tumor cells. Other studies have suggested that glycoalkaloids, such as solamargine and solanine, isolated from SN are the critical compounds inducing apoptosis, specifically through caspase-3 activation; however, glycoalkaloids did not induce autophagy in human leukemia and hepatocellular carcinoma cells.26,38 Compared with AE-SN-induced tumor cell apoptosis, AE-SN-induced LC-3 II accumulation was observed in almost all tested tumor cells, including human endometrial, ovarian, liver, colorectal, and breast cancer cells.22,24,29,31 These findings indicate that LC-3 II accumulation is a common biological feature of AE-SN-treated cancer cells, and LC-3 II can serve as a protein marker for determining the bioactivity among various AE-SN preparations. Overall, recent studies have suggested that AE-SN-induced tumor cell deaths are not the result of a specific chemical compound but might be synergized by various functional compounds that activate programmed cell death via multiple cell death pathways. However, these compounds present in the AE-SN, which activate both autophagic cell death and apoptosis, still require further investigation.
Although the AE-SN exhibited tumor suppression efficacy in ES-2, SKOV-3, and OVCAR-3 human ovarian cancer cells, the IC50 values of the AE-SN were higher than 1 mg/mL in the tested tumor cells, suggesting that the AE-SN alone might not be sufficiently effective in suppressing tumor cell growth. Therefore, we examined the combinatorial effects of the AE-SN and selected drugs to determine whether the AE-SN is capable of enhancing cytotoxicity of the drugs. Cisplatin, an alkylating agent, is a well-studied chemotherapeutic drug that inhibits DNA synthesis and forms DNA adducts in tumor cells.21,39 Doxorubicin, a topoisomerase-II inhibitor, blocks DNA and RNA synthesis in tumor cells.40,41 Docetaxel, a taxane, blocks cell mitosis by stabilizing tubulins in tumor cells.42,43 Treatment using these chemotherapeutic drugs results in cell cycle arrest and activates programmed cell death, mainly caspase-dependent apoptosis in cancer cells. The cytotoxicity of the AE-SN is involved in multiple cell death pathways, including autophagy and apoptosis, and might further enhance the cytotoxicity of chemotherapeutic drugs. In the IC50 evaluation for combined treatment involving the AE-SN and chemotherapeutic drugs, the AE-SN, particularly the 1-mg/mL dose, exhibited enhanced cytotoxicity when combined with cisplatin in all 3 ovarian cancer cells and when combined with doxorubicin and docetaxel in the ES-2 and SKOV-3 cells. Moreover, the analysis of the combinatorial effect indicated that in all the 3 ovarian cancer cells, the synergistic effects mostly occurred in the responsive dose ranges of the dose-response curves of the tested drugs (Figure 3 and Table 2). AE-SN-enhanced cytotoxicity was also observed with chemotherapeutic drugs in other cancer cell types, such as human colorectal cancer cells and human endometrial carcinoma cells.24,29 Overall, data of in vitro experiments in this study indicated that AE-SN treatment enhanced the tumor suppression efficiency of cisplatin, docetaxel, and doxorubicin in human ovarian cancer cells. Examining the combinatorial effects of the AE-SN with dual chemotherapy using platinum analogs and taxanes in additional studies is valuable.
Studies have indicated that AE-SN-induced tumor cell death is based on autophagic cell death and caspase-3-associated apoptosis22,31; however, certain tumor cell types, including endometrial and colorectal cancer cells, exhibited autophagy activation but not cleaved caspase-3 activation after AE-SN treatment.24,29 Consistent with this observation, AE-SN-treated human ovarian cancer cells exhibited various responses to programmed cell death activation. LC3 A/B II induction was detected in ES-2, SKOV-3, and OVCAR-3 cells, whereas caspase-3 cleavage was detected only in SKOV-3 cells. In previous in vitro studies evaluating the combinatorial effects of the AE-SN and chemotherapeutic drugs, the AE-SN has been suggested to enhance chemotherapeutic drug-induced tumor cell death, particularly that based on caspase-3 cleavage, by activating autophagic cell death in tumor cells.24,29 This study showed that AE-SN treatment activated caspase-3 and enhanced cisplatin-, doxorubicin-, and docetaxel-induced caspase-3 cleavage in SKOV-3 cells. This result indicated that the AE-SN induces tumor cell death by activating 2 cell death pathways—autophagy and caspase-3-dependent apoptosis—and further enhances the cytotoxicities of cisplatin, doxorubicin, and docetaxel. To further assess the antitumor effects and unidentified side effects of the AE-SN when used with chemotherapeutic drugs to manage ovarian cancer, advanced preclinical studies using in vivo cancer models are required before clinical trials are conducted.
Conclusion
This study not only demonstrated the tumor suppression efficacy of the AE-SN but also the combinatorial cytotoxic effects of the AE-SN with cisplatin, doxorubicin, and docetaxel in ES-2, SKOV-3, and OVCAR-3 human ovarian cancer cells (Figure 5). These findings suggest that the AE-SN is an antitumor ingredient that can be integrated with chemotherapy by using current antitumor drugs to manage ovarian cancer.
Summary of the combinatorial effect of the aqueous extract of Solanum nigrum (AE-SN) and selected drugs.
Footnotes
Authors’ Note
Chia-Woei Wang and Chun-Liang Chen contributed equally to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Chi Mei Medical Center (101CM-TMU-12-1).
