Abstract
Purpose. Although Shi Quan Da Bu Tang (SQDBT) has been used to treat cancer patients clinically, very few studies evaluating the effectiveness of SQDBT using objective indicators have been published. The study objectives were to examine the effectiveness of SQDBT for alleviating hematotoxicity, as indicated by white blood cell (WBC) counts and hemoglobin (Hb) levels, among patients with breast carcinoma receiving chemotherapy. Methods. The authors identified patients with breast carcinoma who received chemotherapy in a teaching hospital in Taipei in 2008 through a chart review process. Only patients with initial WBC counts of <4000/µL were included. The case group was composed of 47 chemotherapy courses treated with SQDBT, whereas the comparison group included 257 courses without SQDBT. The complete blood count test was done before start of a chemotherapy course and 1 week after chemotherapeutic drugs were given. Results. Age, cancer stage, cancer status, use of granulocyte colony-stimulating factor, and chemotherapy drugs were controlled in the model. Patients who took SQDBT had significantly increased WBC counts, especially neutrophils, and Hb after chemotherapy (adjusted β = 1202.51, 95% confidence interval [CI] 440.45-1964.57 for WBC; β = 834.83, 95% CI = 197.35-1472.31 for neutrophils; β = 0.34, 95% CI = 0.05-0.63 for Hb). There were no significant differences in tumor markers CEA and CA153 between patients given SQDBT or not after chemotherapy. Conclusion. SQDBT is effective in alleviating hematotoxicity among patients with breast carcinoma receiving chemotherapy, without affecting the presentation of tumor markers in the short term. More study is needed to determine long-term outcomes such as recurrence and survival.
Introduction
Of all cancers among women in Taiwan, breast carcinoma is ranked fourth in incidence and mortality. The incidence and mortality was 49.9 per 100 000 women in 2006 and 10.6 per 100 000 women in 2009, respectively. 1 Although these rates are lower than in the United States, yet a trend toward increased incidence and mortality of breast carcinoma is reported in Taiwan.1,2 Most patients with breast carcinoma need to receive chemotherapy for relapse prevention. They often encounter complications from chemotherapy, including hematotoxicity.3,4 Studies showed that the incidence of grade 3 to 4 hematotoxicity was 25% to 57% for leukopenia, 5% to 28% for anemia, and 5% to 20% for thrombocytopenia.3-6 Hematotoxicity has been shown to interfere with the completion of chemotherapy and to cause fatigue and weakness among patients.4,7
In Taiwan, the National Health Insurance system covers both modern medicine and traditional Chinese medicine (TCM) expenses in parallel. More than 70 Western medical hospitals have departments of TCM. It is feasible to treat cancer with an integrative system of modern medicine and TCM. Recent surveys of cancer patients suggested that 9% to 64% patients tried some form of complementary and alternative medicine (CAM).8-13 The classic TCM herbal formula, Shi Quan Da Bu Tang (SQDBT, also known as Juzen-taiho-to, or TJ-48) is a therapeutic formula consisting of 10 different herbs to tonify blood and qi. Studies showed that SQDBT increases white blood cell (WBC) count and hemoglobin (Hb) level.14-17 Some animal studies showed that SQDBT prevented malignant progression and metastasis of tumor cells.15,18,19
To the best of our knowledge, few studies evaluated the effectiveness of TCM among cancer patients using objective indicators and no study examined the effect of SQDBT for alleviating hematotoxicity among human subjects receiving chemotherapy. The purpose of this study was to examine the effectiveness of SQDBT for alleviating hematotoxicity, as indicated by WBC counts and Hb levels, among patients with breast carcinoma receiving chemotherapy.
Materials and Methods
We reviewed the electronic medical charts of patients with breast carcinoma receiving chemotherapy at a university hospital in Taipei City from January to December 2008. The study hospital offered both TCM and Western oncology services. There was no gate-keeping system or formal referral criteria for medical treatment. This study involved no contact with the patients; the patient names were replaced by coded numbers to ensure anonymity during and after data abstraction was completed. Written consent was not required, since patient identifiers were not included in the data. The study was approved by the human subjects committee at the study hospital (approval number TMUH-02-10-02).
