Abstract
Cancer is the final outcome of a plethora of events. Targeting the proliferation or inducing programmed cell death in a proliferating population is a major standpoint in the cancer therapy. However, proliferation is regulated by several cellular and immunologic processes. This study reports the inhibition of proliferation by augmenting immune surveillance, silencing acute inflammation, and inducing p53-mediated apoptosis of skin cancer by 3 promising medicinal extracts. We used the well-characterized model for experimental skin carcinogenesis in mice for 32 weeks to study the chemopreventive effect of the methanolic extracts of Trigonella foenumgraecum, Eclipta alba, and Calendula officinalis. All 3 extracts reduced the number, incidence, and multiplicity of tumors, which was confirmed by the pathologic studies that showed regressed tumors. There was a significant reduction in the PCNA+ nuclei in all treatment groups 32 weeks after the initiation. Mechanistic studies revealed that proliferative population in tumors is diminished by the restoration of the endogenous antioxidant defense, inhibition of the stress-related signal-transducing element NFκB, reduction of inflammation, enhancement of immunosurveillance of the genetically mutated cells, along with silencing of the cell cycle progression signals. Finally, all 3 medicinal extracts induced stable expression of p53 within the tumors, confirmed by the CFDA-Cy3 apoptosis assay. Results of our study confirm that these extracts not only limit the rate of proliferation by inhibition of the processes integral to cancer development but also induce stable cytoplasmic expression of p53-mediated apoptosis, leading to fewer and regressed tumors in mice.
Introduction
A mass of diligent efforts has been marshaled to preclude, regress and/or reverse the pathologic manifestation of carcinogenesis. However, cancer still persists as the toughest challenge that humankind is facing.
The present-day therapies are basically directed to stop/reverse proliferation or induce cellular death within malignant lesions in clinical practice and preclinical testing. 1 The major weakness encountered by these therapies is that they target only the proliferating population, with minimal effect on the systemic/cellular processes that lead to survival of altered cells and allow the progression of the cell cycle resulting in neoplasms. Furthermore, the burden of side effects associated with the existing therapies makes their use further questionable. 2
Carcinogenesis in the skin can be induced by the application of a subthreshold dose of a carcinogen (initiation phase), followed by repetitive treatment with a noncarcinogenic tumor promoter. The initiation phase requires only a single application of either a direct-acting carcinogen or a procarcinogen (which has to be metabolized before being active) and is essentially irreversible; the promotion phase, however, is initially reversible but later becomes irreversible. The window provided by the tumor promotion phase where it is reversible can be harnessed to stop the uncontrolled growth. Not only does this system provide an important model for studying carcinogenesis and bioassaying carcinogenic agents, but it is also one of the best systems for investigating the effects of inhibitors of chemical carcinogenesis. 3
The immunosurveillance of the altered cells and the cellular processes leading to the progression of the cell cycle 4 still remain as selective targets for chemopreventive studies in preclinical or clinical settings. It is well known that croton oil is rich in TPA, 5 which leads to immunosupression 6 mediated by the migration of Langerhans cells (LCs; antigen-presenting dendritic cells that stimulate/regulate immune responses in skin) 7 along with massive inflammation and reactive oxygen species in murine skin, which is independent of the number of times that it is applied. 8 However, the repeated application of promoters after a genetic event forces progression of the cell cycle, 3 and this stage can be segregated into 2 phases biochemically. 9 It seems that failure of the immune reactions toward the altered cells after genetic events allows inflammation and reactive oxygen species to progress the cell cycle.
The inflammatory signals are transduced in the cells via regulated cellular stress-mediating elements. The major protein of this chain is NFκB, 10 which may lead to the induction of the several inflammatory enzymes, such as COX-2. Their role has been already proved in the development of malignancy. 11 One of the various cellular processes that regulate the induction and execution of the chemical-induced carcinogenesis is the activation of protein kinases, including protein kinase C, which is capable of the stimulation of several cell cycle progression molecules, such as p38 MAPK. 12 p53 is the tumor suppressor gene that is downregulated in neoplastic cells; however, induction of p53 expression following chemopreventive strategies represents one of the plausible approaches toward identification of potential chemopreventive agents. 13
A range of epidemiologic studies have highlighted that antidotal strategies can alter oncogenic phenotypes at different sites. Over a period of 2 decades, we have marshaled a range of literature in the direction in a preclinical setting both in vivo and in vitro. Our studies have demonstrated chemopreventive effects of natural agents in skin carcinogenesis, hepatocarcinogenesis, and renal carcinogenesis and colon cancer.14 -19
We have selected 3 plants—namely, Trigonella foenumgraecum, Eclipta alba, and Calendula officinalis—with established medicinal properties, including anticancer properties. The seeds of T foenumgraecum have been demonstrated to inhibit mammary hyperplasia induced by DMBA by induction of apoptosis 20 and reduction of tumor cell growth in the Ehrlich ascites carcinoma model by immune augmentation. 21 Recent studies have also highlighted the anticancer potential and anti-multidrug resistance properties of hydroalcoholic extract of the seeds of E alba.22,23 Similarly, anti-inflammatory, antitumor-promoting activities of the crude extract and the constituents of C officinalis have been reported by two independent studies.24,25 These extracts were standardized for their chemical constitution for their potential chemopreventive efficacy against skin carcinogenesis induced by DMBA and croton oil in mice. T foenumgraecum is particularly rich in saponins (eg, diosgenin) and alkaloids (eg, trigonelline), 26 while E alba contains coumestans such as wedelolactone, polypeptides, flavonoids, and triterpenes. 27 C officinalis mainly contains triterpenoid esters, essential oils, saponins, and flavanol glycosides. 28 Keeping in view these properties, we decided to test these abilities in a murine model for skin carcinogenesis.
The outcomes of the study were rated as decrease in the incidence of tumor, decrease in number of tumors, enhancement of immune surveillance of the altered cells, suppression of inflammation, and silencing of the cellular processes responsible for cell cycle progression and induction apoptosis in established tumors.
