Abstract
Pancreatic carcinoma remains one of the main causes for cancer-related death. Intratumoral application of anticancer agents is discussed as a promising method for solid tumors such as pancreatic cancer. Endoscopic ultrasound provides a good tool to examine and treat the pancreas. European mistletoe (Viscum album L) is a phytotherapeutic commonly used in integrative oncology in Central Europe. Its complementary use seeks to induce immunostimulation and antitumoral effects as well as alleviate chemotherapeutic side effects. Intratumoral mistletoe application has induced local tumor response in various cancer entities. This off-label use needs to be validated carefully in terms of safety and benefits. Here we report on 39 patients with advanced, inoperable pancreatic cancer, who received in total 223 intratumoral applications of mistletoe, endoscopic ultrasound guided or under transabdominal ultrasound control. No severe procedure-related events were reported. Adverse drug reactions were mainly increased body temperature or fever in 14% and 11% of the applications, respectively. Other adverse drug reactions, such as pain or nausea, occurred in less than 7% of the procedures. No severe adverse drug reaction was recorded. Patients received standard first- and second-line chemotherapy and underwent adequate palliative surgical interventions as well as additive subcutaneous and partly intravenous mistletoe application. A median survival of 11 months was observed for all patients, or 11.8 and 8.3 months for stages III and IV, respectively. Due to the multimodal therapeutic setting and the lack of a control group, the effect of intratumoral mistletoe administration alone remains unclear. This retrospective analysis suggests that intratumoral-applicated mistletoe might contribute to improve survival of patients with pancreatic cancer. In conclusion, the application is feasible and safe, and its efficacy should be evaluated in a randomized controlled trial.
Introduction
Pancreatic ductal adenocarcinoma remains a dire prognosis. Although it is the 10th-most common cancer, it ranges from 4th to 6th position for cancer-related deaths in Germany and the United States.1,2 Due to a lack of symptoms, it remains undiscovered, and more than 80% of the patients present advanced cancers at diagnosis with an unresectable tumor or metastatic spread. 1 Resection is the only potentially curative therapy, but even among resectable patients, the 5-year survival is less than 20% in multicenter studies and less than 5% in observational cohorts. 3 The overall survival of patients at all stages is less than 2% after 5 years.3,4
For unresectable tumors, gemcitabine monotherapy became the standard in 1997, as it was superior for clinical benefit response and in median overall survival and 1-year survival when compared to 5-fluorouracil. 5 Many combinations of gemcitabine with other therapeutics have been tested, but no significant increases in overall survival compared to gemcitabine monotherapy have been achieved. 6 Recent studies using gemcitabine in combination with erlotinib, an inhibitor of the epidermal growth factor receptor, 7 or FOLFIRINOX 8 showed significant increases in median survival, response rate, and progression-free survival when compared to gemcitabine monotherapy. The success of gemcitabine and erlotinib is still being carefully evaluated by expert panels, 9 and the role of this combination for general use in patients with metastatic pancreatic cancer is being questioned in international guidelines. 10 Although there was an increase of toxicity with grade 3/4 adverse drug reactions, FOLFIRINOX has been accepted as first-line treatment in patients with good performance status who are younger than 75 years old.11,12
Pancreatic ductal adenocarcinoma shows an early local spreading beyond the anatomic borders of the organ and lymphogenic and neurogenic metastasis. 3 In the cell, rare tumor bed fibroblasts (stellate cells) are the predominant stromal cell type 13 and form a dense stroma tissue due to a strong desmoplastic reaction. 14 This stroma constitutes a dynamic compartment that is involved in cancer initiation, tumor formation, progression, invasion, and metastasis. The typically poor vascularization might be one reason why systemic therapies show limited effects. 15 Therefore, it has long been suggested that intratumoral injection of chemotherapeutic agents might be a promising method for the treatment of advanced pancreatic cancer. 16 The theoretical aim was to deliver high local concentrations of therapeutic agents while minimizing systemic side effects. 17
