Abstract
Pao extract, derived from bark of Amazonian tree Pao Pereira, is commonly used in South American medicine. A recent study showed that Pao extract repressed androgen-dependent LNCaP prostate cancer cell growth. We hypothesize that Pao extract asserts its anticancer effects on metastatic castration-resistant prostate cancer (CRPC) cells. Pao extract suppressed CRPC PC3 cell growth in a dose- and time-dependent manner, through induction of apoptosis and cell cycle arrest. Pao extract treatment induced cell cycle inhibitors, p21 and p27, and repressed PCNA, Cyclin A and Cyclin D1. Furthermore, Pao extract also induced the upregulation of pro-apoptotic Bax, reduction of anti-apoptotic Bcl-2, Bcl-xL, and XIAP expression, which were associated with the cleavage of PARP protein. Moreover, Pao extract treatment blocked PC3 cell migration and invasion. Mechanistically, Pao extract suppressed phosphorylation levels of AKT and NFκB/p65, NFκB DNA binding activity, and luciferase reporter activity. Pao inhibited TNFα-induced relocation of NFκB/p65 to the nucleus, NFκB/p65 transcription activity, and MMP9 activity as shown by zymography. Consistently, NFκB/p65 downstream targets involved in proliferation (Cyclin D1), survival (Bcl-2, Bcl-xL, and XIAP), and metastasis (VEGFa, MMP9, and GROα/CXCL1) were also downregulated by Pao extract. Finally, forced expression of NFκB/p65 reversed the growth inhibitory effect of Pao extract. Overall, Pao extract induced cell growth arrest, apoptosis, partially through inhibiting NFκB activation in prostate cancer cells. These data suggest that Pao extract may be beneficial for protection against CRPC.
Keywords
Introduction
Prostate cancer is one of the leading causes of deaths in men, with the estimation that more than 258 000 men will die from this disease worldwide in 2011. 1 Most deaths from prostate cancer are due to metastases, and usually these lesions become resistant to androgen ablation therapy. 2 Only about 30% of the patients with distant prostate cancer survive 5 years after diagnosis, compared to almost 100% 5-year relative survival rates among patients with localized or regional prostate cancer. 3 Unfortunately, no curative treatment exists for those patients at this stage. As drugs currently used have significant adverse effects, herbal extracts, as well as phytochemicals derived from them, are considered as attractive alternatives.
Pao extract is the extract of the bark of a tree that grows in the Amazon rain forest, Geissospermum vellosii Allemão (familiarly known as Pao pereira), which has been used as a medicine by South American Indian tribes. It is reported that Pao extract has anticancer effects against melanoma and glioblastoma cells in vitro.4-6 Moreover, Pao extract suppresses cell growth and induces apoptosis of androgen-dependent prostate cancer LNCaP cells in vitro and in vivo. 7 These data suggest that Pao extract is a promising agent against cancer. For men with metastatic disease, it is important to determine whether Pao extract also possesses anticancer effects against devastating castration-resistant prostate cancer (CRPC), which may appear following androgen ablation therapy.
Sustained or constitutive activation of NFκB in prostate cancer is inversely correlated with androgen receptor status, linked to androgen independence and therapy resistance.8,9 RelA/p65 and p50 are 2 major subunits of the NFκB complex, which are sequestered in the cytosol through binding to inhibitory subunit IκBα in the absence of stimuli.10-12 The upstream kinase complex, IKK complex, can be activated by proinflammatory stimuli, such as tumor necrosis factor-α (TNFα), or activated PI3K/AKT pathway due to frequent deletion of pten in prostate cancer cells.13-15 Activated IKK complex phosphorylates RelA on serine 536, which is correlated with RelA transcription activity.16,17 Then, RelA:p50 complex translocates from cytosol to nucleus and induces expression of its downstream target genes, such as bcl-2, cyclin D1, mmp9, and VEGFa, involved in the regulation of cell proliferation, apoptosis, and metastasis.
In this study, we used human CRPC PC3 cells to test the hypothesis that Pao extract exerts its anticancer effects through modulation of the NFκB signaling pathway. We report here that Pao extract suppresses PC3 cells in a dose- and time-dependent manner. Pao extract inhibits the activation of AKT/NFκB signaling, retains RelA in the cytosol, and suppresses NFκB downstream target genes, involved in cell growth, survival, and metastasis.