Materials and Methods
Experimental Animals and Diet
Pathogen-free female Swiss albino mice, initially weighing 20 to 25 g, were obtained from Central Animal House governed by the Institutional Animal Ethics Committee, Jamia Hamdard, which is an accredited CSCPEA regulating the use of laboratory animals in India. The animals were acclimatized to standard laboratory conditions, including 22°C ± 2°C temperature, 30% to 50% relative humidity, and a dark:light cycle of 12:12 hours in solid bottom polycarbonate cages (3 animals per cage) with ultraviolet sterilized husk bedding for 1 week before beginning of the study. The animals were fed with a well-defined Constant Nutrition formula pulverized diet (Hindustan Feed) and provided with drinking water ad libitum. The experimental protocol was approved by the ethics committee (AIC) regulating the use of laboratory animals in Jamia Hamdard.
Plant Materials
Dry whole plants were purchased from authorized vendors of botanical material to Jamia Hamdard. These herbs had been collected in northern range in India in August season. Then the plant material was identified by renowned botanist Professor Mohd Iqbal from the Department of Botany, Jamia Hamdard, and the vouchers were collected. Subsequent to identification, the required plant parts were air-dried and milled into fine particles and simultaneously kept in the Hamdard central herbarium:
E alba (voucher EA/JH/SC/TOX/100/2008)
T foenumgraecum (voucher TFG/JH/SC/TOX/101/2008)
C officinalis (voucher CO/JH/SC/TOX/102/2008)
E alba leaves, seeds of Trigonella, and flowers of Calendula plant were used for extraction.
Preparation of Extracts
The extraction procedure was used as described in Didry et al 29 under strict supervision of a pharmacognosy expert. The powdered material (200 g) was subjected to extraction with solvents of increasing polarity—namely, hexane, petroleum ether (60°C-80°C), diethyl ether, ethyl acetate, acetone, and, finally, methanol. The methanolic extract was used for the experiments since the rest of them showed comparatively lower extractive values. The extract thus obtained was cooled at room temperature, evaporated to dryness, and stored at 4°C. All 3 extracts were of slight brown color, and appropriate seals were applied before preserving the extracts.
Serial Standardization of the Plant Extracts by Gas Chromatograph Coupled With Mass Spectrometer
Gas chromatography–mass spectrometry (GC-MS) analysis of the extracts was performed with a Perkin Elmer GC Claurus 500 system and gas chromatograph interfaced to a mass spectrometer equipped with an Elite-1 fused silica capillary column (30 m × 0.25 mm ID ×1 ìMdf, composed of 100% dimethyl poly siloxane). For GC-MS detection, an electron ionization system with ionization energy of 70 eV was used. Helium gas (99.999%) was used as the carrier gas at a constant flow rate of 1 mL/min, and an injection volume of 2 µl was employed (split ratio of 10:1). Injector temperature was 270°C; ion-source was temperature 280°C. The oven temperature was programmed from 110°C (isothermal for 2 minutes), with an increase of 10°C/min to 200°C, then 5°C/min to 280°C, ending with a 9-minute isothermal at 280°C. Mass spectra were taken at 70 eV: a scan interval of 0.5 seconds and fragments. Total gas chromatography running time was 30 minutes. The relative percentage mount of each component was calculated by comparing its average peak area to the total areas. Software adopted to handle mass spectra and chromatograms was Turbo Mass Version 5.2.0. The results were interpreted from Wiley Library for retention time and molecular mass.
GC-MS of the methanolic extract of T foenumgraecum showed 17 peaks (Figure 1A) over a period of 30 minutes per experimental protocol. The peak number 9 as confirmed from the Wiley Library was a natural alkaloid trigonelline (0.31%) with a retention time of 19.32 minutes and a molecular mass of 137.14 g/mol. In similar chromatographic conditions, the methanolic extract of Eclipta alba showed 72 different peaks (Figure 1B); a peak number 21 was conformed to be natural coumestan wedelolactone (1.61%) with a retention time of 15.78 minutes and a molecular mass of 314.24 g/mol. The third extract, C officinalis, showed 67 different peaks (Figure 1C), and peak number 4 was confirmed to be α-cadinol (19.22%) with a retention time of 9.08 minutes and a molecular mass of 222.37 g/mol. These characterized methanolic medicinal extracts were designated as TF (T foenumgraecum), EA (E alba), and CA (C officinalis).

Gas chromatography–mass spectrometry analysis of the methanolic extracts of the 3 medicinal herbs. (A) Chromatogram of Trigonella foenumgraecum with 17 different peaks. Peak number 9 pertains to trigonelline (retention time = 19.32 min). Chromatograms of (B) Eclipta alba (72 peaks) and (C) Calendula officinalis (67 peaks), with identified marker compounds as wedelolactone (peak number 21 and retention time = 15.78 min) and α-cadinol (peak number 4 and retention time = 9.08 min), respectively.
Experimental Drug Preparation
All 3 extracts were freshly mixed with acetone. The dosages of the drugs were based on previous data in mice. TF was administered at 10 mg/kg while the dose for EA was 7 mg/kg and 10 mg/kg for CA. The drug preparations were stored in opaque 20-mL containers at 4°C and usually used for 1 week only.
Induction of Skin Carcinogenesis in Mice and Medicinal Extract Treatment
The potential chemopreventive role of topically applied medicinal extracts was investigated with a well-established and previously published DMBA-initiated and croton oil–promoted 2-stage skin carcinogenesis model in mice 30 with a slight modification. Following an acclimatization period of 1 week with standard diet, mice were randomly divided into 5 groups with 20 animals in each group, based on a power analysis. The first group was topically applied with acetone twice weekly and designated as vehicle-alone group (V). The second group of mice was initially applied with 40 µM of DMBA (initiator [I]). Following a recovery period of 2 weeks, these mice were applied with twice weekly tumor promoter (P), 1% croton oil (in acetone), for 16 weeks and thereafter applied acetone twice weekly for next 16 weeks. Therefore, the total term of the study was 32 weeks. These mice were designated as carcinogenic animals, or I + P group. The medicinal extracts were applied topically (100 µL) 1 hour before the application of croton oil in the respective groups for first 16 weeks and twice weekly without promoter for the next 16 weeks. Therefore, we had 3 groups based on the medicinal extracts—namely, the TF, EA, and CA groups. The study included separate groups for initiator only and all the medicinal extracts given solely for initial data collection. The treatments were applied for all 32 weeks of the study.
