Abstract
Background. Traditional Chinese herbal medicine was associated with improved prognosis in patients with performance score 0-1 at the time of diagnosis of stage IV pulmonary adenocarcinoma (PAC) treated with platinum-based chemotherapy (PBT). Objective. The authors investigated the effect of 1- to 4-month lag time to traditional Chinese medicine (TCM) treatment on the median and 1-year survival of PBT-PAC patients. Methods. The median lag time to treatment was 3 months. In the first 3 months, about 35% of the patients died, but thereafter the survival curve flattened off and the death of the next 35% to 40% of patients took 9 months. Leaving out patients with lag time up to 3 months therefore would be a reasonable choice. To be on the safe side, the effects of leaving out patients up to 4 months were investigated. The Kaplan–Meier survival curves were used. Results. The median and 1-year survival of the PBT-PAC patients was 5.0 months and 27%, respectively. Leaving out patients with 0-1 month follow-up, 0-2, 0-3, and 0-4 months follow-up changed the median and 1-year survival, respectively, to 5.5 months and 32%, 6.5 months and 36%, 9.0 months and 43%, and 10.0 months and 52%. The median survival of PBT + TCM in PAC patients was 22.6 months and 78%. Median lag time to TCM treatment was 3.0 months. With up to 4 months lag time to treatment due to combined patients’ and TCM doctor’s waiting time, the improved survival of PBT + TCM performance score 0-1 (fully ambulant) patients at the time of diagnosis still is significant (P < .01, hazard ratio = 0.51). Moreover, the survival of patients treated by TCM doctors with little or more months lag time to treatment was not different (P = .79). Conclusion. Increasing lag time to treatment up to 4 months improves the median and 1-year survival of PBT patients without TCM but is unlikely to explain the greatly improved prognosis of PBT + TCM treated patients with fully ambulant stage IV PAC.
Keywords
Introduction
Primary lung carcinoma is one of the most common malignant tumors in the world. Unfortunately, the majority of patients are already at an advanced stage by the time of diagnosis 1 leaving radiotherapy, chemotherapy, or targeted therapy (TT) as the only therapeutic options. The median survival time (MST) of advanced non-small-cell lung carcinoma when treated with modern systemic therapy has increased over the past decades. 2 Previous studies have suggested that age-, sex-, performance score 0-1-, diagnosis-, stage-, and diagnosis date–matched patients with metastatic pulmonary adenocarcinoma treated with traditional Chinese medicine (TCM) herbs + chemotherapy may have a much better survival than those with chemotherapy alone.3-5 The same holds for patients with pancreatic cancer, patients with liver metastases, and patients with metastatic lung and colorectal cancer.6-8
However, patients often get the diagnosis of metastatic cancer in different hospitals from where they receive TCM herbal treatment, and even within the same hospital, the TCM doctors may have a (short) waiting list. Moreover, patients may also need time to decide whether they will use TCM herbal treatment. The consequence is that there is a certain lag time to TCM herbal treatment (which is the combined patients’ and doctor’s delay) from 1 week up to several months for certain famous TCM specialists.
An extended lag time to treatment of patients with an overall very poor prognosis may improve the prognosis of the remaining patients, as patients with the worst prognosis already may have died before they ever have been seen by the TCM specialists or started TCM treatment. Patients with the same stage and histologic type of disease seen by TCM specialists therefore may have better survival expectancy than the group of all (consecutive) patients. Figure 1A, showing survival of lung cancer patients treated with chemotherapy without TCM treatment, graphically illustrates the steep decline of the survival curve in the first few months after diagnosis.

This figure explains the hypothesis tested in this study that waiting lists of traditional Chinese medicine (TCM) doctors who will treat patients with performance score 0-1 stage IV pulmonary adenocarcinoma already treated with platinum-based chemotherapy (PBT), could lead to lag time to TCM herbal treatment. This could result in a significantly improved survival, even without any additional adjuvant TCM treatment next to PBT. (A) Survival curve of patients with PBT treatment. Note that in the first 4 months, 40% of all patients died from their disease or roughly 10% monthly. (B) Scatter plot of the follow-up time since diagnosis versus age in relationship to death (triangles) or alive with disease (circles). In the first months, many patients died from metastatic disease. Patients who survive these first months often have longer follow-up (as is clear from the more horizontal shape of the survival curve). As a result, after 4 months, 40% of the patients have died. If the lag time to TCM herbal treatment due to waiting time of the TCM doctors would be 4 months, the remaining 60% of the patients who are still alive and seen by TCM doctors automatically have a significantly better survival than the original total group of all patients. As a result, even without TCM treatment, the survival will be better than that of the original total group of patients.
