Abstract
Purpose. Many botanical immunomodulators are used as adjuvants along with cancer chemotherapy. However, information on the impact of concurrent administration of such botanicals on pharmacokinetics of chemotherapy agents is inadequate. This study investigates inhibitory activities of 3 popular botanical adjuvants: Asparagus racemosus (root aqueous extract; ARE), Withania somnifera (root aqueous extract; WSE), and Tinospora cordifolia (stem aqueous extract, TCE) on human CYP3A4 isoenzyme, responsible for metabolism of several chemotherapy agents. Experimental design. Testosterone 6-β hydroxylation was monitored using high-performance liquid chromatography as an indicator of CYP3A4 catalytic activities. Ketoconazole (positive control) and extracts were studied at their in vivo–relevant concentrations. Results. TCE showed mild inhibition while no significant inhibitory activities were observed in WSE and ARE. TCE was further fractionated to obtain polar and nonpolar fractions. The nonpolar fraction showed significant CYP3A4 inhibition with IC50 13.06 ± 1.38 µg/mL. Major constituents of nonpolar fraction were identified using HPLC-DAD-MS profiling as berberine, jatrorrhizine, and palmatine, which showed IC50 values as 6.25 ± 0.30, 15.18 ± 1.59, and 15.53 ± 1.89 µg/mL, respectively. Conclusion. Our findings suggest that constituents of TCE extract especially protoberberine alkaloids have the potential to interact with cancer chemotherapy agents that are metabolized by CYP3A4 in vivo.
Introduction
It is becoming increasingly evident that cancers depend on a number of altered molecular pathways and therefore, combination regimens may provide the best hope for effective therapies. 1 Botanical immunomodulators are often employed as supportive or adjuvant therapy for their immunoregulatory activities on antitumour immune responses and to overcome undesired effects of immunosuppression. 2 Several botanical immunomodulators such as Echinacea purpurea, Ganodema lucidum, Gingko biloba, and Panax ginseng have shown significant activities, including promoting the function of antigen-presenting cells, phagocytosis, humoral and cellular immunity. 3 Most cancer chemotherapeutic drugs are reported to have narrow therapeutic indices leading to significant change in safety and efficacy profiles because of minute variation in the plasma concentration. Furthermore, several of these anticancer agents are substrates of cytochrome P450 (CYP 450) metabolizing enzymes especially CYP3A4 isoenzyme and therefore, modulation of such enzymes by co-administered botanical drugs possesses the risk for pharmacokinetic (PK) drug interactions.4,5 For instance, total extracts, fractions, and individual phytoconstituents derived from E purpurea, G lucidum, G biloba, and P ginseng have shown CYP3A4 modulatory activities. Such modulation may have potential for clinical PK interactions with CYP3A4 cancer chemotherapeutic substrates having narrow therapeutic indices such as cyclophosphamide, cyclosporine, tamoxifen, and so on.6-8 Alkylamide-rich fraction derived from E purpurea showed potential CYP3A4 inhibitory activity demonstrating potential for PK interaction with CYP3A4 substrates. 9 Several international regulatory agencies such as European Agency for Medicines and US Food and Drug Administration recommend early investigations on CYP3A4 inhibition studies of botanicals and identification of the chemical class of phytoconstituents, which may be responsible for PK interactions.10,11 Such studies on many commonly used botanical adjuvants are needed for rationalizing their use as cancer adjuvant therapy. The present study is an attempt in this direction.
