Abstract
Background and hypotheses. Postcancer cognitive impairment (PCCI) is observed in a substantial number of breast cancer survivors, persisting for as long as 20 years in some subgroups. Although compensatory strategies are frequently suggested, no restorative interventions have yet been identified. This study examined the feasibility of EEG biofeedback (“neurofeedback”) and its potential effectiveness in reducing PCCI as well as the fatigue, sleep disturbance, and psychological symptoms that frequently accompany PCCI. Study design. This was a 6-month prospective study with a waitlist control period followed by an active intervention. Participants were female breast cancer survivors (n = 23), 6 to 60 months postchemotherapy, with self-reported cognitive impairment. Methods. Four self-report outcome measures (Functional Assessment of Cancer Therapy–Cognitive Function [FACT-Cog], Functional Assessment of Chronic Illness Therapy–Fatigue [FACIT-Fatigue], Pittsburgh Sleep Quality Index [PSQI], and Brief Symptom Inventory [BSI]-18) were administered 3 times during a 10-week waitlist control period, 3 times during a 10-week (20-session) neurofeedback training regimen, and once at 4 weeks postneurofeedback. Results. All 23 participants completed the study, demonstrating the feasibility of EEG biofeedback in this population. Initially, the sample demonstrated significant dysfunction on all measures compared with general population norms. Repeated-measures ANOVAs revealed strongly significant improvements (P < .001) on all 4 cognitive measures (perceived cognitive impairment, comments from others, perceived cognitive abilities, and impact on quality of life [QOL]), the fatigue scale, and the 4 psychological scales (somatization, depression, anxiety and global severity index) as well as on 3 of 8 sleep scales (quality, daytime dysfunction, and global). Two of the other sleep scales (latency and disturbance) were significant at P < .01, and 1 (use of medication) at P < .05; 2 were not significant. Improvements were generally linear across the course of training, and were maintained at the follow-up testing. At the follow-up testing, the sample no longer differed significantly from normative populations on 3 of the 4 FACT-Cog measures (impairment, impact on QOL, and comments), FACIT-Fatigue, PSQI sleep quality and habitual efficiency, or any of the BSI-18 measures of psychological disturbance. Conclusions. Data from this limited study suggest that EEG biofeedback has potential for reducing the negative cognitive and emotional sequelae of cancer treatment as well as improving fatigue and sleep patterns.
Keywords
Introduction
As cancer mortality rates decrease, postcancer cognitive impairment (PCCI), frequently called “chemobrain” or “chemofog,” increasingly has emerged as a significant problem affecting survivors. PCCI involves difficulties in several neuropsychological domains, including information and processing speed, attention, and memory retrieval and executive function, and its symptoms are distinct from those seen in neurodegenerative disorders.1,2 Published research (focused largely but not exclusively on breast cancer patients and survivors) suggests that the cognitive and psychological sequelae of cancer treatment (particularly chemotherapy) affect the majority of patients as well as a substantial number of those who have completed treatment.2-6 Notably, a recent study of breast cancer survivors more than 20 years postchemotherapy found impaired performance on cognitive measures, as compared with controls who had never had cancer. 7 Early research on PCCI explored the frequency and validity of patients’ self-reported experience of cognitive and psychological symptoms via objective neuropsychological measures,8-10 subjective patient-reported outcome measures (PROs),5,10 and neuroimaging.11,12 More recently, animal models and functional MRI studies13,14 have suggested candidate mechanisms for chemotherapy-related cognitive change; however, as of yet there is little clarity regarding the interacting contributions of these mechanisms and the differential susceptibility of patients.
Several treatment strategies that focus on symptom amelioration have been tested. These include randomized placebo-controlled trials with dexmethylphenidate, 15 which improved fatigue but not objective cognitive function. Trials with modafinil 16 resulted in improvement of some of the participants’ neuropsychological test scores, but daily functional cognitive outcomes were not examined. Nonpharmacological approaches may provide more effective alternatives with an emphasis on functional improvement. Cognitive-behavioral therapy, also considered an adaptive or compensatory approach, has shown some promise in early studies, but effect sizes have been modest.17,18 Thus, the present focus of interventional research is on potentially restorative approaches, which may address the underlying cause of cognitive dysfunction rather than simply treating symptoms. Treatments reflecting researchers’ new understanding of neuroplasticity across the life span include the computerized Brain Fitness Program19-21 (J. Vardy, PhD, e-mail communication, October 14, 2011), for which preliminary results in elderly adults and PCCI patients have been quite promising.
The present study explored EEG biofeedback as a potentially restorative intervention for PCCI. Although EEG biofeedback has not been tested previously in patients with PCCI, a recent study found changes in EEG patterns among breast cancer patients undergoing chemotherapy, and the authors suggested that “EEG may offer a sensitive means to measure alterations in brain function associated with [chemotherapy].” 22 In our study, participants were breast cancer survivors, specifically those continuing to experience symptoms at least 6 months after the completion of cancer treatment.