Study Participants
Through the electronic chart system, we identified 80 adult patients with breast carcinoma who received chemotherapy in 2008. A total of 823 chemotherapy courses among the 80 patients were identified. Since our research focused on use of SQDBT to treat leukopenia, we excluded patients with WBC counts of ≥4000/µL when SQDBT was started. Patients with chemotherapy courses who did not follow the usual 3-week cycle were also excluded. Finally, the study included 304 chemotherapy courses among 68 patients. Of the 304 courses of chemotherapy, 47 courses among 13 patients included treatment with SQDBT, which comprised the case group, whereas the remaining 257 courses among 66 patients without SQDBT comprised the comparison group. There were 11 patients who were in both the case and the comparison groups. The unit of analysis was one course of chemotherapy.
Chemotherapy Protocol
The type and dosage of chemotherapy drugs were decided by the oncologists in the Division of Hematology and Oncology. The oncologists had undergone training for more than 6 years in the Department of Internal Medicine and were certified by the Taiwan Oncology Society. Usually, patients with breast carcinoma received 3-week cyclic chemotherapy courses. According to the hospital routine, The complete blood count test was done before start of the first chemotherapy course (before CT), and then chemotherapy drugs were administered later at the same day, after complete blood count test results were assessed by the oncologists. The complete blood count test was done again 1 week after chemotherapy drugs were given (after CT). Tumor markers CEA and CA153 were assessed every 3 month. The blood-testing instrument was calibrated daily by qualified technicians at the study hospital.
Shi Quan Da Bu Tang Protocol
Patients were referred by their oncologist or were self-referred for TCM services. The TCM doctors assessed symptoms and the blood-testing data of the patient. If the patient had general weakness and fatigue, and combined leukopenia, neutropenia, or anemia, then SQDBT was prescribed. SQDBT is a traditional multicomponent medication. The dosage of each herb of the SQDBT was based on the Committee on Chinese Medicine and Pharmacy, Taiwan Department of Health. 20 The SQDBT used in this study was in the form of a pill produced by a GMP-certified pharmaceutical company in Taiwan (Ko Da Pharmaceutical Co, Ltd, Taoyuan, Taiwan). This company obtained ISO9001 certification in 2000. Each pill weighed 500 mg and was composed of Poria (36 mg), Rhizoma atractylodis macrocephalae (36 mg), Radix ginseng (36 mg), Radix rehmanniae preparata (36 mg), Radix paeoniae alba (36 mg), Radix glycyrrhizae (36 mg), Radix astragali (36 mg), Cortex cinnamomi (12 mg), Radix angelicae sinensis (36 mg), Rhizoma chuanxiong (36 mg), and Mel (164 mg). Each herb except Mel was cleaned, dried, and ground into powder separately. Then the component powder was mixed together in the ratio listed above. Finally, Mel was mixed with the powder to form a pill. To ensure the quality of the product, thin layer chromatography was performed on sample pills to ensure the existence of each component individually. For example, according to the manufacturer, to detect Radix ginseng of the SQDBT, SQDBT was diluted in methanol. Then SQDBT solution was filtered and analyzed using thin layer chromatography and compared the results with controls (filtered solution of Radix ginseng and filtered solution of SQDBT without Radix ginseng) to confirm the existence of Radix ginseng in the SQDBT. Chemical pattern of SQDBT obtained by the 3-dimensional high-performance liquid chromatography analysis is given in Figure 1. SQDBT was prescribed at a dose of 6 to 10 pills a time, 3 times a day after meals. Based on the consensus of the TCM doctors, the prescribed dose was based on the patient’s WBC count, with 10 pills for WBCs < 2000/µL, 8 pills for <3000/µL, and 6 pills for <4000/µL. No adverse effects have been recorded for SQDBT, but based on our clinical observation; some patients may have oral ulcers or insomnia, which could be caused by chemotherapy drugs and/or SQDBT. The TCM doctors decreased the prescribed dose (but no less than 6 pills) if the patient had oral ulcers and/or insomnia. Studies showed that the effect of SQDBT in increasing WBC and Hb was not evident until 14 days after initiation of use.21,22 Therefore, it was decided to set the start time of SQDBT at 14 days before start of the next course of chemotherapy. Since the chemotherapy courses were cyclical, 14 days before start of the next course of chemotherapy was 1 week after the start of the previous chemotherapy course. Taken together, the SQDBT, chemotherapy, and blood-testing schedule had an integrated course schedule that started with SQDBT for 14 days, then prechemotherapy blood testing was done and chemotherapy was given later the same day. Then, postchemotherapy blood testing was done 1 week later. After 2 weeks, another course of chemotherapy began. Therefore, the integrated course of SQDBT use was 21 days in length. The TCM doctor reassessed the patients at the end of each SQDBT course and decided whether or not to continue the SQDBT therapy.
