Abstract
Aim of Study. Gegen Qinlian decoction (GQLD) is an ancient Chinese medicine formula for treating diseases with inner heat. The aim of this study is to investigate the antitumor effect of GQLD in human renal carcinoma cell (RCC) and its possible mechanism. Method. High-performance liquid chromatography was used to identify and quantify active compounds in GQLD. Inhibition of tumor growth was determined by xenograft model. Cell viability on treatment with the decoction was determined by MTT assay; quantitative real-time polymerase chain reaction and immunoblotting were used to determine gene and protein expression; matrix metalloproteinase-2 (MMP-2) activity was detected by gelatin zymography and in vitro enzymatic reaction assay. Results. Thirteen major peaks were detected in the decoction, 8 of which were identified as berberine, baicalin and baicalein, pueranin, daizidin, liquiritin, wogonoside, and wogonin. GQLD exhibited potent inhibition on xenografted expansion of RCC cells. Interestingly, GQLD treatment did not induce cell death to RCC cells, but blocked the neoangiogenesis in xenografted RCC tumor. Particularly, we found that GQLD significantly inhibited MMP-2 in RCC cells, which was involved as a critical factor in avascular growth of RCC. GQLD directly suppressed the enzyme activity of MMP-2. Radix Scutellariae was the major herbal component that contributed to the potent inhibition of MMP-2. Conclusion. The findings of this study provide experimental evidence of the inhibition of expansion and neoangiogenesis of renal carcinoma by Chinese medicine formula GQLD with involvement of MMP-2 suppression.
Introduction
Renal cell carcinoma (RCC) is the most common type of kidney cancer in adults, with the incidence of cases rising every year all over the world. 1 Although RCC is considered to be relatively resistant to radiotherapy and chemotherapy, 2 the prognosis remains good, and the 5-year survival rate remains 60% to 70% if the tumor is confined to the renal parenchyma. However, survival is lowered considerably when metastases of RCC spread, 3 in which tumor cell–induced neovascularization could promote tumor expansion. Currently, a limited approach is applied in the clinical treatment of RCC. It was reported that sorafenib shows some advances on the outlook of RCC (progression-free survival) without improved survival being demonstrated. 4 This leaves room for improving clinical outcomes of RCC treatment and involving complementary and alternative therapies. Chinese medicine is one of the important complementary and alternative medicines prevalent in Chinese societies. It is believed in Chinese medicine that tumor expansion is partially triggered by the movement of internal heat and Xie (pathogen). Hence, scavenging of internal heat and pathogen is considered as one way to treat cancers in the modern clinical practice of Chinese medicine.
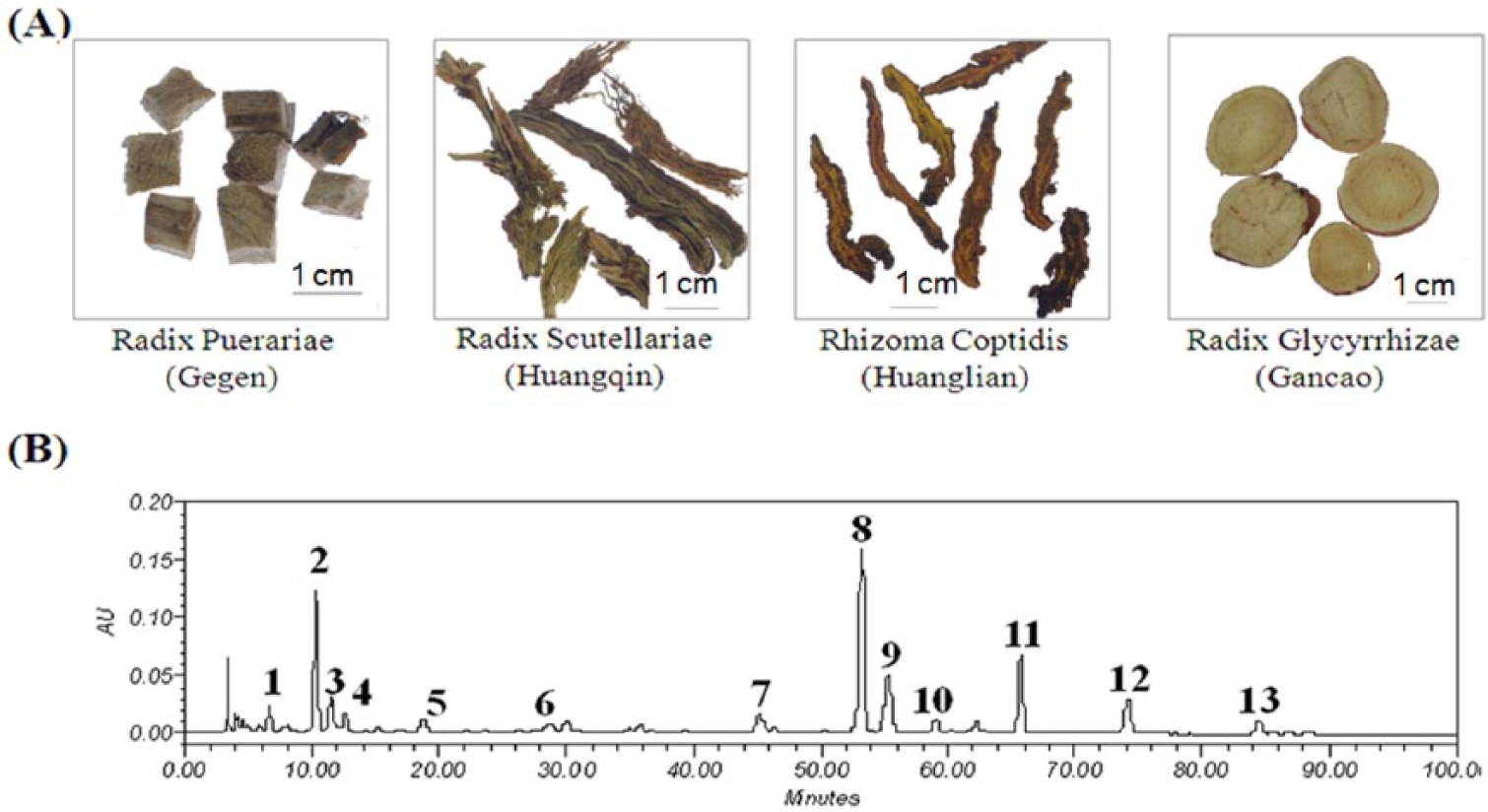
A Chinese medicine formula, Gegen Qinlian Decoction (GQLD), is composed of 4 Chinese medicine herbs: Radix Puerariae (RP; Gegen in Chinese), Rhizoma Coptidis (RC; Huanglian in Chinese), Radix Scutellariae (RS; Huangqin in Chinese), and Radix Glycyrrhizae (RG; Gancao in Chinese; Figure 1A). It was first recorded in Shan-Han-Lun, an ancient Chinese medicine classic, and was applied for clearing interior heat and expelling pathogenic factors in Chinese medicine clinical practice. Identification of the chemical composition of GQLD has been extensively examined. Previous studies used various methods, including high-performance capillary electrophoresis, high-performance liquid chromatography with diode array detection (HPLC-DAD), and liquid chromatography with tandem mass spectrometry, to qualitatively and quantitatively determine the chemical components in GQLD.5 -7 Chemicals including puerarin, daidzein, baicalin, wogonoside, liquiritin, baicalein, wogonin, berberine, palmatine, and jatrorrhizine were identified from the decoction and plasma of rats orally treated with GQLD.8,9 Some other studies also focused on the pharmacological effect of GQLD, reporting its antibacterial, antiviral, antispasmodic, antiarrhythmic, and antihypoxia activities; inhibition on intestine movement; and immuno-modulatory actions. 10 We found in our clinical studies that GQLD alone or in combination with other Chinese medicine could restrict cancer progression. 11 A female patient aged 85, suffering from cholangiocarcinoma, was prescribed GQLD in combination with some minor herbal medicine. She showed progression-free survival for more than 2 years. 12 GQLD was effective in another patient with colon cancers in restricting tumor progression. 13 Clinical observation by us shows that GQLD exhibits potent inhibition on tumor metastasis in patients with renal carcinoma (unpublished data); however, there is no literature published focused on studying the antitumor effect of GQLD in preclinical models or its underlying mechanism. The clinical observations need further experimental investigation as an evidence base.