In mouse models, the feasibility of local application for different agents was proven. 18 Tumor response was described after intratumoral chemotherapy. 19 Better targeting was observed with labeled antibodies given intratumorally,20,21 and antitumoral effects or improved chemosensitivity was demonstrated after administration to genetically transformed cancer cell lines.22 -26 Immunization after vaccination with tumor RNA-pulsed dendritic cells27,28 and increased survival after vaccination with chemotherapy were also observed. 29 In addition, there are successful studies giving chemotherapeutic agents intratumorally by endoscopic ultrasound in a porcine model.30,31
Since 2000 there have been attempts to use endoscopic ultrasound fine-needle injections of biological antitumor agents in humans. 32 Several clinical phase I and II studies tested intratumoral applications of anticancer agents in patients with advanced pancreas carcinoma, with percutaneous transabdominal or transgastric endoscopic ultrasound access showing feasibility and acceptable safety profiles.16,17,33 -37
In a study by Matthes et al, in which 14 pancreatic cancer patients received mistletoe therapy intratumorally, clinical benefits were demonstrated, such as tumor regression (57%), stable disease (36%), and 1-year survival rate (36%), with an overall good tolerance of the treatment. 38 As a next step, we analyzed in this retrospective study all patients with unresectable or metastasized pancreas carcinoma from the Hospital Havelhoehe in Berlin, Germany, and the associated outpatient care center who were treated with intratumoral mistletoe applications. We documented all therapies that were received and analyzed patients’ survival. The aim of this study was to evaluate the effect of mistletoe by intratumoral application and monitor potential adverse drug reactions.
Methods
From 2004 through 2011, the follow-up of all patients of the Department of Gastroenterology of the Hospital Havelhoehe (Berlin, Germany) who were diagnosed with pancreatic carcinoma and consented to intratumoral mistletoe therapy were documented in the Network Oncology. 39 All tumor-related data on diagnostic and therapy were recorded with a special emphasis on the application of mistletoe extracts. Conventional therapies as well as any adverse events related to mistletoe therapy documented in the patients’ files were recorded.
Intratumoral application of mistletoe preparations were administered either transabdominally or transgastrically/transduodenally by endoscopic ultrasound–guided fine-needle application. While the patient was sedated with propofol, the tumor was visualized sonographically, and a Chiba needle (20G) was placed to the distal margin. The mistletoe preparation was administered fractionally by retreating the needle to the proximal border of the tumor; 0.9% NaCl was injected when the needle was removed to avoid backflow of mistletoe into the needle tract to reduce peritoneal irritation. In each session, the tumor was punctured 1 to 3 times. If possible, there was an induction phase of 3 applications, with increasing dosages at the start of the therapy, beginning with 20 mg for Abnoba and 50 to 100 mg for Helixor. Every 2 to 3 days, mistletoe dosage was raised depending on the general performance status under inpatient conditions. Dosage increase was about 20 to 40 mg (Abnoba) or 100 to 200 mg (Helixor), up to a maximum dosage of about 160 mg (Abnoba) or 1400 mg (Helixor). Depending on the chemotherapeutic scheme, all further mistletoe applications were given alternately to chemotherapy in 4-week intervals or more, in line with the S3 guidelines—sedation in gastrointestinal endoscopy. 41 During intervention, blood pressure, heart frequency, and oxygen saturation were monitored, and routine nasal application of 1.5 to 3.1 l/min of oxygen was given. Postintervention, patients were monitored for at least 2 hours. If requested, analgetics (3.25-7.5 mg of piritramid) and antiemetics (eg, 10-20 mg of metoclopramide) were given pre- or postintervention.
Subsequent analyses and graphics were done in R (version 2.14.1, R Development Core Team, 2011). The percentage of adverse events was calculated as the number of events divided by the total number of applications. We calculated Kaplan-Meier estimates for the total patient population and the subgroups of UICC III and IV stages using the survfit function implemented in the survival package (version 2.37-2); 95% confidence intervals (CIs) were calculated based on log (survival).
Results
Data of 39 patients (17 women and 22 men) who received intratumoral mistletoe therapy between 2004 and 2011 were recorded. Median age at first diagnosis was 61 years (women, 64 years; men, 61 years). The minimum age was 39 years, and the maximum was 85 years. Table 1 shows the documented UICC stages of patients at first diagnosis.