Materials and Methods
Cell Lines and Reagents
Human prostate cancer cell lines PC3 were obtained from the American Type Culture Collection (ATCC, Manassas, VA) and routinely maintained in RPMI 1640 (Invitrogen Inc, Carlsbad, CA), supplemented with 10% heat-inactivated fetal bovine serum (Hyclone FBS, Thermo Scientific, Inc, Waltham, MA) and 1% antibiotic (Invitrogen). The cells were cultured at 37°C in a humidified atmosphere of 95% air and 5% CO2. All experiments were conducted with a proprietary extract of Pao extract enriched in β-carboline alkaloids (Natural Source International, Ltd, New York, NY). A single batch of the extract was used for the whole study, which is investigated to demonstrate 54% β-carboline alkaloids in the extract by high-performance liquid chromatography analysis. The extract was dissolved as described previously. 6 Primary antibodies against PARP, RelA, pRelA(Ser536), pAKT(Ser473), and AKT were purchased from Cell Signaling Technologies, Inc (Danvers, MA); p21 and p27 from BD Biosciences (San Jose, CA); Bcl-2, Bax, Cyclin A, Cyclin D1, and PCNA from Santa Cruz Biotechnology (Santa Cruz, CA); XIAP from Anbo Biotechnology Company (Jiangsu, China); Bcl-xL and Lamin B from Proteintech Group, Inc (Chicago, IL); and β-actin from AbMax Biotechnology Company Ltd (Beijing, China).
Cytotoxicity Assay
Cells were seeded at 4000 cells/well in 96-well plates overnight before being treated with Pao extract and then examined every day for 3 consecutive days. MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) solution (10 µL; Sigma Aldrich, St Louis, MO) was added to each well of the plates and incubated for 3 hours at 37°C. MTT lysis buffer was then added to dissolve the formazan. The optical density was measured at 490 nm using a Hitachi SH-1000Lab microplate reader (Corona Electric Co Ltd., Ibaraki-ken, Japan). As for rescue assay, 293T cells were seeded at 3000 cells/well in 96-well plates overnight before being transfected with p3xFlag-CMV and p3xFlag-CMV-RelA. 24 hours later, the cells were treated with different concentration of PAO extract for 24 hours.
Colony Formation Assay
About 500 cells were seeded into 6-well plate and treated with Pao extract with various concentrations for 14-day incubation. Colonies were fixed with methanol and stained with 0.5% crystal violet (Sigma). Only colonies with >50 cells were counted. Each condition per colony formation assay was plated in triplicate.
Flow Cytometry Assay
Cells were incubated with indicated concentrations of Pao extract for 24 hours, before samples were fixed by 70% ethanol. Cells were incubated in 10 mg/mL RNase A containing phosphate-buffered saline for 30 minutes at 37°C, followed by addition of 1 mg/mL propidium iodine to the final concentration of 40 µg/mL (Sigma). Then the cells were analyzed using a fluorescence-activated cell sorting (FACS) Calibur flow cytometer (BD FACS Calibur, BD Biosciences).
Wound Healing Assay and Transwell Invasion Assay
A wound was made by using a 100 µL pipette tip on cell monolayers. Photographs were taken at 0 and 16 hours, after pretreatment of Pao extract for 18 hours. Cell invasion assay was performed as described previously. 18 In brief, 4 × 104 cells were seeded in serum-free medium in the insert, covered by Matrigel (BD Biosciences), and the lower chamber was filled with 10% FBS/RPMI1640 medium. After 18 hours, inserts were stained with Crystal Violet and invading cells were counted for at least 3 different fields.
Western Blotting Assay
Cells were lyzed in RIPA buffer containing protease inhibitor cocktail mini-tablet (Roche Diagnostics, Indianapolis, IN) and phosphatase inhibitor cocktail I (Sigma). Cell lysates (20 µg) was resolved on SDS-PAGE and transferred onto PVDF membrane (Millipore, Billerica, MA).
Real-Time Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR) Assay
Total RNAs were isolated using TRIZOL (Invitrogen). Reverse transcriptions and real-time PCR reactions were performed using PrimeScript RT reagent kit with gDNA Eraser kit (Cat # DRR047A, Takara Biotechnology Co Ltd, Dalian, China). Primers for MMP9 were the following: Forward primer: 5′-TGGCAGAGATGCGTGGAGA-3′, Reverse primer: 5′-GGCAAGTCTTCCGAGTAGTTTT-3′; GROα: Forward primer: 5′-AGGGAATTCACCCCAAGAAC-3′; Reverse primer: 5′-ACTATGGGGGATGCAGGATT-3′. PCR reaction conditions are lidding stage, 95°C for 10 seconds; cycling stage, 95°C for 5 seconds and 60°C for 31 seconds with 40 cycles; followed by melt curve stage, 95°C for 15 seconds, 60°C for 60 seconds, and 95°C for 15 seconds.
Luciferase Activity Assay
The effect of Pao extract on NFκB responsive element containing promoter (6xNFκB-Luc) activity induced by TNFα was analyzed using a luciferase assay. PC3 cells (2.5 × 105 per well) were seeded in 24-well plates. After overnight culture, the cells in each well were transfected by lipofectamine 2000 (Invitrogen) with 0.5 µg of 6xNFκB-Luc. After 24 hours of transfection, the cells were incubated with Pao extract for 24 hours, exposed to 10 ng/mL TNFα for 20 hours, and harvested. Luciferase activity was measured using the dual luciferase assay system (Promega, Madison, WI) and detected using the Victor microplate reader (Perkin-Elmer Life and Analytical Sciences, Waltham, MA).
