Abstract
Tumor metastasis is the most important cause of cancer death and various treatment strategies have targeted at preventing the occurrence of metastasis. Phyllanthus urinaria L is a popular folk medicine and has several proven biological properties, including antioxidant, antihypertension, and anti-inflammatory. This study provides molecular evidence associated with the antimetastatic effects of P urinaria L extracts (PUE), which contained polyphenols including gallic acid, methyl gallate, epicatechin, epigallocatechin-3-gallate, gallocatechin-3-gallate, rutin, epicatechin-3-gallate, and naringin, by showing a marked inhibition on the invasion (P < .001) and migration (P < .001) of highly metastatic A549 and Lewis lung carcinoma (LLC) cells. To further investigate the precise involvement of PUE in tumor metastasis, A549 and LLC cells were treated with PUE at various concentrations and results from zymography and Western blotting showed that a PUE treatment may decrease the expressions of matrix metalloproteinase-2 (MMP-2; P < .001), MMP-9 (P < .001), urokinase plasminogen activator (P < .001), and their endogenous inhibitors, that is, tissue inhibitor of metalloproteinase-2 and plasminogen activator inhibitor-1, in a concentration-dependent manner. Reverse transcription–polymerase chain reaction and MMP-2 promoter luciferase analysis (P < .001) revealed that PUE inhibits the transcription of MMP-2 mRNA. PUE also exerted an inhibitory effect on the DNA-binding activity and the nuclear translocation of NF-κB and AP-1. Furthermore, the inhibitory effects of PUE on the metastasis and growth of LLC cells in vivo were proven. These results indicate that PUE could be applied to be a potential antimetastatic agent.
Keywords
Introduction
Natural compounds, including thousands of plant secondary metabolites, are important for plant biology and human nutrition and have been applied for therapeutic purposes in folk and traditional medicine. During the past decade, it has been shown that Phyllanthus urinaria L has several beneficial biological activities that help in reducing the risks of cardiovascular disease and cancers with antihypertension, antioxidant, anti-inflammatory, antineoplastic, antiangiogenetic, and chemoprotective properties.1-5 P urinaria L, one of the herbal plants belonging to the genus Phyllanthus (Euphorbiaceae), is widely used as a traditional folk medicine in Asian countries such as India, China, Thailand, and Taiwan.
6
Boiling water extracts of P urinaria have been reported to be able to exhibit antiangiogenic and antitumor effects in animal model bearing Lewis lung carcinoma (LLC) cells.
7
Recent studies have shown that the ethanolic extracts of P urinaria exhibited antioxidant and cardioprotective effects against doxorubicin-induced cardiotoxicity via upregulation of catalase and superoxide dismutase enzyme activity.
8
The 50% methanolic extracts of Phyllanthus niruri were reported to exhibit cytotoxic activitiy against human liver carcinoma Hep G2 cells.
9
Several natural compounds were isolated from different Phyllanthus species, including triterpenes, lignans, flavonoids, alkaloids, and tannins.6,10 Hippomanin A, a compound isolated from P urinaria, was reported to have anti-infection activities against herpes simplex virus type 1 (HSV-1) and HSV-2 in vivo.
