Abstract
Managing cancer-related chronic pain is challenging to health care professionals as well as cancer patients and survivors. The management of cancer-related pain has largely consisted of pharmacological treatments, which has caused researchers to focus on neurotransmitter activity as a mediator of patients’ perception of pain rather than the electrical activity during neurobiological processes of cancer-related pain. Consequently, brain-based pain treatment has focused mainly on neurotransmitters and not electrical neuromodulation. Neuroimaging research has revealed that brain activity is associated with patients’ perceptions of symptoms across various diagnoses. The brain modulates internally generated neural activity and adjusts perceptions according to sensory input from the peripheral nervous system. Cancer-related pain may result not only from changes in the peripheral nervous system but also from changes in cortical activity over time. Thus, cortical reorganization by way of the brain’s natural, plastic ability (neuroplasticity) may be used to manage pain symptoms. Physical and psychological distress could be modulated by giving patients tools to regulate neural activity in symptom-specific regions of interest. Initial research in nononcology populations suggests that encouraging neuroplasticity through a learning paradigm can be a useful technique to help treat chronic pain. Here we review evidence that indicates a measurable link between brain activity and patient-reported psychological and physical distress. We also summarize findings regarding both the neuroelectrical and neuroanatomical experience of symptoms, review research examining the mechanisms of the brain’s ability to modify its own activity, and propose a brain-computer interface as a learning paradigm to augment neuroplasticity for pain management.
Technological advances have yielded insight into the mechanisms of brain function and consequently provide new ways of understanding treatment options for patients. In this review, we offer the reader an understanding of the possible relationship between brain activity and pain in cancer patients that may alter current treatment paradigms as well as provide background for further research.
Cancer Pain
Cancer pain, which can enable the progression of metastatic disease, may be an important predictor of survival; consequently, adequate pain relief may not only improve quality of life but also extend life itself. 1 The management of cancer-related chronic pain is an ongoing challenge to health care professionals and patients. Cancer pain management has emotional, social, and physical implications and may be complicated by the multiple factors associated with the disease process. In cancer patients, pain may be caused by tumor progression, tumor invasion, surgeries, systemic treatments, cancer-related infections, inactivity, and generalized fatigue 2 and may involve inflammatory, neuropathic, ischemic, and compression mechanisms at multiple sites. Pharmacological treatments for cancer-related pain often cause myriad side effects, including altered awareness and emotional states, and may contribute to a patient’s sense of loss of control over their body and illness. Patients may also develop tolerance of, and become addicted to, such treatments.
People often experience pain as a threat to their present bodily state, future well-being, and life in general. 3 Acute pain is adaptive and may orient attentional resources toward the stimuli necessary to elicit an appropriate behavioral and/or physiological response 4 and produce temporary analgesia. 5 Chronic pain may have catastrophic consequences: at a minimum, it can diminish quality of life; in the worst case, it can lead to suicide. In patients in whom pain cannot be eliminated, treatment success depends largely on the patient’s ability to adapt to symptoms and self-manage the pain. Consequently, some researchers have focused their efforts on identifying the factors that promote well-being and functioning despite the presence of chronic pain.6,7 The experience of pain is not only emotional and physical but the two are also neurally linked8,9; thus, treating the physical aspect of pain may improve the emotional, and treating the emotional aspects of pain may improve the physical.
