Abstract
Objective. We evaluated the effects of total alkaloids of Rubus alceifolius Poir (TARAP) on the migration and invasion of hepatocellular carcinoma (HCC) and furthermore investigated the possible molecular mechanisms mediating its anticancer activity. Methods. We implanted nude mice with human HCC HepG2 cells and fed them with vehicle (physiological saline) or 3 g/kg/day dose of TARAP 5 days per week for 21 days. We determined the in vitro effect of TARAP on the migration and invasion of HepG2 cells by transwell assay. We evaluated SHH signaling components’ (SHH, PTCH, SMO, and Gli1) expression levels by reverse transcriptase–polymerase chain reaction and immunohistochemistry. Activity of the matrix metalloproteinases (MMPs) in supernatants was analyzed by zymography. The expression of the MMPs and their specific tissue inhibitor (tissue inhibitor of matrix metalloproteinases, TIMP-1, 2) in HCC tissues was detected by immunohistochemistry. Results. We discovered that TARAP inhibited hepatocellular migration and invasion in a dose-dependent manner in vitro. In addition, TARAP decreased the expression of SHH, PTCH, SMO, and Gli1 in HCC mouse tumors at both transcriptional and translational levels. Moreover, TARAP inhibited the activity of MMP2 and MMP9. We found that TARAP reduced the expression of MMP2 and MMP9, as well as the tissue inhibitor of MMPs. Conclusion. Our study showed that TARAP inhibits HCC migration and invasion likely through suppression of the hedgehog pathway. This may, in part, explain its anticancer properties. These results suggest that total alkaloids in Rubus alceifolius may have potential as a novel antimetastasis drug in the treatment of HCC.
Introduction
Hepatocellular carcinoma (HCC) represents a major health problem. It is reported that HCC is the sixth most common malignancy worldwide. 1 After primary diagnosis is made, local therapies can be applied through several effective biomarkers. But patients with HCC still have a high rate of recurrence due to local invasion and intrahepatic metastasis. 2 Total alkaloids is an active ingredient of the natural plant Rubus alceifolius Poir, commonly found in Jiangxi Province, Fujian Province, and Yunnan Province in China, as well as in other Asian countries and Australia. The plant is commonly used for the treatment of liver diseases. Although the role of total alkaloids of Rubus alceifolius Poir (TARAP) in tumor cell proliferation and apoptosis has been previously reported,3,4 the effect of TARAP on cell migration and invasion has not been reported. Moreover, molecular mechanisms governing the antiinvasive activity of TARAP have not been clearly demonstrated.
The hedgehog pathway is essential for embryonic development, tissue polarity, and cell differentiation in animals. 5 The pathway is activated by binding of the ligand Hh to its receptor Patched (PTCH). This binding releases a second receptor, Smoothened (SMO), which moves to the membrane and triggers a series of reactions that result in translocation of transcription activators encoded by glioma-associated oncogenes (Gli1, Gli2, and Gli3) into the nucleus and subsequent transcription of target genes. The hedgehog pathway is critical in the early development of the liver and contributes to differentiation between hepatic and pancreatic tissue formation. The adult liver normally does not have detectable levels of hedgehog signaling.6,7 However, it has been reported that the SHH signaling pathway is activated in the presence of HCC. 8 Research has demonstrated that SHH and IHH as well as PTCH, SMO, and Gl1 are up-expressed in malignant human hepatocellular cell lines HepG2 and Hep3B. Expression and inhibition of SMO decreased Hep3B viability, and SMO RNA abundance is correlated with HCC tumosize. 9 Examination of a collection of 115 HCCs showed that they express components of the Hh pathway. 10 This suggests that the Hh pathway plays an important role in the development of HCC. In this study, we investigated the effect of TARAP on cell migration and invasion and assessed molecular mechanisms with special focus on hedgehog pathway regulation.
