Abstract
Introduction and Objective. Cancer-related fatigue (CRF) is the most common and severe symptom in patients with cancer. The number and efficacy of available treatments for CRF are limited. The objective of this preliminary study was to assess the safety of high-dose Panax ginseng (PG) for CRF. Methods. In this prospective, open-label study, 30 patients with CRF (≥4/10) received high-dose PG at 800 mg orally daily for 29 days. Frequency and type of side effects were determined by the National Cancer Institute’s Common Terminology Criteria for Adverse Events, version 4.0. Scores on the Functional Assessment of Chronic Illness Therapy–Fatigue (FACIT-F) scale, Edmonton Symptom Assessment System (ESAS), and Hospital Anxiety and Depression Scale (HADS) were assessed at baseline, day 15, and day 29. Global Symptom Evaluation (GSE) was assessed at day 29. Results. Of the 30 patients enrolled, 24 (80%) were evaluable. The median age was 58 years; 50% were females, and 84% were white. No severe (≥grade 3) adverse events related to the study drug were reported. Of the 24 evaluable patients, 21 (87%) had an improved (by ≥3 points) FACIT-F score by day 15. The mean ESAS score (standard deviation) for well-being improved from 4.67 (2.04) to 3.50 (2.34) (P = .01374), and mean score for appetite improved from 4.29 (2.79) to 2.96 (2.46) (P = .0097). GSE score of PG for fatigue was ≥3 in 15/24 patients (63%) with median improvement of 5. Conclusion. PG is safe and improves CRF fatigue as well as overall quality of life, appetite, and sleep at night. Randomized controlled trials of PG for CRF are justified.
Introduction
Previous research has shown that patients with cancer develop severe physical and psychosocial symptoms. The frequency of cancer-related fatigue (CRF) varies from 60% to 90%.1,2 Fatigue adversely affects the physical, functional, and psychological domains of quality of life (QOL), resulting in the inability to perform daily activities and affecting mood, social relationships, and work.2-4 Prior studies have revealed that CRF has a multifactorial etiology in these patients. Different mechanisms, directly or indirectly, affect brain function to cause the subjective symptom of fatigue.5-8 Several factors are significantly associated with CRF, including anxiety, depression, pain, anorexia/cachexia, drowsiness, sleep disturbances, cognitive functioning, and performance status. 9 In recent years, several cytokines and other pro-inflammatory mediators that are produced by the host in response to the presence of cancer have been found to be associated with symptoms such as fatigue, pain, cognitive impairment, depression, cachexia, and sleep disorders.10-13 Despite the prevalence and severity of CRF and its effects on the QOL of patients with cancer, the number and efficacy of available treatment options are limited. 14
Ginseng root (Panax ginseng C.A. Meyer, Araliaceae; hereafter “PG”) has a long history of use, particularly in China, in the treatment of fatigue, debility, and declining work capacity and concentration; it is also administered during convalescence.15-17 Currently, PG is widely used in the United States as a traditional medicine to mitigate fatigue in the general population. It has been found to have direct action on cognition/memory, sleep disturbance, anxiety/depression, pain, and the ability to modulate inflammatory cytokines.15,16,18 However, despite evidence of its beneficial effects and safety in the general population, there are no published studies of PG in cancer patients on safety, tolerability and its effects on fatigue and symptoms using validated instruments. The purpose of this preliminary study was to evaluate the safety and tolerability of high-dose, standardized PG extract for the management of CRF.
Methods
The University of Texas MD Anderson Cancer Center Institutional Review Board approved the protocol, and all patients provided written, informed consent.
Patients
Patients were approached by the research nurse in outpatient clinics for palliative care, pain management, internal medicine, and oncology at MD Anderson Cancer Center in Houston, Texas. To be included in the study, patients must have been diagnosed with cancer and currently undergoing outpatient chemotherapy at the cancer center, and experiencing CRF with an average intensity of ≥4 on the Edmonton Symptom Assessment Scale (ESAS; a 0-10 scale) during the 24 hours prior to enrolling in the study. CRF was described as being present every day for most of the day for a minimum of 2 weeks. Other important eligibility criteria were as follows: normal cognition; no infections; hemoglobin ≥8 g/L within 2 weeks of enrollment; Zubrod performance status of ≤2; no current uncontrolled pain or depressive symptoms; no history of psychiatric illness such as major depression, obsessive compulsive disorder, or schizophrenia; no uncontrolled diabetes or treatment with anticoagulants or systemic steroids; no history of hepatitis A, B, or C; no significant history of uncontrolled hypertension or symptomatic tachycardia; and no current use of medications such as ginseng, methylphenidate, modafinil, phenobarbital, diphenylhydantione, primidone, phenylbutazone, monoamine oxidase inhibitors, clonidine, and tricyclic antidepressants.