Ayurveda, a traditional Indian medicinal system describes a special class of rasayana botanicals for immunomodulation and adaptogenic properties. 12 Total polar and nonpolar extracts, and their formulations prepared from botanicals namely, Withania somnifera (WS), Tinospora cordifolia (TC), and Asparagus racemosus (AR), have been reported to exhibit myeloprotection after treatment with cyclophosphamide (CP) indicating their usefulness as adjuvants during cancer chemotherapy therapy. 13 Several formulations containing WS, TC, and AR are widely used as supportive therapy during the cancer chemotherapeutics. Previously, we have reported that aqueous extracts of W somnifera (WSE) and T cordifolia (TCE) with ratio of (80:20) and their nonpolar fractions resulted in protection toward CP-induced myelo- and immune suppression as evident by significant increase in white cell counts, hemagglutinating and hemolytic antibody titers in tumor-bearing mice. 14 Subsequently, several reports have confirmed our findings that TC nonpolar fraction rich with protoberberine alkaloid shows cytototoxic effects on tumor cells by reducing the glutathione concentration and increase in lipid perioxidation simultaneously.15,16 Various in vitro and in vivo studies on protoberberine alkaloids, especially berberine, palmatine, and jatrorrhizine, have suggested their potential as adjuvants in chemotherapy. 17 Other botanicals rich with these alkaloids include Berberis aristata, Berberis wilsoniae, and Thalictrum javanicum. A few traditional Chinese medicine formulations also contain botanicals rich in these alkaloids.
We report here the effects of ARE, WSE, TCE and its polar and nonpolar fractions, and protoberberine alkaloids on CYP3A4 inhibition using human recombinant CYP3A4 isoenzyme system for prediction of possible PK interactions.
Materials and Methods
Chemicals, Reagents, and Solvents
Reference standards of jatrorrhizine, palmatine (ChromaDex, Irvine, CA), testosterone and 6-β-hydroxytestosterone (XenoTech, Lenexa, KS) were used. Berberine hydrochloride was supplied by Dr Patani’s Scientific and Industrial Research Laboratory, Mumbai, India. Methanol, acetonitrile, and ammonium acetate (Merck, Mumbai, India), and all other chemicals used were of analytical grade. Escherichia coli membrane expressing human recombinant CYP3A4 isoenzyme (bactosomes) was purchased from XenoTech LLC (Lenexa, KS) and stored at −70°C until use.
Plant Material
The roots of A racemosus (batch no. AR/060003) and W somnifera (batch no. WS/0603) and stems of T cordifolia (batch no. ACD/48/stem) were procured from Natural Remedies, Bangalore, India and certified as authentic by the National Institute of Science Communication and Information Resources, New Delhi, India (WS, voucher no. NISCAIR/RHM/F-3/2003/413; TC, voucher no. NRPL/SB/2007/374; and AR, voucher no. NRPL/AR/2007/374). Powdered stems were extracted with distilled water as a decoction as per method described in the Ayurvedic Pharmacopoeia of India. The menstruum obtained was concentrated under vacuum at 85°C and 100 mm Hg pressure. The resulting slurry was then spray dried (inlet temperature 180°C, outlet temperature 117°C and vacuum −10 mm Hg). The dried aqueous extracts of A racemosus (ARE), W somnifera (WSE), and T cordifolia (TCE) were stored in aluminum-coated polyethylene bags. The percentage extractive yield was calculated. These extracts were ensured to be free from pathogens, aflatoxins, pesticide residues, and heavy metals to meet World Health Organization guidelines.
Phytochemical Analysis of ARE, WSE, and TCE
Steroidal saponins such as shatavarin IV were used for chemical characterization of ARE using our earlier presented work based on HPLC-ESI-MS/MS (high-performance liquid chromatography–electrospray ionization–tandem mass spectrometry) method. 18 WSE was chemically characterized on the basis of contents of withanolides such as withaferin A and withanolide A using our previously reported HPLC-DAD (HPLC with diode array detection) method. 19 Protoberberine alkaloids such as berberine, palmatine, and jatrorrhizine were used as marker compounds for chemical characterization of TCE using our earlier HPLC-DAD coupled with mass spectrometer method. 20
Polar and Nonpolar Fractions Derived From TCE and Their Chemical Characterization
Tinospora cordifolia extract was subjected to fractionation as polar and nonpolar fractions using earlier reported methodology. 20 Briefly, TCE was extracted with dilute glacial acetic acid (12% v/v) in the ratio of 1:40 followed by centrifugation at 400 × g for 5 minutes. The supernatant was basified to pH 8 with ammonia solution (25% v/v) and partitioned trice with dichloromethane (300 mL each time); both organic and aqueous phases were separated and dried under vacuum. The detailed chemical characterization of derived fractions was carried out using HPLC-DAD coupled with MS method as described in above section.