Methods and Materials
Materials and Reagents
Dulbecco’s modified Eagle medium (DMEM), fetal bovine serum (FBS), penicillin–streptomycin, trypsin-EDTA, antibodies for SHH (Human, pAd), PTCH (Human, pAd), SMO (Human, pAd), Gli1 (Human, pAd), MMP2 (Human, pAd), MMP9 (Human, pAd), TIMP1 (Human, pAd), and TIMP2 (Human, pAd) were obtained from Santa Cruz Biotechnology, Inc (Santa Cruz, CA). All antibodies were diluted 1:100. Matrigel was purchased from BD Biosciences (Bedford, MA). Eight-micrometer pore size transwell inserts were purchased from Corning Corporation (Corning, NY). All other chemicals, unless otherwise stated, were obtained from Sigma Chemicals (St Louis, MO).
Preparation and Content of Total Alkaloids of Rubus alceifolius Poir
TARAP was prepared according to previously described methods. On identification and authentication, alkaloids were extracted and purified as reported. Briefly, the roots of R alceifolius were collected from Anxi in Fujian Province, China, identified and authenticated by experts in our university, and the alkaloids were extracted. The herb powder (1 g) was extracted using 50 mL chloroform–methanol–ammonia solution (15:4:3) for 2 hours in an ice bath, sonicated for 30 minutes, brought to room temperature, and filtered. The filtered solution was collected and desiccated. The resultant residue was dissolved by 2 mL of 2% sulfuric acid solution and filtered. The filter paper and residue were rewashed with 2 mL of 2% sulfuric acid solution and with buffer solution (pH 3.6). The buffer was then added to make a final volume of 50 mL, and the solution was saved for future use. Acid dye colorimetry was used to measure the total alkaloid content. The total alkaloid content was 0.81 mg alkaloid per gram of initial herb powder.
Cell Culture
Human HCC cell lines HepG2 were obtained from the China Type Culture Collection (Shanghai, China). Cells were cultured in DMEM medium containing 10% FBS, 100 units/mL penicillin, 100 ng/mL streptomycin grown under a humidified 5% CO2, 95% air at 37°C.
Animals
Six-week-old athymic BALB/c nu/nu male mice were purchased from SLAC Animal Inc (Shanghai, China) and housed in standard plastic cages under automatic 12 hour light/dark cycles at 23°C, with free access to food and water. All animals were kept under specific pathogen-free conditions. The animal studies were approved by the Fujian Institute of Traditional Chinese Medicine Animal Ethics Committee (Fuzhou, Fujian, China). The experimental procedures were carried out in accordance with the Guidelines for Animal Experimentation of Fujian University of Traditional Chinese Medicine (Fuzhou, Fujian, China).
Methods
Transwell Migration Assay
HepG2 cells were evaluated in 8-µm pore size transwell insert (Corning). The bottom chambers of the transwell were filled with 600 µL of migration-inducing medium (DMEM medium with 10% FBS) and the top of each chamber was seeded with 2 × 104 HepG2 cells treated with different concentrations of TARAP (0, 0.25, 0.5 mg/mL). Cells were allowed to migrate for 24 hours at 37°C, and then removed from the upper compartment of the filter with a cotton swab. The filters were fixed with 4% paraformaldehyde for 30 minutes at room temperature, and then stained with 0.1% crystal violet. Percentage inhibition of migratory cells was quantified and expressed based on untreated control wells.11,12
Matrigel-Based Transwell Invasion Assay
Invasion assay was performed using the same transwell as above. Briefly, the upper surface of the transwell membrane was coated with Matrigel 5 µg while the lower compartment of the chambers was filled with 500 µL DMEM medium containing 10% FBS. HepG2 cells (2 × 105) were placed in the upper part of each transwell and treated with different concentrations of TARAP (0, 0.25, 0.5 mg/mL) for 24 hours. After incubation, cells on the upper side of the filter were removed with cotton swabs. The filters were fixed with 4% paraformaldehyde for 30 minutes at room temperature, and then stained with 0.1% crystal violet. Cells that had invaded through the Matrigel and reached the lower surface of the filter were quantified by counting the number of cells that migrated in 5 random microscopic fields per filter at a magnification of 200×. 11
In Vivo Tumor Xenograft Study
HepG2 cells were grown in culture and then detached by trypsinization, washed, and resuspended in serum-free DMEM. Six-week-old athymic BALB/c nu/nu male mice were given a subcutaneous injection of 4 × 106 HepG2 cells mixed with Matrigel (1:1) in the right flank to initiate tumor growth. When tumor sizes reached 3 mm in diameter mice were randomly divided into 2 groups and gavaged with the following: (a) control group (n = 10), physiological saline (PS); (b) TARAP group (n = 10), 3 g/kg/day dose of TARAP in PS. All treatments were given 5 days a week for 21 days. At the end of the experiment the mice were anesthetized with ether and sacrificed by cervical vertebra dislocation. The tumors were then excised, and tumor segments were fixed in buffered formalin and stored at −80°C for molecular analyses.
