Abstract
Ginkgolide B (GB), the primary active component of Ginkgo biloba extracts, may have antitumor properties. The objective of this study was to determine the effects and possible mechanisms of GB in ovarian cancer cells. In this study, human ovarian cancer cell lines (SKOV3 and CAOV3) were treated with different concentrations of GB alone or in combination with Cis-diaminodichloroplatinum (CDDP). An MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay was performed to determine cell viability. The apoptosis rates of cells were measured by flow cytometric analysis. The expression of apoptosis-associated and proliferation-associated proteins was detected by Western blot. The cytotoxicity of GB was analyzed using a lactate dehydrogenase assay. Treatment with 100 µM GB for 3 days significantly inhibited SKOV3 and CAOV3 cell proliferation by 57.3% and 63.1% compared with control cells, respectively, as determined by MTT assay. Similarly, the apoptotic cell population was increased when treated with GB in a dose-dependent manner both in SKOV3 and CAOV3 cells. These effects were characterized by the upregulation of p21, p27, cleaved capase-3, and cleaved caspase-8 and downregulation of cyclin D1. In addition, a combined treatment of low concentrations of GB and CDDP showed an additive effect on the inhibition of SKOV3 cell proliferation. Furthermore, GB had significantly less cytotoxicity than CDDP in normal human ovarian surface epithelial cells. This study suggests that GB can be proposed as an effective antiproliferative and apoptosis-inducing agent with interesting translational application in ovarian cancers, used in addition to conventional chemotherapy.
Introduction
Ovarian cancer is the fifth most common cause of death from all cancers among women in the world and has the highest mortality of the gynecological cancers. 1 Overall, ovarian cancer has the worst prognosis of all gynecological cancers, in which the 5-year survival rate is less than 40%. 2 The poor survival rate is because of the lack of effective therapies once metastasis has occurred. Therefore, more effective treatment strategies are needed.
In recent years, increasing attention has been focused on plant food–derived phytochemicals as potential anticancer drugs. 3 Approximately 70% of all drugs (ie, paclitaxel, doxorubicin, and camptothecin) used today for the treatment of cancer are derived from or based on natural products. 4 Ginkgo biloba has been used for more than 2 millennia for the general promotion of health. 5 Recent studies conducted with various molecular, cellular, and whole animal models have revealed that leaf extracts of Ginkgo biloba may have anticancer (chemopreventive) properties that are related to their antioxidant, antiangiogenic, and gene or protein regulatory actions.6-9 Cell line studies suggest that Ginkgo may reduce the growth of human breast cancer cells, perhaps through its action on the peripheral-type benzodiazepine pathway that is important in steroid hormone regulation, 10 and it has significant antiproliferative and cytotoxic effects on human hepatocellular carcinoma cells. 11 In vivo experiments suggest that Ginkgo may promote apoptosis of cancer cells by caspase-3/8 activation in oral cancer cells in rats. 12 Our most recent epidemiological and biological data revealed that Ginkgo biloba, perhaps through its ginkgolide B (GB) components, has significant chemopreventive properties against ovarian cancer. 13 However, it remains unclear whether GB could have any direct impact on ovarian cancer cells.
In this study, we report that GB exerts a broad cytostatic effect in ovarian cancer cell lines, including the inhibition of cell proliferation and induction of cell apoptosis. All these properties point to GB as a novel agent with a low-toxic profile and with an interesting therapeutic potential for treating ovarian cancers when used in conjunction with conventional cytotoxics.
Materials and Methods
Chemicals and Reagents
Dimethyl sulfoxide (DMSO), Cis-diaminodichloroplatinum (CDDP), GB (>90% high-performance liquid chromatography grade), and RPMI 1640 cell culture media were purchased from Sigma-Aldrich (St Louis, MO). The MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) cell growth assay kit was purchased from Chemicon (Temecula, CA). The apoptosis detection kit (APO-Direct) was purchased from BD Biosciences (Franklin Lakes, NJ). Lactate dehydrogenase (LDH) release from cells was determined by using an LDH detection kit from Invitrogen (Carlsbad, CA).