CYP3A4 Inhibition Activity Assay
Testosterone was used as a probe substrate and formation of 6-β-hydroxytestosterone was used as a measure of CYP3A4 activity. 21 Briefly, in 24-well plates (Nunc, Sigma-Aldrich, St Louis, MO) containing 205 µL phosphate buffer (pH 7.4), 2.5 µL of test samples, 2.5 µL of testosterone (70 µM), and 25 µL of bactosomes (50 pmol) were added. The reaction was initiated with 12.5 µL of NADPH (2 mM). The reaction mixture was incubated in a shaking water bath at 37°C for 15 minutes. The reaction was stopped by the addition of ice cold 100 µL of acetonitrile and the solution was centrifuged at 3000 rpm for 10 minutes at 8°C. Twenty-five microliters of the supernatant was injected into HPLC system for analysis. Ketoconazole (0.18 µM) and 50% methanol were used as inhibitor and vehicle control, respectively. The assays were performed in triplicate.
Test Material Preparation
Two hundred milligrams of ARE, WSE, and TCE and its polar fraction were dissolved in 2 mL of deionized water. Nonpolar fraction (20 mg) was dissolved completely in 2 mL methanol. Solutions of palmatine, jatrorrhizine (5 mM), and berberine (5 mM) were prepared in 50% and 10% methanol, respectively. All solutions were prior filtered through 0.2 µm of membrane filter. The organic contents were maintained at >1% during the assay.
HPLC Methods for Measurement of 6-β-Hydroxytestosterone
Two different HPLC chromatographic conditions were used for monitoring of testosterone and 6-β-hydroxytestosterone in the assay samples. Briefly, chromatographic separations were carried out on Luna C18 (2) column (250 × 4.6 mm; 5 µm particle size; Phenomenex, Torrance, CA) using isocratic methanol and water (60:40 v/v) as mobile phase (elution I) for 30 minutes with flow rate of 1.2 mL/min for ARE, WSE, TCE, its derived fraction, and palmatine using previously reported method.22,23 For jatrorrhizine and berberine, the following mobile phase with flow gradient was used (elution II): 0 to 3 minutes 25% of B (1.0 mL/min); 3 to 6 minutes 40% B (1.5 mL/min); 6 to 12 minutes 85% of B (1.5 mL/min); 12 to 16 minutes 85% of B (1.5 mL/min), and 16 to 18 minutes of 25% with equilibration for 4 minutes (1.0 mL/min). Autosampler and column oven were maintained at temperatures of 8°C and 30°C, respectively. The analysis was monitored at a wavelength of 240 nm.
IC50 Determinations
Five different concentrations ranging from 10 to 6000 µg/mL of ARE, WSE, and TCE were used for the evaluation of CYP3A4 inhibition activity. These concentrations were selected on the basis of one daily maximum dose of these herbs when diluted in 1 L of gastrointestinal fluid or 56 L of total body fluid to cover the in vivo concentrations. The final concentrations in the range of 26 to 1500, 107 to 6000, and 71 to 4000 were selected for ARE (human dose = 0.5-1.5 g/d), WSE (human dose = 4-6 g/d), and TCE (human dose = 2-4 g/d), respectively. Isolated fractions, pure protoberberine compounds and ketoconazole were used from the concentration range of 1 to 100, 0.3 to 35, and 0.01 to 4.78 µg/mL, respectively. Percentage (%) control activity and % inhibitory activity was calculated using the following formulae: % control activity = (peak area of 6-β-hydroxytestosterone in the presence of TCE or fractions/peak area of 6-β-hydroxytestosterone in control) × 100 and % inhibition activity = 100 − % control activity.