Gene Expression Analysis by RT-PCR
The expression of SHH, PTCH, SMO, and Gli1 genes was analyzed by reverse transcriptase–polymerase chain reaction (RT-PCR). Total RNA was isolated with trizol reagent according to the manufacturer’s instructions. One microgram of total RNA was used to synthesize cDNA using the SuperScript II reverse transcriptase Kit (AMV; TaKaRa, Kyoto, Japan) with random primers in a volume of 10 µL in 10 mM Tris–HCl (pH 8.3), 50 mM KCl, 5 mM MgCl, 1 unit/µL RNase inhibitor, 0.25 unit/µL AMV reverse transcriptase, 2.5 mM random primer, and 1 mM each of dATP, dGTP, dCTP, and dTTP. Reverse transcription was performed for 1 hour at 42°C. The obtained cDNA was used to determine the mRNA amount of SHH, PTCH, SMO, and Gli1 by PCR. GAPDH was used as an internal control.
Immunohistochemistry Analysis
Immunohistochemical staining for SHH, PTCH, SMO, Gli1, MMP2, MMP9, TIMP1, and TIMP2 was performed as previously described. The sections were deparaffinised in xylene and hydrated through graded alcohols. Antigen unmasking was performed using heat treatment in a microwave oven at 750 Watts for 7 minutes in a container with 10 mM sodium citrate buffer, pH 6.0. Sections were allowed to cool in the buffer at room temperature for 30 minutes, and were then rinsed in deionized water 3 times for 2 minutes each. The endogenous peroxidase activity was blocked with 3% (v/v) hydrogen peroxide for 10 minutes. The sections were incubated with 1% bovine serum albumin in order to decrease nonspecific staining and reduce endogenous peroxidase activity. The sections were then incubated with SHH, PTCH, SMO, Gli1, MMP2, MMP9, TIMP1, or TIMP2 antibody, all in 1:100 dilution, at 4°C overnight using a staining chamber. Primary antibodies were diluted (1:100) in phosphate-buffered saline (PBS). After rinsing 3 times in PBS, sections were incubated in biotinylated goat anti-rabbit IgG (Boshide Wuhan, China) followed by avidin-biotin-peroxidasecomplex (Vector). Immunostaining was visualized by incubation in 3,3-diaminobenzidine (DAB) as a chromogen. Sections were counterstained with hematoxylin. The SHH, PTCH, SMO, and Gli1 positive immunostainings were evaluated by the use of Nikon Eclipse 50i microscope (40× objective). The evaluation of SHH, PTCH, SMO, and Gli1 expression was analyzed in 5 different fields, and the mean percentage of SHH, PTCH, SMO, Gli1, MMP2, MMP9, TIMP1, or TIMP2 positive staining was evaluated.
Zymography
Activity of MMP2 and MMP9 in HepG2 cells was estimated by gelatin zymography. HepG2 cells were grown in 100-mm diameter tissue culture plates until 90% confluent. The cells were washed once with serum-free medium and placed in serum-free medium for 2 hours prior to drug treatment. Different concentrations of TAPAP (0, 0.25, 0.5 mg/mL) were added to the plate and the plate was incubated in the 5% CO2 incubator at 37°C for 24 hours. The medium was collected, concentrated, and subjected to zymography. Ten microliters of the sample was electrophoresed in 8% polyacrylamide gel containing 0.1% gelatin in the presence of SDS. After electrophoresis, the gel was washed 2 times, 20 minutes each, in 2.5% Triton X-100 and incubated overnight at 37°C in a solution containing 50 mM Tris–HCl, pH 8, 5 mM CaCl2, 10 mM ZnCl2, and 0.02% NaN3. The gel was then stained for 30 minutes with 0.5% Coomassie Brilliant Blue R-250 prepared in 30% methanol/10% acetic acid solution and destained with several washes with water. Nonstaining regions of the gel corresponding to MMP2 and MMP9 were quantified by densitometry using Chemi Doc XRS+ image analyzer (Bio-Rad).