This has suggested the hypothesis that the improved survival of stage IV pulmonary adenocarcinoma (PAC) patients treated with PBT (platinum-based chemotherapy) + TCM versus PBT alone is due to lag time to treatment bias (combined patients’ delay and doctor’s delay). However, it is unknown how strong the influence of this phenomenon is, if there is any influence at all. We have therefore performed a computer simulation exploring the effect of an artificial lag time on the survival of PAC patients with PBT treatment. The median and 1-year survival of patients with an artificial lag time to treatment of up to 4 months have been studied. We also compared these survivals with a group of PBT patients treated with additional TCM treatment, with little or 1- to 2-months’ lag time to treatment.
Patients and Methods
The PBT patients for the analyses were selected from an earlier study described in detail elsewhere. 5 Briefly, in that study, the PBT-only patients were selected from a large database of patients diagnosed with radiologically and pathologically confirmed stage IV adenocarcinoma of the lung and treated at the Respiratory Hospital, Yangpu district, Shanghai, China. These patients had been selected by matching them on age, sex, and diagnosis date with all new consecutive fully ambulant patients (Eastern Cooperative Oncology Group performance status 0, 1, or 2) with the same performance, stage, and diagnosis, treated with the same PBT but in addition also TCM herbal therapy by Professor Jia Xiang Liu between January 2006 and January 2009 at the TCM outpatient clinic of the Longhua Hospital, Xuhui district, Shanghai, China. The age, sex, treatment, and follow-up of these patients were known and they had not been treated with exploratory surgery. The patients (PBT-PAC group) currently studied therefore all had had stage IV, pathologically proven adenocarcinoma of the lung with performance status (PS) 0-1, and fully ambulatory PS-2. The sex and age distribution, median and 1-year survival (5.0 months and 27%, respectively) were in agreement with the data from large international studies on PBT patients with stage IV disease. 1 However, while doing the analyses for the current study, an article was published showing that more than two thirds of Chinese cancer patients believed that combining integrated Chinese and Western medicine was effective and 63% of those receiving chemotherapy used Chinese medicine before/during/after Western medicine treatment. Interestingly, nearly two thirds did not tell their physicians that they were using traditional Chinese medicine herbs. 9 We therefore considered that the same may also have occurred with the patients from the Respiratory Hospital. One of us (HG) therefore personally contacted each patient or their relatives and discovered that 35 of the 65 patients from this hospital also “secretly” used TCM herbal treatment provided by different TCM specialists (ie, had consulted TCM specialists elsewhere in Shanghai outside the supervision of the treating Western medicine specialist). There were no differences (P > .10) in the diagnosis month, age, sex, or performance status of these 2 subgroups of patients with chemotherapy alone versus chemotherapy plus TCM herbal treatment.
As we wished to evaluate the effect of lag time to treatment by a leaving-out simulation experiment, the question was how many months treatment delay should be studied. The median lag time to treatment was 3 months. In the first 3 months, about 35% of the patients died, but thereafter the survival curve flattened off and the death of the next 35% to 40% of patients took 9 months (Figure 1). Leaving out patients up to 3 months therefore would be a reasonable choice. To be on the safe side, we investigated the effect of leaving out patients up to 4 months and created from the original PBT-PAC patients 4 new groups of patients, as follows: the same as the original patients, but leaving out the patients with <1 month follow-up, <2 months follow-up, <3 months follow-up, and <4 months follow-up. All patients by definition had metastases at the time of diagnosis (being stage IV) and were carefully controlled by the database management department of the Respiratory Hospital. Death was confirmed in all cases. These “4 groups of new patients” were then added to the original database. The PBT + TCM treatment group was also added to this new database. This resulted in 312 “patients.”
Using SPSS version 18 (SPSS, Chicago, IL), Kaplan–Meier survival curves were made for the different patient groups. The median and 1-year survival of each group were calculated and the differences between the different groups evaluated with the log rank test. The hazard ratio (HR) and 95% confidence intervals were calculated by Cox regression.