Prediction of Possible Clinical Interactions
IC50 of testosterone oxidation activity was calculated graphically by nonlinear regression analysis of logarithmic inhibitor concentration (log conc.) versus % of inhibitory activity plot using GrapPadPrism5 (GrapPad, San Diego, CA). The data are expressed as mean ± standard deviations. Ratio of I/Ki was used to predict possible clinical interaction and >0.1 was considered for possible clinical interactions. 24 [I] is mean maximum surrogate plasma concentration (Cmax) at steady state after administration of the highest clinical dose of inhibitor in human and used from previously published literature. Inhibitor constant (Ki) was calculated using the equation Ki = IC50/1 + [S]/[KM] where [S] and [KM] are substrate concentration and Michaelis constant, respectively. In our experiment, [S] and [KM] values were same, that is, 70 µM. [I] values for berberine and ketoconazole were used as 0.0003 and 1 to 5 µg/mL (5 µg/mL was used in the experiment), respectively.25,26 It was not possible to calculate I/Ki ratio for ARE, WSE, TCE, derived fractions, palmatine, and jatrorrhizine, because of absence of human pharmacokinetics data especially [I] values.
Results
Phytochemical Standardization of ARE, TCE, and WSE
Asparagus racemosus extract was chemically characterized on the basis of the steroidal saponin, shatavarin IV using HPLC-ESI-MS/MS in positive ionization mode method. Shatavarin IV content was found to be 1.301 ± 0.07 µg/mg. Withanolides such as withaferin A and withanolide A was estimated in WSE by HPLC-DAD analysis and result showed 0.4585 ± 0.054 and 0.4785 ± 0.038 µg/mg, respectively. TCE was standardized on the basis of protoberberine alkaloids such as berberine, palmatine, and jatrorrhizine, as shown in Figure 1. Quantitative estimation of berberine and palmatine content in TCE were found to be 0.039 ± 0.007 and 0.020 ± 0.005 µg/mg, respectively. Jatrorrhizine peak (peak 1) in TCE did not show base to base separation and therefore was not quantified. However, identity of peak 1 was confirmed using MS analysis.

Representative HPLC-DAD chromatograms of TCE, its derived fractions, and reference marker compounds. Jatrorrhizine (1), palmatine (2), and berberine (3) in TCE (chromatogram B) and TCE nonpolar fraction (chromatogram D) were identified using retention time and spectral overlay matching (a, b, and c, respectively, given in window X). TCE polar fraction (chromatogram C) showed absence of jatrorrhizine, palmatine, and berberine. Chromatogram A represents reference marker compounds. Chemical structures of these alkaloids are shown in window Y.
Chemical Characterization of Fractions Derived From TCE
Nonpolar and polar fractions were obtained from TCE with extractive yields in the range of 0.50% to 1.00% and 99.00% to 99.50%, respectively. These fractions were chemically characterized for the presence of protoberberine alkaloids using in-house HPLC-DAD method. Nonpolar fraction showed the presence of 3 alkaloids, namely jatrorrhizine, palmatine, and berberine, as shown in Figure 1. Protoberberine alkaloids were identified in TCE using HPLC-DAD based external standard calibration approach wherein retention time (tR) and ultraviolet spectral matching were used to confirm the presence of jatrorrhizine (peak 1), palmatine (peak 2), and berberine (peak 3). The quantitative estimation of berberine and palmatine in nonpolar fraction resulted in 3.86 ± 0.08 and 2.01 ± 0.30 µg/mg, respectively. The polar fraction did not show the presence of these alkaloids.
CYP3A4 Inhibitory Activities and HPLC Method Optimization
Recombinant human CYP3A4 isoenzyme was incubated with test drugs and rate of formation of metabolite 6-β-hydroxytestosterone were monitored using HPLC methods. Two different mobile phase elusion programs were optimized to avoid interferences of phytoconstituents during the analysis of testosterone and 6-β-hydroxy testosterone as shown in Figure 2. Simple isocratic elution (elution I) with water and methanol was used for ARE, WSE, and TCE, its derived fractions and palmatine, which did not show any interference with testosterone (tR = 14.22 minutes) and its metabolite 6-β-hydroxytestosterone (tR = 5.0 minutes) as shown in Figure 2A. In this isocratic condition, berberine, and jatrorrhizine resulted in the interferences with 6-β-hydroxytestosterone (tR = 5.0 minutes). Therefore, mobile phase with flow gradient elution program (elution II) was optimized for berberine and jatrorrhizine. The gradient resulted in the separation of berberine (tR = 9.12 minutes) and jatrorrhizine (tR = 9.89 minutes) from testosterone and 6-β-hydroxytestosterone as represented in Figure 2B and C, respectively. Thus, the optimized chromatographic conditions were found to be specific for the analysis of testosterone and 6-β-hydroxytestosterone.