11
Geraniin and 1,3,4,6-tetra-O-galloyl-β-
The spread of malignant cells from the primary tumor and subsequent growth of secondary tumors in distant tissues and organs may complicate the clinical management and may even lead to death. 14 Despite advances in cancer therapy during the past few years, most cancer mortalities result from metastatic disease. 15 Suppression of extracellular protease activity or expression has been demonstrated to contribute to the inhibition of the invasion of various types of cancer cells. 16 Considerable interest has been raised in natural compounds that inhibit the activities of proteases, such as plasminogen activator system and matrix metalloproteinases (MMPs), thereby preventing cancer metastasis. MMP-2 and MMP-9 are members of a unique family of zinc-binding endopeptidases that are thought to play an essential role in the process of cancer angiogenesis, invasion, migration, and metastasis.17,18 Urokinase-type plasminogen activator (u-PA), a serine proteinase, may initiate the activation of an enzymatic cascade that involves the activation of plasminogen to plasmin, which in turn could cleave and activate MMPs. 19 The activation of these enzymes enables the degradation of extracellular matrix (ECM) by cancer cells, allowing their access to the vasculature, invasion into the target organ and development of tumor metastasis.19-21 Moreover, u-PA or MMPs gene expression is primarily regulated at the transcriptional level through nuclear factor-κB (NF-κB) and activating protein-1 (AP-1), which are the downstream factors of the mitogen-activated protein kinase (MAPK) and PI3K-Akt pathways. At the posttranscriptional levels, u-PA and MMPs can be further modified by their activators or inhibitors depending on their surface localization.22,23 Hence, based on the functional importance of MMPs and u-PA in cancer metastasis, these molecules and their governing signaling pathways have been considered as primary targets for the development of therapeutic and chemopreventive agents.21,24
Although it is quite clear that P urinaria L may inhibit the growth of various cancers by inducing cancer cells toward apoptosis, limited studies are available concerning the effect of P urinaria L in antimetastasis. Therefore, the purpose of the present study was to characterize the inhibitory effects of PUE on cell invasion of highly metastatic lung carcinoma cell lines. The changes of cell physiology or molecular events involved in cell invasion and the effect on tumor cell metastasis in vivo were also investigated.
Materials and Methods
Chemicals
Phyllanthus urinaria L (Euphorbiaceae) was obtained from a plantation of the Green Health Biotechnology Corporation (Yunlin, Taiwan) and identified by Professor Yih-Shou Hsieh, Chung Shan Medical University. Dulbecco’s modified Eagle’s medium (DMEM), basal medium Eagle (BME), glutamine, Giemsa, and 3-(4,5-dimethylthiazol-2-y1)-2,5-diphenyltetrazolium bromide (MTT), were obtained from Sigma Chemical Co (St Louis, MO) and Matrigel was purchased from BD Biosciences (Bedford, MA). Rabbit polyclonal antibodies against c-Jun and c-Fos were purchased from Biosource (Camarillo, CA); a rabbit polyclonal antibody against tissue inhibitor of matrix metalloproteinase-2 (TIMP-2) was purchased from Serotec (Oxford, UK). Monoclonal antibodies against NF-κB and C23; rabbit polyclonal antibodies against extracellular signal-regulated kinase 1/2 (ERK1/2), p38, and Akt, the total and phosphorylated protein, were purchased from Santa Cruz Biotechnology, Inc (Santa Cruz, CA). A monoclonal antibody against plasminogen activator inhibitor (PAI) was obtained from American Diagnostics, Inc (Greenwich, CT). ECL Plus detection kit was obtained from Amersham Life Sciences, Inc (Piscataway, NJ). Lightshift kit was purchased from Thermo (Rockford, IL).
Preparation of Phyllanthus urinaria L. Extracts
The P urinaria L extracts (PUE) were prepared as described previously. 25 Briefly, 100 g of air-dried leaves were boiled at 60°C for 24 hours with 500 mL of 50% ethanol. The extraction procedure was repeated twice. Then, solvent was removed from the combined extract with a vacuum rotary evaporator. The filtrate was then lyophilized and stored at −20°C. Furthermore, the chemical profile of PUE was analyzed by using high-performance liquid chromatography (HPLC)–mass spectrometer. Briefly, PUE was analyzed by HPLC–mass spectrometer using a HPLC (Waters 600 with a 2998 Photodiode Array detector). Samples (10 µL) were injected into a Merck LiChrospher 100 RP-18 column (4 mm × 250 mm). The analyses were carried out using the mobile phase composed of 2 solvents: solvent A (0.05% acetic acid/water) and solvent B (acetonitrile). The flow rate was 1 mL/min. The elution was carried out in a programmed gradient elution as follows: 0 to 30 minutes, with 0% to 60% B; 30 to 35 minutes, isocratic with 100% B; 35 to 40 minutes, isocratic with 100% A. Absorbance was monitored at 254 nm. The molecular masses of the peaks were determined from electrospray ionization mass spectra using multiply charged ion profile based on the modified method of Chu et al. 26
Cell and Cell Culture
A549 (human lung adenocarcinoma), LLC (murine lung carcinoma), and MRC-5 (normal human fetal lung fibroblast) cell lines obtained from ATCC (Manassas, VA) were cultured in DMEM (for A549 and LLC) or BME (for MRC-5) supplemented with 10% fetal bovine serum, 2 mM glutamine, 100 U/mL penicillin, and 100 µg/mL streptomycin. All cell cultures were maintained at 37°C in a humidified atmosphere of 5% CO2.