UICC Stages at First Diagnosis and Kaplan-Meier Estimates. a

Given are the number of patients, the number of deaths (events), and the medians of overall survival, along with lower and upper 95% confidence intervals based on Greenwood standard error. The latter could not be calculated for UICC II. For 2 patients, UICC stage was not recorded at first diagnosis.
Conventional Therapies
Three UICC stage II patients received a pylorus-preserving pancreatoduodenectomy as primary tumor resection. Intratumoral administration of mistletoe extracts was first applied to these patients after they experienced an inoperable relapse. Further surgeries were diagnostic or explorative laparotomies in 13 cases. Fourteen patients received a port system, and 1 patient had a Denver shunt implanted. Nine patients received palliative surgeries as follows: percutaneous endoscopic gastrostomy tube, percutaneous endoscopic gastrostomy tube and gastrostomy, biliodigestive anastomosis, posterior gastroenterostomy with discarded loop, median longitudinal laparotomy and bilateral adnexectomy, laparoscopic adnexectomy, partial resection of the ileum with ileoileostomy, ascendo-transversostomy, and ileocoecal resection including 1 m of ileum.
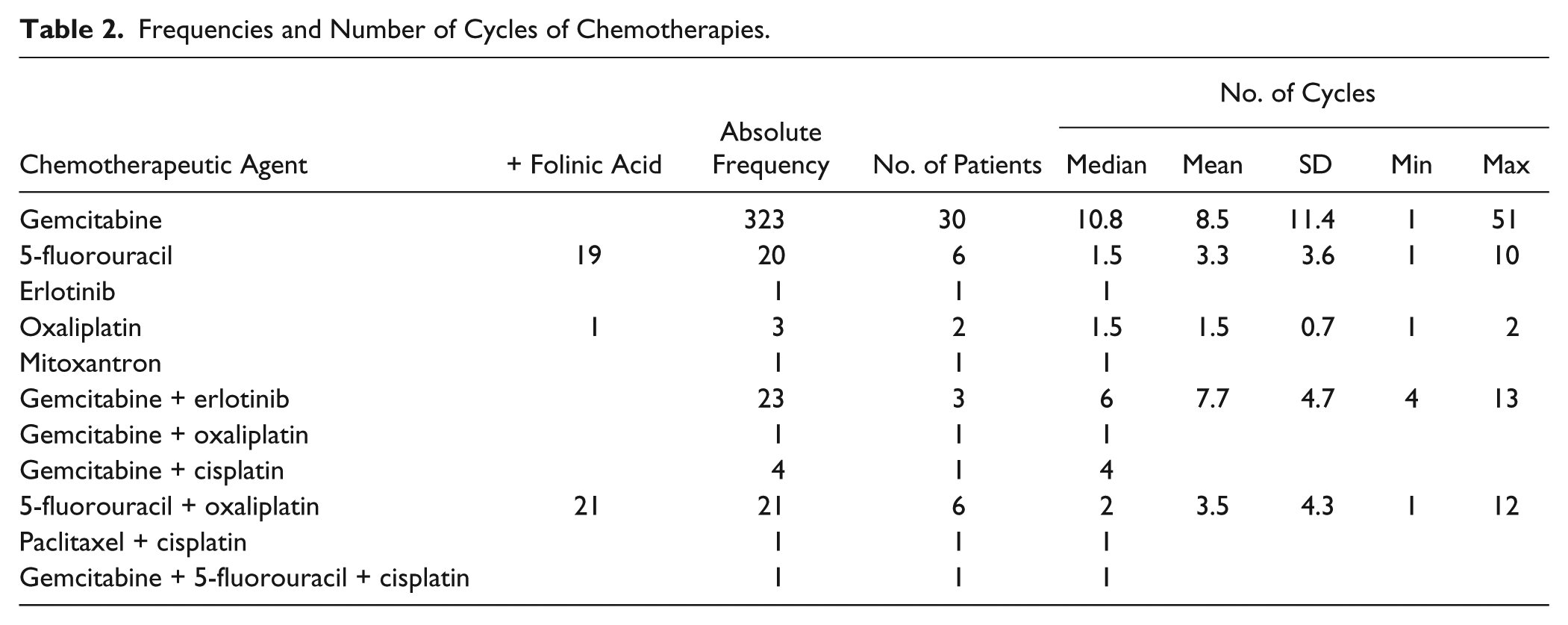
Two patients received radiation therapy due to bone metastases. In total, 33 patients were treated with first-line chemotherapy; 10 received second-line; and 1, third-line. Thirty patients received gemcitabine monotherapy; 11 had combined regimes of 2 or more chemotherapeutic remedies (Table 2).