HPLC-DAD representative chromatograms showing the absence of interference due to phytoconstituents present in the study samples analyzed using 2 different chromatographic conditions as described in the method suggesting the specificity of the methods. In chromatographic condition I, TCE, its nonpolar fraction, and palmatine did not show any interference with testosterone (tR = 14.22 minutes) and its metabolite 6-β-hydroxytestosterone (tR = 5.0 minutes) as shown in (A). In condition II, berberine (tR = 9.12 minutes) was well resolved from testosterone (tR = 11.55 minutes) and 6-β-hydroxytestosterone (tR = 8.10 minutes) represented in (B) and in the same condition, jatrorrhizine (tR = 9.89 minutes) was also distinguished from testosterone and 6-β-hydroxytestosterone as shown in (C). Chromatograms a, b, c, d, and e are representative chromatograms of recombinant human CYP treated without any test materials (control), with ketoconazole, TCE, TCE nonpolar fraction, and palmatine respectively, as shown in A. Chromatograms f, g, h, i, j, and k, l, m, n are the representative chromatograms of recombinant human CYP treated with different concentration of berberine (B) and jatrorrhizine (C), respectively.
Effects on CYP3A4 Inhibitory Activities
ARE, WSE, TCE, TCE-derived fractions, and protoberberine alkaloids were studied on recombinant human CYP3A4 isoenzyme using a testosterone 6-β-hydroxylase assay. Ketoconazole, a known CYP3A4 inhibitor, was used as positive control. All samples were assayed in triplicate and the percentage inhibition versus logarithmic concentration graphs were plotted for the calculation of IC50 values as shown in Figure 3. The calculated IC50, Ki, and [I]/Ki ratio are presented in Table 1. Ketoconazole completely abolished CYP3A4-dependent activity at 0.531 µg/mL with IC50, Ki, and [I]/Ki ratios of 0.036 µg/mL, 0.018 µg/mL, and 277.77, respectively. These findings were consistent with previous reports.27,28 TCE showed dose-dependent inhibitory activities from 100 to 3200 µg/mL with IC50 values of 594.10 ± 76.53. ARE, WSE, and polar fraction derived from TCE did not show significant inhibitory activities even at high doses of 1500, 6000, and 4000 µg/mL, respectively. IC50 values for ARE, WSE, and TCE polar fractions were considered >1500, 6000, and 4000 µg/mL, respectively. In contrast, nonpolar fractions showed significant inhibition of CYP3A4 with IC50 value at 13.06 µg/mL. In the same assay system, berberine showed the lowest IC50 value of 6.25 ± 0.030 µg/mL with [I]/Ki value of 0.0001. Palmatine and jatrorrhizine showed IC50 values at 15.53 ± 1.89 and 15.18 ± 1.59 µg/mL, respectively. Ki and [I]/Ki ratios for ARE, WSE, TCE, its derived fractions, palmatine, and berberine could not be calculated because of absence of human pharmacokinetics, especially [I] values.