EEG Biofeedback
In EEG biofeedback (neurofeedback), a real-time display of the brain’s electrical activity, fed back as visual or auditory information, enables the user to modify that brainwave activity. In a 2010 keynote address to the International Society for Neurofeedback and Research, Doidge 23 suggested that the misperception that the adult brain was fixed and unchangeable “led scientists to doubt the claims made by the pioneers of neurofeedback.” 23 Only with the discovery of neuroplasticity did the work of neurofeedback investigators and clinicians begin to find acceptance among other researchers. Still, for some time, there existed limited evidence as to whether EEG biofeedback directly affects neuroplasticity, resulting in ongoing skepticism about its potential as a restorative therapeutic modality. However, several recent studies utilizing transcranial magnetic stimulation and functional magnetic resonance imaging (fMRI) have demonstrated objective, temporally direct changes in cortical activation and connectivity as a result of neurofeedback.24,25
In 2000, Clinical EEG and Neuroscience published a special issue on the topic of neurofeedback. The editor introduced the topic by writing,
The literature, which lacks any negative study of substance, suggests that EBT (EEG biofeedback therapy) should play a major therapeutic role in many difficult areas. In my opinion, if any medication had demonstrated such a wide spectrum of efficacy, it would be universally accepted and widely used.26(pv)
Neurofeedback has since been shown to be an effective intervention for traumatic brain injury, 27 and there have also been clinical reports (though no formal studies) of neurofeedback providing symptom relief for patients with multiple sclerosis (S. Othmer, PhD, e-mail communication, November 12, 2012). These findings provide a rationale for studying neurofeedback as an intervention for PCCI because traumatic brain injury and multiple sclerosis share features with PCCI: that is, demyelination and an expanded extent of brain activation for short-term memory tasks, suggesting “compensatory recruitment of additional brain regions in order to perform the task successfully” (p. 27). 28
Similar to trends in cognitive neuroscience, current neurofeedback strategies reflect 2 different but complementary directions: one driven by a focus on localization and the other by a focus on global brain function. The more common approach, with its roots in the localization school of neuroscience, could be characterized as a “diagnosis and treatment” approach, in which abnormalities in brainwave frequencies at particular locations are identified, ordinarily by means of a quantitative EEG. Researchers and clinicians have identified EEG patterns commonly associated with particular symptoms, and the neurofeedback equipment can be programmed to reward the brain for shifting its activity away from the symptom-associated patterns. For example, attention-deficit hyperactivity disorder (ADHD) in children frequently is associated with slow (theta) wave to fast (beta) wave ratios greater than 3:1 along the cingulate gyrus, located on the innermost surface of each hemisphere above the corpus callosum. A child with ADHD evidencing this pattern would be trained over a series of sessions to lower his/her theta wave amplitude. 29
The present study used a newer approach to neurofeedback, rooted in the global view of brain function. The NeurOptimal system, developed by the Zengar Institute (www.Zengar.com) is designed to train the brain as a whole, without reference to particular locations or frequencies. Unlike classical neurofeedback approaches, in which the participant engages actively and/or consciously with the software and is rewarded for producing prescribed EEG patterns, the participant in the Zengar approach simply “lets go” and allows the brain to use the feedback—provided as brief interruptions to the music he or she is hearing—to enable its own innate capacity for self-organization.
The feedback delivered by the Zengar system is systemic—based on the whole brain’s dynamic activity over time, not its achievement of prescribed states in prescribed locations. The fundamental assumption is that lowering the amplitude of any specific frequency (eg, 8-12 Hz in the left prefrontal cortex) will, by necessity, affect other frequencies in other parts of the brain in the same way that strengthening a single muscle group will affect alignment in other parts of the body, and so it is more realistic to train the brain as a whole system rather than focus on a single location or set of frequencies. This approach recognizes that the brain has a natural tendency toward self-regulation and resilience, allowing flexible cognitive and behavioral responses to a challenging and changing environment.
We are aware that oncology clinicians will be curious about the biological mechanism by which this form of neurofeedback might ameliorate PCCI. This is particularly true, given that the studies verifying PCCI and attempting to identify its causes have used a different paradigm than the one that underlies this form of neurofeedback. However, the existing evidence that PCCI exhibits variable neuroimaging findings and affects multiple neuropsychological domains (particularly in complex frontal-subcortical networks) does suggest that PCCI is not a clearly localizable phenomenon, which points to the importance of developing management strategies that respect the brain as a complex and highly integrated system. Our suspicion is that the concepts of nonlinear dynamical systems theory (eg, self-organization of complex systems, sensitive dependence on initial conditions, basins of attraction, and the importance of feedback), best understood by theorists and researchers in the fields of complexity science and systems theory, 30 ultimately may be more productive in explaining both the changes in cognition seen during and after cancer treatment and also the mechanism underlying this form of neurofeedback.
Some work is already being done to bridge the fields of complexity science and neuroscience. In Modeling Phase Transitions in the Brain, Freeman asserts that “abrupt global reorganizations by phase transition in larger brain systems implement a wide variety of intellectual and intentional brain functions . . . including the switch from prodrome to epilepsy and from from sleep to wake or REM. . . . In each aggregate [of neurons] there are certain conditions that specify a critical point in the phase space at which the system is particularly susceptible to transit from one phase to another, as when the neurons in the sensory cortex transit from a disorganized state of expectation to an organized state of categorization, from noise to signal” (p. v, p. vii). 31 Freeman acknowledges that this view of brain function is as yet unproven, but it is currently a focus of study among computational neuroscientists, 32 and Freeman advocates the development of a detailed theory of nonlinear neurodynamics.
The Zengar system is rooted in this view of brain organization. Its software detects phase state changes, the precursors to phase transitions. Alerted by feedback that a phase transition is imminent, the brain is able either to reorganize to return to its prior phase (as when the mind refocuses on a task after wandering) or to transit to a new phase (as in the movement from wakefulness to sleep). Neither phase is preferred, or sought, or avoided by the software. Instead, feedback simply is given when the phase transition is about to occur. Because there is no diagnosis required for this form of neurofeedback, and no specific protocol is developed on the basis of that diagnosis, this approach is considered to be training the brain in flexibility and resilience rather than treating particular symptoms. As a result, the Zengar system is not a controlled medical device and, therefore, the developers have not sought FDA approval.
We fully acknowledge the speculative aspects of Freeman’s work and its application to the Zengar approach to neurofeedback. However, if future research confirms the mechanism and significance of phase transitions, the Zengar neurofeedback system, rooted in an understanding of the brain as a nonlinear dynamical system, may be situated uniquely to remediate PCCI.