Frequencies and Number of Cycles of Chemotherapies.
Mistletoe Therapy
Thirty-one patients were treated with the mistletoe preparation Helixor, 30 patients with abnobaVISCUM, 9 with Iscador, and 2 with Iscucin. For intratumoral application, 26 patients had Helixor; 18 had abnobaVISCUM; and 1 had Iscucin. There was a total of 223 intratumoral applications, made up of 139 applications of Helixor, 81 of abnobaVISCUM, and 3 of Iscucin (Table 3). Twenty patients had intratumoral Helixor Mali exclusively, while 13 patients had abnobaVISCUM exclusively. In addition to their intratumoral therapy, 32 patients had subcutaneous and 21 had intravenous mistletoe comedication.
Number of Intratumoral Applications and Dosage Information.

Iscucin, n = 3; total, n = 223.
Safety and Complications During Intratumoral Mistletoe Application
The most frequently reported side effect was elevated body temperature (≤ 38°C). For 23 patients, adverse drug reactions were observed, including fever (> 38°C), pain, nausea, generalized skin irritations, changes in blood cell count, circulatory problems, and headaches (Table 4). During 223 applications, 6 procedure-related difficulties and 2 application errors were described. Procedure-related difficulties were appearances of “bad presentability,” “air in the abdomen,” or “gut in the puncture line.” Application errors were 1 injection into a peritumoral venous conglomeration and 1 into the duodenal wall. Both showed spontaneous healing and had no further clinical consequences. Data about analgesics at first diagnosis were available for only 23 patients at admission.
Frequencies of Observed Adverse Drug Reactions During Intratumoral Mistletoe Therapy.

UICC Staging and Survival
The median overall survival time was calculated for all patients and with respect to UICC stage (Table 1). The median survival for all patients was 11 months (95% CI: 7.8, 16.1). Figure 1A shows survival of patients at all stages, while Figure 1B shows survival for UICC stages III and IV. The median survival periods of patients at stages III and IV were 11.8 months (95% CI: 8.2, 22.8) and 8.3 months (95% CI: 6.4, 15.2), respectively, but were not significantly different (log rank test, χ2 = 0.8, df = 1, P = .379). One-year survival was achieved by 42% of all patients, or 27% and 33% of patients at UICC stages III and IV, respectively.

Kaplan-Meier survival estimates for (A) all patients and (B) patients with UICC stages III and IV. The vertical lines indicate the median survival times.