Food and water intake as well as behavioral changes was monitored every day, and the body weight of the animals was recorded every 8 weeks. All animals were sacrificed following DMBA or vehicle injection—that is, at 32 weeks following commencement of the study. The animals were fasted overnight before sacrifice. The rationale for using this specific protocol relates to identifying agents that intervene in initiation and progression of skin cancer.
Tumor Number, Incidence, and Multiplicity
After the completion of the treatment regimen, mice were removed from the ambient housing, and observations were made under bright light with 1 group at a time. The papillomas developed—hereafter designated as “tumors”—were described as light to dark pink nodes on the dorsal skin of the mice. The tumors were counted in each mouse of the group. The data were maintained as number of tumor-bearing mice in total mice and total number of tumors in each group. Tumor multiplicity was deducted by dividing the total number of tumors in each group by the number of tumor-bearing mice per group. The initiator-only group and extract-only applied groups were also studied.
Pathologic Studies on Paraffin-Embedded Tissue Sections
The skin samples were processed for histologic investigation with hematoxylin and eosin staining. The formalin-fixed skin samples were dehydrated with graded ethanol (Merck) and embedded in paraffin (Hi-Media Labs, Mumbai, India) after rinsing with tap water. The samples were cut by microtome at 5 µm and mounted on glass slides. The sections were dewaxed with xylene (BDH) solution (mixture of isomers) and graded ethanol and stained with hematoxylin and eosin (Hi-Media Labs).
Assessment of Free Radical Generation and Antioxidant Defense
Skin Processing for Fractions of Cellular Lysates
Animals were sacrificed by ether narcosis 32 weeks after completion of 2-stage chemical carcinogenesis regimen. Skin from dorsal area was removed and cleaned free of extraneous tissue. Skin homogenates were prepared in chilled phosphate buffer (0.1M, pH 7.4) with polytron homogenizer (PT-3100, Kinematica Inc, USA) and filtered through muslin cloth. The cellular lysates so obtained were centrifuged (C-24BL, Remi, India) at 3000 × g for 6 minutes at 4°C to obtain the pellet (containing the membranes), which was immediately stored at −20°C, while the supernatant was spun at 10 500 × g for 30 minutes at 4°C to obtain the postmitochondrial supernatant, which was stored at −80°C till further analysis.
Peroxidation of Membranes
Estimation of lipid peroxidation was done according to the method of Wright et al. 31 The reaction mixture consisted of 0.58 mL of phosphate buffer (0.1M, pH7.4), 0.2 mL of lipid-rich fraction, 0.2 mL of ascorbic acid (1mM), and 0.02 mL of ferric chloride (100mM) in a total volume of 1 mL. The mixture was incubated at 37°C in a shaking water bath for 1 hour; then, 1 mL of 10% trichloroacetic acid and 1 mL of 0.67% thiobarbituric acid was added. All the tubes were placed in boiling water bath for 20 minutes. The tubes were then placed in ice bath and centrifuged at 2500 × g for 10 minutes. The amount of malondialdehyde formed in each of the samples was assayed by measuring the optical density of the supernatant at 535 nm. The results were expressed as nanomole of malondialdehyde formed per hour per gram of tissue based on a molar extinction coefficient of 1.56 × 105 per M/cm.
Estimation of Nonprotein Thiols
Nonprotein thiols (GSH) were measured by the method of Jollow et al 32 with few modifications. postmitochondrial supernatant (1 mL) was precipitated with 1mL of sulfosalicylic acid (4.0%). The samples were kept at 4°C for 1 hour and then centrifuged at 1200 × g for 15 minutes at 4°C. The assay mixture contained 0.2 mL of filtered aliquot, 2.6 mL of sodium phosphate buffer (0.1M, pH 7.4), and 0.2 mL of DTNB in a total volume of 3 mL. The absorbance of the reaction product was read at 412 nm. The GSH content was expressed as nanomole of GSH per gram of tissue.
Determination of GPx Activity
GPx activity was assayed according to the method of Mohandas et al. 33 The assay mixture consisted of 1.44 mL of sodium phosphate buffer, 0.1 mL of EDTA (1 mmol/L), 0.1 mL of sodium azide (1 mmol/L), 0.05 mL of GR (1 IU/mL), 0.1 mL of GSH (1 mmol/L), 0.1 mL of NADPH (0.02 mmol/L), 0.01 mL of H2O2 (0.25 mmol/L), and 0.1 mL of postmitochondrial supernatant (10%) in a total volume of 2 mL. Oxidation of NADPH was recorded spectrophotometrically at 340 nm. The enzyme activity was calculated as nanomole of NADPH oxidized per minute per milligram of protein, based on a molar extinction coefficient of 6.22 × 103 per M/cm.
Estimation of GR Activity
GR activity was assayed by the method of Carlberg and Mannervik. 34 The assay system consisted of 1.65 mL of phosphate buffer (0.1M, pH 7.6), 0.1 mL of EDTA (0.5mM), 0.05 mL of oxidized glutathione (GSSG, 1mM), 0.1 mL of NADPH (0.1mM), and 0.1 mL of postmitochondrial supernatant (10%, w/v) in a total volume of 2.0 mL. The enzyme activity was measured at 25°C as disappearance of NADPH at 340 nm and was calculated as nanomole of NADPH oxidized per minute per milligram of protein based on a molar extinction coefficient of 6.223 × 103 per M/cm.
MPO Activity
Skin MPO (enzyme present in neutrophils) is measured by homogenizing the dermal tissue in 50mM K2HPO4 buffer (pH 6.0) containing 0.5% hexadecyl trimethyl ammonium bromide (Sigma), and enzyme activity was measured by the method of Bradley et al. 35 After 3 cycles of sonication and freezing-thawing, the samples were centrifuged at 2500 × g for 30 minutes at 40°C. MPO activity in supernatant (0.1mL) was assayed by mixing it with 2.9 mL of phosphate buffer (50mM, pH 6.0) containing 0.167 mg/mL of o-dianisidine dihydrochloride (Sigma) and 0.0005% hydrogen peroxide (Sigma). The change in absorbance resulting from decomposition in H2O2 in the presence of o-dianisidine dihydrochloride was measured at 460 nm for 5 minutes. The results are expressed as units per minute per milligram of protein. One unit of MPO activity was defined as that degrading 1 mol of peroxide per min. Peroxidase is used as standard.