Biopsychosocial Model of Cancer Pain
Biopsychosocial models of chronic pain predict pain and subsequent behavioral responses better than solely biological models do. 10 For example, the high levels of depression and anxiety related to a cancer diagnosis, cancer pain, or treatment side effects can not only increase emotional suffering but also contribute to physiological changes such as heightened central nervous system activity, vasoconstriction, and muscle spasm. Conversely, the physical pain from cancer and its treatments can cause feelings of anxiety, depression, fear, anger, helplessness, and hopelessness. 10
Cancer, from its diagnosis and treatment through the survivorship phase, is described as a chronic stressor because it poses a ubiquitous threat to life. Even before a cancer diagnosis is made, cancer testing and screening can promote fear, anxiety, and other psychological distress. 11 In a review of 19 studies of cancer pain and psychosocial factors, Zaza and Baine 12 found that 14 studies reported a significant association between psychological distress and pain. Several other studies have emphasized the role fear plays in the cancer experience. Ashing-Giwa et al, 13 in a study of 1377 cancer survivors of various ethnic backgrounds, found that “fear of finding cancer” was the most frequently cited explanation for delays in diagnostic testing. Klikovac and Djurdjevic 14 found that 85% of participants reported fear, 65% reported anger and anxiety, and 90% reported nervousness and irritability as psychological aspects of a cancer diagnosis. Lemay et al, 15 after controlling for depression and physical symptoms, found that fear of pain predicted limitations in function in cancer patients but not patients with chronic pain who did not have cancer. 15 Although a conventional model of pain causation predicts that pain precedes distress, studies in humans and animals have revealed that stress can increase nociception, and experiencing distress prior to pain exposure predicts worse pain outcomes.16-18 This is consistent with the notion that similar neural structures are involved in perceiving emotional and nociceptive pain, and one type of pain sensitizes the other within similar neural networks.
Brain Activity and Pain
Many factors influence the way in which pain at the periphery is perceived in the central nervous system. Contrary to the predictions of traditional cause-and-effect models, pain perception often is not linearly related to the activity of the brain.19-21 From a neurobiological perspective, pain perceived in regions of the body has direct spinal inputs to the lower brainstem and limbic (emotional processing) structures. Spinothalamic pathways are involved in the regions of the brain that process body state (insular cortex), attention, and response priorities (anterior cingulate cortex [ACC]). The ACC receives a major serial input from a somatosensory-limbic pathway that contributes to varying degrees of cognitive evaluation of pain affect. 3 Because of its integrated network connections, the ACC is involved in various sensory, emotional, and cognitive functions, including pain. Neurons in the ACC interconnect with neurons in the amygdala, a structure critical to experiencing fear and anxiety, emotional states that increase the synaptic strength in the lateral amygdala. 22 In addition to its association with changes in synaptic strength, increased amygdala activity has been associated with decreased insular and ACC activity, signifying a decreased ability of the amygdala to regulate pain perception owing to an interruption in a common network of regulation. In pain paradigms, brain activation occurs along the somatosensory cortex, insula, amygdala, and finally ACC, where increased insular and ACC activity may indicate an early stage in the transition from acute to chronic pain.23,24
Chronic pain is a persistent stressor that indirectly affects the feedback loop of the hypothalamic-pituitary-adrenal (HPA) axis by involving brain regions in the limbic system. 25 The hippocampus inhibits, whereas the amygdala excites, the neurons in the HPA axis, thereby controlling the activity of the axis. Prolonged exposure to a chronic stressor such as pain may damage hippocampal neurons and inhibit neurogenesis. 26 The HPA axis also activates in response to psychological stressors such as depression and anxiety. 25
To summarize, attentional networks and networks underlying pain processing, emotional processing, learning, and memory have overlapping areas of cerebral activity; therefore, brain activity across many symptoms and behaviors relies on a network model of operation. This suggests that a network approach to the evaluation and treatment of chronic pain will elicit the best outcomes.