Discussion
In this retrospective study, we present data of 39 patients with advanced unresectable pancreatic carcinoma receiving intratumoral mistletoe applications. Additionally, patients received systemic mistletoe therapy and standard chemotherapy. In German-speaking countries, there is a high prevalence of complementary and alternative medicine,41,42 and European mistletoe extracts (Viscum album L.) are the mostly frequently prescribed herbal remedies.43-48 An overview on the mode of action of mistletoe extracts is given by Bar-Sela 49 and has been well summarized by Zänker et al. 50 In vitro, it showed antitumoral effects, such as induction of apoptosis,44-48,51 inhibition of cell proliferation,47,48,52 potentiating anticancer effects of TNF-alpha,53,54 activation of immunocompetent cells,54-57 decreasing angiogenesis,50,53 and retention of chemotherapeutic agents in cancer cells by modulating the transport of cell-toxifying substances. 58 Whereas systemic mistletoe therapy primary aims to stimulate the immune system, intratumoral application focuses on the local cytotoxic effect. Anticancer activity on human pancreatic cancer xenograft was firstly shown in a mouse model after intratumoral injection of lectin-rich mistletoe extract, resulting in partial or complete remissions in three-quarters of the cases. 59 Tumor reduction following intratumoral mistletoe application was reported in patients with varying carcinomas.60 -64 Even complete remission was documented in patients with different cancer localities.65 -68
In the initial study, which documented intratumoral application of mistletoe in 14 patients with pancreatic cancer, 38 a tumor response rate of 57% and a local tumor control rate in 93% were reported. In the present follow-up data, we did not have sufficient information on the local response rate, which, as an outcome parameter, has been questioned for some tumors, such as pancreatic carcinoma, 69 due to early local spreading. Additionally, the sensitivity and specificity of ultrasound and computed tomography under local treatment can be largely affected by the application itself, edema, and inflammation. 70 Hence, the measurement of the local tumor response is complex 71 and may not accurately reflect the extent of the anticancer effect of treatment. 35 As intratumoral application of mistletoe extracts is an off-label use, safety issues are of essential interest. Out of 223 applications, 2 erroneous transabdominal punctures occurred: 1 in the duodenal wall, causing pain postinterventionally, and 1 in a peritumoral venous conglomeration. Both showed spontaneous regression and had no further clinical implications. Procedure-related difficulties were described in 2.7% of applications but could be resolved. Whereas Hecht et al 35 had 2 perforations in the duodenal bulb due to the stiffness of the endoscope and no perforations occurred after the protocol was changed to transgastric injections, we had no technical alterations in endosonographic-guided procedures. Also, they reported on 2 cases (out of 21) with sepsis before the introduction of prophylactic oral antibiotics. We saw no cases of sepsis without any prophylactic antibiotics. Furthermore, no procedure-related pancreatitis was observed, and no needle tract seeding was seen.
In a phase I/II study involving 37 patients, a replication-deficient adenovector containing the human TNF-alpha-gene, regulated by a radiation-inducible promoter (TNFerade), was injected endoscopically or transabdominally into tumors. 17 In addition to continuous 5-fluorouracil infusions and radiation, patients received intratumoral application weekly over a 5-week period, with up to 4 applications per treatment session. The endoscopic ultrasound group and the transabdominal group were compared and showed no difference in clinical benefit. Procedure-related adverse events occurred in 24% of patients in the endoscopic ultrasound group, compared to 56% of patients in the transabdominal group, making the endoscopic ultrasound–guided application more favorable.
Adverse effects in other studies, such as fever and nausea, were thought to be associated with the applied antitumoral agent rather than the application procedure.17,34 Elevated body temperature, fever, pain, nausea, general skin irritation, and headaches observed in the present study have been reported with systemic mistletoe treatment and are comparable to data from systematic reviews showing local reaction rates from 0.9% to 43% and systemic reaction rates between 0.45 and 4%.41,72 Changes in blood count and circulatory problems were not otherwise specified or classified in terms of Common Terminology Criteria for Adverse Events v3.0 (CTCAE). Data about the incidence of adverse drug reactions in response to mistletoe therapy range widely, and there is an ongoing discussion concerning whether certain reactions are actually adverse drug reactions or desired immunoreactions. Elevated temperature is a well-known and even desired reaction in mistletoe treatment. According to product information, it is part of the dose-finding strategy in systemic therapy, and mistletoe-induced fever is described as an immunostimulation. 73 Whereas a range of immunotherapy strategies in cancer have been long under investigation,74,75 prospective data on survival and fever induction are lacking. In line with the literature, the presented results show that intratumoral application is feasible, well tolerated, and safe. Intratumoral application, especially via endoscopic ultrasound, offers new opportunities in the treatment of pancreatic cancer. There are some drawbacks, however, as it is not universally available, due to equipment costs and the level of training required to perform procedure. 32
Administered first- and second-line chemotherapies were performed according to international guidelines. With a median of 10.8 cycles for gemcitabine monotherapy and only 3 cycles in combined regimes, chemotherapy duration was generally rather short. FOLFIRINOX was not established at the treatment time. While a high number of cancer patients report concurrent use of herbal drugs under chemotherapy, 76 possibly harmful herb-drug interactions have been discussed.77 -79 There is growing experimental and clinical evidence suggesting that some botanicals interfere with molecular targets and processes involved in cancer. 80 In a phase I/II dose escalation study, Mansky et al 81 investigated the combination of mistletoe and gemcitabine in terms of safety, toxicity, and interference. Neutrophil recovery, mistletoe lectin antibodies, plasma concentrations of cytokines, and gemcitabine were measured in 44 patients with advanced solid cancers of various types who received dose-escalating mistletoe-gemcitabine therapy. Gemcitabine pharmacokinetics were unaffected by mistletoe, and no significant effects on cytokines were observed, while neutrophil values showed a trend to increase. The authors concluded that addition of mistletoe may increase the nadir and even allow higher doses of gemcitabine to be given, demonstrating a beneficial effect of mistletoe, with no reduction in chemotherapy effectiveness.