Statistical Analysis
All data are the means of 3 determinations. The data were analyzed using the SPSS package for Windows (Version 11.5). Statistical analysis of the data was performed with Student’s t test and ANOVA. Differences with P < .05 were considered statistically significant.
Results
Migration and Invasion Are Inhibited by TARAP in HepG2 Cells
To evaluate the influence of TARAP on cell migration and invasion, HepG2 cells were treated with 0 to 0.5 mg/mL of TARAP to do transwell migration and Matrigel-based transwell invasion assay. Our results indicated that TARAP could significantly inhibit HCC cell migration (Figure 1A) and invasion (Figure 1C) in a dose-dependent manner, suggesting a crucial role for TARAP in suppressing HCC metastasis. In the untreated control, HepG2 cells showed high rates of migration and invasion; the rates were significantly reduced to 57 ± 6.27%, 32 ± 4.16% (Figure 1B), and 39 ± 4.68%, 23 ± 3.08% (Figure 1D), when HepG2 cells were treated with TARAP at 0, 0.25, and 0.5 mg/mL concentrations, respectively.

Effect of TARAP on the migration an invasion of HepG2 cells.
SHH Signaling Components Were Downregulated by TARAP in HCC Tissue
Certain components of the SHH signaling pathway have been reported to be overexpressed in HCC compared to normal tissue. 6 To evaluate the role of TARAP on the SHH signaling pathway, we tested the expression of SHH, PTCH, SMO, and Gli1 using RT-PCR and immunohistochemistry. We found that all components of the pathway tested (SHH, PTCH, SMO, and Gli1) were downregulated by TARAP at transcriptional levels. Figure 2A and B shows that TARAP reduced the relative mRNA expression of SHH, PTCH, SMO, and Gli1 to 0.293 ± 0.031, 0.341 ± 0.029, 0.578 ± 0.043, 0.642 ± 0.052, respectively. Results of immunohistochemical (IHC) assay consistently showed that the protein expression patterns of SHH, PTCH, SMO, and Gli1 were similar to their respective mRNA levels. The percentage of SHH, PTCH, SMO, or Gli1 positive cells in control group was 76.83 ± 14.06%, 50.83 ± 6.56%, 70.19 ± 11.32%, or 56 ± 7.39%, whereas that in TARAP-treated mice was 26.33 ± 4.51%, 18.67 ± 2.67%, 27 ± 3.83%, or 32.83 ± 4.93% (Figure 2C and D). Collectively, it is suggested that TARAP inhibits the HCC cell migration and invasion via suppressing the SHH signaling pathway.