Immunohistochemistry of Langerin, NFKB, p38 MAPK, COX-2, p53, and PCNA
The processed skin tissues were obtained and preserved in the 4% paraformaldehyde overnight, followed by dehydration in 10%, 20%, and 30% sucrose solution successively till 3 days, and postfixed after that in formaldehyde fixative until immunochemical staining. Sections of paraffin-embedded tissues (5-10 µm thick) were cut with grading-type lieca microtome, and after running through xylene and different percentages of ethanol (ranging from 30% to 100%), the sections were boiled in 0.1M citrate buffer (pH 6.0) at 75°C for 20 minutes for antigen retrieval process and briefly incubated in 0.3% H2O2 in methanol, followed by incubation in blocking buffer containing 0.1M PBST, 0.04% Triton X-100, and 10% NGS (normal goat serum). Sections were incubated in anti-mouse p53 antibody raised in rabbit (1:100) diluted in tris buffered saline, anti-NFκB (1:150), anti-Cox-2(1:300), and anti-p38 MAPK (1:300) overnight at 4°C. After buffer rinsing, sections were processed with a 3-layer peroxidase staining kit from Thermo scientific system. The peroxide complex was visualized with 3,3-diaminobenzidine (pH 7.2). Last, the slides were counterstained with hematoxylin for 5 to 15 seconds. Slides were then cleaned in xylene, gradually dehydrated with ethanol, cover-slipped in mounting medium, and photographed under a epifluorescent upright Olympus Microscope (BX51).
Assessment of Apoptosis in Tumors
Dissociation of Tumors
Mice were deeply anesthetized, and all surfaces were sterilized by 70% Et-OH for about 30 minutes. Finally, 2 to 4 tumors from respective treatment groups were pooled in autoclaved petri dishes in DMEM under a sterilized class II hood. In case of control animals, clippered skin was excised from the dorsal surface and stored in the same conditions as described above. After the DMEM was poured off, tissues were chopped with crossed scalpels in a solution containing collagenase and trypsin with EDTA. The released cells from each group were collected in conical tubes after adding 2 to 4 volumes of DMEM. The cells were spun at 800 × g for 6 minutes, and excess DMEM was decanted. The process was repeated to clean the cells. The cells so obtained were maintained in serum-free DMEM in 5% CO2 incubators at 37°C overnight till assay.
Assessment of Apoptosis Within Tumor Cells
Cells obtained after tumor dissociation and final storage were spun again and suspended in 1 mL of PBS at a concentration of 2 × 106 cells per 1 mL. Then 50-µL cell suspension was incubated with 50 µL of double-staining solution containing 1 µg/mL AnnCy3 and 500 µM 6-CFDA in 1× Binding Buffer on poly-L-lysine-coated slide as suggested by the manufacturer’s instructions. Then the cells were immediately observed under fluorescent microscope after appropriate filter adjustment. Images were recorded, and the phosphatadylserine-Cy3 conjugate (+) cells were counted by computer assisted image analysis.
Image Analysis
Image analysis in case of PCNA+ and p53+ nuclei and Cy3+ cells was carried out by a person blind to the experimental protocol. Typically, we selected 3 slides per group and counted nuclei or cells using a particle counter jar with cell-type recognition ability integrated to the image analysis software (Image J, 3.1, freeware from the National Institutes of Health, USA). The data were presented as mean ± standard deviation of 3 independent readings and used for the final data compilation.
Estimation of Protein
Protein estimation, wherever needed in the experimentation, was carried out per the previously published method of protein dye binding. 36
Expression of Results, Data Analysis, and Statistical Significance
All other data are expressed as mean ± standard deviation. One-way analysis of variance was used to estimate overall significance, followed by post hoc analysis with the Tukey-Kramer multiple comparisons test. A value of P < .05 was required for results to be considered statistically significant. A commercial software program (Instat 3.2, USA) was used for statistical analyses and representation of data.
Results
Food and Water Intakes
During the entire study period, we did not observe any differences in food or water intake among the various experimental groups. Food and water intakes as monitored were 8.2 to 8.5 g per 100 g of body weight per day and 2.12 to 4.28 mL per 20 to 24 g of body weight per day, respectively, for all groups.
Mortality
There were no treatment-related deaths of mice from any group before the termination of the study (ie, 32 weeks).
Body Weight Analysis
No statistical differences were noticed between the growth rates of any of the treatment groups and normal groups. The final weight of the vehicle-alone (V) group was 42 ± 1.01 g, while that for the I + P group was 40 ± 2.33 g. The final body weight of the plant extract–treated groups was as follows: TF, 41 ± 2.65 g; EA, 42 ± 2.55 g; and CA, 40 ± 0.29 g.
Effects of TF, EA, and CA
On the Number, Incidence, and Multiplicity of DMBA-Initiated Croton Oil–Promoted Tumors
At the termination of the experiment, the tumor incidence and number of tumors (papillomas) per mouse was compared to compare the effect of the medicinal treatments on murine skin carcinogenesis. There were no visible tumors on the skin of the vehicle-only group (group V), DMBA-only (initiator) group (group I), and all the medicinal plant extract–treated groups (data not shown). However, a significant tumor multiplicity (P < .001) in the form of visible tumors of light to deep pink color arose on the backs of the various DMBA (I) + croton oil (P) groups in comparison to the vehicle-alone or initiator-only groups. These data reflect the authenticity of the murine model used, since initiator only was not able to induce tumor growth in mice despite the long window of the experiment (ie, 32 weeks). Table 1 summarizes tumor incidence, total number of tumors, and average number of tumors per tumor-bearing mice (tumor multiplicity) of the various experimental groups. Topical application of all 3 medicinal plants was able to reduce the tumor incidence in the I + P group, suggesting direct inhibition of the tumor growth by these extracts. We calculated a very significant (P < .001) reduction in tumor multiplicity by TF in the tumors induced by DMBA and croton oil, followed by EA (P < .01) and CA (P < .05).
Effect of Methanolic Extracts Treatment of TF, EA, and CA on the Development of Skin Tumors in Mice After DMBA Initiation and Subsequent Promotion by Croton Oil.

Abbreviations: CA, Calendula officinalis; EA, Eclipta alba; I, initiator, P, promoter; TF, Trigonella foenumgraecum.
Total no. of mice = 20.