Practically, a single sensor for each hemisphere is placed at C3 on the left and C4 on the right, midway between the top of the ear and the crown of the head. The sensors simultaneously analyze the EEG activity at 8 clusters of frequencies within each hemisphere. The identification of phase state changes occurs through the analysis of 16 clusters of frequencies (eg, 1-6 Hz, 9-19 Hz), 8 for each hemisphere. The software developer established the 16 frequency clusters through analysis of the EEG data associated with 20,000 neurofeedback sessions, half named by trainer and client as having had positive outcomes and half as not having had positive outcomes. The use of this general criterion was necessitated by the clients having undertaken neurofeedback for a variety of reasons and on all available neurofeedback equipment, making more specific outcome measures impossible to devise. The developer reports that this approach did reveal distinct differences in filtering characteristics between the more-effective and less-effective sessions and led directly to the frequency clusters now used in the Zengar software (V. Brown, PhD, e-mail and oral communication, December 18, 2012).
The participant sits quietly, listening to soothing music via earbuds or speakers. Brief interruptions in the music signal to the participant that the software is providing feedback. (Visual feedback can also be delivered, but was not used in the study.) The participant does not need to respond in any active way to the feedback or even attend to it consciously because the brain—a complex adaptive system—uses the feedback for its own process of self-organization without reliance on conscious intervention.
Methods
Sample
Participants were recruited through a newspaper article describing chemobrain and mentioning the study as well as 2 additional brief notes in the Health section of the same newspaper. Flyers were also available in several locations, and 1 local cancer center referred 2 patients.
Inclusion criteria were as follows: (a) female breast cancer survivor, (b) age 40 years or older, (c) 6 to 60 months postchemotherapy, (d) self-reported cognitive impairment since cancer diagnosis or treatment, (e) able to read English, (f) able to provide written informed consent. Exclusion criteria were as follows: (a) current evidence of cancer (based on patient self-report) and (b) self-reported history of severe emotional problems continuing to the present time (patients in active treatment with a mental health professional, for a condition the patient did not consider to be stable, were excluded).
In all, 29 women came to an initial interview with one of the investigators. No patients were excluded based on the above criteria; 6 met inclusion criteria but decided not to participate because of the time or travel demands of the study or because they concluded that their impairment was not severe enough to warrant participation. The remaining 23 participants continued to the end of the study. All participants were Caucasian; the median age was 56 years (range = 43-70 years). The median time elapsed since last chemotherapy was 24 months (range = 9-59 months). All had had a mastectomy and/or one or more lumpectomies; 19 participants (83%) had had radiation treatments. None had undergone autologous bone marrow transplantation. Also, 7 participants (30%) reported having taken paclitaxel, 4 (17%) were taking tamoxifen, 7 (30%) were taking aromatase inhibitors, 6 (26%) were taking antidepressants, and 9 (39%) were taking sleep medications at least 3 times a week.
Procedure
Prospective participants met with the principal investigator (PI), who explained the purpose of the study, its timeline, and the time commitment involved. If the prospective participant continued to be interested, the PI gathered basic information about her cancer history—dates of diagnosis and last chemotherapy treatment, chemotherapeutic agents, and other treatments—and the cognitive symptoms she was experiencing. The participant then provided informed consent.
Because we anticipated recruiting only a small number of participants for this preliminary study, we chose to have participants serve as their own waiting list controls. The pilot neurofeedback schedule involved twice-weekly sessions (once weekly and twice weekly are the most common training schedules for this equipment) for 10 weeks, so the control phase was also 10 weeks, with no active involvement other than baseline self-report scales administered at the clinic at weeks 1, 5, and 10. Sham neurofeedback was not used during this phase because of the lack of an algorithm at the time when this study was conducted; one is currently being designed by the software’s manufacturers.
Immediately following testing at week 10, participants began neurofeedback, with scales administered during the 4th, 7th, and 10th week of sessions. All participants attended all 20 sessions. The median interval between scale administrations was 28 days, except for 25 days between the fifth and sixth testings. Measures were administered immediately prior to neurofeedback on those visits. Participants returned again 4 weeks after the conclusion of the neurofeedback regimen for a final testing session. The procedure and informed consent were approved by the institutional review board of Quietmind Foundation of Plymouth Meeting, PA.
Neurofeedback was administered twice a week—in most cases, at the same time of day—for 10 weeks, using the Zengar NeurOptimal Professional System, version 1.98, with a zAmp encoder, also manufactured by the Zengar Institute, and sensors were placed at C3 and C4. These sessions consisted of 30 seconds of monitoring to ensure good sensor connections followed by 33 minutes of neurofeedback, proceeding through 4 different periods. In these 4 periods, different algorithms were used to identify moments at which feedback would be delivered, using the 64-dimensional matrix (8 frequency clusters in each hemisphere) to provide feedback on a broad spectrum of the brain’s networked activity. Although version 1.98 was used in this study, the 2.0 upgrade allows administration without constant monitoring. The PI was present throughout neurofeedback but did not provide any verbal feedback during sessions.
Assessment Instruments
Studies of PCCI use various measures of cognitive function. In some studies, neuropsychological tests are used largely for their objectivity. However, neuropsychological testing after cancer treatment may appear to show average or even above-average cognitive function, but without a pretreatment baseline, it is not possible to determine whether—for this particular participant—the current level actually reflects a decline in function. 33
In other studies,8,34,35 PROs of cognitive function are used. Although they are not objective, they may portray more accurately the individual’s own experience of cognitive impairment. In fact, some researchers assert that “for many cancer-related symptoms, given their subjective nature, self-report is the only way an assessment can be conducted”36(pW33) (emphasis in original).
A third approach to assessing PCCI involves the use of neuroimaging methodologies. A notable example is the Ferguson et al.8 study of identical twins, one of whom was a breast cancer survivor, whereas the other was not. Neuropsychological tests showed only small differences between the twins, whereas their PROs and fMRI showed striking differences. This finding suggests that neuropsychological tests alone, although they can distinguish between PCCI and neurodegenerative diseases, may be insufficient for diagnosing the presence of PCCI.