Systemic mistletoe therapy has been used in a large number of cancers for decades and has been reported to increase health-related quality of life and attenuate adverse effects of conventional therapies.43,82-85 Whether mistletoe therapy can contribute to survival is still the issue of a controversial debate. 86 Recent studies have indicated a possible influence of mistletoe on survival in patients with colorectal or pancreatic carcinomas.87,88 Matthes et al 87 demonstrated a lower adjusted hazard ratio of dying in patients with pancreatic carcinoma with subcutaneous mistletoe therapy combined with gemcitabine versus the control group who received gemcitabine alone. 80 A randomized controlled trial yielded prolonged survival from 2.7 to 4.8 months in patients with advanced pancreatic cancer not eligible for antineoplastic therapies who were treated with subcutaneous mistletoe in comparison to best supportive care. 87 In our data, complete tumor remissions did not occur, and 34 of 39 patients died during the observation period. A median survival of 11 months was observed for all patients, or 11.8 and 8.3 months for stages III and IV, respectively. Results of phase III studies with intratumoral application of antitumor agents in patients with pancreatic cancer are not available. Overall, survival rates from other innovative approaches of phase I/II studies are difficult to compare. After intratumoral application of an replication-selective adenovirus that preferentially replicates in and kills malignant cells, combined with a gemcitabine chemotherapy, the overall survival in 21 patients was 7.5 months. 36 After intratumoral application of a replication-deficient adenoviral vector that expresses TNF-alpha and additive radiochemotherapy and 5-fluorouracil chemotherapy over 5 weeks in stage III patients, the overall survival was 9.8 months. 17
Although it is difficult to compare data due to different UICC stages and other factors, our results seem comparable and possibly better than those of other anticancer agents currently being investigated. Methodological limitations in our data set are caused by the retrospective approach and the integrative oncologic setting, including different dosages and duration of intratumoral, subcutaneous, and intravenous mistletoe therapy. In addition, adjuvant and palliative surgical strategies and standard chemotherapy were applied. Even if the presented result points to a possible influence on survival, these data have to be interpreted carefully, as we lack a proper control group. In conclusion, the effect of intratumoral mistletoe administration in our results alone remains unclear, as in such a multimodal therapy concept, outcome is influenced by various interventions and additive factors might play a role.
Conclusion
This retrospective analysis of 39 patients with advanced pancreatic ductal adenocarcinoma undergoing intratumoral mistletoe therapy in a multimodal oncologic treatment concept documents an (encouraging) overall survival of 11.8 (stage III) and 8.3 months (stage IV). The direct injection of mistletoe extracts, transabdominally or transgastrically, was feasible, well tolerated, and safe. To evaluate efficacy of intratumoral mistletoe treatment, a prospective randomized control study, including measurement of health-related quality of life, is warranted.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H. Matthes is member of the administrative board of the Weleda AG, Arlesheim. The other authors declare that they have no competing interests. By contract, researches were independent from the funder.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The NO database project was funded by the Software AG Stiftung Darmstadt, Weleda AG, Arlesheim, Abnoba GmbH, Pforzheim, and Helixor GmbH, Rosenfels, Germany.