Results
Figure 2 shows the survival curves of the original PBT patients and the PBT + TCM treated patients. Note that the median survivals are 5.0 and 22.6 months, a difference of nearly 18 months. The 1-year survivals were 27% and 78%, respectively. The different panels in Figure 3 show the survival curves of the patients with PBT, and with increasing lag times to treatment (simulated by leaving out from the PBT-treated patients those with follow-up of <1 month, <2 months, <3 months, and <4 months). Table 1 shows the median and 1-year survival of each group. Note that with up to 2 months lag time to treatment bias, there is hardly any effect on the median and 1-year survival, and the differences are not significant. Only after 3 months lag time does the difference become (just) significantly different from the original total group of PBT-treated patients; improved survival becomes clinically impressive only after 4 months lag time to treatment. However, even then, the PBT + TCM group still has significantly better survival (P = .01, HR = 0.51 for up to 4 months lag time to treatment; Table 1, last column; Figure 4). Moreover, there is no survival difference between general TCM specialists (with little lag time to treatment) and a famous TCM specialist (with more lag time to treatment; P = .79, Figure 5).
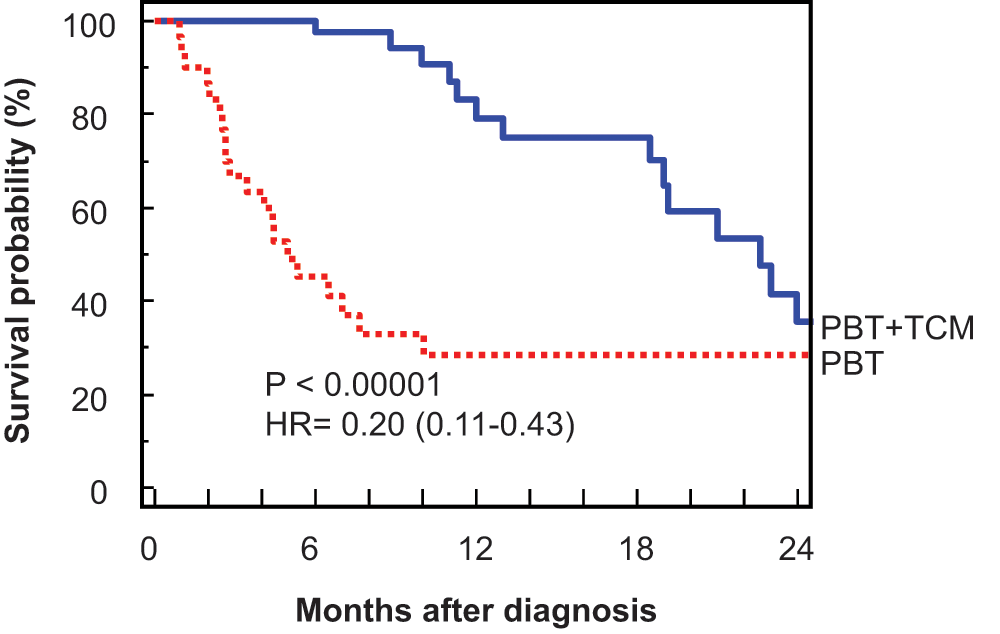
Survival curves of the original platinum-based therapy (PBT) patients and the PBT + TCM (traditional Chinese medicine) treated patients. Note that the median survivals are 5.0 and 22.6 months, a difference of nearly 18 months. The 1-year survivals were 29% and 78%, respectively, for the PBT and PBT + TCM treated patients

Panels A to D show the survival curves of the patients with platinum-based chemotherapy (PBT), and with increasing lag times to treatment (simulated by leaving out from the PBT-treated patients those with less than 1, less than 2, less than 3, and less than 4 months’ follow-up)
The Influence of Lag Time to Treatment on the PBT and PBT + TCM Groups

Abbreviations: PBT, platinum-based chemotherapy; TCM, traditional Chinese medicine; HR, hazard ratio; CI, confidence interval.