Nuclear Extracts Preparation
PC3 cells were seeded in 10-cm dishes. PC3 were stimulated by 10 ng/mL TNFα for 2 hours after treatment with 250 µg/mL Pao extract for 1 hour. Cells were carefully washed with ice-cold PBS/phosphatase inhibitors (Nuclear and Cytoplasmic Protein Extraction Kit, Cat. 40010, Active Motif, Carlsbad, CA). The nuclear extracts were prepared according to the manufacturer’s protocol. Protein concentration was measured by Bradford Assay (Sangon Company, Shanghai, China).
NFκB DNA-Binding Activity Assay
NFκB/RelA binding to its consensus oligonucleotide was quantified using the enzyme-linked immunosorbent assay (ELISA)-based TransAM NFκB Transcription Factor assay kit (Cat. 40096, Active Motif, Carlsbad, CA) according to the manufacturer’s instructions. Diluted nuclear extract from PC3 cells was added to a 96-well plate with bound oligonucleotides. We used the rabbit anti-NFκB/RelA monoclonal antibody to target RelA binding to its consensus oligonucleotide. Binding activity was measured via colorimetric absorbance at 450 nm on a microplate reader with a reference wavelength 655 nm. The absorbance was normalized by protein concentration.
Zymography
Conditioned media from the cell cultures were analyzed for MMP gelatinolytic activities by gelatin zymography as described previously. 19 The volume of supernatant collected was normalized to the protein concentration to ensure equal loading.
Statistics Analysis
Data shown are the mean ± standard deviation. Difference between means were compared by the 2-tailed Student t-test and considered significantly different at P < .05.
Results
Pao Extract Suppresses Cell Proliferation of Human CRPC PC3 Cells
To evaluate the effects of Pao extract on cell viability of CRPC cells, we treated human CRPC PC3 prostate cancer cells with Pao extract. We observed that Pao extract strongly suppressed PC3 cell growth in a dose- and time-dependent manner (Figure 1A), as manifested by floating and roundup of cell morphology (Figure 1B). The MTT assay indicated that at a concentration of 125 µg/mL Pao extract shows cytotoxic effects on PC3 cells. The IC50 value of Pao extract for PC3 is 122.08 µg/mL at 72 hours. To test its long-term effects, we carried out colony formation assays. As shown in Figure 1C and D, PC3 cells lost their colony formation capacities when treated with Pao extract. Compared with the vehicle-treated group, 125, 250, and 500 µg/mL of Pao reduced colony formation efficiency in PC3 cells from 100% to 47.35%, 20.87%, and 0%, respectively.
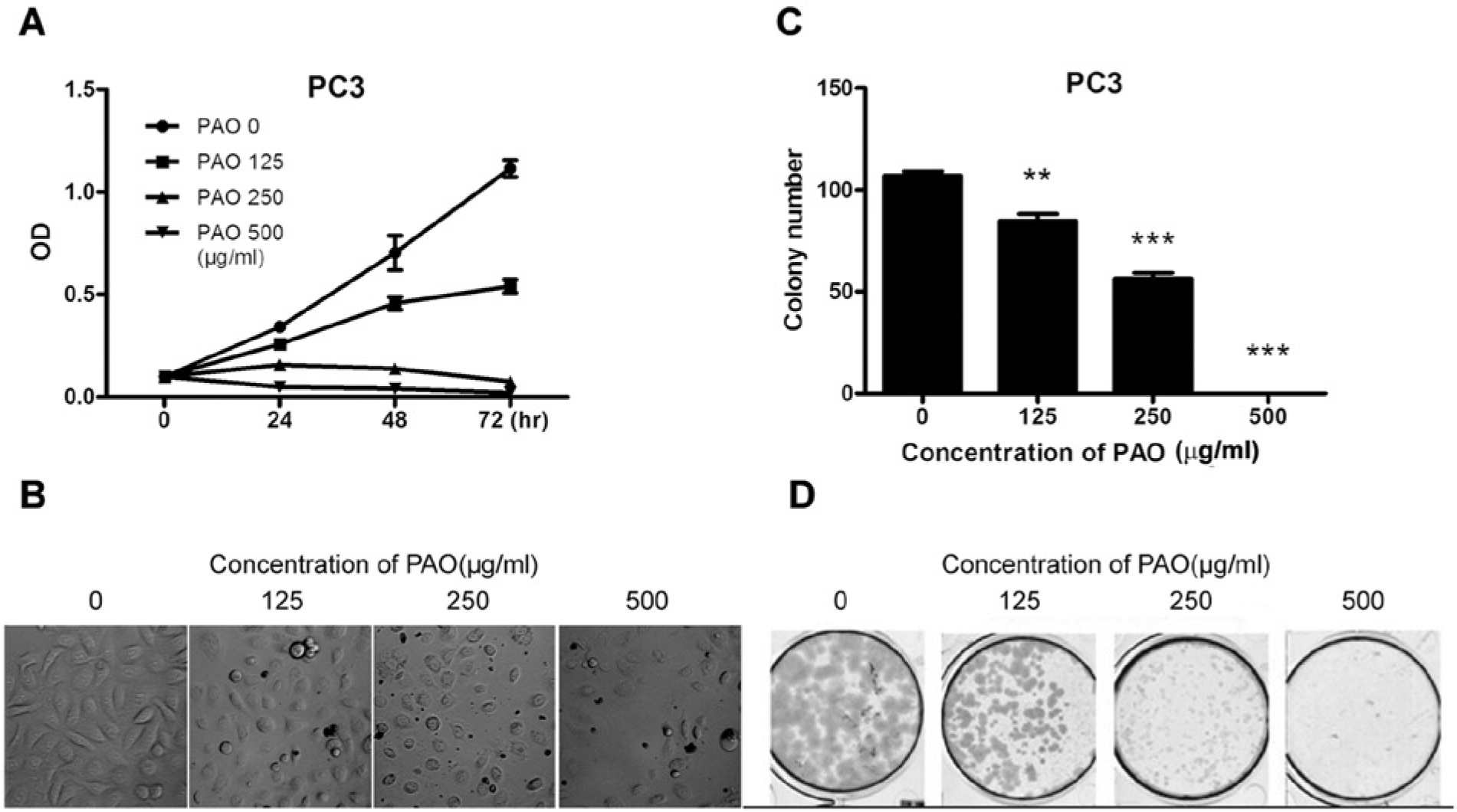