Electrophysiological Measurement of Brain Activity
Electroencephalography (EEG) is a measurement of electrophysiological activity in the cortex, which comprises the upper 6 neuronal layers of the brain. The summation of synchronous neural activity oscillates at many frequencies and is classified according to the range of the frequencies. These frequency ranges are referred to as delta (1-3 Hz), theta (4-7 Hz), alpha (8-11 Hz), beta (12-32 Hz), and gamma (32 Hz and above) bandwidths. Brainwave frequencies change with brain states. For example, delta may be predominant during certain stages of sleep; theta is correlated with drowsy states, whereas alpha activity is associated with a relaxed state. Beta activity is associated with an engaged state as reflected during attention tasks and cognitive processing, and gamma is associated with cognitive processing, working memory, and intelligence.27-29 This measured electrical activity normally fluctuates according to stimuli from the environment, though the overall EEG is stable over time. 30
EEG activity correlates with neuronal activity in patients who experience acute or chronic pain; it has been hypothesized that the activity measured in various EEG bandwidths is representative of the underlying mechanisms that trigger the emotional experience and physical perception of pain. Therefore, pain may be thought of as a mental perception that physiologically changes the brain’s resting state or organized baseline activity by modifying the functional frequencies of groups of pyramidal cells throughout various regions of the cortex. In a chronic pain state, cortical restructuring may occur, making sensory input neither sufficient nor necessary for the experience of pain, suggesting that the brain plays a complex role in the experience of chronic pain. 31
Pain without a known sensory input includes tinnitus, phantom limb pain, and neuropathy. Therefore, quantifying EEG findings to explore the potential signatures the brain exhibits when symptomatic is an important step to understanding how the brain works under pathological conditions. Researchers could use brain-mapping techniques such as quantitative EEG to delineate regions of interest and site-specific electrical activity to determine an individualized, mechanistic approach to treating cancer pain that targets both psychological and physical distress. Such an approach has already been used in nononcology populations. For example, in a summary of 8 EEG studies, Jensen et al 32 found that although intense and painful stimulation (ie, acute pain) increases all EEG frequencies, the relative increase of power within beta frequencies is greater than that of other bandwidths, whereas the relative power of alpha frequencies (8-12 Hz) is lower than that of other bandwidths. Therefore, relief from acute pain may increase relative alpha activity and decrease relative beta activity. 33
Conversely, Sarnthein et al 34 found that theta band (7-9 Hz) EEG activity in all electrodes in patients with chronic neurogenic pain was higher than that in a healthy control group, suggesting that chronic pain is associated with heightened activity in lower-frequency bands. This effect was seen in patients who were taking centrally acting medications as well as in those who were not. Like patients with acute pain, patients with chronic pain may demonstrate excessive amplitudes of beta activity 32 ; however, additional research is needed to determine what, if any, differences exist between patients who show increases in fast-wave EEG activity and those who show increases in slow-wave EEG activity.
To summarize, EEG studies of pain report varied frequency band activity across brain regions. The reason for varying outcomes often depends on the chronicity of symptoms; however, influences such as genetics, environment, and experience also contribute to individual brain activity. Additionally, in the oncology population, patients may have specific neurophysiological changes as a result of chemotherapy and other cancer treatments.
Neuroplasticity, Brain-Computer Interface, and Learned Control of Cortical Activity
The nervous system’s fundamental feature is its neuroplasticity, that is, its ability to adapt to changing environmental conditions. 35 As neuronal activity reflects environmental stimuli, the activity-dependent neuronal plasticity gradually tunes neural networks to optimally code for environmental information. Each time a person accesses the state-dependent memory, learning, and behavior processes that encode a problem, he or she has an opportunity to reassociate and reorganize that problem in a manner that resolves the problem. 36 Control over the endogenous pain modulatory system could enable a unique mechanism for control over pain. 37 Because the brain experiences pain based in part on cortical restructuring, pain may also be alleviated by similar cortical reorganization via a mechanism utilizing repetition, learning, and memory.
One technique that can help in cortical reorganization is neurofeedback. Neurofeedback enables patterns of neuronal activity to change via a brain-computer interface in which a patient studies a visual representation of his or her own brain activity. Auditory and visual modalities are used to reward the patient when the presence of the desired brainwave is detected by the EEG in specific brain regions. Over time and after a number of sessions, the brain becomes flexible enough to call on alternative pathways that may make pain and its concomitant emotional complexities more manageable. At a neuronal level, this reorganization strengthens or changes existing pathways to accommodate new learning and may facilitate coherent neuronal oscillations that correlate with perception and affect systemic synaptic plasticity. Brain-computer interface learning modalities such as neurofeedback can be used to modify patterns of cortical activation in the EEG and are under voluntary control as demonstrated in human and animal research.38-40 In neurofeedback, electrical physiology is paired with knowledge of neuroanatomy to design treatment protocols. Neurofeedback facilitates endogenous change in which plastic changes in the brain can be shown after 1 session, 41 although there is a temporal correlation with training and long-term change. In terms of neurofeedback, neuroplasticity may equal persistent change in neurotransmission. To illustrate changes in a nonpain patient, Figures 1 and 2 show brain maps before training and then after 50 sessions of EEG-based neurofeedback.