Intervention
Eligible patients who agreed to participate in the study were given a 29-day supply of 400-mg PG capsules. They were directed to take 1 capsule orally twice a day for 4 weeks. The patients were instructed to take 1 capsule of the study medication in morning and 1 capsule prior to 3
Panax ginseng extract capsules (PG) were supplied by Indena S.p.A. (Milan, Italy). PG which is commercially available, was prepared from Panax ginseng C.A. Meyer root (drug extract ratio [DER] 1:3–5) and standardized 19 to contain ≥7.0% of ginsenosides and malonylginsenosides (≥0.9% Rg1 ≤1.4%; ≥1.7% Rb1 ≤3.0%). The manufacturing percentage range (mean ± standard error of the mean) of ginsenosides was 12% ± 3%. The extract was prepared through hydroalcoholic extraction (EtOH 70%).
Ginseng Dosage Justification
According to monographs of the World Health Organization and German Commission E, the current recommended dosage of PG is 2 to 3 g per day of dried plant material or 300 to 800 mg per day of a standardized extract that contains 4% to 7% ginsenosides.20,21 However, the Pharmacopoeia of the People’s Republic of China recommends dosages of 3 to 9 g of the root as a tea, with higher dosages recommended for more debilitated patients. 22 Several clinical trials23-37 (Table 1) have been performed using various doses from 40 to 800 mg of ginseng extract and 2 to 6 g of plant material. 38 In this study, we used a dosage of 800 mg per day to evaluate the safety and tolerability of PG in cancer patients for CRF. Furthermore, these dosages were confirmed as reasonable, with regard to efficacy and safety, on consultation with Mark Blumenthal (founder and executive director of the American Botanical Council and editor/publisher of HerbalGram), Dr Lorenzo Cohen (Professor and Director of the Integrative Medicine Program at MD Anderson Cancer Center), and Dr Moshe Frenkel (founder and director Integrative Oncology Consultants).
Dosages of Panax ginseng Previously Used in Clinical Trials.

Dried root.
Standardized ginseng extract of the dried root.
Outcome Measures
Patients’ demographic data, including age, sex, ethnicity, cancer diagnosis, and education level were recorded at the time of study entry.
The safety of PG was assessed by the research nurse at baseline (prior to initiation of treatment), treatment day 15, day 29, day 43, and day 57, in accordance with the National Cancer Institute’s Common Terminology Criteria for Adverse Events, version 4.0.
A research nurse also supervised the patients’ completion of the Functional Assessment of Chronic Illness Therapy (FACIT) fatigue subscale (FACIT-F), ESAS, and Hospital Anxiety and Depression Scale (HADS) at baseline, day 15 and day 29. The patient also completed Global Symptom Evaluation questionnaire (GSE) at the day 29.