Whereas neuroimaging may be the gold standard for diagnosing PCCI, PROs may be preferable to neuropsychological tests as a cost-effective alternative. In this study, PROs were utilized as the outcome measures given their low cost and real-world validity and given the absence of baseline precancer neuropsychological testing with which to compare objective data.
In addition to measures of cognitive function, some of the studies of PCCI37-39 have included psychological measures and measures of sleep quality and fatigue because these symptoms are also associated with PCCI. 40 In this study, we assessed all these areas, using the following validated instruments:
Functional Assessment of Cancer Therapy–Cognitive Function (FACT-Cog), 36 Version 3, is a self-report scale measuring 4 domains of subjective cognitive function over the past week. These domains are perceived cognitive impairments (items reflecting memory, attention, word finding, processing speed, and multitasking, among others), comments from others (items reflecting what other people have said about the participant’s speech, memory, and thought process), perceived cognitive abilities (items reflecting which of the above areas the participant considers to be intact), and impact on quality of life (QOL). In each of the domains, a higher score indicates better functioning.
Functional Assessment of Chronic Illness Therapy–Fatigue (FACIT-Fatigue), 41 Version 4, is a self-report scale measuring perceived fatigue, energy level, and QOL over the last week. A higher score indicates better functioning.
Pittsburgh Sleep Quality Index (PSQI) 42 is a self-rated scale that assesses sleep quality and disturbances over the past month; 19 individual items generate 7 component scores—subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleeping medication, and daytime dysfunction. In these components, a lower score indicates better functioning. For diagnostic purposes, component scores usually are reported on ordinal scales, which lose precision. Therefore, analyses below also include 5 continuous individual items from this instrument for greater accuracy of measurement.
Brief Symptom Inventory–18 (BSI-18) 43 is a measure of somatization, depression, and anxiety frequently used with oncology patients. On these dimensions, a lower score indicates better functioning.
Participants completed the 4 instruments 7 times during the course of the study: 3 times during the control phase, 3 times during the neurofeedback protocol, and once 4 weeks postneurofeedback. The average time required for completing the 4 instruments was initially about 15 minutes and decreased to about 10 minutes as participants became more familiar with them. In accordance with standard procedure for these instruments, participants did not keep a log of their symptoms but were simply asked to recall their experience during a designated timeframe. Participants did not see or hear about their results until they had completed the study, but during their exit interview, the PI did review their results with them.
Analysis Plan
We had intended to average scores on the 3 pretests to provide a more stable baseline from which to assess improvement. However, as described below, because of some unexpected improvement during the control phase, we used the scores at Time 3 as the baseline instead. The main analyses consisted of 1-way repeated-measures ANOVAs. It was hypothesized that participants would improve more or less steadily over the course of the training and that improvement would be maintained at the follow-up testing. All analyses were conducted using SPSS 17.0 (SPSS Inc, Chicago, IL).
Results
Outcome Measures
Table 1 shows the means and standard deviations of measures at the first and third testings (times 1 and 3). Table 1 also displays comparisons of this sample’s scores with normative data (not including the PSQI continuous item scores, for which normative data are not available). These comparisons indicated significant dysfunction on all but one of the subscales, suggesting seriously impaired cognitive and emotional function and increased levels of fatigue and sleep disturbance.
Scores on Measures at First Testing, Compared With Available Norms.
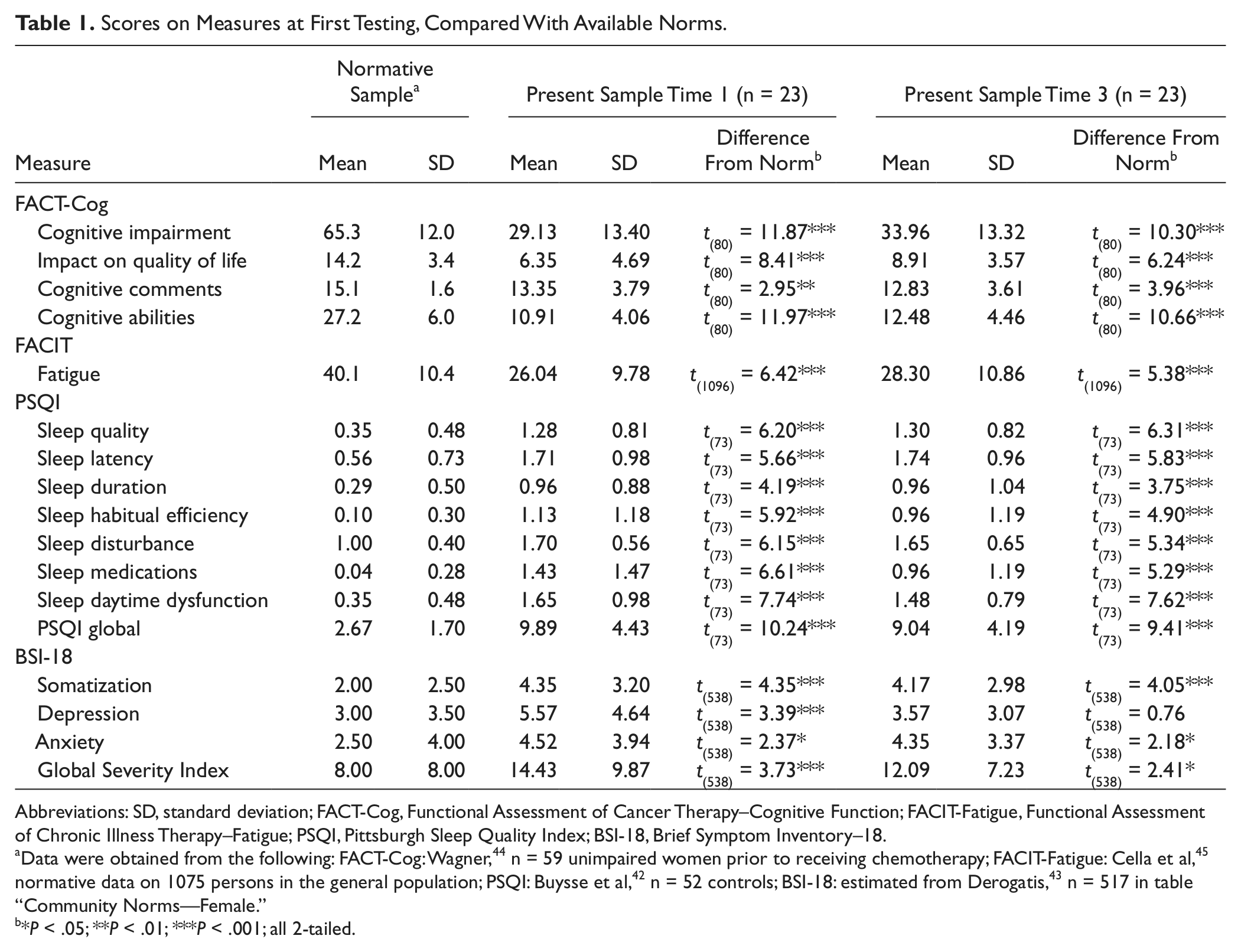
Abbreviations: SD, standard deviation; FACT-Cog, Functional Assessment of Cancer Therapy–Cognitive Function; FACIT-Fatigue, Functional Assessment of Chronic Illness Therapy–Fatigue; PSQI, Pittsburgh Sleep Quality Index; BSI-18, Brief Symptom Inventory–18.