P < .001, vehicle vs I + P group.
P < .001, TF, EA, or CA vs I + P group.
P < .01, TF, EA, or CA vs I + P group.
P < .05, TF, EA, or CA vs I + P group.
As the treatment of mice with all the 3 extracts resulted in a significant inhibition of DMBA–croton oil skin tumor development, further studies on molecular targets or chemopreventive mechanism were performed with these doses of plant extracts, and data were compared with the treated but DMBA–croton oil group. Since the initiator and extract groups showed no tumorigenic activity, these were eliminated from further studies.
On the Pathologic Changes
Following the evaluation of the tumor data, pathologic changes were studied on the paraffin-embedded coronal tissue sections (Figure 2A). The vehicle-treated animals in the control group showed normal stratification of the skin tissue with well-differentiated epidermis, dermis, and subcutis. The cells revealed normal granulated cytoplasm and small uniform nuclei. Mice in the I + P group (viz, tumor group) depicted irregular cytoplasm and considerably large and hyperchromatic nuclei. In addition, this group showed dermal penetration of large tumors (Figure 2B) with irregular/regular boundaries with loss of epidermal stratification and dermal differentiation. Figure 2C depicts 2 adjacent large epidermal tumors, T1 and T2 (bordered with red dotted lines). The treatment of the mice following the tumor induction reduced the incidence and number of tumors in mice (Table 1). Histologic investigation revealed regression of the tumor size and concomitant normal stratification of the skin tissue, which was highest in the TF mice in comparison to the EA or CA mice, 32 weeks after the initiation of the protocol.

Pathologic studies on the skin biopsies from mice. The normal mouse skin has a clear stratification (A) with well-defined thin epidermis, uniform spongy dermis, and supporting subcutaneous tissue. In contrast, the dermal tissue (€; B) of carcinogenic mice are invaded by large epidermis tumors (T1 and T2; C). Photomicrographs of the mice treated with Trigonella foenumgraecum (D; red dotted line underlining the reorganized epidermis), Eclipta alba (E), and Calendula officinalis (F). Note the regression in the dermal tumors (black arrows) with reorganized epidermis layer (white arrows). Original magnification 10×.
On the Free Radical Generation and Antioxidant Defense
The free radicals generated by the continuous application of TPA following initial initiation by DMBA can induce inflammation and cellular proliferation within mouse skin. We studied a significant amount of the peroxidation of the lipid membranes within the I + P group, which is seldom indicative of the free radical generation (Table 2). However, the concomitant application of the plant extracts was able to induce reducing equivalents in the skin, evidenced by the marked upregulation of reduced glutathione and glutathione-dependent enzymes. The possible hydroxyl ion overload was significantly reduced by the higher levels of superoxide dismutase. It may be noted that TF was able to restore the endogenous defense to the TPA-induced free radical stress to a better extent than that of EA and CA (Table 2).
Effect of Medicinal Extracts Peroxidation of Biomembranes and Glutathione-Dependent Enzymes in Mouse Skin After 2-Stage Carcinogenesis.

Abbreviations: CA, Calendula officinalis; EA, Eclipta alba; I, initiator, P, promoter; TF, Trigonella foenumgraecum.
P < .001, V vs I + P group.
P < .001, TF, EA, or CA vs I + P group.
P < .01, TF, EA, or CA vs I + P group.
P < .05, TF, EA, or CA vs I + P group.
On the Cellular Proliferation (Proliferating Nuclear Antigen)
The proliferation of cells is the most reliable marker used in the tumor studies. We performed immune staining of PCNA to reveal the effect of the various treatments on the DMBA-TPA-induced skin tumors in mice. The vehicle-treated mice showed very little or no PCNA expression in the dermal as well as the epidermal areas in the paraffin-embedded serial coronal sections (Figure 3A). However, the I + P group depicted intense PCNA expression that was diffuse from epidermis (Figure 3B) to dermis (Figure 3C). Moreover, dermis showed established and growing tumors with somewhat discrete borders, lined with PCNA+ nuclei. The different treatment regimens decreased the PCNA expression considerably, with the lowest expression in the TF animals. Morphometric analysis of the slides from all the groups revealed a massive increase (P < .001) in the expression of the PCNA+ nuclei (approximately 74%; Figure 3G) in the I + P group when compared to vehicle group. TF reduced the population significantly (P < .01), while EA (Figure 3E) and CA (Figure 3F) were able to suppress the expression of PCNA to an equal extent (P < .05). These results confirm that treatments were successful in reducing cell proliferation after chemical-induced skin carcinogenesis in mice.

Antibody binding to PCNA and detection by DAB in mouse tumors. Presence of significantly higher number of PCNA+cells in epidermis (B) and dermis (C) of carcinogenic (I + P) mice (black arrows); £ represents the PCNA - cells in comparis to the vehicle control (A). Topical application with medicinal extracts TF, EA, and CA (D -F, respectively) reduced the PCNA+ nuclei in comparison to the carcinogen-alonetreatedanimals, as depicted in the histograms (G). CA, Calendula officinalis; EA, Eclipta alba; I, initiator, P, promoter; TF, Trigonellafoenumgraecum; V, vehicle.
On the Tissue Inflammation After Induction of Skin Carcinogenesis
Inflammation in murine skin following exposure to TPA is a well-established fact. We were typically interested in investigating whether inflammation within the TPA-exposed skin is mediated by systemic stimulation or modulation of cellular prostaglandins by the stress-responsive intracellular element NFkB. It is quite evident from the results that the inflammation in the skin tissue is mediated by the intracellular stress signaling as well as the systemic stimulation evidenced by the expression of NFkB, which was intense in the epidermal tissue as well as the dermal tissue (Figure 4B). It is worth mentioning that COX-2, synthesized from the local prostaglandins, also showed very intense expression in the epidermal and dermal tissues (Figure 4G).

Local inflammatory responses in mouse skin after chemical-induced carcinogenesis. The local inflammatory response is generated by the promoter by sudden upregulation of NFκB (red arrows) and linked overexpression of COX-2 (black arrows) in epidermal (B and G, respectively). Medicinal plant extracts TF, EA, and CA effectively reversed the local and systemic immune response by limiting the NFκB (C, D, and E, respectively) and COX-2 (H, I and J, respectively). CA, Calendula officinalis; EA, Eclipta alba; I, initiator, P, promoter; TF, Trigonella foenumgraecum.