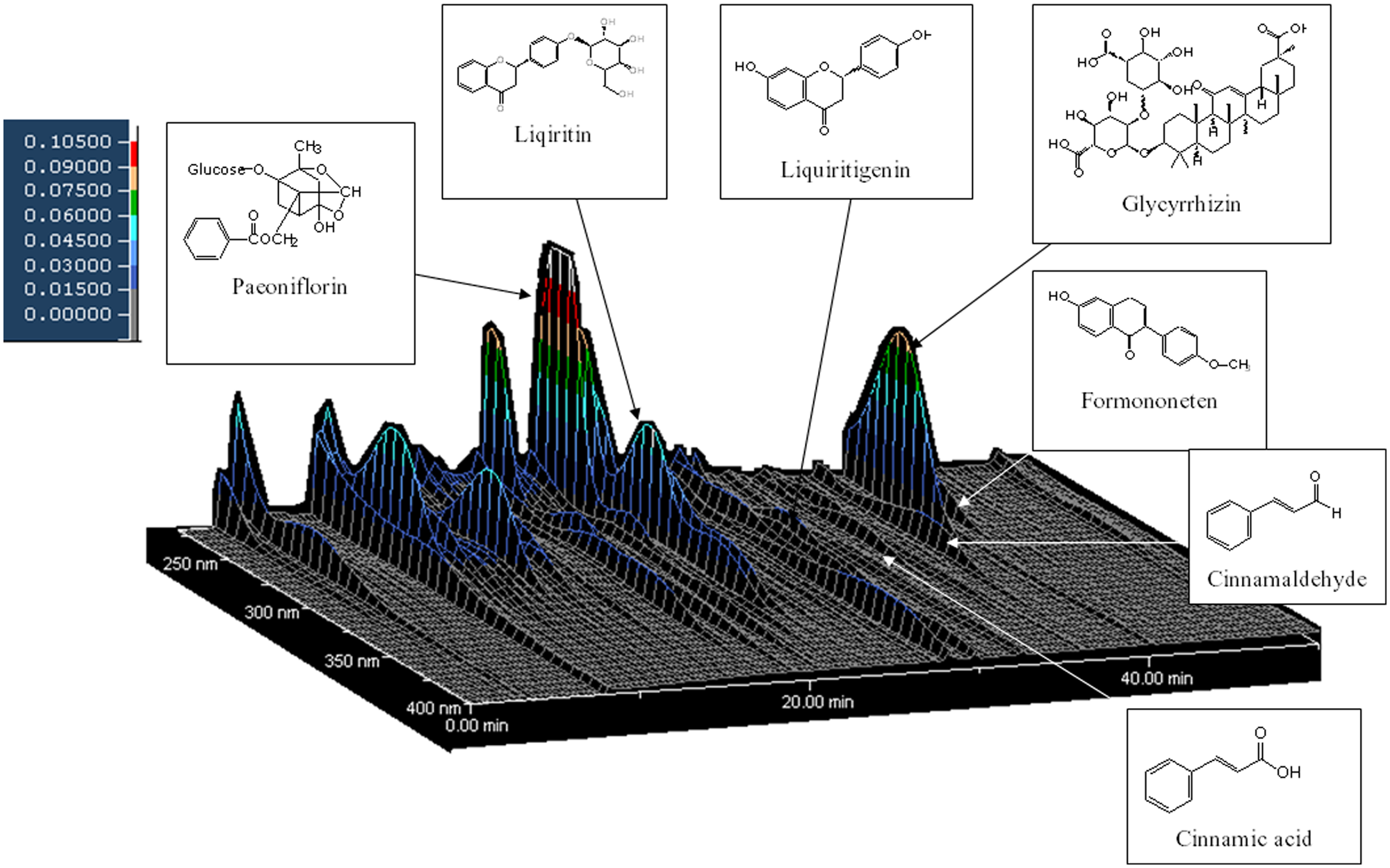
Chemical profile of SQDBT analyzed by 3-dimensional HPLC. Each peak of SQDBT in the HPLC profile was identified by comparison of the retention times and VB spectra of chemically defined standard compounds. HPLC condition was as follows: column, Mightysil RP-18, GP 250 × 4.6 mm (5 µm); carrier A, acetonitrile; carrier B, 0.03% H3PO4; gradient, 0% to 60% carrier A linear in 65 minutes, 40% to 100% carrier B linear in 65 minutes; flow rate, 1.0 mL/min; injection volume, 10 µL; detector, Hitachi D-7000 interface, L-7100 pump, L-7455 diode array detector, L-7200 autosampler
Measurements
A structured questionnaire was used to abstract data from the medical charts. The data included demographics (age and disease history), disease and treatment information (cancer stage and cancer status, chemotherapeutic drugs, chemotherapy course, use of granulocyte colony-stimulating factor [GCSF], and use of SQDBT), and blood test results (WBC counts, Hb, CEA, and CA153) before and after chemotherapy. The hematologic toxicity of chemotherapeutic drugs was determined based on the National Cancer Institute–Common Terminology Criteria for Adverse Events, version 3.0. 23
Data Analysis
Statistical analyses were performed using Predictive Analytics Suite Workstation, version 18.0, software (IBM Corp, Somers, NY). Individual variables were examined using percentages, means, and standard deviations. Differences between the case and comparison groups were explored using χ2 statistics, Student’s t tests, and one-way analysis of variance. The generalized estimating equations (GEE) model was used to analyze the independent effect of SQDBT on alleviating hematotoxicity, as indicated by the WBC and Hb levels, given potential confounders were adjusted in the model. The GEE approach accounted for correlated data resulting from multiple observations of the same individual at different time points. 24 In all analyses, a 5% significance level was used.
Results
Characteristics by the Case–Comparison Status
We compared patient characteristics by case–comparison group status (Table 1). There were no significant differences between the case and comparison groups for mean age and cancer status. The comparison group had a greater number of patients with stage III and IV cancers than the case group (63.0% vs 34.1%). All patients used chemotherapeutic drugs with hematotoxicity. There were no significant differences between the case and comparison groups in use of chemotherapeutic drugs except that epirubicin and docetaxel were used more often in the comparison group.
Characteristics by the Case–Comparison Group Status (n = 304)

Abbreviations: SQDBT, Shi Quan Da Bu Tang; GCSF, granulocyte colony-stimulating factor; CT, chemotherapy.
t and χ2 are respective statistical values from Student’s t test and χ2 test. The significance level was set to P < .05.
All patients used chemotherapeutic drugs with hematotoxicity.