Cell Cultures
Human ovarian cancer cell lines (SKOV3 and CAOV3) were obtained from the American Type Culture Collection (Rockville, MD). Normal human ovarian surface epithelium (NOSE) cells were provided as a generous gift by Dr Bin Ye from Harvard Medical School, Boston, MA. Cells were cultured in sterile 75-cm2 cell culture flasks in MCDB 105 and medium 199 supplemented with 10% fetal bovine serum (Gemini Bioproducts, Calabasas, CA) and 1% antibiotic (200 mM/L L-glutamine, 10 000 units penicillin, and 10 mg/mL streptomycin). Cells were maintained at 37°C under 5% CO2/95% air in a high-humidity chamber. Cell monolayers at 60% to 80% confluence were enzymatically removed using trypsin/EDTA and plated in 96-well flat-bottomed plates at a concentration of 1 × 103 per well.
Cell Proliferation Assay
Cell proliferation was assayed with an MTT test. SKOV3 and CAOV3 cells were seeded in 96-well plates (1 × 103 per well) and serum-starved for 24 hours before adding CDDP (2 µg/mL) as a positive control, with GB (25, 50, and 100 µM) or DMSO (1:2500 dilution) as a negative control. For the drug combination treatment, we used CDDP (1 µg/mL), GB (50 µM), or CDDP (1 µg/mL) plus GB (50 µM) to treat SKOV3 cells for the MTT assay.
Cell growth was assessed at day 3 after CDDP or GB treatment. Ten microliters of the MTT dye solution was added to each well, and the plates were incubated at 37°C for 4 hours in a humidified chamber. After incubation, 100 µL of the solubilization/stop solution was added to each well. One hour after the addition of the solubilization solution, the contents of the wells were mixed and read by the 96-well plate scanning spectrophotometer (µQuant) and quantitative software (KC-junior, Bio-Tek Instruments, Inc, Winooski, VT) at an absorbance of 630 nm for quantitative analysis. Data were collected from at least 3 separate experiments, and each treatment condition was repeated 8 times.
Annexin V–Fluorescein Isothiocyanate (FITC)/Propidium Iodide (PI) Double Staining for Flow Cytometric Analysis (FCM)– Assessed Apoptosis
The SKOV3 and CAOV3 cells were seeded into 75-cm2 culture flasks and treated with DMSO, 25 µM, 50 µM, and 100 µM of GB for 3 days, or in a combination of GB 50 µM with CDDP 1 µg/mL. Apoptotic cells were differentiated from viable or necrotic cells by the combined application of Annexin V-FITC and PI (BD Biosciences Clontech, Palo Alto, CA). Apoptotic and necrotic cells were distinguished on the basis of Annexin V-FITC reactivity and PI exclusion. Briefly, the cells were trypsinized, washed twice with ice-cold phosphate-buffered saline (PBS), adjusted to a concentration of 1 × 106 cells/mL. Volumes of 100 µL of the suspensions were added to each labeled tube, and 10 µL Annexin V-FITC and 10 µL PI (20 µg/mL) were added and incubated for at least 20 minutes at room temperature in the dark. Then, 400 µL PBS binding buffer was added to each tube without washing and analyzed using the computer program Multicycle from Phoenix Flow System (San Diego, CA).
Western Blotting
Cells were washed twice with ice-cold PBS and scarped into 1 mL of ice-cold NP40 lysis buffer (10 mM Tris–HCl [pH 8.0], 150 mM NaCl, 1% [vol/vol] NP40, 1 mM EDTA, 50 mM NaF, 5 mM Na PPI, 1 mM phenylmethylsulfonyl fluoride, 1 mg/mL leupeptin, and 1 mg/mL pepstatin A). Cells were then sonicated for 5 seconds at 5 W. Insoluble debris was removed by centrifugation at 1000g for 15 minutes. Total proteins (50 µg) were analyzed by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis. The detection primary antibodies were sheep anti-p21/p27 (1:1000; Oncogene Science, Inc, Cambridge, MA), mouse anti-cyclin D1 (1:1000; BD Transduction Laboratories, Inc, Lexington, KY), rabbit anti-caspase-3/cleaved caspase-3 and caspase-8/cleaved caspase-8 (1:1000; Santa Cruz Biotechnology, Inc, Santa Cruz, CA), and rabbit anti-β-actin (1:2000; AbCam, Cambridge, MA). Blot quantitation was done with a Molecular Dynamics laser densitometer (model PSD, Mount Holly, NJ) and the Image Quant version 3 software.