Determination of Cell Viability (MTT Assay)
Cells were incubated with 0.5 mg/mL MTT in culture medium for an additional 4 hours; the blue formazan crystals of viable cells were dissolved by lysis buffer and measured spectrophotometrically at 570 nm. 27
Transwell Cell Invasion and Migration Assays
After a pretreatment with PUE for 24 hours, cells were harvested and seeded to BD Falcon cell culture inserts (BD Biosciences, Bedford, MA) at 5 × 104 cells/well in serum-free medium and then incubated for another 12 hours at 37°C. For invasion assay, 100 µL Matrigel (1 mg/1 mL) was applied to the membrane filters with a pore size of 8 µm and the bottom chamber contained standard medium. Filters were then air-dried for 5 hours in a laminar flow hood. The invaded cells were fixed with methanol and stained with Giemsa. Cell numbers were counted under a light microscope, and migration assay was carried out as described in the invasion assay, with no coating of Matrigel. 28
Determination of MMP-2, MMP-9, and u-PA by Zymography
The activities of MMP-2 and MMP-9 in conditional medium were measured by gelatin zymography protease assays as previously described. 20 Briefly, collected media of an appropriate volume (adjusted by vital cell number) were prepared with SDS (sodium dodecyl sulfate) sample buffer without boiling or reduction and subjected to 0.1% gelatin–8% SDS-PAGE (SDS-polyacrylamide gel electrophoresis). After electrophoresis, gels were washed with 2.5% Triton X-100 and then incubated in reaction buffer (40 mM Tris–HCl, pH 8.0; 10 mM CaCl2 and 0.01% NaN3) for 12 hours at 37°C. Gel was then stained with Coomassie brilliant blue R-250. Visualization of u-PA activity was performed as previously described. 20 Briefly, 2% casein (w/v) and 20 µg/mL plasminogen were added to 8% SDS-PAGE gel and then performed as described for gelatin zymography.
Immunoblotting Analysis
Samples were separated in a 10% polyacrylamide gel and transferred onto a nitrocellulose membrane as previously described. 24 The blot was subsequently operated with standard procedures and probed with primary and secondary antibodies. The protein expression was detected by chemiluminescence using an Immoblon Western Chemiluminescent HRP Substrate kit.
Reverse Transcription–Polymerase Chain Reaction
For reverse transcription (RT), 2 µg of total RNA were used as templates in a 20 µL reaction containing 4 µL dNTPs (2.5 mM), 2.5 µL Oligo dT (10 pmole/µL), and 200U RTase. The appropriate primers 5′-GGCCCTGTCACTCC TGAGAT-3′ and 5′-GGCATCCAGGTTATCGGGGA-3′ for MMP-2 (473 bp), 5′-TTGCGGCCATCTACAGGAG-3′ and 5′-ACTGGGGATCGTTATACATC-3′ for u-PA (351 bp), 5′-GGATCCAGCCACTGGAAAGGCAACATG-3′, and 5′-GGATCCGTGCCGGACCACAAAGAGGAA-3 for PAI-1 (254 bp); 5′-GGCGTTTTGCAATGCAGATGTAG-3′ and 5′-CACAGGAGCCGTCACTTCTCTTG-3′ for TIMP-2 (496 bp); and 5′-CGGAGTCAACGGATTTGGTCGTAT-3′ and 5′-AGCCTTCTCCATGGTTGGTGAAGAC-3′ for GAPDH (glyceraldehyde-3-phosphate dehydrogenase; 305 bp) were used for polymerase chain reaction (PCR) amplifications. PCR was performed using Platinum Taq polymerase (Invitrogen, Carlsbad, CA) with the condition: 25 cycles of 94°C for 1 minute, 55°C (u-PA and PAI-1) or 63°C (MMP-2, TIMP-2, and GAPDH) for 1 minute, 72°C for 2 minutes followed by 10 minutes at 72°C.