White Blood Cell Counts
There were no significant differences in mean WBC counts between the case and comparison groups at the time of SQDBT start (Table 1). The case group had lower mean WBC and neutrophil counts than the comparison group before chemotherapy (3754.47/µL vs 4658.60/µL, P = .011 for WBCs; 2302.68 vs 2873.18, P = .066 for neutrophils). One week after chemotherapy, the case group had significantly higher WBC and neutrophil counts than the comparison group (3165.53/µL vs 2436.97 µL, P = .001 for WBCs; 2002.87 vs 1373.99, P < .001 for neutrophils; Table 1). In terms of grades of hematotoxicity, the case group had higher incidences of grades 3 to 4 leukopenia and neutropenia before chemotherapy (17.0% vs 2.3% for leukopenia; 17.6% vs 5.3% for neutropenia), but the case group had lower incidences of grades 3 to 4 leukopenia and neutropenia after chemotherapy (14.9% vs 44.0% for leukopenia; 19.6% vs 44.8% for neutropenia; Table 2). These results were sustained regardless of whether or not GCSF was used (Table 2).
Grades of Hematotoxicity Before and After Chemotherapy by Case–Comparison Status and Use of GCSF (n = 304) a

χ2 is the respective statistical value from the χ2 test; the significance level was set at P < .05.
Missing data = 13.
Missing data = 19.
The GEE results showed that patients who had concomitant SQDBT with chemotherapy had significantly increased WBC and neutrophil counts after chemotherapy compared with the comparison group (β = 1202.51, P = .002 for WBCs; β = 834.83, P = .010 for neutrophils) given that age, cancer stage, cancer status, and chemotherapeutic drugs were controlled in the model (Table 3).
GEE Results of the Effect of SQDBT on WBC, Neutrophil, and Hb Level Given Age, Cancer Stage, Cancer Status, and Chemotherapeutic Drugs Were Controlled in the Model a

Abbreviations: GEE, generalized estimating equations; SQDBT: Shi Quan Da Bu Tang; WBC, white blood cell; Hb, hemoglobin; 95% CI, 95% confidence interval; GCSF, granulocyte colony-stimulating factor; CT, chemotherapy; ×, interaction.
The references were comparison status, no use of GCSF, and before CT. The significance level was set to P < .05.
Hemoglobin Level
The mean Hb level did not differ significantly between the case and comparison groups before or after chemotherapy (Table 1). Before chemotherapy, the case group had higher incidences of grades 2 to 4 anemia than the comparison group (27.7% vs 17.5%, P = .014), but there were no significant differences in the incidences of grades 2 to 4 anemia after chemotherapy (Table 2).
The GEE results showed that patients who had concomitant SQDBT with chemotherapy had significantly increased Hb levels after chemotherapy compared with the comparison group (β = 0.34, P = .021), given that age, cancer stage, cancer status, and chemotherapeutic drugs were controlled in the model (Table 3).
Tumor Markers
We further examined whether tumor markers, CEA and CA153, differed between the case and comparison groups after chemotherapy to address the concern that SQDBT may interfere with the effect of chemotherapy. Given that age, cancer stage, cancer status, GCSF, and chemotherapy drugs were controlled for in the model, the GEE results showed that CEA and CA153 did not differ in patients who had concomitant SQDBT with chemotherapy as compared with the comparison group (β = −4.09, P = .574 for CEA; β = −1.21, P = .917 for CA153; Table 4).
GEE Results of the Effect of SQDBT on CEA and CA153 Given Age, Cancer Stage, Cancer Status, and Chemotherapeutic Drugs Were Controlled in the Model a

Abbreviations: GEE, generalized estimating equations; SQDBT: Shi Quan Da Bu Tang; 95% CI, 95% confidence interval; GCSF, granulocyte colony-stimulating factor; ×, interaction.
The references were comparison status, no use of GCSF, and before SQDBT use. The significance level was set to P < .05.
Discussion
This study showed that 3-week intervention of SQDBT was effective in alleviating hematotoxicity among patients with breast carcinoma receiving chemotherapy, as manifested by the finding that the SQDBT cases had significantly higher WBC and Hb levels (or WBC and Hb decreased less) 1 week after chemotherapy. Although WBCs, neutrophils, and Hb levels were lower in the case group than the comparison group before chemotherapy, these levels decreased significantly less in patients receiving SQDBT 1 week after chemotherapy. The effect of SQDBT in alleviating the hematotoxicity of chemotherapy could be related to several mechanisms. Hisha et al 21 suggested that fatty acids contained in the SQDBT might help in promoting the proliferation of hematopoietic stem cells. Ohnishi et al 22 suggested that SQDBT could stimulate spleen colony-forming units, therefore alleviating hematopoietic injury induced by the anticancer drugs. Further study is needed to examine the mechanism linking SQDBT to increased WBC and Hb levels.