The role of systemic stimulation was revealed by the higher levels of infiltrating neutrophils evidenced by myeloperoxidase (Figure 5). Inflammation, being a chemopreventive target, was ameliorated by the topical application of all 3 extracts by decreasing the amount of infiltrating neutrophils and concomitant silencing of the cellular stress signal NFkB (Figure 4C-4E) along with inhibition of prostaglandin metabolism (Figure 4H-4J). It may be noted that TF was able to reduce the inflammation to higher extent (P < .001), followed by EA (P < .01) and CA (P < .05) when compared to the I + P group.
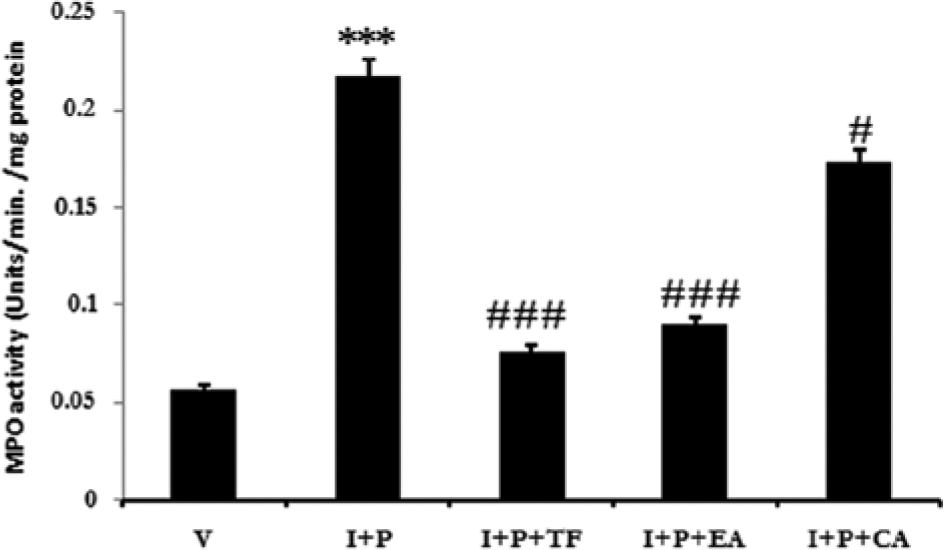
The systemic inflammatory response to the promoter is revealed by the significant infiltration of neutrophils quantified by the myeloperoxidase activity (MPO) in the carcinogenic mice. Note the lesser number of infiltrating neutrophils in the medicinal extract–treated groups. CA, Calendula officinalis; EA, Eclipta alba; I, initiator, P, promoter; TF, Trigonella foenumgraecum; V, vehicle.
On the TPA-Induced LC Migration in Skin
Recent findings into the mechanism of TPA-induced skin hyperproliferation have indicated the significant migration of the epidermal antigen-presenting LCs out of the epidermis, leading to the survival of the initiated/altered cells by compromised surveillance. We confirmed these findings and recorded decreased expression of LCs in the epidermis in the I + P group in comparison to the vehicle-treated mice (Figure 6A, 6B), proving the compromised immune surveillance of the DMBA-initiated genetically damaged cells. As the essential part of the study, we studied the LC expression in epidermis after treatment with all 3 medicinal extracts. It may be noted that there was a significant amount of LC retention in the epidermal tissues, revealed by antibody binding to the langerin after treatment with TF (Figure 6C). The remaining 2 extracts also showed considerable retention of the LCs (Figure 6D, 6E) in the epidermis with a lower degree as compared to TF. These results demonstrate the role of these extracts in enhanced immune surveillance in DMBA-initiated TPA-promoted sites.

Migration of antigen-presenting Langerhans cells out of the mice skin with tumors. The repeated topical application of tumor promoter led to the remarkable migration of Langerhans cells (black arrows) from epidermis (B) in comparison to vehicle alone control mice (A), which was however stopped by the application of medicinal extracts Trigonella foenumgraecum (C), Eclipta alba (D), and Calendula officinalis (E).
On the Cell Cycle Progression
The initiation and promotion (I + P) scheme adopted in the study leads to the hyperproliferation demonstrated by the increased PCNA+ cells in the I + P group. However, the molecular standpoint regulating the proliferation of the cells was not clear. We noticed a remarkable increase in the p38 MAPK expression in the epidermal (Figure 7B) and dermal (Figure 7C) regions of the I + P mice, which indicated the cell cycle progression in both parts of the skin. Therefore, cell cycle progresses in conjunction with migration of LCs and massive inflammation. It is worthwhile noting that the treatment with all 3 extracts (TF, EA, CA) was able to slow the cell cycle progression directly or modulate the LC migration and silence the inflammatory signals. TF was better able to reduce the p38 MAPK expression compared to EA or CA.

Silenced cell cycle progression in tumor mice after treatment. No or very little expression in vehicle-treated mice (A) yet remarkable increase in the expression of p38 MAPK (black arrows) in carcinogen-only-applied mouse epidermis (B). Treatment with medicinal extracts Trigonella foenumgraecum (C), Eclipta alba (D), and Calendula officinalis (E) silenced the expression of p38 MAPK.
On the p53 Expression
One of the chemopreventive targets of our study was the expression of p53 tumor suppressor protein in tumors. The cytoplasmic stabilization of the p53 remains one of the signals of the apoptotic cell death. We studied a very low expression of p53 in the dermal tumors (Figure 8C) in the I + P group (P < .001), which is caused by the chemical-induced carcinogenesis. Furthermore, treatment with TF induced ectopically high expression of p53 in dermal tumors (P < .001; Figure 8D). p53 was highly expressed by the invading tumor in basal dermis. EA (Figure 8E) and CA (Figure 8F) were also able to express p53 (P < .05) within the tumors but with a lower intensity than TF.