Measurement of MMP-2 Promoter Activity
A 460-bp (−218 to +243) segment from the 5′-promoter region of the MMP-2 gene was cloned. Briefly, a 0.46-kb segment of the 5′-flanking region of the human MMP-2 gene was amplified by PCR using specific primers with restriction enzyme site from the human MMP-2 gene (accession no. NM_004530): 5′-GGTACCCAGATCG CG AGAGAGGCAAGTGG (forward/KpnI) and 5′-AAGCTT TGGTTGGAGCCTGCTCCGCGGCG (reverse/HindIII). The pGL3-Basic vector, containing a polyadenylation signal upstream from the luciferase gene, was used to construct the expression vectors by addition of PCR-amplified DNA with restriction enzyme site of the MMP-2 promoter to the KpnI/HindIII site of this vector. The PCR products (pGL3-MMP-2) were confirmed by size, as determined by electrophoresis and DNA sequencing. pGL3-MMP-2 plasmid was transfected into A549 cells using the PolyJet reagent (SignaGen Laboratories, Gaithersburg, MD) according to the manufacturer’s instructions. After incubation with PUE, cells were collected and disrupted by Luciferase Assay System (Promega, San Diego, CA). After centrifugation, aliquots of the supernatants were tested for luciferase activity using the Luciferase Assay System. Firefly luciferase activities were standardized for β-galactosidase activity. 29
AP-1 and NF-κB Binding Assay
Binding of AP-1 and NF-κB in nuclear extracts was assessed by electrophoretic mobility shift assay (EMSA) with biotin-labeled double-stranded AP-1 or NF-κB oligonucleotides, and EMSA was carried out by using the Lightshift kit. Briefly, binding reactions containing 10 µg of nuclear protein, 10 mM Tris, 50 mM KCl, 1 mM DTT, 5 mM MgCl2, 2 µg poly (dI-dC) and 2 pmol of oligonucleotide probe were incubated for 20 minutes. Specific binding was confirmed by using a 200-fold excess of unlabeled probe as specific competitor. Protein–DNA complexes were separated by using a 6% nondenaturing acrylamide gel electrophoresis and then transferred to positively charged nylon membranes and crosslinked in a Stratagene crosslinker. Gel shifts were visualized with streptavidin-horseradish peroxidase followed by chemiluminescent detection. 30
Measurement of Lung Metastasis and Tumor Growth in LLC-Bearing Mice
C57BL/6 male mice (National Taiwan University Animal Center, Taiwan), 6 weeks old, were implanted with 2 × 106 LLC cells (0.1 mL/mouse) via a subcutaneous injection. On the following day (day 1), mice were randomly divided into 2 groups (n = 8 for each group) to be fed by oral gavage with water (control) and PUE (0.5 g/kg/d by body weight). The growth of tumors was measured daily during the study. On day 30, tumor-bearing mice were sacrificed by euthanizing with CO2, and the primary tumors were separated from the surrounding muscles and dermis, and then weighed. Their lungs were quickly removed by surgical operation and metastatic nodules of lung were counted using stereomicroscopy. 31 At the termination of the experiment, tumor samples were added with lysis buffer (50 mM Tris–HCl, pH 7.4, 150 mM NaCl, 1% IGEPAL CA-630, 1 mM EDTA, 1 mM PMSF, 1 mM Na3VO4, 1 mM NaF, 1 mg/mL leupeptin, and 1mg/mL aprotinin), homogenized at 4°C and kept on ice for 30 minutes followed by centrifugation at 10000 × g for 30 minutes. Cell lysates were processed for Western blot analysis.