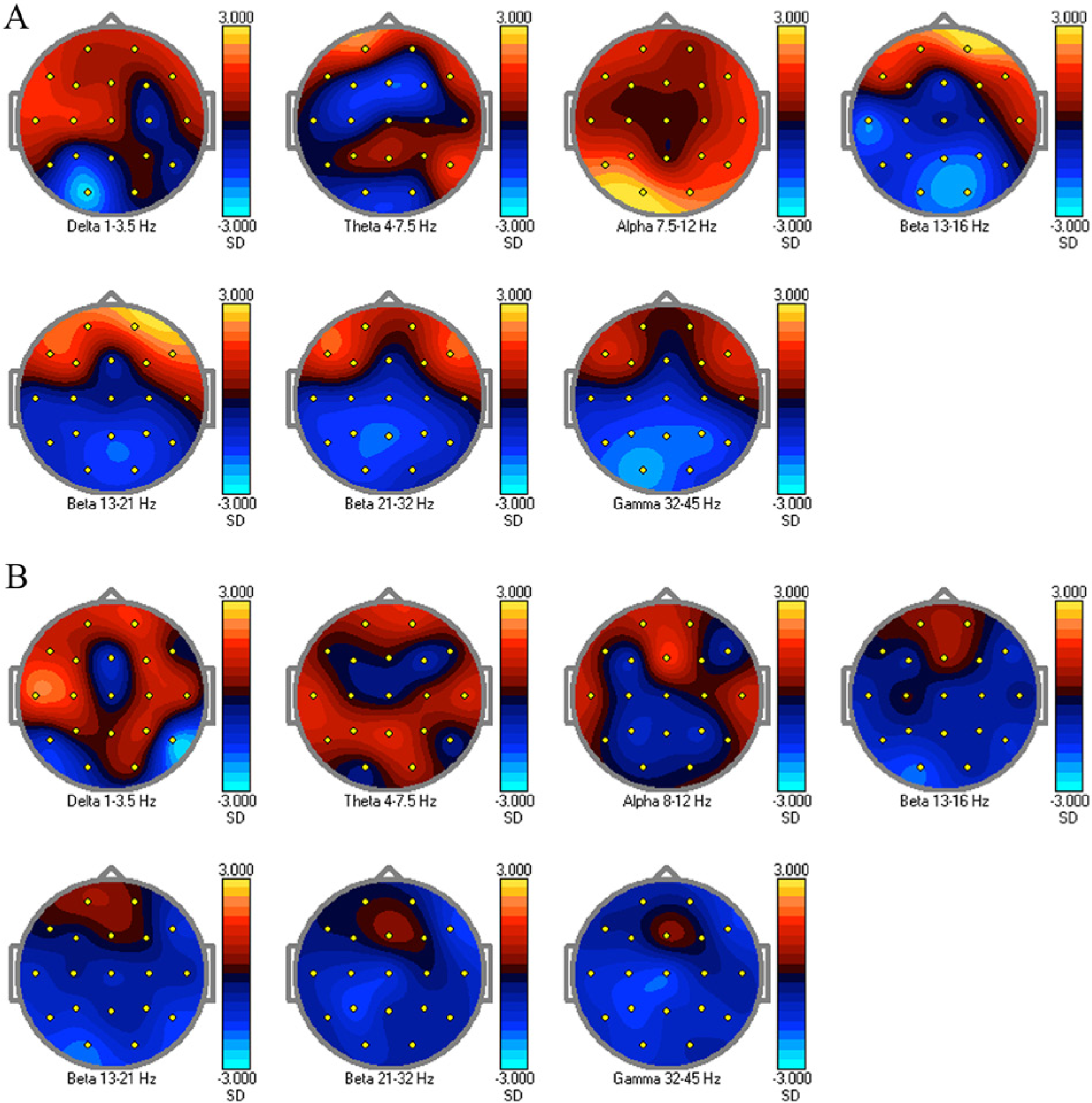
A. Baseline cortical activity: neurofeedback protocols may be designed from quantitative EEG maps. For this patient, alpha (7.5-12 Hz) in the posterior portion of the head was inhibited, whereas beta (13-21 Hz) was also inhibited in the frontal lobes. B. Cortical activity after neurofeedback showing a change in alpha and beta activity. The remapping was done after approximately 12 weeks of training.