Pao extract inhibited human CRPC PC3 cells. (A) PC3 cells were seeded in 96-well plates and exposed to 0 to 500 µg/mL Pao for 3-day treatment period. The cytotoxicity was measured by MTT assay. Photographs were taken by microscopes (B). (C, D) Colony formation assay of PC3 cells, treated with 0 to 500 µg/mL Pao for 14 days and subsequently fixed and stained with Crystal Violet. Vertical bars indicate the mean cell count ± SD in each treatment group. **P < .01; ***P < .001 as compared on the vehicle-treated group.
Pao Extract Induced G1 Arrest and Apoptosis
Since both apoptosis and growth arrest account for the overall growth inhibitory effect of Pao extract, we performed cell cycle analysis by flow cytometry. The data showed that treatment with Pao induced G1 arrest under 125 µg/mL of Pao extract (Figure 2A and B), while higher dosage induced obvious apoptosis, as manifested by the appearance of subG1 peak (Figure 2C). The PC3 cell population in G1 phase increased in a dose-dependent manner (Figure 2A and B).
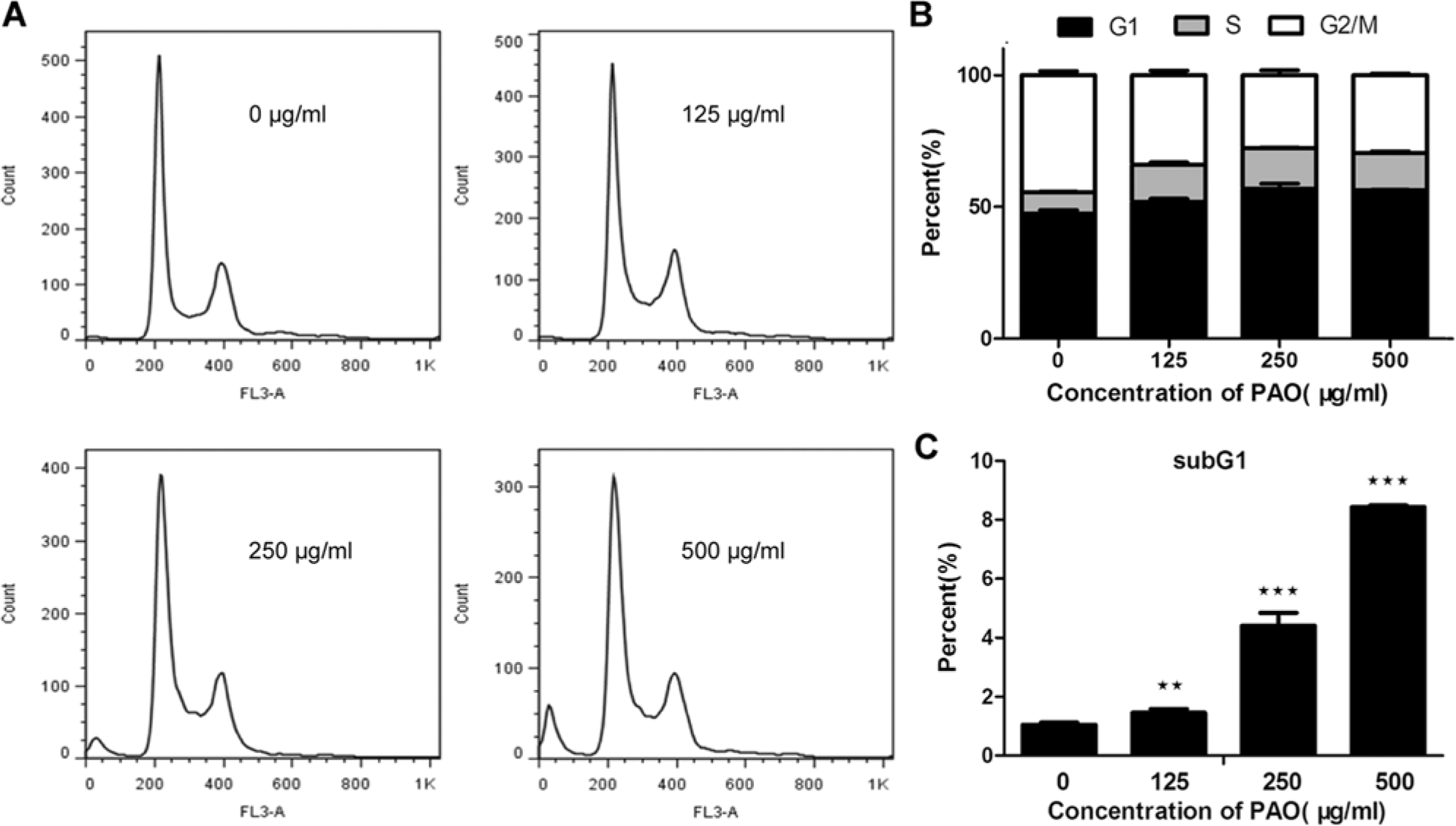
Pao extract induced cell proliferation arrest and apoptosis. (A) Histogram of flow cytometry analysis. PC3 cells were plated at a density of 60% in 6-cm dishes. After cancer cells were treated by Pao ranging from 0 to 500 µg/mL for 48 hours, cell cycle profile and Sub-G1 ratios were assessed by flow cytometry. (B) Cell cycle distribution. (C) The rate of apoptosis. Data represent the mean ± SD of triplicate samples. **P < .01; ***P < .001 as compared to the vehicle-treated group.
Pao Extract Suppresses Metastatic CRPC PC3 Cell Migration and Invasion
Since CRPC cells are prone to metastasize, we analyzed the anti-metastasis effect of Pao extract on PC3 cells using Matrigel invasion and wound healing assays. As shown in Figure 3A and B, PC3 cells treated for 18 hours with Pao extract (250 µg/mL) showed at least 65.38% reduced invasion capability, compared to the vehicle treated control. The concentration 500 µg/mL of Pao extract showed more than 90.9% suppression (Figure 3A and B). This inhibitory effect on cell invasion was not the result of cell growth inhibition induced by Pao extract as the number of viable cells added into the invasion chamber was the same. Wound healing assay also demonstrated that 500 µg/mL of Pao extract inhibits cell migration across the denuded area in a dose-dependent manner (Figure 3C and D). These data indicate that Pao extract is able to inhibit prostate cancer cell migration and invasiveness.