Functional Assessment of Chronic Illness Therapy–Fatigue
The FACIT-F is a well-validated QOL instrument widely used for the assessment of CRF in clinical trials. It consists of 27 general QOL questions classified into 4 domains (physical, social, emotional, and functional), which constitutes FACT-G and the 13-item FACIT-F subscale. 39 FACIT-F allows patients to rate the intensity of their fatigue and its related symptoms on a scale of 0-4 (0 = not at all, 4 = very much). We chose the FACIT-F score as the primary outcome measure because it has been widely used in CRF treatment trials by our team and others.40-43 Test-retest reliability coefficients for the FACIT-F have ranged from .84 to .90. 44 This scale has been shown to have strong internal consistency (α = .93-.95) and has a sensitivity of 0.92 and specificity of 0.69. 44
Edmonton Symptom Assessment System
The ESAS was used to assess 10 symptoms commonly experienced by cancer patients during the previous 24 hours: pain, fatigue, nausea, depression, anxiety, drowsiness, dyspnea, anorexia, sleep disturbance, and feelings of well-being.45,46 The severity of each symptom was rated on a numerical scale of 0-10 (0 = no symptom, 10 = worst possible severity). we categorized the ESAS symptoms as follows: ESAS physical, sum of pain, shortness of breath, appetite, nausea, fatigue, and drowsiness scores; ESAS psychological, sum of anxiety, and depression scores; and ESAS symptom distress, sum of pain, dyspnea, appetite, nausea, fatigue, drowsiness, anxiety, depression, and well-being scores. The ESAS is both valid and reliable in the assessment of the intensity of symptoms in cancer patients.47,48
Hospital Anxiety and Depression Scale
Depression and anxiety were assessed using HADS. This 14-item questionnaire has been validated in a number of clinical situations and has been widely used in patients with cancer.49,50
Global Symptom Evaluation
Patients were asked about their fatigue (worse, about the same, or better) after starting study medication. If their answer was “better,” patients were asked to rate how much better their symptoms are (hardly any better at all, a little better, somewhat better, moderately better, a good deal better, a great deal better, a very great deal better). If their answer was “worse,” patients were asked to rate how much worse their symptoms were (hardly worse at all, a little worse, somewhat worse, moderately worse, a good deal worse, a great deal worse, a very great deal worse). 51
Statistical Analysis
We summarized the demographic and clinical characteristics of all patients enrolled in the pilot study. Means and standard deviation or medians and interquartile ranges were calculated. We also performed exploratory analyses on the changes in FACIT-F subscale (primary outcome measure) from baseline to day 29. Similar analyses were conducted for the secondary outcomes (FACT-G, ESAS symptoms, HADS. Based on whether the data were normally distributed (Shapiro-Wilk test) we used paired t test. We also summarized fatigue scores, other secondary outcomes including ESAS items, FACT-G and HADS at each point in time, and the frequency and type of side effects as determined by National Cancer Institute’s Common Terminology Criteria for Adverse Events, version 4.0.
Results
A total of 129 patients were approached to participate in this study. Of those, 69 were not eligible owing to low symptom burden (n = 23) or contraindications (n = 46); 16 refused because they did not have enough time or were unable to complete follow-up assessments; and 12 refused because they were not interested or were not feeling well. Two patients signed the consent and withdrew consent prior to the start of the study because of refusal to complete study assessments. Of 30 patients who took at least one dose of PG, 24 (80%) were evaluable. The patients who were not evaluable had disease progression (3), did not comply with the study protocol (2), or were lost to follow-up (1).
The characteristics of the 30 patients enrolled in the study are summarized in Table 2. The median age was 58 years, 50% were females, 26/30 (84%) were white, and 4/30 (16%) were African American. The most common cancer type was genitourinary cancer (31%). Table 3 shows that no grade 3 or higher adverse events related to the study drug were reported. Table 4 shows ESAS scores (mean, standard deviation [SD]) for feeling of well-being improved from 4.67 (2.04) to 3.50 (2.34) (P = .013) and scores for appetite improved from 4.29 (2.79) to 2.96 (2.46) (P = .0097). Figure 1 shows significant improvement of ESAS physical, psychological, and symptom distress scores. Of 24 evaluable patients, 21 (87%) had improved FACIT-F scores (≥3 points) by day 15. The improvement in the GSE score for fatigue was 3 points or greater in 15/24 patients (63%), and the median improvement was 5 points (1 = hardly any better, 7 = a very great deal better).
Patient Characteristics at Baseline (N = 30).

Abbreviations: FACIT-F, Functional Assessment of Chronic Illness Therapy–Fatigue; HADS, Hospital Anxiety and Depression Scale; ESAS, Edmonton Symptom Assessment Scale; SD, standard deviation.
Summary of Types of and Severity of Adverse Events Experienced by Patients Who Received Panax ginseng (N = 18).

Changes From Baseline in Symptom Scores at Day 29 and Day 15.