Data were obtained from the following: FACT-Cog: Wagner, 44 n = 59 unimpaired women prior to receiving chemotherapy; FACIT-Fatigue: Cella et al, 45 normative data on 1075 persons in the general population; PSQI: Buysse et al, 42 n = 52 controls; BSI-18: estimated from Derogatis, 43 n = 517 in table “Community Norms—Female.”
*P < .05; **P < .01; ***P < .001; all 2-tailed.
Table 2 shows correlations among each of the subscales. Notably, correlations of subscales within each test (triangular sections of the table) tended to be high, but those among different tests (rectangles) were low and mostly nonsignificant. For example, the top triangular section shows intercorrelations among the 4 domains of the FACT-Cog. They range from r = 0.37 to 0.69, all significant except for the lowest one. The middle rectangle on the left, in contrast, shows correlations between the FACT-Cog domains and the PSQI categorical components. Of the 4 × 7 = 28 correlations, only 2 are significant. FACIT-Fatigue was an exception to this pattern, indicating that fatigue commonly is associated with the other dysfunctions. Interestingly, psychological disturbance as measured by the BSI-18 was almost entirely independent of the other measures, suggesting that cognitive impairment in this sample could not be attributed simply to depression or anxiety. All significant correlations were in the expected direction, indicating that persons with greater dysfunction in one area tended also to be doing poorly in other areas.
Correlations Among Measure Subscales (n = 23). a
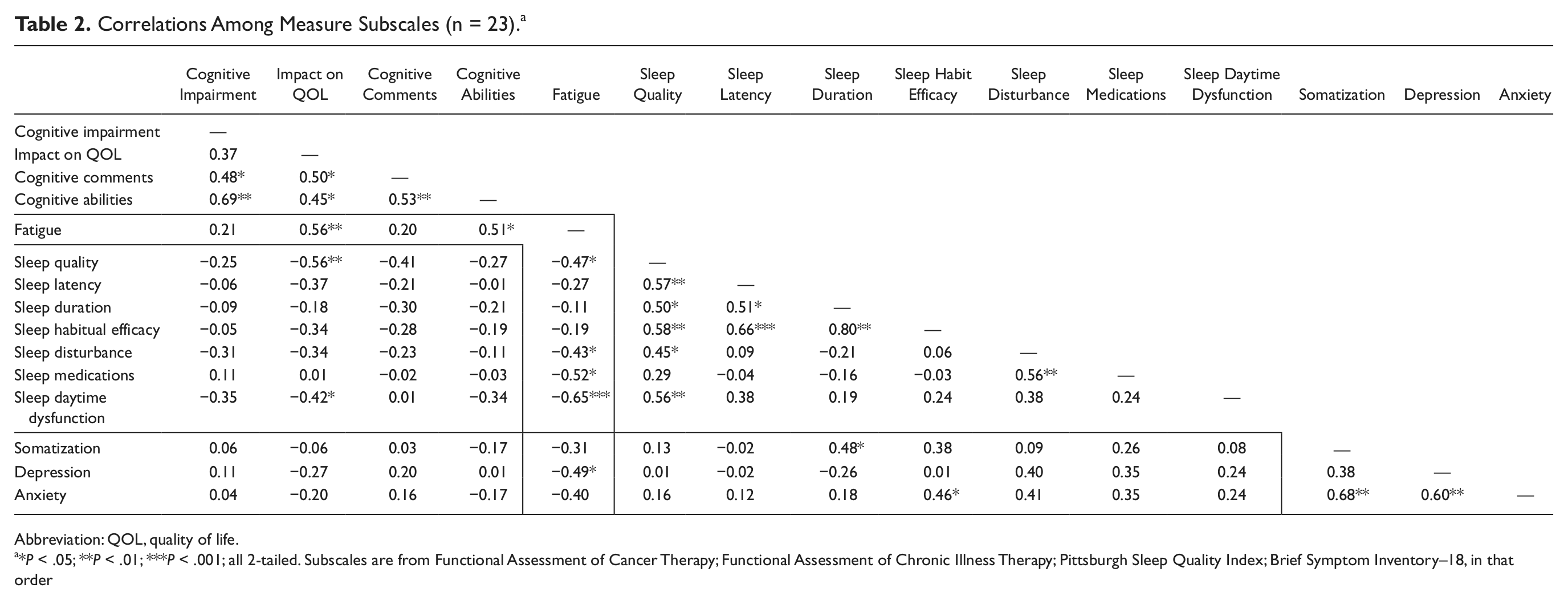
Abbreviation: QOL, quality of life.