Quality control of GQLD.
In the present study, we report for the first time the antitumor potential of GQLD on human renal clear cell carcinoma. Chemical analysis examined the major compounds in GQLD, and in total, 8 compounds were identified and quantified. GQLD exhibits potent inhibition on the xenografted expansion of RCC cells without inducing significant cytotoxicity to the human renal carcinoma cells ACHN and Caki-1. Interestingly, we observed the potent inhibition of GQLD on the neoangiogenesis in xenografted tumor of RCC. GQLD reduced the presence of CD31-positive vascular endothelial cells. Particularly, we observed that GQLD could inhibit matrix metalloproteinase-2 (MMP-2) in RCC cells, which is involved in avascular expansion of RCC tumors. In vitro enzymatic reaction has shown that GQLD can directly suppress the enzyme activity of MMP-2. In addition, RS was demonstrated to be the major active component in GQLD inhibition of MMP-2. Our findings provide experimental evidence on the antitumor effect of GQLD.
Materials and Methods
Chemicals and Herbs
Acetonitrile was purchased from Duksan Pure Chemicals Co, Ltd (Korea). Triethylamine, potassium dihydrogen phosphate, and phosphoric acid was from Sigma-Aldrich (St Louis, MO). Berberine, baicalin, and baicalein were purchased from Sigma-Aldrich. Pueranin, daizidin, liquiritin, wogonoside, and wogonin were from Hong Kong Jockey Club Institute of Chinese Medicine (Hong Kong).
RP, RC, RS, and RG were collected from the clinical center of the School of Chinese Medicine, The University of Hong Kong. The herbs were authenticated by Dr Feng Yibin under the guidance of the Pharmacopeia of China (2010) and stored at the School of Chinese Medicine, The University of Hong Kong. The pictures of chopped herbs are shown in Figure 1A. The raw herbs were then washed with distilled water and cut into small pieces before use.
Sample Preparation
To prepare the aqueous extract of GQLD, the 4 crude herbs were mixed and boiled in 20 times of distilled water (w/v) at 100°C for 90 minutes. The solvent was then filtered and collected. This extraction step was repeated 3 times and the filtrate mixed together and then evaporated to dryness, and the powder was stored at −20°C until use. One of the 4 herbs in the decoction was removed to prepare the respective sham control extracts. All samples are dissolved in water–methanol (50:50) for chemical analysis and in water–DMSO (10:90) for biological tests.
Chemical Analysis
HPLC was performed under the following conditions: solid phase: Alltima HP C18 (250 mm × 4.6 mm, 5 µm; Alltech Associates, Inc); mobile phase: acetonitrile (A)–H2O (containing 0.05% KH2PO4, 0.05% triethylamine, adjusting pH 3.0 with phosphoric acid; (B) system was used to separate compounds in GQLD—0 minutes: 12% A; 17 minutes: 12% A; 18 minutes: 14% A; 24 minutes: 14% A; 59 minutes: 25% A; 60 minutes: 27.5% A; 68 minutes: 27.5% A; 70 minutes: 35% A; 95 minutes: 35% A. Wavelength: 280 nm; flow rate: 1.0 mL/min. The analysis was performed at room temperature.
Cells and Cell Culture
Human renal carcinoma cell lines ACHN and Caki-1 were purchased from ATCC. ACHN cells were maintained in modified Eagle medium; Caki-1 were maintained in McCoy’s 5A medium. All the media were supplemented with 10% fetal bovine serum (FBS) and 1% antibiotics, and cells were incubated in 5% CO2 at 37°C.
Cell Viability Assay
Cells were cultured in 96-well cell culture plates. Each well contains 10 000 cells for attachment overnight. Then a series of concentrations of GQLD was added and cells were incubated for 24 or 48 hours. Then 10 µL of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; 5 mg/mL, Sigma) was added to each well 4 hours before the end of treatment and incubated at 37°C. The medium was then discarded and 100 µL DMSO was added to dissolve the crystals with gentle pipetting. The absorbance was read at 575 nm with a Multiskan MS microplate reader (Labsystems, Finland).