A. Low-resolution brain electromagnetic tomography (LORETA) imaging at baseline for the patient in Figure 1. B. LORETA imaging after neurofeedback showing a change in activity of targeted frequency bands.
Neurofeedback to elicit electroencephalographic changes dates back to the 1960s when Sterman’s original research showed that animals could be taught to modify brainwave activity. 42 Interest grew in the use of neurofeedback as a treatment modality after, for example, it was demonstrated that operant training the sensory motor rhythm over the motor cortex served as a protective mechanism against epileptic seizures in animals. 43 Neurofeedback has since been used to address a variety of psychological and physical conditions, including pain, addiction, attention-deficit hyperactivity disorder, and traumatic brain injuries.44-47 It has also been used to enhance academic and athletic performance.48-50 Although neurofeedback has been used in many other settings, it is a novel approach to treating cancer-related pain.
Few studies have investigated the use of neurofeedback to alter the perception of pain. However, as early as the 1970s, Gannon and Sternbach 51 found that increasing alpha activity (8-12 Hz) in a headache patient decreased the intensity and duration of the headaches. 51 Studies examining the effects of neurofeedback on chronic pain conditions such as fibromyalgia, trigeminal neuralgia, and complex regional pain syndrome type 1 reported reductions in pain intensity, fatigue, depression, and anxiety.52-55 Ibric et al 56 found that of 74 patients who had been unsuccessfully treated for pain from chronic disease, injury, surgery, or other sources, 68 (92%) reported a clinically significant improvement in their pain following at least 19 sessions of neurofeedback. One patient in the study who had been diagnosed with leukemia and colon cancer reported a 50% reduction in pain, better control of anxiety and depression, better sleep, and decreased use of medications for 2.5 years following her last neurofeedback session. Other studies report neurofeedback eliciting similar decreases in pain symptoms. Caro and Winter 52 found that neurofeedback treatment significantly decreased physician-assessed tenderness, pain, attention, and fatigue in 15 fibromyalgia patients. In a randomized controlled trial of neurofeedback versus escitalopram for the treatment of fibromyalgia symptoms, both groups showed improvement in all outcome measures; however, therapeutic efficacy for those receiving neurofeedback reached a maximum effect at week 4 of treatment compared with the control group, for whom it did not reach maximum effect until week 8. 53 A retrospective analysis of 18 patients who had complex regional pain syndrome 1 and underwent neurofeedback training as a part of a multidisciplinary pain treatment program revealed a statistically and clinically significant decrease in pain intensity at the primary pain site posttraining, statistically significant improvements in pain intensity at 2 additional sites, and improvements in muscle spasm, muscle tension, perceived deep ache, and overall well-being. 57 Although previous research has demonstrated that neurofeedback improves various types of pain in varying patient populations, most of these studies were case reports or small, single-arm trials. 53
Cortical reorganization is a potential treatment for psychological pain, specifically depression. Rosenfeld 58 investigated whether the EEG patterns of patients who were treated twice weekly with both neurofeedback and psychotherapy (50% time dedicated to each modality) could be modified and, if so, whether those patterns could cause a change in mood. The results of the study indicated that asymmetry between left and right hemispheres was indeed modifiable in 9 of the 13 “normal” participants, and because they were subject to the influence of phasic psychological states under a participant’s self-control, sources of variance in frontal cortical activation asymmetry were also associated with state and trait characteristics. Prior to this study, EEG patterns in patients with depression were thought to be “trait” patterns and thus potentially less apt to be modified using neurofeedback. In terms of pain management, parietal and sensory motor areas integrate somatosensory input with learning and memory and are at the origin of a pathway that converges on the same cortical and subcortical limbic structures (the ACC, insular cortex, and amygdala) that receive direct input from spinal pain pathways. 59 This example of overlapping physical and emotional neural routes may anatomically model the networks responsible for pain reduction via neurofeedback.