*P < .05; **P < .01; ***P < .001; all 2-tailed. Subscales are from Functional Assessment of Cancer Therapy; Functional Assessment of Chronic Illness Therapy; Pittsburgh Sleep Quality Index; Brief Symptom Inventory–18, in that order
It was observed that over the course of the 3 pretests, before neurofeedback was begun, some of the outcome measures indicated improvement. Specifically, 5 of the 22 measures (FACT-Cog impairment, impact, and cognitive abilities; PSQI medications; and BSI depression) showed improvement between time 1 and time 3 by paired-samples t tests. However, all but one of these differences were the result of a large change between time 1 and time 2, and change between time 2 and time 3 was significant only for depression (t(22) = 2.71; P < .05). None of the other differences between time 2 and time 3 approached significance (all P > .15). We speculated that improvement on these measures may have occurred either because of participants’ optimism that neurofeedback would relieve their symptoms or because acknowledgment of their symptoms and acceptance into the study may have diminished their need to emphasize the extent of their impairment. We considered adjusting subsequent improvement scores based on the changes seen prior to training, but this proved complex and would have distorted the reported means, sacrificing comparability to other research. We also observed that even at time 3, scores indicated severe impairment, as shown in Table 1. Therefore, instead of using the mean of the pretests as a baseline, we decided to measure subsequent improvement starting from time 3. It should be noted that this was a conservative decision because scores at time 3 generally indicated less dysfunction than the mean of all 3 pretests.
Improvement During and Following Neurofeedback
Table 3 displays means and standard deviations of all measures at all 7 testings. Figures 1, 2, and 3 illustrate how the means showed improvement from the baseline measure over the course of the intervention and through the posttest, 1 month following the last neurofeedback session. Figure 1 presents means for FACT and FACIT scales for tests 3 through 6 and the posttest. Figure 2 displays mean PSQI categorical component scores across the tests (the PSQI global score—the sum of the 7 component scores—was omitted to reduce clutter). These also show a general trend toward improvement over time, although with less regularity. Figure 3 displays similar mean scores for the BSI-18 dimensions.
Scores on Measures at Each Testing (n = 23).

Abbreviations: SD, standard deviation; QOL, quality of life.
These continuous items contribute to the Pittsburgh Sleep Quality Index components but are reported separately here for their greater precision.

Means of Functional Assessment of Cancer Therapy–Cognitive Function and Functional Assessment of Chronic Illness Therapy–Fatigue subscales across tests.

Means of Pittsburgh Sleep Quality Index categorical components across tests.

Means of Brief Symptom Inventory–18 dimensions and Global Severity Index (measures of psychological disturbance) across tests.
Table 4 summarizes the results of 1-way repeated-measures ANOVAs conducted on each of the 22 measures independently. The overall F tests indicate whether the 5 means on each repeated measure differed significantly from one another. However, this could be true whether or not there was a general trend toward improvement; that is, wild swings in the mean values up and down could also yield significant results. As Table 4 shows, with only 3 exceptions (all of them PSQI scores), all measures revealed significant overall and linear effects. That is, improvement over time was more or less constant, as suggested by the relatively straight lines in Figures 1 to 3. Significant values for quadratic effects are also shown. These indicate that some of the lines included a significant curved component; in every case, this could be attributed to less improvement