Pao extract suppressed prostate cancer invasion. (A) Transwell invasion assays. PC3 cells were seeded on transwell filters, precoated with 50 µL Matrigel. Basal medium contained 10% FBS and 0, 250, and 500 µg/mL Pao. (B) Cells invaded were counted from at least 3 random fields at 100× magnification using a phase-contrast microscope. (C) Pao extract inhibits PC3 cells motility. Cell migration was visualized by light microscopy and photographed with a digital camera. (D) Width of denuded areas versus Pao extract was plotted in pixels. Each experiment was done in triplicate. Columns, mean; bars, ±SD; ***P < .001.
Modulation of the Expression of Cell Proliferation Proteins and Apoptosis Proteins
To evaluate the mechanisms underlying Pao extract–induced cellular effects we observed above, we analyzed cell proliferation and apoptosis related proteins by Western blot assay. As shown in Figure 4A, cell proliferation markers, PCNA, Cyclin A, and Cyclin D1, were reduced and cell cycle inhibitors, p21 and p27, were increased under the treatment of Pao extract (Figure 4A). Furthermore, we found that Pao extract induced cleavage of PARP (Figure 4B), which is consistent with previous flow cytometry data (Figure 2A). Moreover, we saw a clear increase in pro-apoptotic Bax protein level in Pao extract–treated PC3 cells and reduction of anti-apoptotic proteins, such as Bcl-2, Bcl-xL, and XIAP, in a dose-dependent manner (Figure 4B).

Pao extract inhibited cell proliferation–induced apoptotic proteins and suppressed AKT/NFκB signaling pathway. (A) PC3 cells were treated with 0 to 500 µg/mL Pao extract for 24 hours. Cell lysates were immunoblotted with specific antibodies to PCNA, Cyclin A1, Cyclin D1, p21, and p27. (B) Pao extract–induced apoptosis. Western blot assays were performed using antibodies to PARP, Bax, Bcl-2, Bcl-xL, and XIAP. (C) Pao extract suppressed phosphorylation levels of AKT(Ser473) and NFκB/p65(Ser536) signaling pathway and NFκB downstream target proteins, VEGFa and MMP9. Unphosphorylated proteins of AKT, NFκB/p65, and Actin were used as a loading control. (D and E) Total RNA was extracted from PC3 cells treated with 0 to 500 µg/mL Pao extract for 24 hours and subjected to qRT-PCR using specific primers for 2 NFκB/p65 direct downstream target genes, GROα and MMP9. (F) Nuclear fractions of PC3 cells were prepared after being treated with different concentration of PAO for 24 hours. The effect of Pao extract on NFκB DNA binding activity was examined by TransAM assay. (G) NFκB reporter assay for PC3 cells exposed to Pao extract. Cells were transfected with 0.5 µg of 6xNFκB-Luc. After 24 hours of transfection, the cells were treated with Pao extract for another 24 hours. *P < .05; **P < .01; ***P < .001 compared with control.