Statistical Analysis
Statistical significances were analyzed by one-way analysis of variance with post hoc Dunnett’s test. P value <.05 was considered statistically significant (Sigma-Stat 2.0, Jandel Scientific, San Rafael, CA).
Results
Effects of PUE on the Cell Viability of Lung Cancer Cells
To evaluate the bioactive compound of P urinaria L, we successively extracted the P urinaria with 50% ethanol. Chromatographic patterns from HPLC analysis of PUE showed peaks corresponding to the retention times. Absorbance was monitored at 254 nm (Figure 1A). Figure 1 and Table 1 show that gallic acid, methyl gallate, epicatechin (EC), epigallocatechin-3-gallate (EGCG), gallocatechin-3-gallate (GCG), rutin, epicatechin-3-gallate (ECG), and naringin are contained in PUE. The cytotoxic effects of various concentrations of PUE (0 to 100 µg/mL) on A549 and LLC cells are shown in Figures 2A and 2B, respectively. Although a treatment of PUE up to 100 µg/mL had no significant cytotoxic effect on A549 cells, the viability of LLC cancer cells was decreased by PUE in a dose-dependent manner (P < .001). Results from the same procedures performed on normal human fetal lung fibroblast cell lines, MRC-5, revealed that this compound did not have any significant cytotoxicity on these cells (Figure 2C).
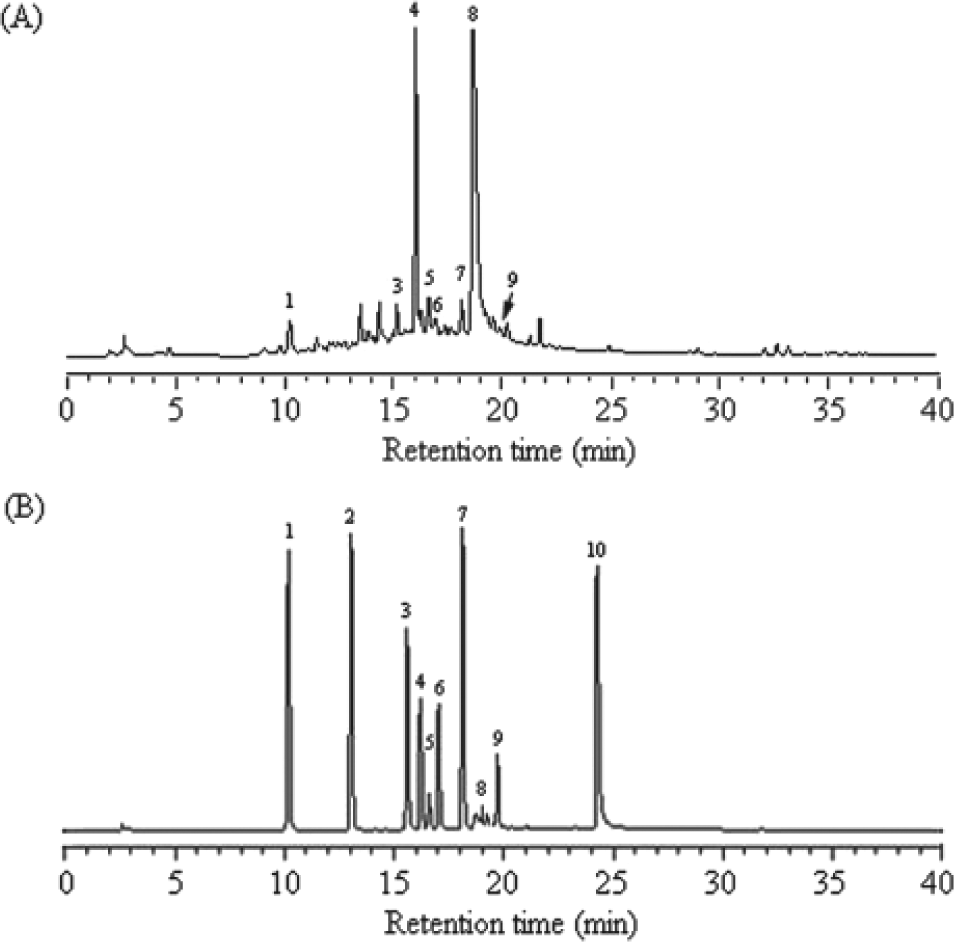
The chemical profile of PUE was analyzed by HPLC–mass spectrometry. (A) HPLC chromatogram of 10 kinds of standard compound. Peaks: 1, 1 µg of gallic acid; 2, 1 µg of protocatechuic acid; 3, 1 µg of methyl gallate; 4, 1 µg of EC; 5, 1 µg of EGCG; 6, 1 µg of GCG; 7, 1 µg of rutin; 8, 0.5 µg of ECG; 9, 1.5 µg of naringin; 10, 1 µg of quercetin. (B) Chromatographic patterns from HPLC analysis of PUE extracts showed peaks corresponding to the retention times (minutes). Absorbance was monitored at 254 nm
Characteristics of Phenolic Compounds of PUE

Abbreviations: EC, epicatechin; EGCG, epigallocatechin-3-gallate; GCG, gallocatechin-3-gallate; ECG, epicatechin-3-gallate.