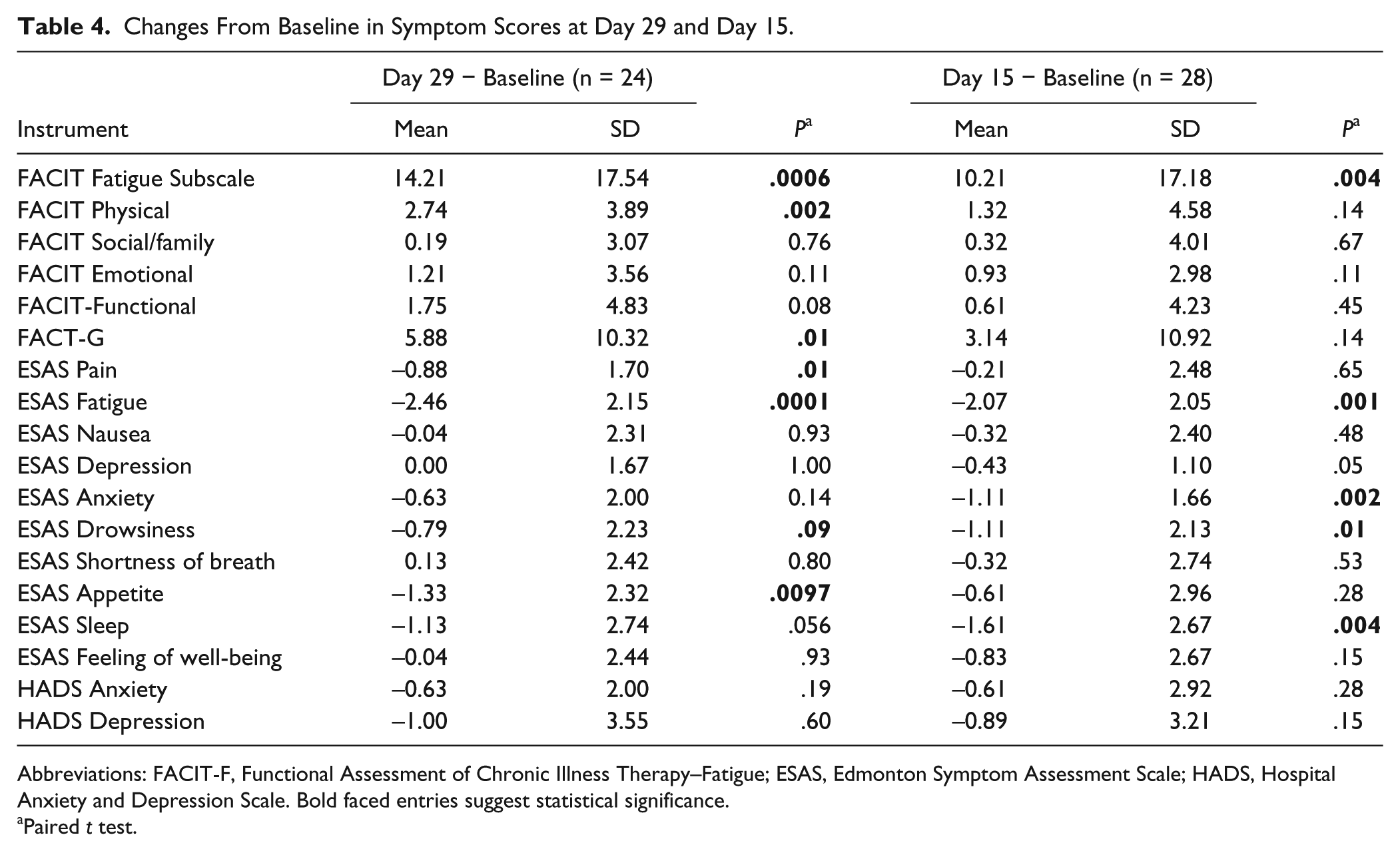
Abbreviations: FACIT-F, Functional Assessment of Chronic Illness Therapy–Fatigue; ESAS, Edmonton Symptom Assessment Scale; HADS, Hospital Anxiety and Depression Scale. Bold faced entries suggest statistical significance.
Paired t test.

Change in symptom distress scores*
Discussion
The results of this preliminary study showed that high-dose PG was safe and tolerable, and no adverse events related to the study drug were reported. The study also showed that CRF and other symptoms including pain, appetite, and overall QOL improved with PG treatment for 4 weeks. Our results confirm the results of previous studies using PG in noncancer populations.17,52,53
Panax ginseng significantly improved QOL scores possibly as a result of its effects on CRF and related symptoms such as pain, sleep disturbances, and anorexia. Prior studies in animal models have shown that PG modulates pro-inflammatory cytokines54-56 owing to the effects of the active ingredients such as ginsenosides and polysaccharides contained in the ginseng extract. Hence, further studies are needed to understand the mechanism of action of PG on CRF. Recent studies suggest the dysregulation of inflammatory cytokines cause and intensify CRF and its related symptoms.11,12,57-59 In our study, 63% of the patients perceived moderate to vast improvement of improvement in CRF with PG treatment.