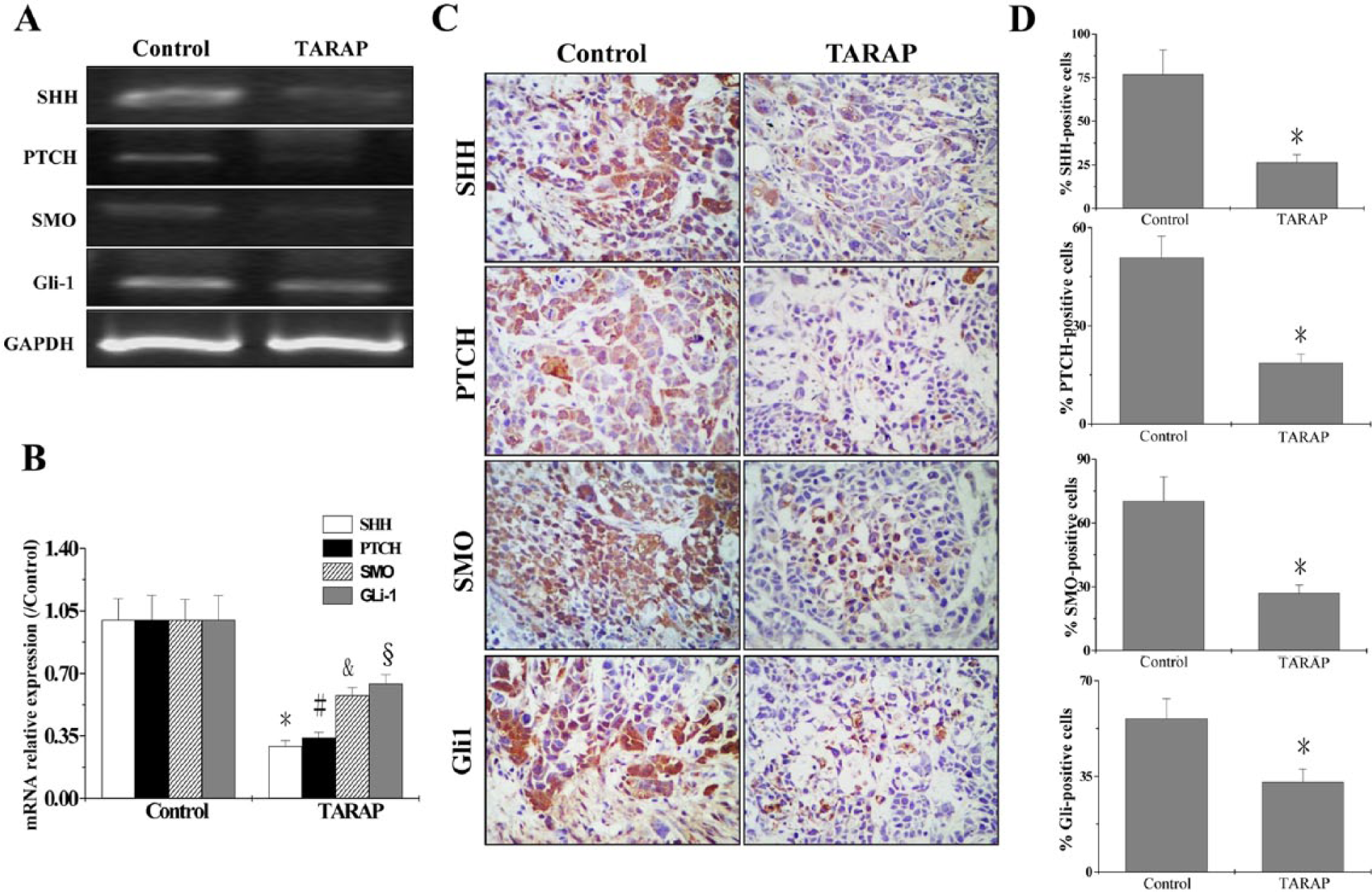
TARAP treatment profoundly reduced expression of signaling molecules.
The Gelatinolytic Activity of MMP2 and MMP9 Was Inhibited by TARAP
To investigate whether the antiinvasive activity of TARAP was a result of inhibition of MMPs, we examined the effect of TARAP on the expression and activity of MMP2 and MMP9 gelatinases in HepG2 cells. We found treatment of HepG2 cells with different concentrations of TARAP (0, 0.25, 0.5 mg/mL) inhibited MMP2 and MMP9 gelatinolytic activity in a zymography assay. The inhibition was pronounced in a dose-dependent manner. Figure 3A and B shows the inhibition of MMP2 and MMP9 activity 0.73 ± 0.102, 0.51 ± 0.066, and 0.87 ± 0.103, 0.54 ± 0.06 at 0, 0.25, and 0.5 mg/mL concentrations, respectively.

Analysis of supernatants of HepG2 cells incubated with TARAP.