Invasion Chamber Assay
To analyze the drug action on invasion of the cells through the extracellular matrix (ECM), the invasion chamber assay was conducted with Millicell Cell Culture Insert (24-well PCF 8.0 µL, Millipore). Briefly, the inserts standing in 24-well cell culture plates were coated with 100 µL 1 mg/mL Matrigel Matrix (BD Falcon) in serum-free DMEM medium and air-dried. A total of 100 µL serum-free medium containing 5 × 104 cells were added to the insert. A total of 0.5 mL of medium containing 10% FBS and indicated concentrations of drug was added to each well of the 24-well plate. This follows incubation in 5% CO2 at 37% for 48 hours. Then the noninvading cells on the upper surface of the Matrigel matrix were removed by cotton swabs. The cells that invaded across the collagen to the lower surface of the membrane were fixed by ice-cold 100% ethanol and stained by 2% Crystal Violet (Sigma Aldrich). Photographs of the stained invaded cells (5 random fields/culture) were taken under an inverted microscope at 400× and the mean number of cells of the 5 fields was recorded.
Gelatin Zymography
Gelatin zymography was performed to determine the activity of MMP-2. Briefly, medium was collected and mixed with 3× loading buffer. Protein in medium was then separated in 10% SDS-page gel containing 1 mg/mL gelatin. After running, the gel was incubated in 2.5% triton-X in deionized water for renaturing with gentle agitation for 30 minutes at room temperature. Then the gel was incubated in developing buffer (50 mM Tris–HCl, 0.2 M NaCl, 5 mM CaCl2, 0.02% Brij35) overnight with gentle shaking. The gel was stained Coomassie Blue R-250 for 30 minutes and then washed. The gel was visualized under a chemiluminescence imaging system (Bio-Rad) and the figure was captured.
In Vitro MMP-2 Activity Assay
The MMP-2 activity dynamics were evaluated using MMP-2 colormetric drug discovery kits (Enzo) under the manufacturer’s instruction. In brief, the human recombinant MMP-2 was mixed with testing drugs with indicated concentrations. The mixture was incubated at 37°C for 45 minutes and then the substrate was added. The plate was immediately read at 405 nm by Multiskan MS microplate reader (Labsystems). Continuous recording was made at 2-minute intervals for 10 minutes. The dynamic changes of MMP-2 were determined by the slope of line fit to liner portion. The experiment was conducted in duplicate.
Immunoblotting
Cells were lysed with radioimmunoprecipitation assay (RIPA) buffer with complex cocktailed proteinase inhibitor (Roche) and phosphatase inhibitor (1 mM Na3VO4 and 1 mM NaF) on ice for 30 minutes followed by centrifugation at 14 000 rpm at 4°C for 15 minutes. The supernatant was transferred and protein concentration was determined by BSA assay (Bio-Rad). Equal yield of protein was separated on SDS-PAGE and transferred onto a polyvinylidene fluoride membrane (PVDF, Bio-Rad). The membrane was then blocked in buffer containing 5% BSA, Tris (10 mmol/L, pH 7.4), NaCl (150 mmol/L), and Tween 20 (1%) at room temperature for 1 hour with gentle shaking. The membrane was then incubated with primary antibodies at 4°C overnight followed by incubation with appropriate secondary antibody (Abcam) at room temperature for 1 hour. The immunoreactivites were detected using ECL advanced kit (GE Healthcare) and visualized using a chemiluminenescence imaging system (Bio-Rad).
Xenograft Model
Six-week-old male BALB/c nu/nu athymic nude mice received 1 × 106 ACHN cells injection subcutaneously at the left side of the waist. One week after injection, mice were randomized into 2 groups (n = 4). The control group received the same volume of PBS, while the other group received 150 mg/kg GQLD via gavage daily for 3 weeks. Tumor volume and body weight were measured every week. At the end of the experiment, the mice were sacrificed with an overdose of phenobarbital (200 mg/kg) and the tumor was dissected out. All animals received human care and study protocols complied with the guidelines of the Laboratory Animal Centre of The University of Hong Kong and were approved by the Committee on the Use of Live Animals in Teaching and Research (CULATR) of the University of Hong Kong. Moreover, animals were processed according to the suggested international ethical guidelines for the care of laboratory animals throughout the experiments.
Statistical Analysis
Data obtained were analyzed by Student’s t test. Data were expressed as mean ± SD of the mean, and values of P < .05 were considered to be statistically significant.
Results
Phytochemical Analysis
The chemical profile of GQLD was established with HPLC-DAD using conditions in literature with some modification. 6 To optimize the condition, we compared different elution rates and detection wavelengths, and we found the current separation and detection condition provides adequate information for the quality control of GQLD. A total of 13 major chromatographic peaks were obtained (Figure 1B), and by comparison with standard chemicals, 8 peaks were identified. Quantification of each identified peak was also conducted, as shown in Table 1.
Identification and Quantification of Compounds in Gegen Qinlian Decoction.