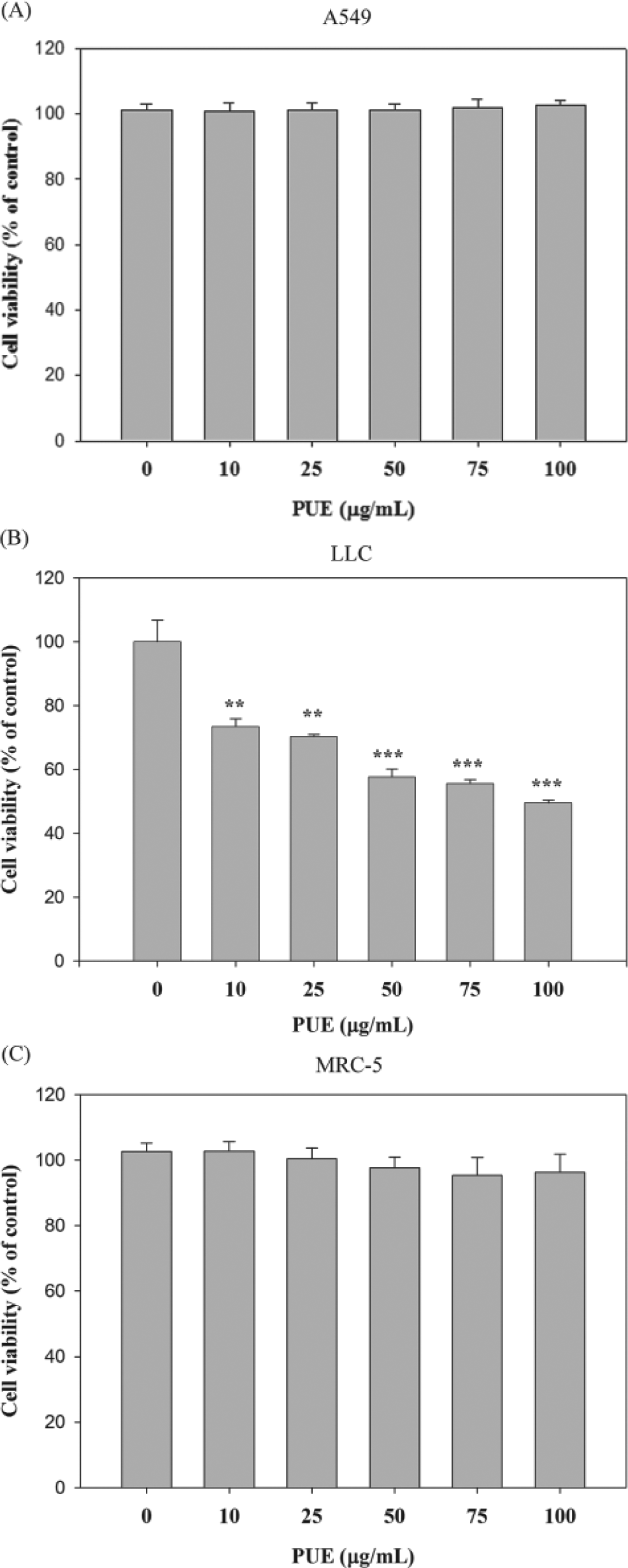
The effect of Phyllanthus urinaria L extracts (PUE) on the viability of A549, LLC, and MRC-5 cells. (A) A549, (B) LLC, and (C) MRC-5 cells were treated with PUE for 24 hours and then subjected to MTT assay for cell viability. The values are presented as mean ± SD of at least 3 independent experiments (**P < .01; ***P < .001)
Inhibition on Invasion and Migration of A549 and LLC Cells by PUE
Most deaths of cancers are due to metastasis, and the treatment for metastasis is still far from satisfactory. Therefore, the effects of PUE on cell invasion of lung cancer cell lines (A549 and LLC cells) were investigated. Using a cell invasion and migration assay with Transwell, it was shown that PUE significantly reduced the invasion and migration of A549 cells in a dose-dependent manner (P < .001), with only 20.7% and 25.5% remaining after a treatment of 100 µg/mL PUE, respectively (Figure 3A). Consistent inhibitory effect of PUE on LLC cells is shown in Figure 3B (P < .001).

The effects of Phyllanthus urinaria L extracts (PUE) on cell invasion and migration of A549 and LLC cells. (A) A549 and (B) LLC cells were pretreated with PUE at an indicated concentration for 24 hours, and then equal numbers of cell were subjected to analyses for invasion and migration as described in the Materials and Methods section. Data are presented as mean ± SD with that of control being 100%, and the statistical significance of results was analyzed by using one-way analysis of variance with post hoc Dunnett’s test (*P < .05; **P < .01; ***P < .001)
Inhibitory Effect of PUE on Matrix Metalloproteinase Activity
Via gelatin zymography assays, it was found that PUE effectively reduced MMP-2 and MMP-9 activity in a concentration-dependent manner on A549 and LLC cell, with 71.7% and 68.7% of inhibition, respectively, by 100 µg/mL of PUE, as shown in Figures 4A and 4B, respectively (P < .001).