Inhibition of the SHH Pathway Regulates Matrix Metalloproteinases and the Tissue Inhibitor of Metalloproteinase
To further evaluate the effect of SHH pathway on migration and invasion, the MMPs and their tissue inhibitor of metalloproteinase were tested by IHC. As Figure 4A and B shows, the percentage of MMP2, MMP9, TIMP1, TIMP2 positive cells in control was 54.17 ± 7.53%, 41.83 ± 7.24%, 26.5 ± 3.63%, and 16.17 ± 2.13%, whereas in cells treated with TARAP it was 24.33 ± 3.36%, 18.17 ± 2.6%, 45.83 ± 6.14%, and 56 ± 7.78%, respectively. These data together demonstrate that TARAP inhibits the SHH pathway and further regulates the MMPs and their tissue inhibitor of metalloproteinase.
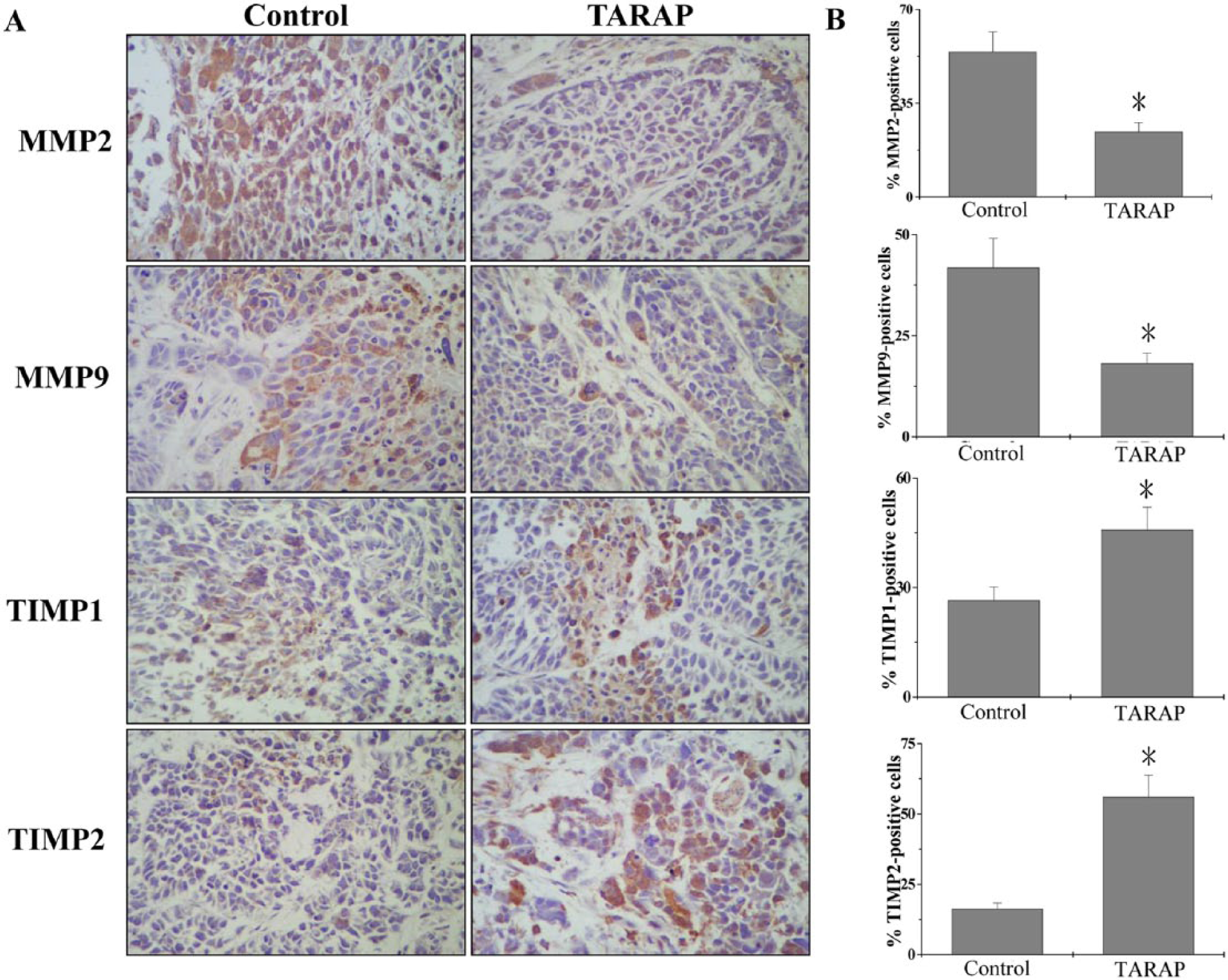
Immunohistochemical staining of tumor tissues for MMP2, MMP9, TIMP1, and TIMP2.