GQLD Inhibits Xenografted RCC Expansion Without Inducing Cell Death
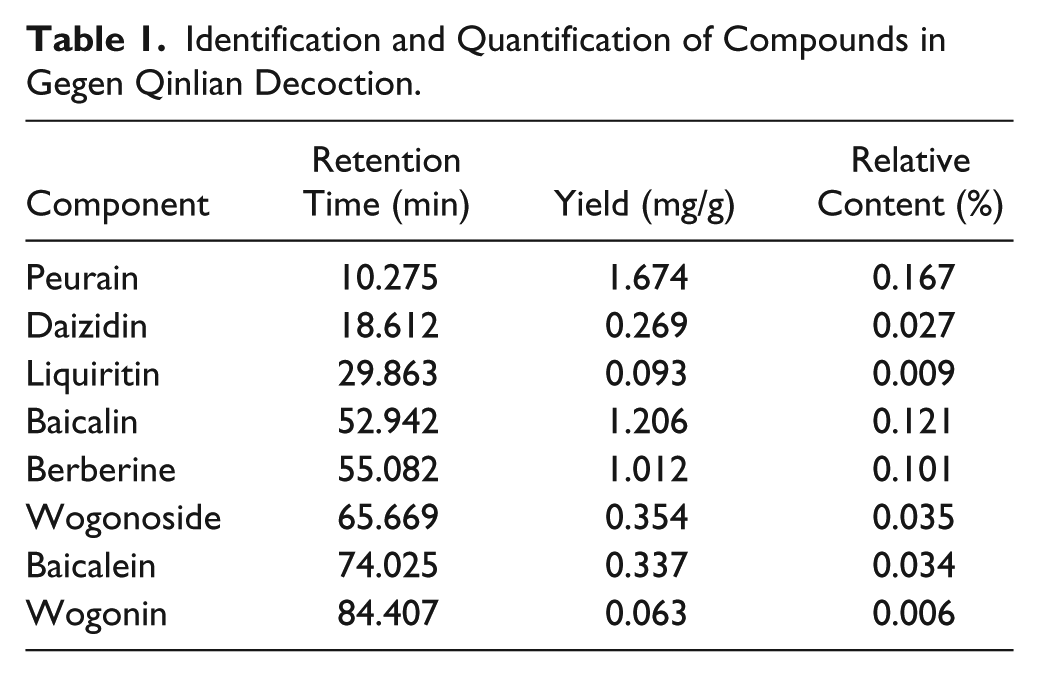
To examine the antitumor effect of GQLD on human RCC, we established a xenograft model with nude mice. No significant body weight loss or abnormal gain could be found in GQLD-treated mice, indicating that this decoction did not bring observable side effects to the mice (Figure 2A). Furthermore, the growth of xenografted tumor was retarded by 150 mg/kg administration of GQLD (Figure 2B). At the end of the study, mice were sacrificed and the tumor dissected. It was observed that the tumor size was significantly reduced in the GQLD-treated group (Figure 2C). These observations confirmed that GQLD may exhibit remarkable antitumor effect on xenografted RCC. To further our knowledge, we examined if GQLD could directly induce death of RCC cells. The cell viability was determined in renal carcinoma cells with GQLD treatment. Metastatic renal carcinoma cell lines ACHN and Caki-1 were used in this study. No remarkable cell death could be observed in the 2 cell lines with exposure to GQLD for 24 or 48 hours (Figure 2D), indicating no direct inhibition of GQLD on tumor cell growth and proliferation at up to 1 g/mL. Immunoblotting showed no potent increase of cleavage of Poly (ADP-ribose) polymerase (PARP), the cell death marker, in cells with GQLD intervention (Figure 2E). Our findings indicate that GQLD exhibits potent inhibitory effect on RCC without inducing cell death at the dose lower than 1 g/mL.
GQLD inhibits tumor expansion without inducing tumor cell death at the dose of up to 1 g/mL in vitro.
GQLD Blocks Neoangiogenesis of Tumor With Involvement of MMP-2 Downregulation
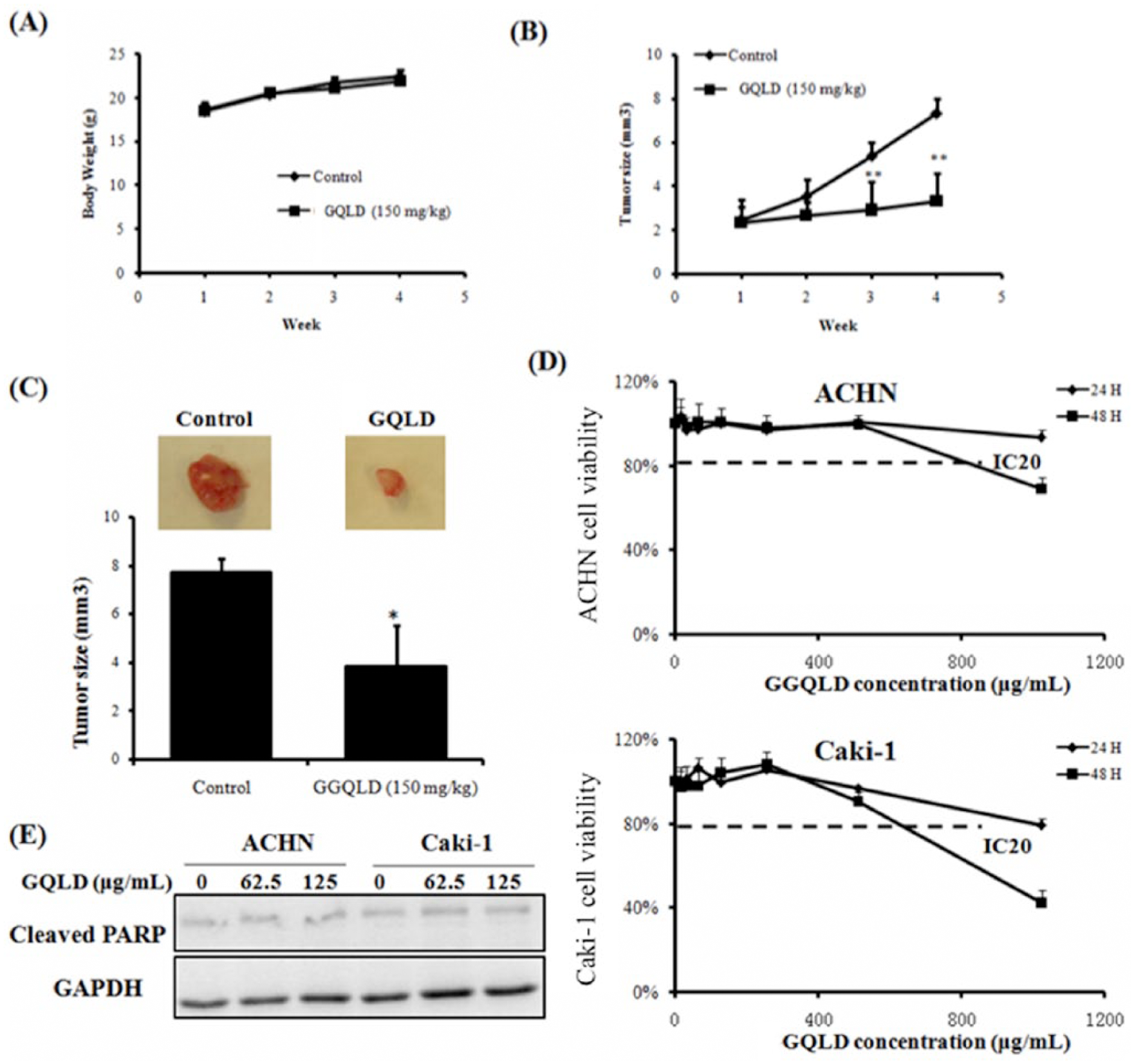