The effects of PUE on the protein and mRNA activities of MMP-2, MMP-9, and u-PA. Cells were treated with PUE for 24 hours and then subjected to gelatin zymography, casein zymography, and Western blotting to analyze the activities of MMP-2, MMP-9 (A and B), u-PA (C and D), TIMP-2, and PAI-1 (E) as described in the Materials and Methods section. For mRNA levels, A549 total RNAs were extracted and subjected to a semiquantitative RT-PCR for MMP-2, u-PA, TIMP-2, and PAI-1 with GAPDH being an internal control (F). (G) Luciferase activity was measured in transiently transfected A549 cells using pGL3-MMP-2. Data are presented as mean ± SD with that of control being 100%, and the statistical significance of results was analyzed by using one-way analysis of variance with post hoc Dunnett’s test (*P < .05; **P < .01; ***P < .001)
Inhibitory Effect of PUE on u-PA Activity
After PUE treatment, via casein zymography assays, it was shown that PUE tremendously reduced u-PA activity both on A549 and LLC cells in a dose-dependent manner (Figures 4C and 4D; P < .001), with only 51.3% and 9.9% of u-PA activity being left in A549 and LLC cells, respectively, after treatment with 100 µg/mL PUE.
PUE Exerts an Inhibitory Effect on the Protein Levels of TIMP-2 and PAI-1
Physiological activity of MMP-2 and u-PA is greatly related to that of their specific endogenous inhibitors, TIMP-2 and PAI-1, respectively, therefore Western blotting was employed to see the effect of PUE on TIMP-2 and PAI-1 expression, and results showed that TIMP-2 and PAI-1 protein levels were gradually decreased along with the concentration of PUE in A549 as shown in Figure 4E (P < .001).