or to a slight regression toward the baseline during the period following the completion of neurofeedback. As this suggests, participants improved on all subscales over time and at a fairly constant rate, except for the interval between the last neurofeedback session and the posttest, in which scores remained essentially flat. Two measures also showed significant cubic (S-shaped) effects. It was noted also that using the continuous score measures for some of the PSQI components yielded generally stronger results. In particular, sleep duration and habitual efficiency individual item scores yielded significant improvement results, whereas the standard categorical scores did not (although this was reversed for sleep latency). Of note, the proportion of patients requiring sleep medications decreased from 39% (9 participants) to 17% (5 participants) from the beginning to the end of the study.
Results of 1-Way Repeated-Measures ANOVAs (n = 23) on Each Measure. a
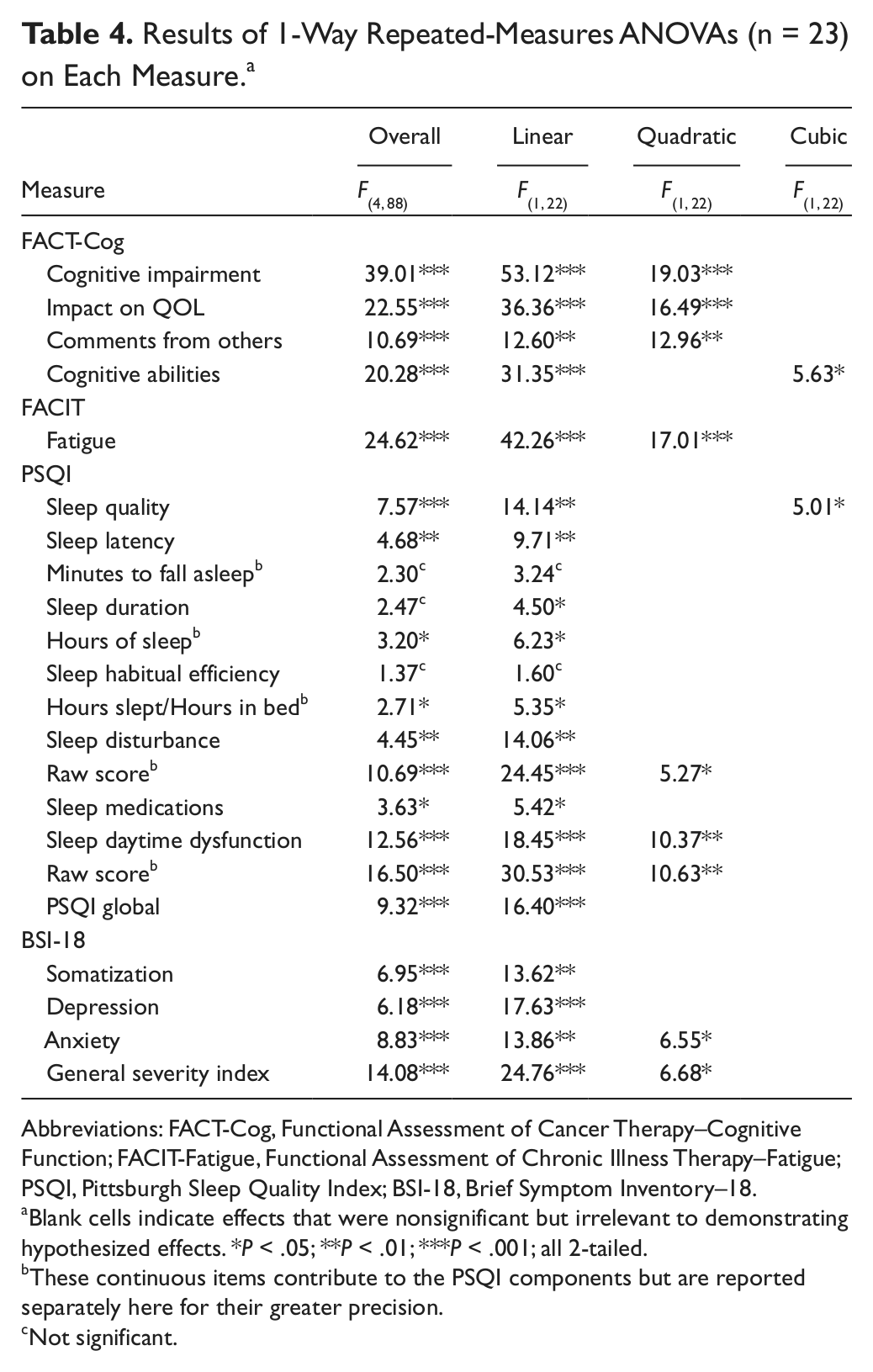
Abbreviations: FACT-Cog, Functional Assessment of Cancer Therapy–Cognitive Function; FACIT-Fatigue, Functional Assessment of Chronic Illness Therapy–Fatigue; PSQI, Pittsburgh Sleep Quality Index; BSI-18, Brief Symptom Inventory–18.
Blank cells indicate effects that were nonsignificant but irrelevant to demonstrating hypothesized effects. *P < .05; **P < .01; ***P < .001; all 2-tailed.
These continuous items contribute to the PSQI components but are reported separately here for their greater precision.
Not significant.
Certain baseline participant characteristics were related to baseline self-report measures. In particular, BSI-18 somatization, anxiety, and GSI were correlated significantly with months elapsed since the end of chemotherapy (respectively, r = −0.63, P < .001; r = −0.45, P < .05; and r = −0.54, P < .01), such that psychological disturbances were less strong the longer it had been since completion of chemotherapy, as one might expect. Spontaneous reports of anxiety during the intake interview were also associated with significantly poorer scores on several PSQI measures of sleep quality (although not, interestingly, with BSI-18 anxiety scores). Those who had reported previously receiving paclitaxel scored more poorly on cognitive function than others. Patients taking tamoxifen had significantly poorer scores on PSQI measures of sleep quality and categorical disturbance. Therefore, these baseline participant characteristics were included as covariates in repeated-measures ANCOVAs on outcome measures with which they had been associated. Overall, results were very similar to those reported above; that is, participant characteristics did not appear to be correlated with improvement over the course of the study.
Initially, the sample showed serious dysfunction on all measures compared with population norms (Table 1). These comparisons were repeated with the means and standard deviations from the posttest (Table 5). It was found that at this time, our sample no longer differed significantly from the normative populations on 3 of the 4 FACT-Cog measures (impairment, impact on QOL, and comments), FACIT-Fatigue, PSQI sleep quality, or any of the 4 BSI-18 dimensions.
Scores on Measures at Posttesting Compared With Available Norms.
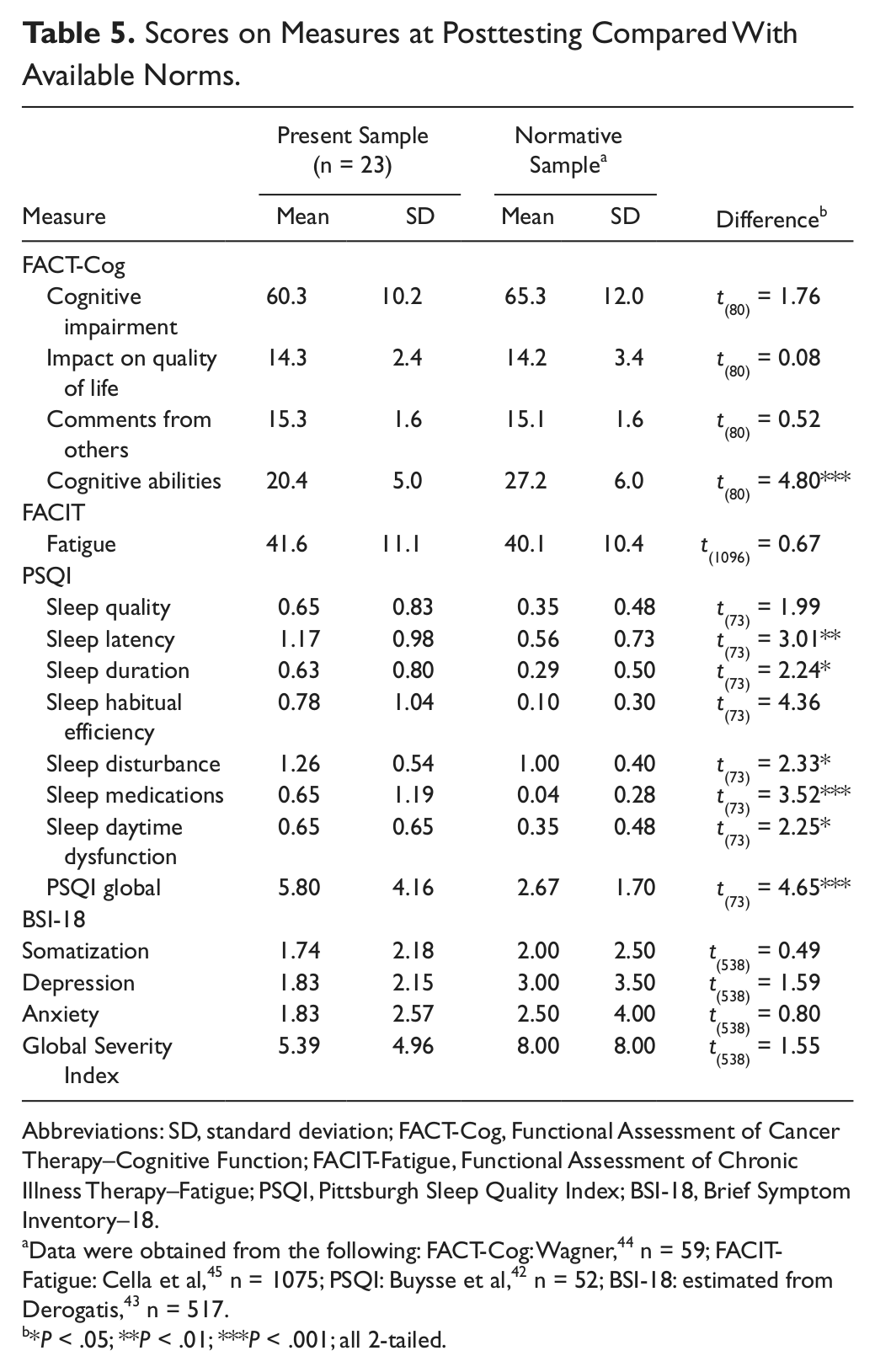
Abbreviations: SD, standard deviation; FACT-Cog, Functional Assessment of Cancer Therapy–Cognitive Function; FACIT-Fatigue, Functional Assessment of Chronic Illness Therapy–Fatigue; PSQI, Pittsburgh Sleep Quality Index; BSI-18, Brief Symptom Inventory–18.
Data were obtained from the following: FACT-Cog: Wagner, 44 n = 59; FACIT-Fatigue: Cella et al, 45 n = 1075; PSQI: Buysse et al, 42 n = 52; BSI-18: estimated from Derogatis, 43 n = 517.
*P < .05; **P < .01; ***P < .001; all 2-tailed.
Finally, neurofeedback was well tolerated, and when questioned before and after each session, none of the 23 participants reported any side effects or other adverse events.