To examine the possible mechanism that underlies tumor inhibition by GQLD, we examined if GQLD could reduce the tumor expansion by blocking abnormal blood vessel formation in xenografted tumor. By immunostaining with CD31, the endothelial cell marker that labels blood vessel cells, we observed potent neoangiogenesis in xenografted tumor of RCC. Interestingly, treatment of GQLD reduced blood vessel formation in RCC tumor. The size and density of tumor-induced blood vessels were both potently reduced by GQLD treatment (Figure 3A). This evidenced that GQLD could retard the neoangiogenesis in RCC xenograft. Previous studies have reported the clinical significance of MMP-2 in the prognosis of human RCC, in which MMP-2 may promote the RCC expansion and angiogenesis and lead to poor prognosis in patients.14,15 To further elucidate the action of GQLD, we examined if expression and activity of MMP-2 could be suppressed by GQLD. To examine if GQLD could suppress the expression of MMP-2 in ACHN and Caki-1 cells, we used quantitative real-time polymerase chain reaction (PCR) and immunoblotting to determine the mRNA and protein expressions, respectively. The results showed that MMP-2 mRNA transcript could be downregulated on GQLD treatment in a dose-dependent manner, though no statistical significance in differential expression of MMP-2 mRNA could be found (Figure 3B). This result was further confirmed by immunoblotting assay, in which we found the protein expression of pro-MMP-2 was significantly reduced by GQLD treatment (Figure 3C). We then further examined if GQLD could inhibit the gelatinase activity of MMP-2, which is reported to be critical for tumor cells digesting ECM to facilitate the neoangiogenesis in xenografted tumor. Interestingly, dose-dependent suppression of GQLD on MMP-2 activity could be observed in gelatin zymography assay (Figure 3D). In both ACHN and Caki-1 cells, GQLD treatment could decrease the gelatinase activity of MMP-2 as well as its expression. Our findings indicate that suppression of MMP-2 may be the possible mechanism of the antimetastatic action of GQLD.
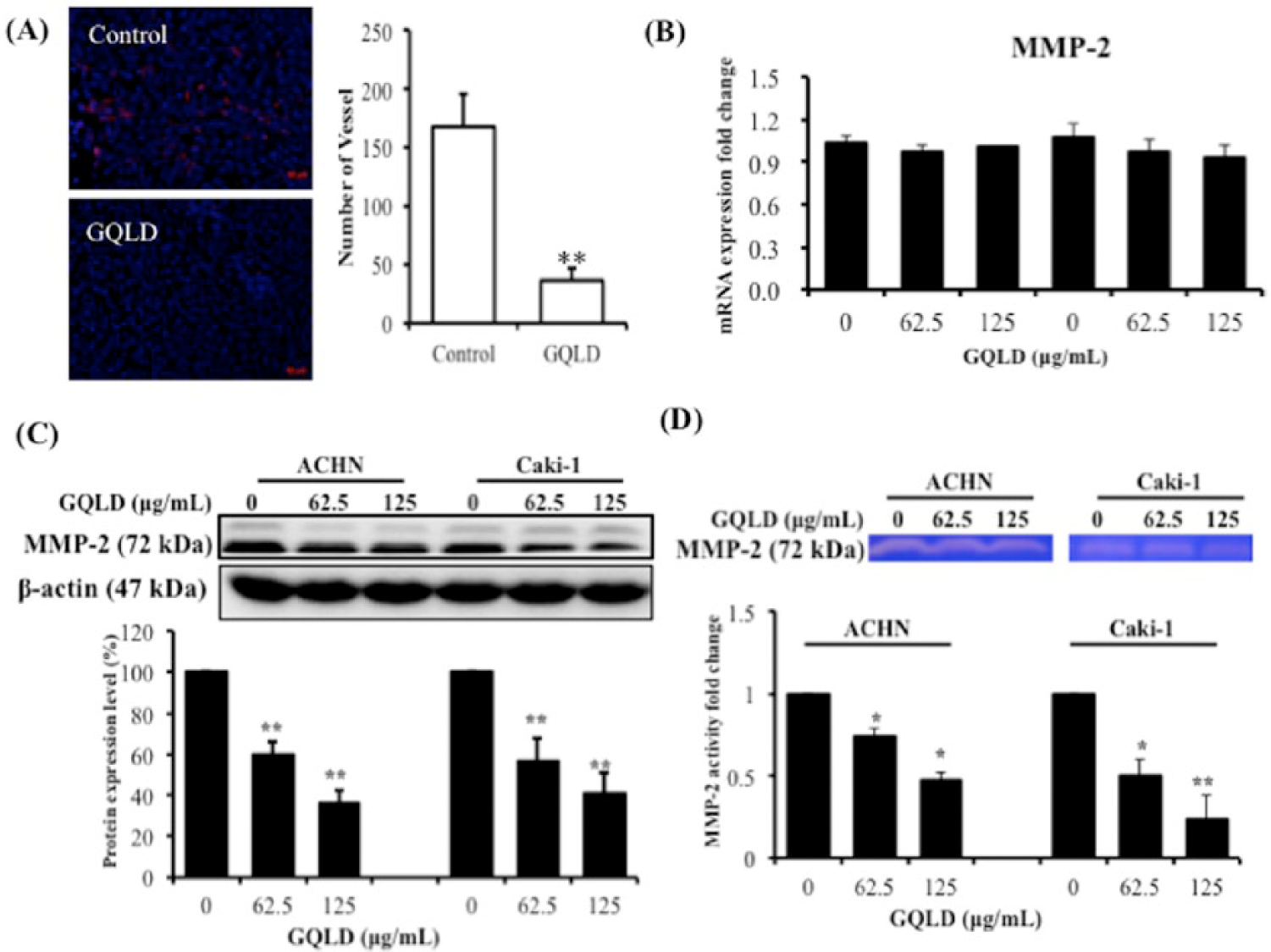
GQLD suppresses neoangiogenesis of xenografted RCC tumor.