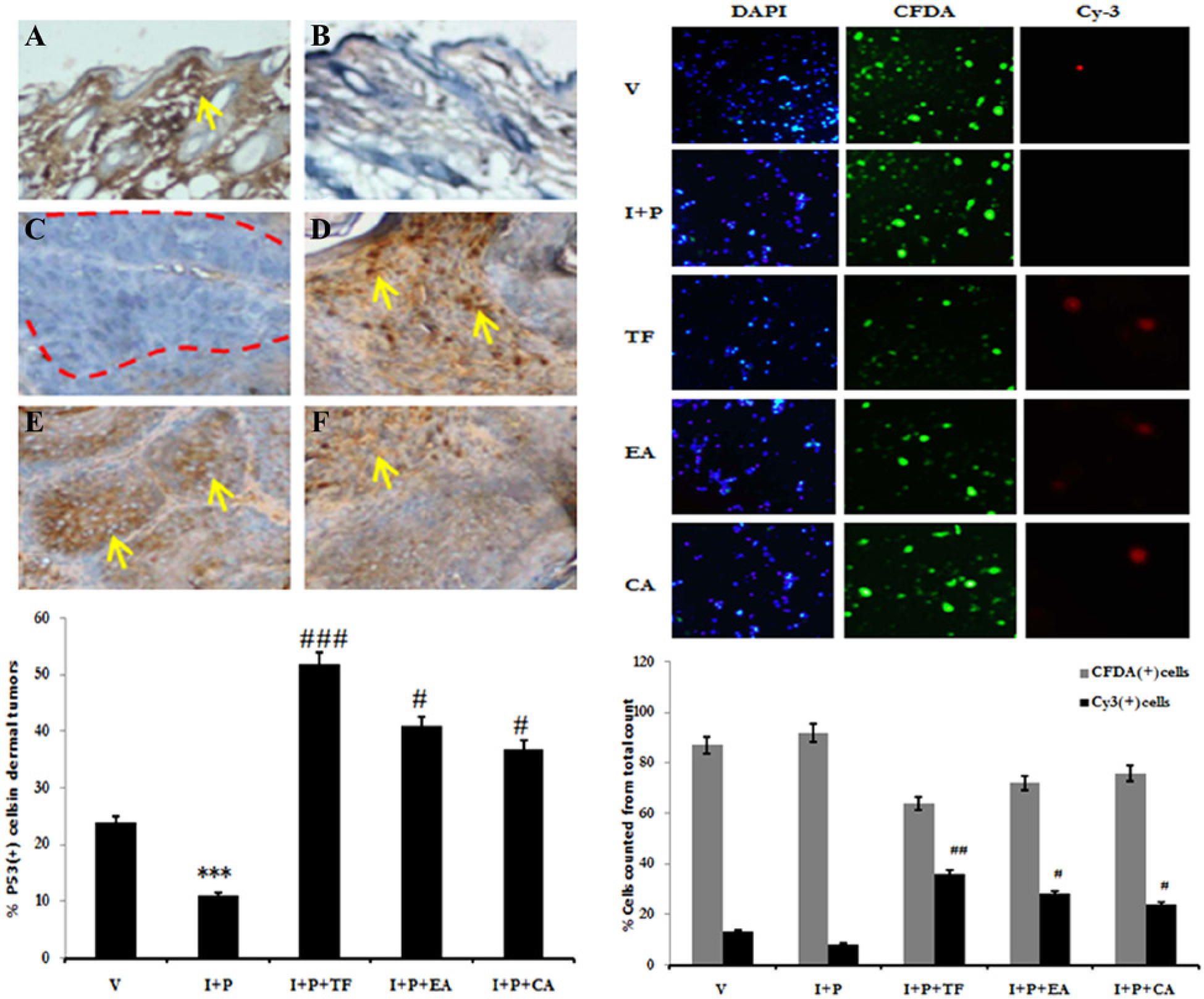
Apoptosis in tumors after cytoplasmic stabilization of p53 protein. p53 protein expression was suppressed after chemical-induced carcinogenesis in the epidermis (B) and dermis (C) when compared to the vehicle alone treated mice (A). Regression in the tumors after treatment with medicinal extracts TF (D), EA (E), and CA (F) coincided with the stable cytoplasmic re-expression of p53 evidenced by increasing Cy3+ cells in the treatment groups. CA, Calendula officinalis; EA, Eclipta alba; I, initiator, P, promoter; TF, Trigonella foenumgraecum; V, vehicle.
On Apoptotic Cell Death Within Tumors
An in vitro method was adopted to assess the induction of apoptosis by the medicinal extracts within the tumors induced by DMBA and croton oil in mice skin (Figure 8, right panel). The activation of p53 is direct evidence of the regulated apoptosis within tumors. The I + P tumor cells group demonstrated a health state with maximum CFDA immunofluorescence, and almost nil cells were Cy3+ apoptotic. In comparison, vehicle-treated mice skin cells demonstrated few Cy3+ cells. Groups treated with TF (P < .01), EA (P < .05), and CA (P < .05) showed considerably higher numbers of Cy3+ cells and fewer CFDA+ healthy cells. Moreover, CA was not as competent as TF or EA in inducing apoptosis in tumors induced by DMBA-TPA.
Discussion
Chemically induced skin carcinogenesis in mice is the best-characterized model for cellular initiation and promotion in preclinical testing of anticarcinogenic potential of synthesized or natural molecules. DMBA-initiated and croton oil–promoted preneoplastic and neoplastic tumors in mice skin closely mimic nonmelanoma skin cancer in humans. Therefore, the data on the anticarcinogenic potential at preclinical stages can be extrapolated for human evaluation. In this study, we have made an attempt to investigate the effect of 3 promising medicinal herbs on the appearance of skin tumors in mice and elucidation of the potential chemopreventive strategy adopted by these herbs.
The attenuation of tumor growth by dietary restriction has gained renewed interest. 37 It has been observed that calorie restriction causing body weight loss may also be associated with reduction in tumor volume. 38 The ability of dietary and caloric restriction in decreasing tumor incidence and arresting tumor growth has been established in chemically induced as well as spontaneous tumors in rodents.39 -42 We did not notice any significant difference in the food and water intake as well as the mean body weight among various mice groups, suggesting a similar nutritional status of the experimental animals. However, there was a significant reduction in the tumor multiplicity in the treated groups in comparison to the carcinogen-only exposed animals. As all the mice in the present study grew equally irrespective of dietary condition, it can be concluded that the observed inhibitory effects of TF, EA, and CA on tumor growth is not associated with an impairment of nutritional status of the carcinogen-exposed animals.