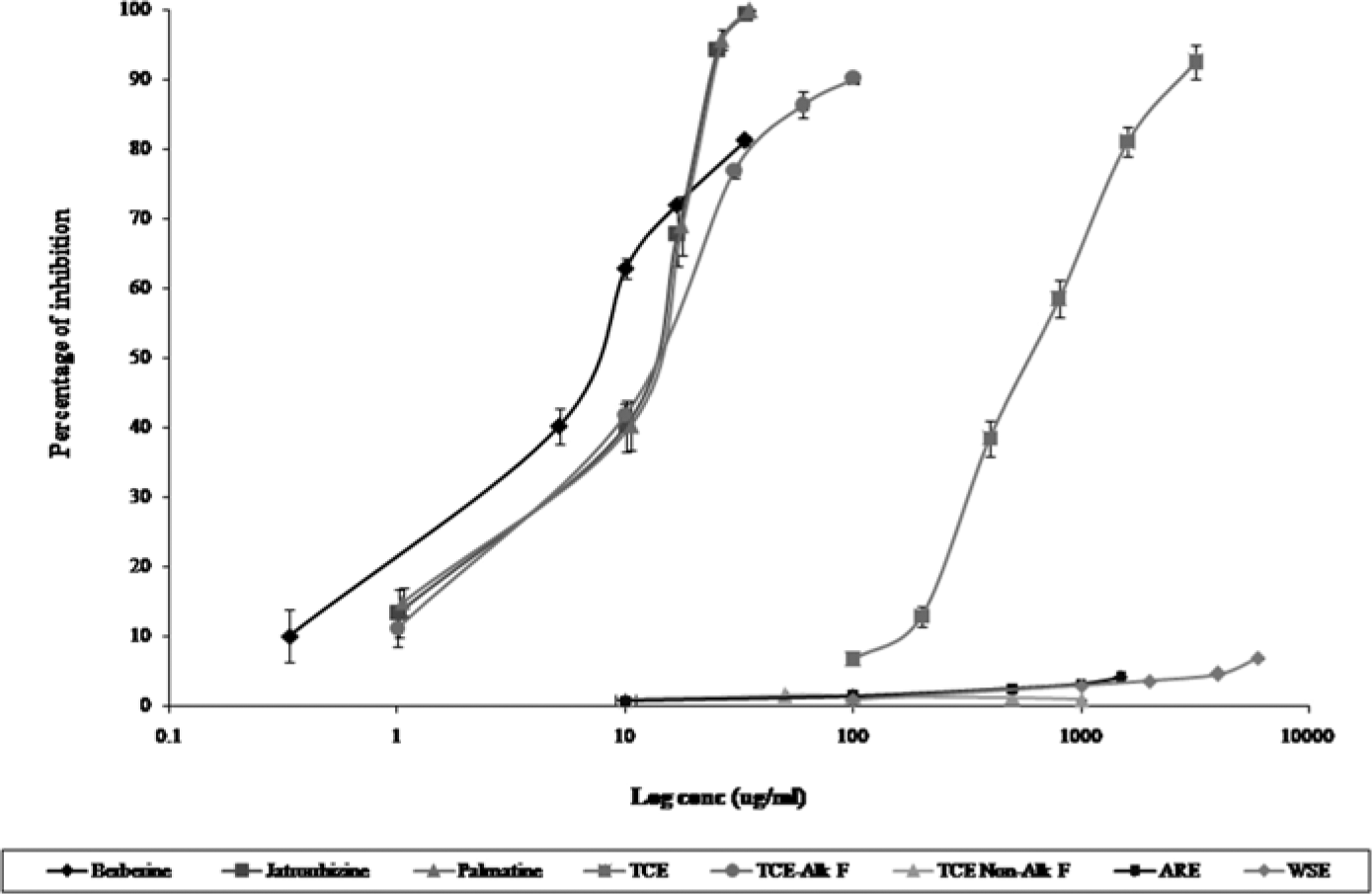
Effect of different concentrations of ARE, WSE, TCE, its derived fractions, and individual protoberberine alkaloids on CYP3A4-mediated metabolism of testosterone presented as percentage of inhibition. ARE (10-1500 µg/mL), WSE (100-6000 µg/mL), TCE, and TCE polar fraction with concentrations ranging from 1 to 3200 µg/mL were used. TCE nonpolar fractions with concentrations from 1 to 100 μg/mL were used. Concentration ranges of berberine, palmatine, and jatorrhizine were 0.33 to 33.6, 1.0 to 35.2, and 1.0 to 33.8 µg/mL, respectively. Percentage inhibition versus logarithmic concentration plot was used for IC50 determination using nonlinear regression method. Values are expressed as mean ± standard deviation.