GQLD Is an Enzyme Inhibitor of MMP-2
We further observed if GQLD could directly suppress the enzyme activity of MMP-2 by conducting in vitro enzymatic reaction assay. Using thiopeptide as the substrate, we observed the rate at which recombinant MMP-2 digests the substrate was potently reduced by the presence of GQLD (Figure 4A). This may indicate that GQLD could be a direct MMP-2 inhibitor in vitro. Furthermore, since MMP-2 serves as an important factor in cancer cell movement by digesting the ECM, we further confirmed the action of GQLD on MMP-2 in RCC cells by the invasion chamber assay. Matrigel matrix was used to mimic ECM in tumor microenvironment, and we observed that movement of ACHN and Caki-1 cells through Matrigel was suppressed in a dose-dependent manner (Figure 4B). These data confirmed that the presence of GQLD could reduce the MMP-2-driven ECM degradation and RCC cell movement.
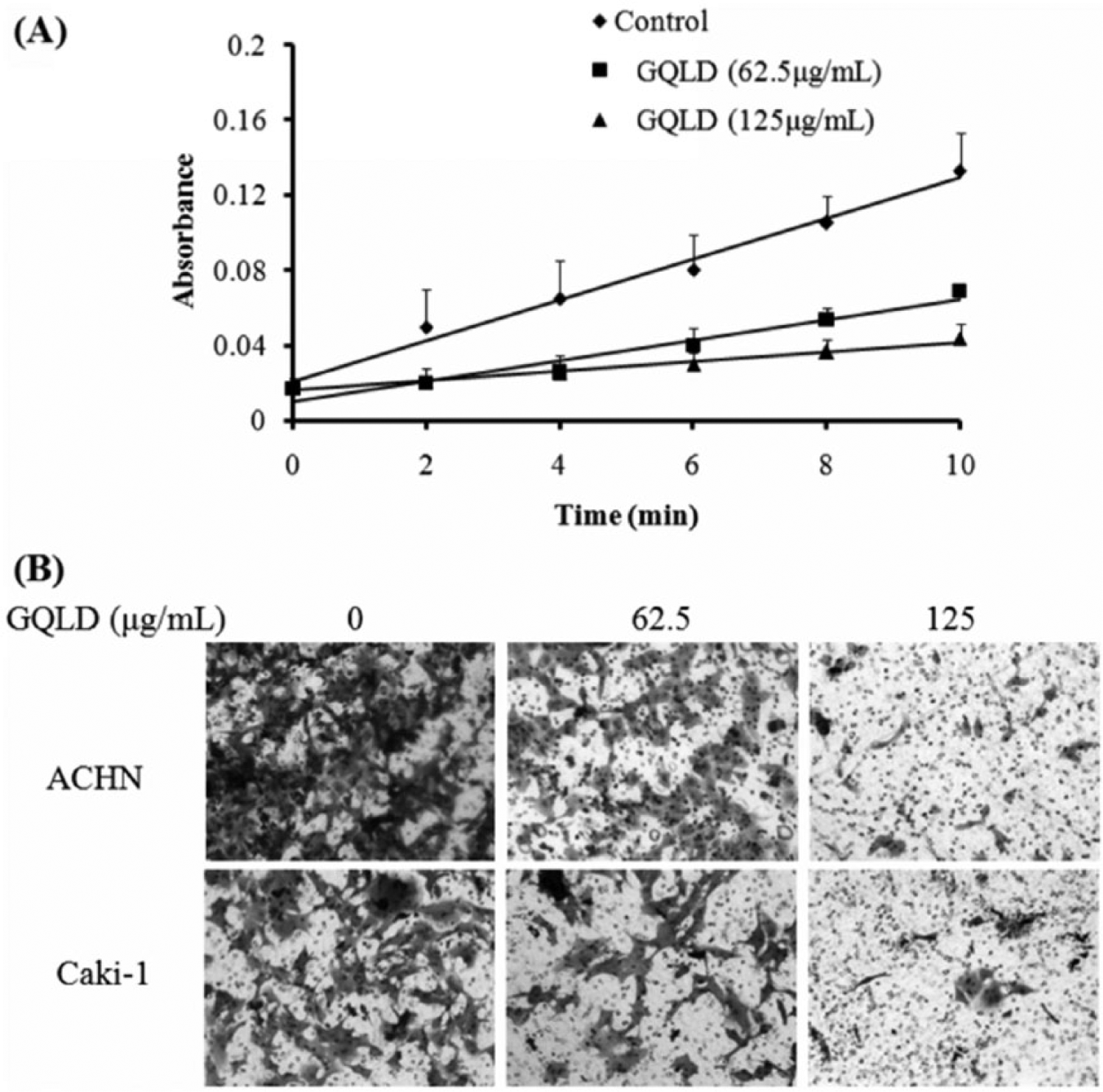
GQLD is a direct inhibitor of MMP-2.
Radix Scutellariae Is the Major Active Component in GQLD
To further identify the active component in GQLD, enzyme activity of MMP-2 was examined as a criterion for screening the most active herbal component in this formula. We observed that the water extract of RS exhibited potent inhibition on MMP-2 activity, while extracts from other herbs had minimal effect (Figure 5A). This may indicate that RS is the major active component in this formula. To further validate the role of RS in the antitumor potential of GQLD, we removed RS from the formula and prepared a sham control containing only RC, RG, and RP. This decoction could not retard the enzymatic reaction of MMP-2 in vitro (Figure 5B). Furthermore, we observed that the aqueous extract of RS could particularly downregulate MMP-2 activity in renal carcinoma cells, which is consistent with the action of GQLD. Decoction without RS has no inhibition on MMP-2 (Figure 5C). Our results indicate that RS is the major active component in GQLD.