Lactate Dehydrogenase (LDH) Release Test
LDH release from cells was determined by using an LDH detection kit (Invitrogen) as an index of cytotoxicity. NOSE cells were seeded at 3 × 103 cells/well 1 day before and cultured for 24 hours with GB (100 µM) or CDDP (1 µg/mL) in 96-well plates. DMSO was used as a control. The assay procedure was performed according to the manufacturer’s instruction.
Statistical Analysis
Analysis of variance and Student’s t tests were used to determine the statistical significance of the differences between experimental groups. Intergroup differences were analyzed using 1-way analysis of variance for the comparison of 3 or more groups. P < .05 was regarded as significant.
Results
Ginkgolide B Inhibits Ovarian Cancer Cell Proliferation and Modulation of Relevant Molecular Effectors
To investigate whether GB could modulate the proliferation rate of SKOV3 and CAOV3 cells, we assessed the growth rate of GB treated cells by MTT assay. As shown in Figure 1, we found that in the presence of 25 µM of GB, cell proliferation of SKOV3 and CAOV3 was decreased to 70% and 90%, respectively, compared with the control cells. With the addition of 50 µM of GB, the SKOV3 and CAOV3 cell proliferation was decreased to 67% and 83%, respectively, compared with controls (P < .05). SKOV3 and CAOV3 cell proliferation in the presence of 100 µM of GB was significantly decreased to 54% and 60%, respectively, compared with the control cells (P < .01; Figure 1A and B).

Ginkgolide B (GB) inhibits ovarian cancer cell proliferation and modulation of relevant molecular effector.
Western blots revealed that SKOV3 cells constitutively expressed higher levels of p21 and p27 proteins than control cells after treatment with an increasing concentration of GB. We also found that the cyclin D1 protein expression was significantly decreased in GB-treated SKOV3 cells (Figure 1C and D).
Ginkgolide B Increases Apoptosis of Ovarian Cancer Cells and Modulation of Relevant Molecular Effector
Because GB exerted an antiproliferation effect, we examined the apoptotic population induced in SKOV3 and CAOV3 cells by GB treatment. SKOV3 and CAOV3 cells were incubated with increasing concentrations of GB (25, 50, and 100 µM) in growth medium for 3 days. FCM analysis revealed the effect of GB on the induction of apoptotic cell death to be an increase in cells in the sub-G1 phase. As shown in Figure 2A and B, the percentage of the sub-G1 fraction in GB-treated cells increased in a dose-dependent manner. After treating with 25 µM of GB, the percentage of cells in the sub-G1 phase increased significantly, to 13.4% for SKOV3 cells and to 17.3% for CAOV3 cells. After treatment with 50 µM of GB, the percentage of cells in the sub-G1 phase was 14.1% for SKOV3 cells and 19.8% for CAOV3 cells. For 100 µM of GB, the percentage of cells in the sub-G1 phase was high, at 48.2% for SKOV3 cells and 31.3% for CAOV3 cells, whereas that of the DMSO-treated cells was low, at 7.9% for SKOV3 and 11.7% for CAOV3. Meanwhile, we examined the cleaved caspase-3 and cleaved caspase-8 expression by Western blot, which revealed an increase after treatment with GB (Figure 2C and D). These data demonstrate that GB is able to increase apoptosis in ovarian cancer cells in vitro.

Ginkgolide B (GB) increases apoptosis of ovarian cancer cells and modulation of relevant molecular effector.
Combination Treatment of GB With CDDP Enhances Their Inhibitory Effects on Ovarian Cancer Cell Proliferation
We further examined the effect of combined treatment of GB with CDDP on SKOV3 cell proliferation by MTT assay. We found that with the addition 1 µg/mL of CDDP or 50 µM of GB, cell proliferation of SKOV3 cells decreased to 64.6% and 67.5%, respectively, by 5 days, compared with the control cells (Figure 3A). In the presence of a combination of low concentrations of CDDP (1 µg/mL) with GB (50 µM), the SKOV3 cell proliferation decreased to 54.1% compared with controls (P < .01). By FCM, as shown in Figure 3B, after treating with 50 µM of GB, 1µg/mL of CDDP, or in a combination of GB with CDDP for 5 days, the percentage of apoptotic cells increased significantly. These data indicate that the combination treatment of GB with low concentrations of CDDP has an additive effect on the inhibition of SKOV3 cell proliferation.