Pao Extract Suppresses AKT/NFκB and NFκB Target Gene Expression
NFκB signaling pathway has been implicated in prostate cancer, especially in CRPC, which may account for the upregulation of anti-apoptotic, proliferative, and metastatic gene products. 13 To dissect the underlying mechanisms, we performed Western blot assays. We observed that Pao extract treatment of PC3 cells decreased active phosphorylation of AKT at Ser473 and NFκB/p65 at Ser536 (Figure 4C). Besides the well-known NFκB target proteins, Cyclin D1, as well as Bcl-2, Bcl-xL, and XIAP, involved in cell proliferation and survival (Figure 4A and B), we also found that Pao extract suppressed NFκB target genes, such as VEGFa and MMP9, involved in metastasis (Figure 4C). Last, we confirmed that Pao extract repressed MMP9, as well as another metastatic gene, GROα, at mRNA level (Figure 4D and E). Consistently, we found that Pao extract treatment significantly inhibited NFκB/RelA/p65 DNA binding activity by ELISA-based TransAM kit (Figure 4F) and its transcriptional activity by luciferase reporter assay (Figure 4G) at the concentration as low as 125 µg/mL. All these data support our hypothesis that Pao extract suppresses the NFκB signaling pathway due to inhibition of its transcriptional activity.
Effect of Pao Extract on the Subcellular Localization of NFκB
Since TNFα can specifically induce the activation of NFκB signaling, we treated PC3 cells with TNFα to determine whether Pao extract specifically abolishes the activation of NFκB signaling. We found that 10 ng/mL of TNFα induced nuclear translocation of NFκB in PC3 cells. However, Pao extract blocked NFκB nuclear translocation induced by TNFα (Figure 5A). Western blot assay and DNA binding assay also revealed that Pao extract reduced TNFα-induced nuclear NFκB/RelA/p65 protein level (Figure 5B) and its DNA binding activity (Figure 5C). Congruently, we detected that Pao extract treatment of PC3 cells resulted in a decrease in TNFα-induced NFκB responsive element containing promoter activity by luciferase assay (Figure 5D). Furthermore, the Transwell assay revealed that Pao extract also suppresses TNFα-induced PC3 cell invasion (Figure5E), presumably by reducing MMP9 activity, indicated by zymography assay (Figure 5F).

Pao extract suppressed TNFα-induced NFκB activation. (A) Pao extract inhibited TNFα-induced nuclear translocation of NFκB. Cells were plated onto slides for 24 hours, fixed, and permeabilized. Immunofluorescence staining was performed with primary antibodies against NFκB/p65 followed by FITC (green)-conjugated secondary antibodies. Inset, localization of NFκB/p65 in one representative cell. (B) PC3 cells were stimulated by 10 ng/mL TNFα for 2 hours after treatment with 250 µg/mL PAO extract for 1 hour. The amount of nuclear NFκB/p65 level was analyzed by fractionation and Western blotting assay. Lamin B was used for normalization. (C) NFκB DNA binding activity assay for PC3 cells exposed to Pao extract with and without TNFα treatment. (D) Pao extract suppressed NFκB transcription activity by luciferase assay. (E) Pao extract suppressed TNFα-induced cell invasion by Transwell assay. Quantification of invaded cell number per field was shown in the plot. (F) Pao extract reduced TNFα-induced MMP9 enzymatic activity by zymography assay. *P < .05; **P < .01; ***P < .001, compared to control treatment group. #P < .05; ##P < .01; ###P < .001, compared to TNFα treatment group.
Forced Expression of RelA/p65 Reverses the Growth Inhibitory Effect of Pao Extract
To test whether NFκB plays a causal role in cytotoxic effects of Pao extraction, we forced the expression of RelA/p65 by transfection in 293T cells. Western blotting demonstrated that the overexpression of RelA/p65 increased the expression levels of 2 NFκB target genes, Cyclin D1 and Bcl-xL, and conferred cells resistance to Pao extract (Figure 6). Hence, these results indicated that Pao extract exerts an inhibitory effect on cell growth via inactivation of NFκB activation.