Previous studies support the effectiveness of GCSF for increasing neutrophil counts.25-28 Our model showed that GCSF significantly increased the levels of WBCs and Hb before chemotherapy, but that these levels significantly decreased after chemotherapy. This is consistent with our observations that patients that ever used GCSF to treat neutropenia tended to continue to use GCSF in their following chemotherapy courses. We found that the SQDBT cases used less GCSF after use of SQDBT, whereas the comparison group had a correspondingly similar rate of GCSF use during the period. In addition, the effects of SQDBT on WBCs and Hb levels were not influenced by GCSF use, as indicated by the insignificant interaction term. Integrated use of GCSF and SQDBT may help keep neutrophil levels higher among patients with breast cancer receiving chemotherapy. Furthermore, use of SQDBT may decrease the need for GCSF.
We demonstrated that SQDBT alleviated hematotoxicity without counteracting the effect of chemotherapy, as indicated by the lack of significant differences between the case and comparison groups for CEA and CA153 levels in the short term. Several in vitro and animal studies suggest that SQDBT has antitumor effects,18,19,29,30 but we did not find significantly decreased tumor marker CEA and CA153 expression among SQDBT cases. Whether SQDBT can decrease tumor cell growth or prevent relapse among patients with breast cancer is outside the scope of the current study and warrants further studies with a longer period of follow-up.
Limitations
The allotment of case and comparison status was based on patient’s decisions to seek TCM treatment with or without an oncologist’s referral in this study. Therefore, there might be a potential selection bias. Nonetheless, lower mean WBC and neutrophil levels before chemotherapy in the SQDBT case group suggested that the SQDBT group could be at higher risk for developing leucopenia and neutropenia after chemotherapy. In addition, as the prescribed SQDBT was taken home by the patients, we were unable to determine the actual intake of SQDBT and thus the patients may not have complied with the planned treatment. If that occurred, then our study could have underestimated the effect of SQDBT and our results could be viewed as a conservative scenario. We used a chemotherapy course as the unit of analysis. Since each patient received many courses of chemotherapy, it was not feasible to use individual patients as units of analysis. We used the GEE model to control the effects of study covariates and to account for correlated data. This approach could enhance the comparability of the case and control courses and increase data efficiency, since different courses of the same individual could serve either as cases or controls depending on whether SQDBT was used. Nonetheless, number of patients was small in the study and future study with larger sample size is needed to further validate our results. Since this study excluded patients that did not follow the usual 3-week cycle of chemotherapy, we could not examine whether SQDBT can prevent a delay in chemotherapy because of low levels of WBCs, Hb, and neutrophils. Patients with delayed chemotherapy usually had complicated underlying conditions, which could have confounded the study results. Further study is needed to address this issue. This study only examined the combined effects of the multiple components of SQDBT on alleviating hematotoxicity. The effect of individual ingredients of SQDBT is not clear. We only collected short-term data on tumor markers and the patients took SQDBT only during chemotherapy. Though some animal studies have shown that SQDBT could prevent malignant progression and metastasis of tumor cells,18,19 yet the potential anticancer effect of SQDBT is beyond the scope of this study. Whether continuously taking SQDBT after completion of chemotherapy could prevent malignant progression merits further studies.
Conclusions
Shi Quan Da Bu Tang alleviated hematotoxicity among patients with breast carcinoma receiving chemotherapy, without affecting tumor marker expression in the short term. Randomized controlled trials are needed to confirm the effects of SQDBT for cancer patients receiving chemotherapy.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Committee on Chinese Medicine and Pharmacy, Department of Health, Taiwan (CCMP100-RD-026).