Combination treatment of ginkgolide B (GB) with CDDP enhances their inhibitory effects on ovarian cancer cell proliferation.
GB Has a Lower Cytotoxicity Than CDDP in NOSE
To compare the cytotoxicity of GB with CDDP in normal cells, we measured LDH release from NOSE cells treated with CDDP and GB for 24 hours using an LDH detection kit as an index of cytotoxicity. GB showed a significantly less cytotoxicity than CDDP in NOSE (P < .01; Figure 4). These data confirmed that GB was less toxic to normal cells compared with the traditional chemotherapy agent, CDDP.
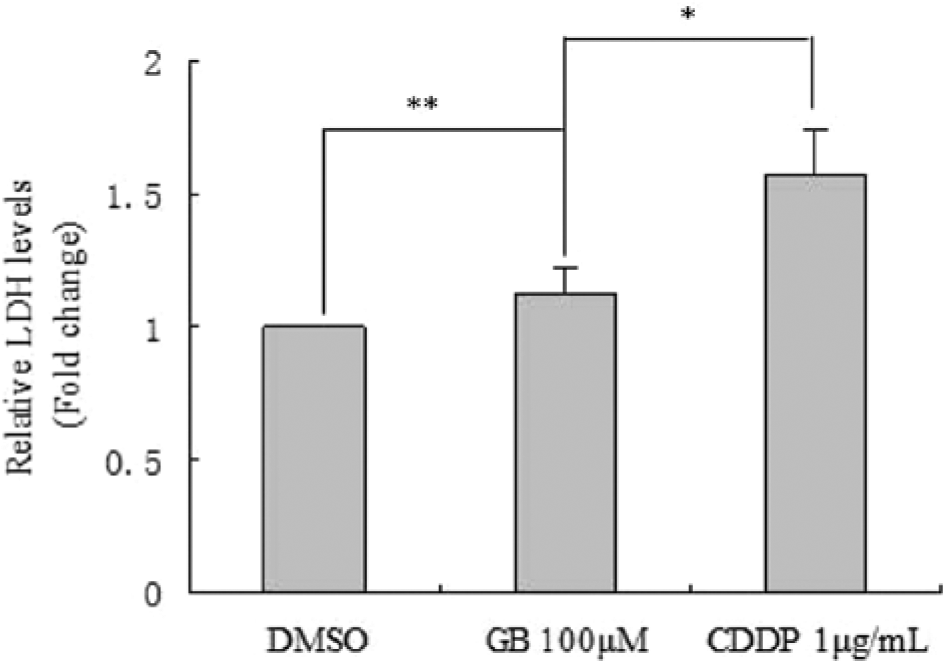
Ginkgolide B (GB) has a lower cytotoxicity than CDDP in NOSE.
Discussion
In the present study, we demonstrated that GB effectively inhibits ovarian cancer cell growth and induces cell apoptosis in vitro in a dose-dependent manner. GB also showed an additive effect with the most commonly used chemotherapy agent, CDDP, and demonstrated significantly less cytotoxicity than CDDP.
New adjuvant therapeutic agents would be extremely beneficial for the control of unresectable and recurrent lesions of ovarian cancer. 14 Ginkgo biloba extract has been shown to provide antiproliferative effects in different systems based on its striking inhibition of diverse cellular events associated with tumor pathogenesis. The inhibitory effect of GB on cell proliferation may be attributed to its antioxidation capacity and cytotoxic action. Furthermore, GB inhibits the activity of several proteins involved in cell growth and signal transduction pathways, including tyrosine phospho-EGFR, Src, FAK, Paxillin, and cdc25 phosphatase.15,16
In the current study, the effects of GB on SKOV3 and CAOV3 cell viability and proliferation were determined by MTT test. GB significantly inhibited cell proliferation of these cell lines with a peak inhibition at 100 µM in 4 days, while it showed more potent growth inhibition in SKOV3 cells than in CAOV3 cells. Our results indicate that the antiproliferative effect of GB in ovarian cancer cells may reflect changes in the expression of several proteins related to the cell cycle and apoptosis induction.