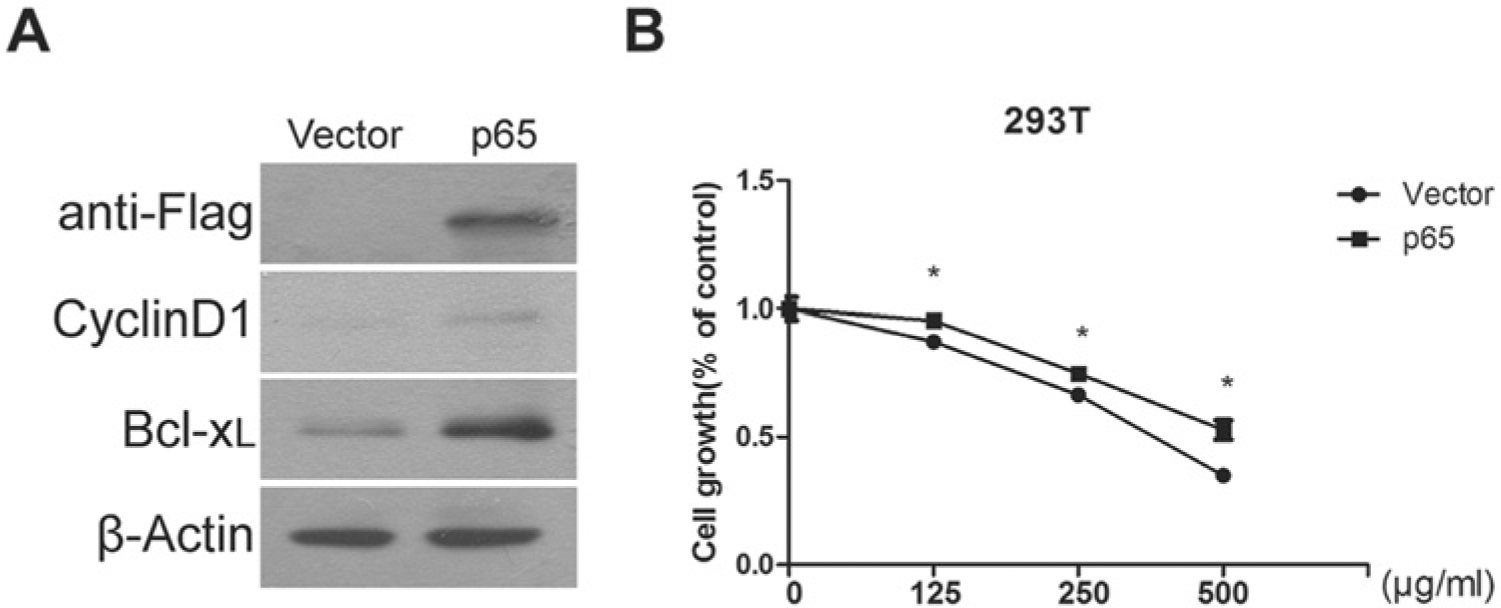
NFκB activation through overexpression of RelA/p65 confers Pao extract resistance. (A) Activation of NFκB signaling through transfection of RelA/p65 expression palsmid. The expression levels of RelA and its targets, Cyclin D1 and Bcl-xL, were examined by Western blotting assay. (B) 293T cells were tranfected with p3xFlag-CMV or p3xFlag-CMV-RelA plasmids, respectively, for 24 hours. The viability of 293T cells under Pao extract with the indicated concentrations was examined by MTT assay.
Discussion
CRPC is the most difficult form of prostate cancer to treat, particularly in the presence of bone metastasis. The development of metastatic CRPC is a multistep process, including cell proliferation/survival, angiogenesis, proteolytic digestion of extracellular matrix (ECM), cell migration to the circulatory system, and recolonization at distal sites. NFκB is frequently activated in CRPC cells after androgen deprivation therapy.7-9 Its activation can be induced by either proinflammatory stimulus, such as TNFα, and/or PI3K/AKT signaling pathway, due to frequent loss/mutation of pten gene in prostate cancer. Therefore, NFκB signaling is a potentially central drug target for metastatic CRPC.