A recent study found similar results in a related adaptogen, Panax quinquefolius (American ginseng), which differs from PG having a lower ratio of active ginsenosides Rb1 and Rg1. In this multisite, double-blind trial, Barton et al 60 randomly assigned 364 cancer survivors with fatigue to 2000 mg of American ginseng extract or placebo for 8 weeks. The primary endpoint was the score on the general subscale of the Multidimensional Fatigue Symptom Inventory–Short Form at 4 weeks. The results of this study suggested changes from baseline in the patients’ scores on the general subscale of the Multidimensional Fatigue Symptom Inventory–Short Form were 14.4 (SD = 27.1) in the ginseng arm versus 8.2 (SD = 24.8) in the placebo arm at 4 weeks (P = .07). A statistically significant difference was seen at 8 weeks with a change score of 20.0 points for the ginseng group and 10.3 points for the placebo group (P = .003). Greater benefit was reported in patients who were actively receiving cancer treatment than in those who had completed treatment. There were no discernible toxic effects associated with the treatment. Our study also showed preliminary evidence of improvement in CRF levels and trends toward improvement in other common cancer-related symptoms such as drowsiness and sleep disturbances (as assessed via the ESAS) most likely because of its psychostimulant effects similar to those of methylphenidate at 2 and 4 weeks of treatment. 61 However, randomized, placebo-controlled studies are needed to confirm these findings.
There have been few case reports of breast tenderness, postmenopausal vaginal bleeding, and menstrual abnormalities associated with PG use, suggesting PG has estrogenic properties.62-64 Also, in vitro studies to investigate the estrogenic properties of Ginsenosides suggest that Ginsenoside-Rh1 activates estrogen receptor in human breast carcinoma MCF-7 cells. 65 A recent large double-blind trial of Panax quinquefolius in cancer patients enrolled 206/364 (56%) breast cancer patients, however, did not report any estrogen-like side effects. 60 Based on these reports, administration of PG in patients with hormone-receptor positive breast cancer should be done with caution until further long term clinical studies are available to show PG is safe in this patient population. In addition, there were recent reports of potential interaction of PG with targeted agents such as imatinib. 66 One possible mechanism is inhibition PG of cytochrome P450 3A4 enzyme system which is the primary enzyme involved in the metabolism of imatinib. Therefore, caution should be used in administration of PG in the cancer patients on therapy with potential interacting medications and continuous monitoring of liver function tests is important.
The study has several limitations. The primary purpose of our study was to evaluate for the safety and tolerability and therefore the data regarding efficacy on CRF, other symptoms, and QOL should be interpreted with caution. Most of the enrolled patients (Table 2) in the study were receiving cancer therapy. As CRF varies according to the cycle and duration of cancer therapy (ie, chemotherapy or targeted therapy or radiation therapy),67,68 the cancer therapy can be a confounder in this study. Also because this study had no placebo arm, the efficacy of PG may have been due to a placebo effect. 69 However, the lack of adverse events related to the study drug, tolerability of the medication regimen, along with the patients’ report of benefit in improvement of CRF (as assessed by GSE) on using the study drug warrant further randomized, placebo-controlled studies and long-term safety trials. In addition to studies aimed at determining the efficacy of PG, studies to identify the mechanisms by which it improves CRF, such as its role in modulating pro-inflammatory cytokines, are needed.
We conclude that high-dose PG is safe and tolerable and rapidly improves CRF. Our findings also suggest that PG can improve symptoms such as pain, appetite, sleep disturbances, and overall QOL. Randomized, placebo-controlled trials of PG are justified.
Footnotes
Acknowledgements
The authors sincerely thank Mark Blumenthal, founder and Executive Director of the American Botanical Council (ABC) and Editor of HerbalGram, Dr Moshe Frenkel, MD, and Alison Deanna Pawlus, PhD, for their kind contribution in identification of dose and guidance in determination of standardized preparation. We also thank Markeda L. Wade for manuscript review.
Authors’ Note
The Panax ginseng capsules and placebo were supplied by Indena Spa in Milan, Italy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Preparation of this article is supported in part by the MD Anderson Cancer Center support grant CA 016672; American Cancer Society (RSG-11-170-01-PCSM) [SY].