Retarded cell cycle progression was accompanied by diminished cyclin D1 and enhanced p21 and p27 expression levels. 17 Cyclin D1 is considered to regulate the G1/S transition, its loss being associated with an increase of cells in G0/G1 and a decrease of cells in S-phase. 17 This is consistent with our results demonstrating a decrease of cyclin D1 expression level in GB-treated SKOV3 cells. Parallel to cyclin downregulation, enhanced expression of p21 and p27 has been detected in GB treated cells. P21 and p27 are potent cyclin-dependent kinase inhibitors and upregulation of both p21 and p27 attenuates SKOV3 cell proliferation. Recent experiments have demonstrated that enhanced p21 and p27 expression correlates with reduced levels of cyclin D1, 18 which is in accordance with our results.
Our results demonstrate that GB-suppressed SKOV3 and CAOV3 cell viability may also occur by inducing apoptotic cell death. Annexin/PI staining validated GB-induced apoptotic cell death in SKOV3 and CAOV3 cells after treatment with GB. Apoptosis and its related signaling pathways have a profound effect on the progression of cancer.19,20 Induction of apoptosis is, therefore, a highly desirable goal for cancer control. Thus, apoptosis regulators have been targets for cancer therapy for several decades. 21 However, chemotherapeutics usually have many side effects, such as severe bone marrow depression, which are often undesirable for patients. Recently, research in apoptosis induction therapy has been performed with medicinal plants that were known to be chemopreventive candidates with few side effects.22-24 Caspases, cysteine-containing aspartate-specific proteases, play an important role in apoptosis. 25 Activation of caspases is central to the execution of apoptosis. 26 Moreover, activation of caspase-3 is often considered as the point-of-no-return in the apoptotic signaling cascade. 27 The death receptor–dependent apoptotic pathway is triggered at the cell surface and requires activation of caspase-8, whereas the mitochondria-dependent pathway is initiated by the release of mitochondrial cytochrome C into the cytoplasm. 28 Subsequently, caspase-8 can activate caspase-3, which in turn targets and degrades specific and vital cellular proteins, ultimately resulting in nuclear DNA degradation and apoptotic cell death. 29 Our Western blot data indicated that GB stimulated the expression of cleaved caspase-3 and cleaved caspase-8 in SKOV3 cells in a dose-dependent manner. These results suggest that GB could activate caspase-8 via the mitochondria-dependent pathway and that caspase-8 could then activate caspase-3. Caspase-3 in turn cleaves cytoskeletal and nuclear proteins, which finally induces apoptosis.
Importantly, we demonstrated that the combination of a low concentration of CDDP and GB had an additive effect on the inhibition of SKOV3 ovarian cancer cell proliferation, and GB had significantly less cytotoxicity than CDDP, thereby providing a rationale for the clinical use of GB as an adjuvant therapy for ovarian cancer. In the current study, we demonstrated for the first time that GB is a potent inhibitor of ovarian cancer cells with relatively low toxicity to normal cells. Because GB has different mechanisms of action and lower toxicity to normal cells compared with other clinically used chemotherapy agents such as CDDP, it could be used as adjuvant therapy in combination with CDDP in ovarian cancer patients.
In summary, we demonstrated that GB inhibits ovarian cancer cell growth and induces apoptosis. These results are of significance because GB may have clinical applications in the treatment of ovarian cancer because of its direct inhibitive effects and for its ability to enhance the effects of other chemotherapeutics with a low toxicity. Combination therapy of GB with chemotherapy agents such as CDDP may provide a new and more effective strategy for the treatment of ovarian cancer.
Footnotes
Authors’ Note
Wei Jiang and Qing Cong contributed equally to this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Shanghai Municipal Health Bureau Youth Research Project (No. 2008Y001) and Dana-Farber Cancer Center Starr Foundation and ovarian cancer Specialized Programs of Research Excellence (1P50-CA105009-01).