Comparative IC50, Ki, and I/Ki Ratios of TCE, Its Derived Fractions, and Protoberberine Alkaloids. a

Abbreviations: ARE, Asparagus racemosus extract; WSE, Withania somnifera extract; TCE, Tinospora cordifolia extract; IC50, half maximal inhibitory concentration; SD, standard deviation; NP, not possible.
Inhibitor constant (Ki) was calculated using the equation Ki = IC50/1+[S]/[KM], where [S] and [KM] are the substrate concentration and Michaelis constant, respectively. In our experiment, same value for [S] and [KM] were used (70 µM). I/Ki ratio was used to predict possible clinical interaction and ratio >0.1 was considered for possible clinical interactions. [I] is the mean maximum surrogate plasma concentration (Cmax) at steady state after administration of the highest clinical dose in human and these values were considered from the reported literature. For berberine and ketoconazole [I] values were considered as 0.0003 and 5 µg/mL, respectively. It was not possible (NP) to calculate I/Ki ratio for TCE, derived fractions, palmatine, and jatrorrhizine because of absence of human pharmacokinetics data, especially [I] values.
Our study indicates that ARE, WSE, and TCE and its polar fraction did not show significant CYP3A4 inhibition and may not have serious PK interactions especially with CYP3A4 substrate drugs. Whereas, the nonpolar fraction derived from TCE showed significant inhibition indicating potential for in vivo PK interactions when concurrently administered with cancer chemotherapeutics specifically with CYP3A4 substrates. The observed inhibition in the nonpolar fraction may be attributed to presence of protoberberine alkaloids such as berberine, palmatine, and jatrorrhizine.
Discussion
Ayurvedic rasayana botanicals such as A racemosus, W somnifera, and T cordifolia have been extensively used as immunomodulators and considered as promising adjuvants for the treatment of cancer. However, studies on CYP3A4 inhibitory potential on these botanical immunomodulators were missing. Effects of these botanicals and their fractions on the recombinant human CYP3A4 inhibitory potential were carried out using probe drug substrate assay. In present work, testosterone, one of the substrate drugs mentioned in the US Food and Drug Administration guidance, was used and the rate of formation 6-β-hydroxytestosterone was monitored as marker for CYP3A4 activity. Ketoconazole, a known CYP3A4 inhibitor, was used as positive control. Ki value for ketoconazole was found to be 0.018 µg/mL with [I]/Ki ratio of 277.77 suggesting possibility of clinically significant PK interactions with CYP3A4 substrates. This finding is consistent with previously reported clinically significant PK interactions with CYP3A4 substrates such as ranolazine, ritonavir and imatinib.29,30 ARE, WSE, and the polar fraction derived from TCE did not show significant inhibitory activities even at high doses up to 6000 µg/mL. IC50 values for these substances were considered at >1000 µg/mL suggesting low chances of PK interactions with CYP3A4 substrates. TCE showed modest inhibitory activity with IC50 values of >500 µg/mL and suggested need for identification of possible inhibitory fractions/phytoconstituents toward monitoring quality and safety of its products. It is known that pharmacological activities of T cordifolia are attributed to polar and nonpolar components. 31 Nonpolar fraction using either chloroform or dichloromethane has shown cancer cell cytotoxic activities in several in vitro systems whereas polar extracts rich with polysaccharides have resulted in immunoregulatory activities.32,33 Therefore, simple liquid–liquid extraction using dichloromethane was used to separate 2 biologically distinguished fractions from T cordifolia for the evaluation of CYP3A4 inhibitory potential in dose-dependent manner. The nonpolar fraction showed significant inhibition with IC50 value at 13.06 µg/mL. The observed IC50 value was 1.5-fold higher than the concentration required for maximum inhibition (8 µg/mL) of cancer cells as reported earlier suggesting CYP3A4 inhibitory potential at therapeutic concentration. 