Discussion
Intrahepatic metastasis is the major characteristic of HCC malignancy, and it remains a risk in some patients even when tumor mass is surgically removed at an early stage of disease. Many patients retain residual disease after surgery, which can eventually lead to metastasis. Tumor cells’ ability to migrate and invade healthy tissue is the foundation of tumor metastasis. If migration and invasion can be prevented, the prognosis of HCC is much improved. Unfortunately, many antitumor agents currently in use are intrinsically cytotoxic. This toxicity to normal cells limits the effectiveness of current cancer therapies. Natural products have relatively few side effects and have been used clinically for thousands of years to treat various types of diseases, including cancer.13,14 As a natural product, Rubus alceifolius Poir has shown strong therapeutic potential for liver cancer.3,4 However, the precise mechanism by which it limits cancer growth has remained largely unclear.
The hedgehog signaling pathway plays an important role in regulating growth, proliferation, and apoptosis of HCC.7,8,15 The invasion and metastasis of tumor cells are tightly regulated by the Hh pathway, 16 and many studies have shown that Hh signaling pathway inhibitors have a cytotoxic effect on tumor cells.17-19 Recently, much research showed that the Hh signaling pathway (including PTCH, SMO, Gli, and others) is tightly associated with tumor invasive and metastasis.19-22 We therefore assessed the effect of TARAP on the expression of key mediators of the SHH pathway using RT-PCR and IHC analyses. Our data in this study showed that TARAP blocked the SHH signaling containing the expression of SHH, PTCH, SMO, and Gli1 in HCC mouse tumors to inhibit the growth of HCC tumors. These results suggest that the in vivo inhibitory effect of TARAP on tumor migration and invasion could be mediated by the suppression of SHH signaling pathway.
Local invasion and distant metastasis are one of the most important factors in the prognosis of malignant tumors. Degradation of the extracellular matrix (ECM) that surrounds tumor cells is one of the essential steps that may be mediated by MMPs and their specific tissue inhibitors (TIMPs).23-26 MMPs are zinc-dependent enzymes that play an important role in this proteolytic degradation of the ECM. Various types of MMPs have been reported to date, and these MMPs are subdivided into several groups. MMP2 and MMP9 are considered to be particularly important targets for the development of anticancer drugs because they are associated with aggressive, invasive, or metastatic tumor phenotype.27,28 Nagai et al have shown that Gli1 contributes to the invasiveness of pancreatic cancer through MMP9 activation. 29 Our results demonstrate that TARAP inhibited expression of MMP2 and MMP9 both by zymography and IHC. In addition, TARAP promoted the expression of TIMP1 and TIMP2 by IHC analysis.
In the present study, we demonstrated that TARAP inhibited the migration and invasion of human HCC HepG2 cells in a dose-dependent manner. We also discovered that TARAP blocked the SHH signaling pathway, a likely mechanism by which TARAP acts to further inhibit the migration and invasion of disease cells. In addition, TARAP regulated the MMPs and their tissue-specific inhibitors to inhibit degradation and cellular invasion of the ECM and basement membrane. We believe the data in this study provide new information about the antimetastatic potential of TARAP beyond its demonstrated antitumor activity.
Footnotes
Acknowledgements
We would like to thank Clarity Manuscript Consultants, LLC for their help in the editing of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by the National Natural Science Foundation of China (No. 81303125) and Nature Science Foundation of Fujian Province of China (No. 2010J01191 and 2010J01194); and the project was sponsored by the Medical Originality Foundation of Fujian Province of China (No. 2009-CX-18).