The field of neurofeedback is expanding, with novel methods to facilitate and augment neuroplasticity as a mechanism of brain change. One such technique is EEG low-resolution brain electromagnetic tomography (LORETA). LORETA enables researchers to localize the electrical activity in the brain based on scalp potentials from a multiple-channel EEG and determine the relative activity of regions in the brain using surface electrodes. 57 LORETA neurofeedback utilizes this localization in real-time training of specific regions of interest. Although few studies have investigated the use of LORETA neurofeedback, it has been shown to be an effective means of modifying the brain activity implicated in pain processing and perception. Cannon et al 60 found that healthy participants could use LORETA neurofeedback to increase low beta activity (specifically 14-18 Hz) in the cognitive division of the anterior cingulate gyrus. The researchers also found that LORETA neurofeedback of the anterior cingulate gyrus and right and left dorsolateral prefrontal cortices, which are implicated in the processing of both emotional and physical pain, could be used to enhance functioning as measured by the Working Memory Index and Processing Speed Index of the Wechsler Adult Intelligence Scale in a network of related brain regions. These findings suggest that LORETA feedback is a neural-specific form of neurofeedback. Exploring the differences between LORETA and conventional neurofeedback is an area of current investigation; early studies suggest that one advantage LORETA has over conventional neurofeedback is that it requires less training time before differences in patient-reported measures are noted. Because LORETA has demonstrated efficacy in training brain regions identical to those implicated in pain processing, the modality’s utility in treating pain is encouraging.
In addition to EEG biofeedback approaches, real-time functional MRI (rtfMRI) training of neuronal structures may be effective in modulating pain perception. In one study, investigators used rtfMRI to guide the training of the ACC; participants were able not only to change the perception of pain when a noxious stimulus was applied (ie, acute pain) but also reported decreases in the level of chronic pain after training. These studies of LORETA and rtfMRI, though preliminary, provide examples of self-regulation of pain through direct neuromodulation. 37
The next step toward utilization of neuromodulatory approaches for symptom management is the identification of brain regions specific to types of cancer pain. For example, neuropathic pain may have neurophysiological markers different from postsurgical pain. If characteristic EEG patterns are found in cancer pain, these patterns may be corrected by common neurofeedback protocols. For example, it is possible that the central nervous system is somehow “changed” as a result of cancer treatments such as chemotherapy and that these changes may be predictable. However, because of the dynamic properties of the brain and unique responses to treatment, it is likely that different personalized protocols resulting from quantitative EEG analysis will be the most helpful. Because pain is a unique perceptive experience for each cancer patient, even though similarities across patients may exist in regional brain activity, the challenge will be to quantify these unique perceptive experiences, so that replicability of study results is possible.
Summary
Pain is an intricate interaction of psychophysiological components that involve the peripheral and central nervous systems. Neurophysiological studies of the cerebral effects of biopsychosocial factors such as fear and stress have revealed similarities in the neural networks involved in the emotional and physical experiences of pain. In patients with cancer pain, these factors may be present throughout diagnosis, treatment, recovery, and survivorship. Neurofeedback is a potentially effective and economical tool with which to manage cancer pain through modulating neural pathways. By harnessing quantified electrical neurophysiological patterns and the brain’s neuroplastic ability to learn and change, researchers could use neurofeedback to affect many factors—especially those related to mental and physical pain—that are particularly problematic for oncology patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the National Institutes of Health through MD Anderson’s Cancer Center Support Grant CA016672, the American Cancer Society (PF-11-169-01-PCSM), and The Hille Foundation.