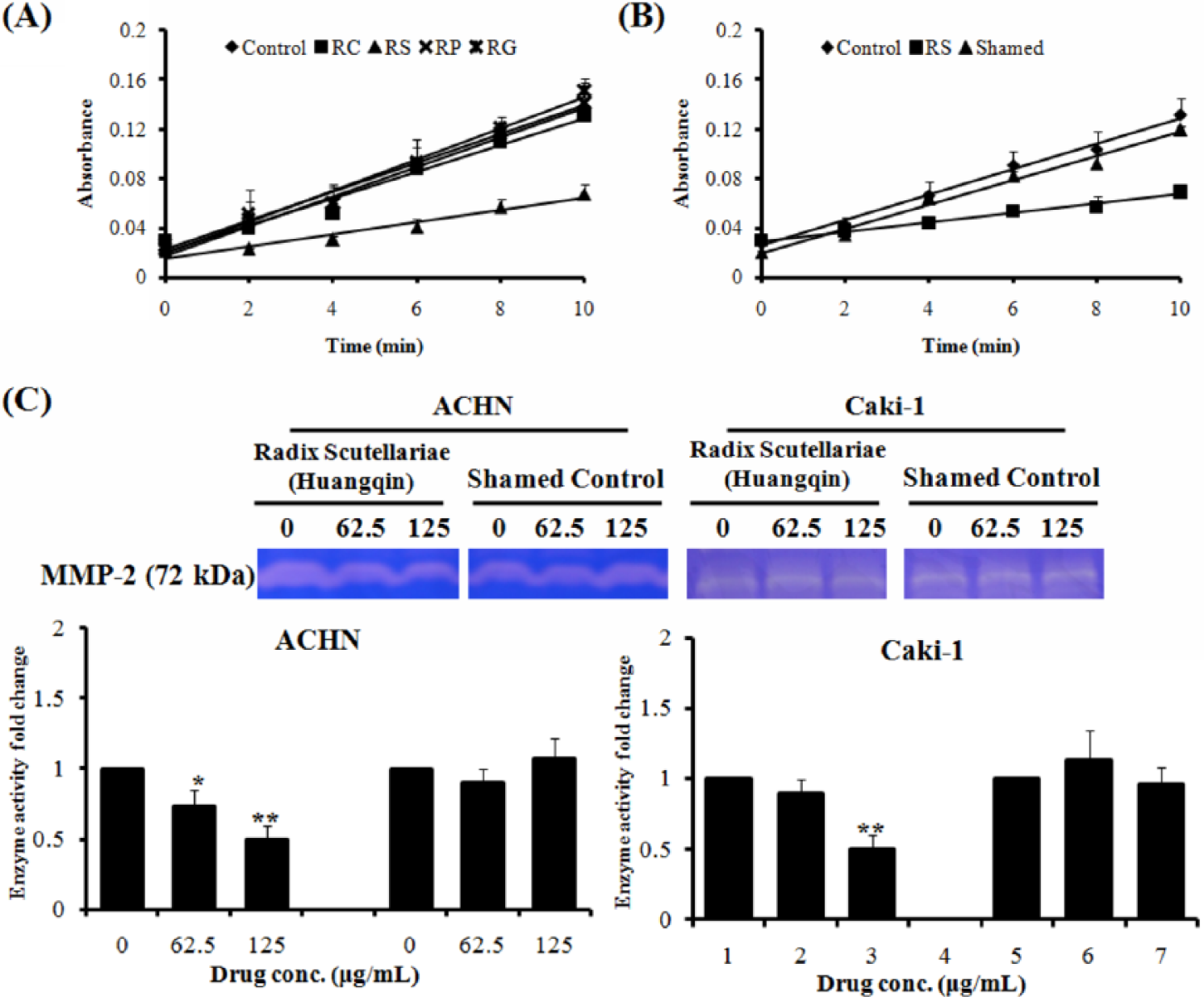
Radix Scutellariae (Huangqin) is the major active component herb in GQLD.
Discussion
GQLD is composed of 4 Chinese medicine herbs including CR, RS, RG, and RP. It was traditionally used in Chinese medicine clinical practice to clear Xie (pathogen) and to scavenge internal heat. Modern clinical applications of GQLD exhibit its potential as an antipuretic and antimicrobic agent. 16 An earlier study reported the use of GQLD in attenuating enteritis induced by radiotherapy in patients with cancers, 17 indicating the potential use of GQLD in reducing the side effects of radiotherapy. However, there is no study reporting any beneficial action of GQLD in preventing tumor expansion. In this study, we report for the first time that GQLD significantly suppressed the expansion and neoangiogenesis of human renal carcinoma cells without exhibiting potent toxicity. Oral administration of GQLD is capable of suppressing growth of RCC xenograft in mice by reducing the abnormal formation of blood vessels. GQLD inhibited MMP-2 expression and activity, which is the critical factor that contributes to the tumor neovascularization and avascular expansion. Our observation reveals that GQLD may have potential to be one of the complementary treatments in human RCC therapy.
Degradation of ECM protein by cancer cells is required during tumor invasion, and special proteinases, the matrix metalloproteinases (MMPs), are necessary for tumor metastasis. 18 An MMP family containing 24 human proteolytic enzymes has been identified that could remodel all of ECM components. 19 Among the MMPs members, the gelatinases MMP-2 are considered as critical molecules involved in the degradation of ECM by tumor cells and could promote tumor cell invasion and metastasis. The activity and expression of MMP-2 is upregulated in most of the human cancers. 20 Interestingly, the role of tumor cell–derived MMP-2 in neoangiogenesis was extensively examined in many studies. A previous study with clinical samples has revealed that the higher level of MMP-2 that carcinoma cells presented, the greater angiogenesis would be. Expression of MMP-2 appears to confer, in this study, vascular endothelial proliferation. 21 It was found that deficiency of MMP-2 reduced the angiogenesis of tumor in mice, and the tumor progression in MMP-2-deficient mice was retarded, which may indicate that MMP-2 inhibitor could be a potential anticancer candidate. 22 This was supported by another study, which claims that MMP-2 could not only degrade the ECM to allow vascular tube formation but also allow release of ECM-bound pro-angiogenic factors such as bFGF, VEGF, and TGF-β to facilitate tumor angiogenesis. Furthermore, MMP-2 could initiate integrin signaling to allow proliferation of vascular endothelial cells. 23 A recent study already showed that an MMP-2 inhibitor could as well block angiogenesis in cancer, 24 which further showed that MMP-2 suppression could result in abrogation of angiogenesis. In human renal carcinoma, MMP-2 exhibits high expressing profile with strong enzymatic activity, and is correlated to the expansion of RCC cells. 25 Therefore, a potent MMP-2 inhibitor may be suitable to retard RCC expansion and could potentially benefit the clinical outcome of RCC treatment. Our study reveals that GQLD may act as an MMP-2 inhibitor to reduce RCC tumor expansion and angiogenesis. As a complementary and alternative approach in cancer treatment, the use of this decoction may slow tumor progression to allow time for other therapeutics to kill RCC cells.