Pao extract, an alkaloid-rich herbal extract, has recently gained our attention for its potential antitumor activity in human androgen-dependent prostate cancer cells. We are the first to report that Pao extract has anticancer effects against highly malignant metastatic CRPC PC3 cells. We observed that Pao extract increased expression levels of cell cycle inhibitors, p21 and p27, and reduced expression of proliferative proteins, such as Cyclin D1, Cyclin A, and PCNA (Figure 4A). The Western blot results were consistent with the finding of G1 arrest in Pao extract treatment group (Figure 2). Bax functions as proapoptotic protein, forming a heterodimer with anti-apoptotic protein Bcl-2, and thereby neutralizing apoptotic effects of Bcl-2. The alteration in the ratio of Bax/Bcl-2 is a decisive factor to determine whether cells will undergo apoptosis. 20 The Western blot data showed that Pao extract significantly increased Bax/Bcl-2 ratio in PC3 cells; meanwhile, another 2 anti-apoptotic proteins, Bcl-xL and XIAP, were also reduced under Pao treatment (Figure 4B). Consequently, activation of Poly(ADP-ribose) polymerase (PARP) protein cleavage was observed. These data demonstrated that Pao extract induced apoptosis in human CRPC PC3 cells, besides androgen-dependent LNCaP cells, shown previously. 7
Deregulation of AKT/NFκB signaling is frequently detected in CRPC. PC3 cells show strong activation of RelA/p65, with the activation of AKT signaling, due to deletion of the pten gene. 21 We employed PC3 cells to examine the effects of the Pao extract. Our result demonstrated that Pao extract showed anticancer effects at least in part through the inhibition of AKT/NFκB signaling. First, Pao extract suppressed phosphorylation of AKT at Ser473 and phosphorylation of p65 on Ser536, which correlates with p65 transcriptional activity (Figure 4C). Second, we observed that Pao extract inhibited RelA DNA binding activity and transcriptional activity at the concentration as low as 125 µg/mL (Figure 4F and G). Consistently, Pao extract also inhibited TNFα-induced NFκB/p65 nuclear translocation (Figure 5A and B). Pao extract decreased NFκB/RelA/p65 transcription activity and TNFα-induced NFκB activation (Figure 5C and D). Third, Pao extract inhibited NFκB downstream target gene expression involved in proliferation (Cyclin D; Figure 4A), survival (Bcl-2, Bcl-xL, XIAP; Figure 4B), and metastasis (VEGFa, MMP9, and GROα/CXCL1; Figure 4C-E). Though Pao extract is not decreasing TNFα-induced NFκB activation below the untreated control level (Figure 5C and D), which might suggest a bystander effect of NFκB, the forced expression of RelA/p65 resulted in the partial rescue of growth inhibition by Pao extract in 293T cells (Figure 6), revealing that Pao extract plays a partial role in inhibiting the NFκB pathway. As a result, we observed that Pao extract induced cell cycle arrest, apoptosis, and suppressed cell invasion of human CRPC PC3 cells in a dose- and time-dependent manner.
Secretion of cytokines and chemokines are essential for cancer cell metastasis and survival. VEGF, CXCL1/GROα, and MMP9 are all higher in conditional media of aggressive CRPC cell lines, C4-2B and PC3, compared to normal PrEC cells. 22 Our data showed that Pao extract significantly reduced the expression levels of these 3 genes (Figure 4C-E). As one of the metastatic proteins related to NFκB signaling we detected, VEGFa is essential for angiogenesis and metastasis. Its overexpression is associated with increased blood vessel stabilization and elevated levels of VEGF and Ang-2. VEGF/VEGFR signaling is one of the promising drug targets for the treatment of CRPC, though single inhibition of VEGF signaling does not increase overall survival, even with significant improvements in tumor response and progression-free survival. 23 Moreover, MMP9 is associated with prostate cancer progression, as it is responsible for degrading type-I and type-IV collagens. MMP9 promoter region contains one NFκB binding site, which can be regulated by TNFα. Our data demonstrated that Pao extract not only inhibited MMP9 expression in a dose-dependent manner but also repressed TNFα-induced MMP9 activity, through NFκB signaling.
Besides VEGFa and MMP9, GROα belongs to CXC chemokine subfamily (CXC chemokine ligand 1, CXCL1), functioning as chemoattraction, cell survival, angiogenesis, cancer metastasis, and chemoresistance through the 7-transmembrane G-protein-coupled receptor CXCR2. 24 In cancer cells, GROα is regulated by NFκB, which can be suppressed by NFκB inhibitor, curcumin. 25 A recent report demonstrated that stromal-derived TNFα, which is induced by chemotherapy, boosts GROα expression in breast cancer cells. Secreted GROα further recruits CD11b+Gr1+ myeloid cells into the tumor, producing chemokines including S100A8/9 to enhance cancer cell survival and chemoresistance. 26 Moreover, GROα treatment increases CRPC PC3 and DU145 cell invasion through activating AKT/NFκB signaling and inducing NFκB/HDAC1 complex formation to silence gene expression of fibulin-1 involved in cell adhesion. 27 These evidences suggest that GROα and NFκB pathway can mutually regulate each other in cancer cells. Pao extract may suppress GROα through NFκB to inhibit their vicious cycle in CRPC cells.
Collectively, our findings outline the potent antitumor effects of Pao extract on CRPC cells, which promotes cell growth arrest, apoptosis, and impairs cell invasion. The inhibition of NFκB plays a partial role in the PAO effect observed in PC3 cells. The role of Pao extract on the suppression of NFκB activation and NFκB-dependent proliferative, survival, and metastatic gene expression warrants its consideration as a potential therapeutic candidate in treating CRPC patients with constitutively active NFκB signaling pathway.
Footnotes
Authors’ Note
Jimin Gao, Hongqian Guo, and Jun Yan contributed equally as co-corresponding authors. The study sponsors had no involvement in the experimental design, data collection, analysis, and interpretation of the study described in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by MOST (2011CB944104), the National Natural Science Foundation (81172009, 81372168), Doctoral Fund of Ministry of Education of China (20110091120028), Fundamental Research Funds for the Central Universities (090314340001), and Natural Source International, Ltd, to JY.