Cellular proliferation is integral to multistage skin carcinogenesis, and the risk of cancer development has been associated to the rate of the cellular mitosis. 43 Therefore, the agents that can arrest proliferation or rate of proliferation can be used as chemopreventive strategies against the skin carcinogenesis. We studied the immunolocalization of a 36-kDa nuclear protein, PCNA (proliferating nuclear antigen), that is expressed during the late G1 and S phases of the cell cycle. With pathologic data, PCNA can be used in detection and staging of tumors. 44 As a step in the chemopreventive strategy used by TF, EA, and CA against the multistage skin carcinogenesis, we studied the cellular proliferation by immunohistochemical detection of PCNA in carcinogen alone (I + P group) and extract-treated mice skin. The data suggested a significantly higher rate of proliferation in the number of PCNA+ nuclei in the mice in the I + P group, which was lowered after treatment with TF, EA, and CA. We had previously used quantification of radioactive thymidine for the assessment of cellular proliferation. 44 However, PCNA allows a better study of the genesis of the epidermal tumors as well as the dermal tumors and indicates the development of the locus very early.
The contribution of inflammatory processes in the development of the cancer is an ongoing debate. A range of studies has demonstrated the involvement of persistent inflammation in cancers. 45 Several studies have also highlighted the role of inflammation in the development of skin tumors in mice after croton oil treatment. 46 However, inflammation is a broad term, and at a particular site like skin, it has both local and peripheral components. The repeated topical application of the promoter on the skin can initiate cellular processes leading to inflammation and can concomitantly recruit circulating neutrophils to the site. Our results confirmed the assumption, and there was a significant increase in the infiltrating neutrophils along with the hyperexpression of COX-2, which is synthesized by the local prostaglandins. The in situ trigger for the cellular processes of inflammation was discovered to be NFκB, which can transduce multidimensional signals, including inflammation. 47 However, treatment with TF, EA, and CA was successful in reducing the local and peripheral components of inflammation. These results are in line with the previously published data suggesting the anti-inflammatory role of plant-based extracts or the constituent molecules.
Survival of the genetically damaged/altered cells by the application of a potent tumor promoter TPA leads to the progression of the cell cycle machinery and, finally, the genesis of a clone of genetically modified cells. However, efficient recognition of the altered cells can reduce the chances of their survival. Recent literature suggests that LCs are antigen-presenting cells that can regulate or stimulate the immune surveillance in the skin. 48 LCs migrate out of the epidermal tissues following single or repeated application of the tumor promoter TPA. Bearing in mind these considerations, we studied the effect of TF, EA, and CA on the immune surveillance of altered cells due to the single topical application of DMBA. We noted a marked retention of LCs in the epidermis following treatment with TF, EA, and CA. However, the LC retention ability of TF was more in comparison to EA or CA, which showed more or less comparable results.
p38 MAPK is a cell cycle regulation switch that is sensitive to various mitogenic signals, including tumor promoters. 49 The activation and downregulation of MAPKs are major deciding factors in cellular survival and proliferation. 49 As discussed earlier, TF, EA, and CA limit inflammation and lead to the retention of antigen-presenting LCs in mouse skin following TPA exposure. These results were further strengthened by the reduction of p38 MAPK in the treatment groups, suggesting either elimination of the altered cells or silencing of the cell cycle progression. It is worthwhile to note that downregulation of inflammation and LC retention may be responsible for the silencing of the cell cycle progression.
It is a well-known fact that human carcinogenesis, as well as its experimental counterparts represented by a range of animal models, leads to the suppression of tumor suppressor gene p53. 50 Therefore, strategies that can elevate its expression within the tumors may prove to be fruitful in chemopreventive testing. 51 During carcinogenesis, the p53 gene gets suppressed, leading to the survival of aberrant cells. Conversely, its expression can check the proliferation within tumors. We studied the expression of p53 protein in the dermal tumors in the I + P group and other treatment groups. There was a negligible expression of p53 with the tumors in carcinogen-only exposed mice. However, all the experimental extracts reexpressed p53 within tumors, and this ability was demonstrated in the mice treated with TF.
The expression of p53 within the tumors raises 2 important inferences. First, the expression may have increased due to the direct effect of experimental extracts on the p53 gene expression. Another possibility is the p53 expression in response to the induced regulated apoptosis, since eukaryotic cells express p53 under genetic instability. 52 p53 protein gets stabilized within the cytoplasm, leading to enhanced mitochondrial permeability and numerous downstream targets of apoptosis. 53 To address this issue, we studied the CFDA-Cy3 fluoro tagging of the healthy and apoptotic cells within the cells dissociated from tumors from the experimental animal groups. Our results confirmed the second possibility, as there was a significant increase in the apoptotic cells in the various treatment groups and the carcinogen-only group showed no apoptotic cells.
Our studies are further strengthened since medicinal extracts described herein or their active constituents have already demonstrated anticancer properties. Particularly, the constituents identified in the extract standardization have potential medicinal values. A recent study reports that trigonelline (an alkaloid) found in T foenumgraecum can sensitize pancreatic cancer cells toward apoptosis by inhibiting proteasomal gene expression. 54 Wedelolactone (a coumestan) from E alba has been seen to inhibit the DNA topoisomerase IIα activity apart from the inhibition of transcription factor NFkb, 55 which can lead to dramatic growth arrest. Wedelolactone has further been reported to induce apoptotic cell death in cancer cells. 56 Alpha-cadinol (an alcohol) found in variety of plants, including C officinalis, can induce cytotoxicity in cancer cells of different origins. 57
In summary, the genesis of tumors is not mere hyperproliferation but a process perturbed by a plethora of individual events that include inflammation, altered cell survival, and progression of cell cycle. Medicinal herbs TF, EA, and CA can prevent or intercept these standpoints. However, extensive research is needed to uncover the active principles and mechanisms involved behind the above-said effects.
Footnotes
Acknowledgements
We are thankful to the University Grants Commission of the Government of India, New Delhi, for providing a Senior Research Fellowship to Farrah Ali during the tenure of the study.
Author Contributions
FA designed and performed the experiments. AL, AQK, RK, and MTM helped in data acquisition, and SS edited and reviewed the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: University Grants Commission of the Government of India, New Delhi, for providing a Senior Research Fellowship to Farrah Ali during the tenure of the study.