Discussion
In this study, breast cancer survivors demonstrated significant baseline impairments in self-reported cognitive function, fatigue, sleep quality, and psychological well-being as compared with a normal population. After 10 weeks (20 sessions) of neurofeedback, their performance in these areas had improved to levels indistinguishable from population norms. Additionally, participants were continuing to improve on most measures through the end of the training period. Therefore, it is possible that a longer period of neurofeedback training would have resulted in even greater improvement. Despite a time-intensive training regimen, there were no dropouts from the study. The results thus suggest that EEG biofeedback deserves further study as a novel method of addressing PCCI that may be safe, effective, and acceptable to patients.
Several considerations strengthen our conclusion that the positive training effects were valid and reliable. First, analyses focused on improvement following an initial series of 3 pretests, during which some placebo or optimism effects presumably had already elevated scores somewhat. Second, all measures were obtained before training at each session, typically 3 to 4 days following the previous session. Thus, responses did not reflect merely short-term, posttraining effects. Third, most measures showed a leveling off of improvement or a slight reversion to previous levels at the posttest approximately 4 weeks following completion of training. This indicates that improvement was not merely a result of the nonspecific supportive intervention of visiting the clinic or of natural improvement over time. Fourth, there was improvement in 4 distinct clusters of symptoms (cognitive function, fatigue, sleep, and emotional well-being), which were not highly correlated at intake.
Several limitations of the present study should be addressed in future research. Our sample was small, middle-aged, and exclusively Caucasian and female, limiting generalizability to other cancer populations. Baseline (precancer) cognitive measures were unavailable; serial neuropsychological testing and functional neuroimaging were not performed, preventing any conclusions about the effect of neurofeedback on objective cognitive parameters. The study does not allow us to draw firm conclusions on the optimal number or length of neurofeedback sessions or on whether chemotherapy or hormonal agents may be associated with PCCI or interact with the training. In addition, the study does not provide definitive insight into the specific mechanisms by which EEG biofeedback might exert an effect on cognitive function. Possible mechanisms remain speculative and include, in addition to a specific effect of the software’s training algorithms, residual placebo effect, nonspecific relaxation effect produced by listening to soothing music, and positive effect of metacognitive monitoring (ie, simply paying close attention to one’s own cognitive patterns). Finally, results must be interpreted conservatively given the lack of a rigorous double-blind placebo-control design. The positive results obtained through this form of neurofeedback nonetheless suggest that further study of this intervention for PCCI is warranted. In particular, in a future study, we hope to include both neuropsychological assessment and fMRI scans before and after the neurofeedback regimen to identify more precisely the nature of the changes occurring. A study of neurofeedback delivered during cancer treatment would help determine whether the incidence and/or severity of PCCI can be reduced through this intervention. Finally, to establish whether or not the training itself is truly causal, a double-blind, fully controlled study, including an active control group receiving sham neurofeedback, should be undertaken.
Footnotes
Acknowledgements
We acknowledge the helpful comments of David Silbersweig, MD, on the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