32 The detailed HPLC-PDA-MS based chemoprofiling has been done for identification of inhibitory constituents present in the nonpolar fraction derived from TCE. The data based on retention time, ultraviolet and mass spectra matching with reference compounds has confirmed the presence of major protoberberine alkaloids such as jatrorrhizine, palmatine, and berberine in the fraction (Figure 1). To study the role of TCE and its nonpolar fraction for CYP3A4 inhibition potential, individual alkaloids were tested in dose-dependent manner in the same assay system. Berberine showed the highest inhibition as seen from the lowest IC50 value of 6.25 ± 0.030 µg/mL. Palmatine and jatrorrhizine showed IC50 values of 15.53 ± 1.89 and 15.18 ± 1.59 µg/mL, respectively. Previous reports on these alkaloids suggest IC50 values of 0.31 ± 0.10 and 0.70 ± 0.23 µg/mL for palmatine and jatrorrhizine, respectively. 34 The variations in the IC50 values might be because of atypical nature of CYP3A4 isoenzyme or change in probe substrate. 35 Prediction of clinically possible PK interactions based on inhibitor constant (Ki) and mean maximum surrogate plasma concentration at steady state (I) has been well documented for individual compounds. 36 The [I]/Ki ratio >0.1 suggested possible clinical drug interactions and requires in vivo evaluation. The Ki value for berberine was found to be 3.12 µg/mL with [I]/Ki values of 0.0001 suggesting absence of clinically significant interactions. This observation is consistent with previously reported PK interaction study, where subchronic treatment of berberine with pre- and post-oral administration of cyclosporine A showed no significant changes in PK parameters. 37 In contrast to this observation, a report on concurrent use of berberine with cyclosporine A in renal transplant patient showed significant increase in Area Under the Curve (AUC) (by 34.50%), tmax (by 1.7 hours), t1/2 (by 2.7 hours) and decrease of apparent oral clearance (by 40.40%). 38 The suggested speculative mechanism of these interactions could be attributed to decreased liver or intestinal metabolism through CYP3A4 inhibition leading to increase in cyclosporine A bioavailability. These observations advocate the need for more reliable screening tools to predict metabolic clinical herb–drug interactions during disease conditions. Therefore, concurrent administration of berberine, berberine rich fractions or formulations may require close monitoring for clinically significant PK interactions with CYP3A4 substrates. In support, another report on the berberine containing botanical supplement (Hydrastis Canadensis) has shown significant PK interactions with midazolam; which is a known CYP3A4 substrate. 39 Palmatine and jatrorrhizine also showed CYP3A4 significant inhibition in our in vitro assay system. However, it was difficult to predict possible clinical interaction due to inadequate data on human PK especially on steady state concentration [I] at highest clinical dose of these alkaloids. Thus, our study highlights need for herbal PK profiling for prediction of clinically relevant herb–drug interactions. 40
In summary, chemically standardized aqueous extracts of A racemosus, W somnifera, and T cordifolia prepared as per traditional method did not show significant CYP3A4 inhibition suggesting safe use as dietary supplements or adjuvant during cancer chemotherapeutics. This may be because of the protective effects of other compounds or the presence of low amounts of protoberberine alkaloids.
Conclusion
Botanical immunomodulators are frequently used as adjuvants or dietary supplements during the cancer chemotherapeutics. Sufficient data are not available on possible herb–drug interaction through CYP3A4 inhibition potential. Our work suggests that A racemosus, T cordifolia, and W somnifera prepared as per traditional procedures did not show any significant CYP3A4 inhibition even at the concentration equivalent to the highest clinical dose after oral administration. This suggests that they may be safely used as adjuvant concurrently with cancer chemotherapy agents linked to CYP3A4 substrates. Interestingly, the nonpolar fraction containing berberine, palmatine, and jatrorrhizine from TCE showed significant inhibition, which needs to be monitored for safety when concurrently used with such substrates. More studies, especially in vivo, are necessary to confirm these observations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This work was partially supported by Department of Science and Technology, Government of India under the research grant number VIIPRDSF/13/04-05/TT dated 13.02.2008.