Comparison of the survival of patients treated with platinum-based chemotherapy (PBT, dotted line), PBT + traditional Chinese medicine herbs (PBT + TCM, continuous line), and PBT with leaving out patients with less than 4 months’ follow-up (PBT + 4 months, double-dotted line). The difference in the survival of patients with PBT + TCM at 1 year (78%) is still significantly better than that of patients with diagnosis to TCM treatment interval of 4 months lag time to treatment (52%; P = .01). It is therefore likely that adding TCM herbal treatment does indeed improve the prognosis of performance score 0-1 patients with stage IV adenocarcinoma of the lung

Comparison of patients with platinum-based chemotherapy + traditional Chinese medicine (PBT + TCM), treated by a famous TCM specialist with more lag time to treatment (“doctor A,” continuous line) and different general TCM specialists with little lag time to treatment (“doctors B,” broken line). There is no difference in the treatment results (P = .79)
Discussion
It has previously been observed that patients with metastatic PAC treated with TCM herbs + chemotherapy have a much better survival than those with chemotherapy alone.3-5 However, while presenting these data the hypothesis was forwarded that lag time to treatment might perhaps explain the improved survival of the patients with the additional TCM herbal treatment along with PBT. We found in this computer simulation study that with up to 2 months lag time to treatment bias, there is hardly any effect on the median and 1-year survival, and the differences are not significant. Only at 3 months lag time, the difference becomes (just) significant, but this improvement is clinically hardly meaningful. After 4 months lag time to treatment, the clinical improvement in survival becomes meaningful. However, even then, the PBT + TCM group still has significantly better survival (P = .01, HR = 0.51). As such a lag time to treatment as a result of the time patient needs to decide for TCM treatment plus the doctor’s waiting lists (TCM doctor’s delay) in reality for general TCM specialists is rarely more than 1 month, it is unlikely that the improved survival of TCM treatment plus PBT of fully ambulant stage IV PAC patients with performance score 0-1 at the time of diagnosis is due to lag time to treatment. This is confirmed by the lack of difference in survival between patients treated by general TCM specialists (with little lag time to treatment) and a famous TCM specialist (with 2 to 8 weeks waiting list and hence longer lag time to treatment). We therefore feel that the hypothesis that the survival improvement of patients with stage IV PAC treated with PBT + TCM herbal treatment is due to lag time to treatment, should be rejected.
It must be emphasized that the results of this retrospective study cannot give a definitive answer on the influence of patients’ and doctor’s delay on the prognosis. A prospective study is needed, in which the patients’ and doctor’s delay are both registered and studied. Moreover, although the results are strongly suggestive, a computer simulation study of course cannot give the final answer whether additional TCM treatment next to PBT in fully ambulant stage IV PAC patients improves the prognosis. For this, a prospective randomized clinical trial (RCT) of consecutive patients is required, but organizing this is practically very difficult. In much of the Western world, Chinese herbs are not legal, and in other countries, obtaining regulatory authorization for an RCT of a clinical trial of herbal medicines is quite burdensome, especially for prescriptions of multiple herbs typically used in TCM. Moreover, prescription of TCM herbs is based on TCM syndrome differentiation and may vary over time in the same patient. TCM medical specialists with oncology experience, especially in lung cancer, may not always be available in the Western world. It may therefore seem easier to attempt to set up an RCT in China or Taiwan. However, a previous study showed that many Chinese patients using chemotherapy secretly use TCM herbs, without informing the Western medical oncologists treating them, 9 something confirmed in our previous study. 5 In a prospective randomized trial supervised by a TCM oncology specialist, it would be questionable if patients would reliably report use of alternative TCM herbs, either those used by a placebo or control group or herbs used by an herbal treatment group in addition to those prescribed by the TCM specialist. A prospective RCT should include urine or blood tests to search for metabolic by-products of other herbs used than the ones prescribed, but such tests currently are also not totally reliable because of the great variety of herbs that could possibly be used. Development of reliable metabolic urinary and blood markers for many of the TCM herbs used are therefore of the utmost importance.
There are other challenges in developing an RCT. First, a phase I trial documenting safety of a proposed treatment would be necessary before starting any randomized trial. Development of an appropriate placebo or other control treatment would also be a challenge. Another question in a prospective RCT is the use of single or multiple TCM herbs. From a Western scientific point of view, one would use as few herbs as possible, as this would make it easier to trace the effect of the herbs investigated. Even when only one herb is used, this may contain many biologically active compounds, which may not be easy to study. Importantly, however, such treatments would differ markedly from the suggestive results seen with the epidemiological studies we discuss, and may have little relevance to actual TCM clinical practice.
In conclusion, the current results of this computer simulation study make it unlikely that lag time to treatment bias is a significant contributor to the improved survival of ambulant (ie, performance score 0-1 at the time of diagnosis before chemotherapy started) stage IV PAC patients treated with TCM herbs along with PBT.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