We noticed that the major active component of GQLD in inhibiting RCC expansion and neoangiogenesis is RS. A previous study showed that the aqueous extract of RS could significantly downregulate the expression of Cyclin D1 and suppress the motility of human lung cells, which is correlated with the decreased MMP-2 activity on RS extract treatment. 26 Herbal formula Sihoga-Yonggol-Moryo-Tang containing RS was found to present anti-migration and anti-invasive effect to human hepatocellular carcinoma (HCC). With potent downregulation of MMP-2/9 activities on treatment of the decoction, HCC cells exhibit decreased motility and invasion in a dose-dependent manner. 27 In our study, the water extract of RS is capable of suppressing the MMP-2 enzyme activity in vitro, which is comparable to GQLD. Removing RS from GQLD abolishes its inhibitory effect on MMP-2 activity in vitro as well as renal carcinoma cells. Baicalein and its glycoside baicalin, the major compounds in RS, were intensively reported for their antimigration and antiinvasion properties. Previous studies show that baicalein and baicalin are capable of inhibiting MMP-2 in human hepatoma cells, 28 breast cancer cells, 29 and endothelial cells. 30 Suppression of MMP-2 contributes to the antiinvasiveness of baicalein and baicalin. In addition, a previous study reported that baicalein could increase cell adhesion and abolish cell movement through upregulating Erzin expression. Silencing Erzin expression attenuates baicalein’s antimigration action in human lung cancer cells. 31 In our study, we found that extracts of GQLD and particularly RS inhibit MMP-2 in RCC cells. Although the expression of MMP-2 was also reduced by GQLD and RS treatment, the property of RS-containing decoction as an MMP-2 inhibitor predominantly contributes to the antitumor effect of the decoction in RCC. It was noticed that though RS plays a major role in inhibiting MMP-2, while other herbs in the formula have no direct effect on MMP-2 expression and activity, suppression of MMP-2 in RS-treated RCC cells is not as potent as that in GQLD-treated RCC cells. This may indicate that other herbs in the formula could potentiate RS treatment. The exact mechanism needs further investigation. The overall scheme of the regulation of GQLD on renal carcinoma metastasis is shown in Figure 6.
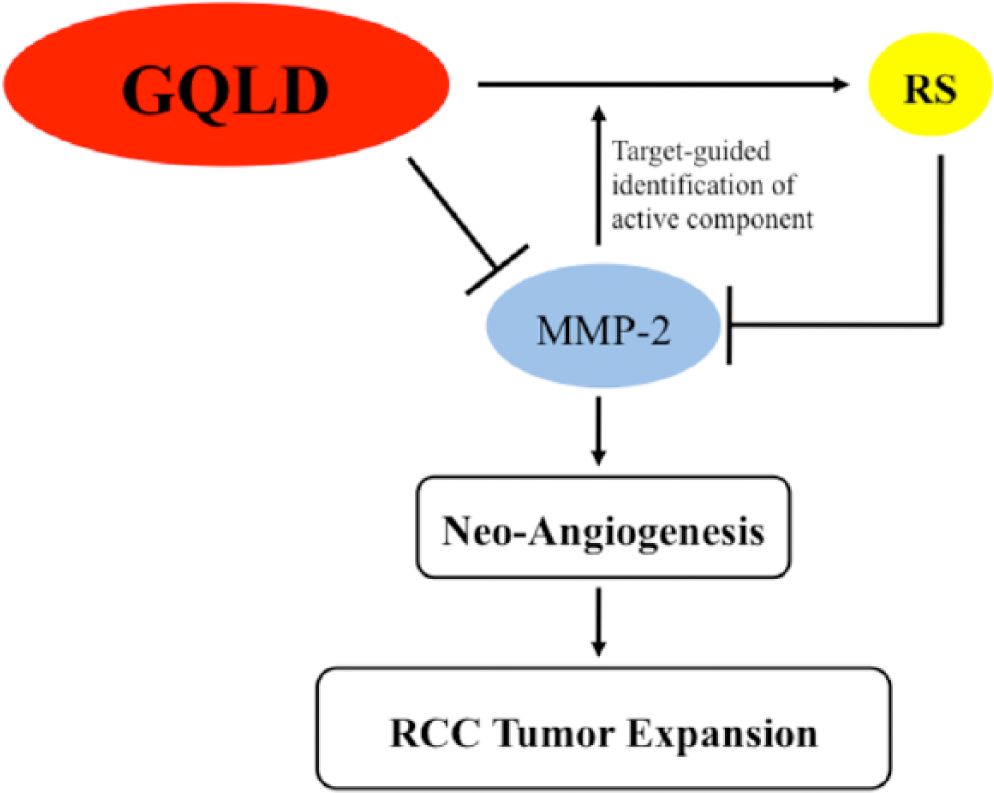
The overall regulatory network of GQLD on renal carcinoma expansion.
Conclusion
In conclusion, we reported for the first time that GQLD may suppress human renal carcinoma cell expansion and neoangiogenesis. The phytochemical analysis showed that GQLD contains 13 major compounds, 8 of which have been identified as berberine, baicalin and baicalein, pueranin, daizidin, liquiritin, wogonoside, and wogonin. GQLD exhibits potent inhibition on the xenografted growth of RCC cells in mice, but presents minimal effect on the viability of RCC in vitro. Treatment of GQLD could reduce the blood vessel formation in xenografted tumor of RCC, as evidenced by the decreased presence of CD31-positive vascular endothelial cells in tumor. MMP-2 inhibition was observed in GQLD, and the decoction may act as a particular inhibitor of MMP-2 to block its enzyme activity. In addition, RS is shown as the major active component in GQLD in inhibiting MMP-2. Our findings provide the first experimental evidence on the antitumor effect of GQLD on renal carcinoma.
Footnotes
Acknowledgements
We are grateful to the support of Professors Yung-Chi Cheng, Chi-Ming Che, Yao Tong, Sai-Wah Tsao, and Allan SY Lau. The authors would like to express thanks to Ms Meifen Zhu, Ms Oi-Yee Chow, Ms Cindy Lee, Mr Keith Wong, and Mr Freddy Tsang for their technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financially supported by Wong’s donation to Modern Oncology of Chinese Medicine (Project Code: 20006267), The Research Grants Council (RGC) of Hong Kong SAR of China (Project Code: 10500362), and The University of Hong Kong (Project Code: 104002320 [to Dr Feng Yibin] and Project Code: 104002587 [to Dr Wang Ning]). We would like to thank The Hong Kong Jockey Club Charities Trust (HKJCCT) for funding the project of “R&D Laboratory for Testing of Chinese Medicines.”