Inhibitory Effect of PUE on the mRNA Levels of Protease and Their Endogenous Inhibitors
To further examine the regulatory effects of PUE on MMP-2, u-PA, TIMP-2, and PAI-1, a semiquantitative RT-PCR analysis was performed. With GAPDH as an internal control, mRNA levels of MMP-2, TIMP-2, u-PA, and PAI-1 were significantly reduced, with only 50.1%, 27.7%, 58.4%, and 83.7% remaining after a 100 µg/mL PUE treatment in A549, respectively (Figure 4F).
Inhibitory Effect of PUE on the Transcriptional Activity of MMP-2 Promoter in A549 Cells
To evaluate the effects of PUE on the MMP-2 promoter, we performed a transient transfection with the pGL3-MMP-2 promoter and analyzed the luciferase activities. As shown in Figure 4G, the luciferase activity of the transfectants treated with PUE was reduced in a dose-dependent manner. These results confirmed the inhibition by PUE on the gene expression of MMP-2 in PUE-treated A549 cells.
Inhibition of Akt Phosphorylation by PUE
As we have shown that a treatment of A549 cells with PUE inhibited the cell invasion and activities of MMP-2 and u-PA, the underlying mechanisms were further investigated. As shown in Figure 5, PUE significantly inhibited the activation of Akt, whereas it has no significant effect on p38 and ERK1/2 activity. Moreover, no significant change in the total amount of ERK1/2, p38, and Akt proteins was observed.

The inhibitory effect of Phyllanthus urinaria L extracts (PUE) on the phosphorylation of Akt proteins. A549 cells was treated with PUE for 24 hours, and then cell lysates were subjected to SDS-PAGE followed by Western blotting with (A) anti-p38, anti-phosho-p38, anti-ERK1/2, anti-phosho-ERK1/2, (B) anti-Akt, and anti-phosho-Akt antibodies. Signals of proteins were visualized with an ECL detection system. Results from 3 repeated and separated experiments were similar
Inhibitory Effect of PUE on the Activation of NF-κB, c-Jun, and c-Fos
The expressions of ECM degrading proteases and their inhibitors are regulated by several transcription factors, such as AP-1 and NF-κB. To examine whether the inhibitory effect of PUE on MMPs and u-PA expression was linked to NF-κB and/or AP-1 activities, nuclear extract was analyzed by EMSA for AP-1 and NF-κB DNA binding activity, and it was found that a pretreatment with PUE suppressed the AP-1 (Figure 6A) and NF-κB (Figure 6B) binding activity. Subsequently, Western blot was performed to further confirm these results, and it was found that a pretreatment of PUE suppressed the nuclear levels of NF-κB, c-Jun, and c-Fos with C23 being the internal control (Figure 6C).

The effects of Phyllanthus urinaria L extracts (PUE) on the activation of NF-κB, c-Jun, and c-Fos. A549 cells were treated with PUE and then nuclear extracts were analyzed for DNA binding activity of AP-1 (A) and NF-κB (B) using biotin-labeled NF-κB and AP-1 specific oligonucleotide by electrophoretic mobility shift assay (EMSA). The last lane represented nuclear extracts incubated with unlabeled oligonucleotide (Comp) to confirm the specificity of binding. Nuclear extracts were subjected to SDS-PAGE followed by Western blotting with anti-NF-κB, c-Fos, c-Jun, or C23 antibodies (C). Signals of proteins were visualized with an ECL detection system. The experiments were repeated 3 times with similar results
The Antimetastasis Effects of PUE in Animal Model
To verify the in vivo antimetastatic effects of PUE, LLC-bearing C57BL/6 mice were treated with water or PUE. Lung metastases of animals treated with PUE have decreased by 38.8% compared with the water-treated group (Figure 7A; P < .001). Small solid tumors were observed on the eighth day after the cell inoculation and a 2.9-fold reduction in PUE-treated animals was seen on day 29, compared with control animals (Figure 7B; P < .001). Moreover, by day 30, PUE feeding induced a 2.2-fold reduction in tumor weight (Figure 7C; P < .001) without any apparent signs of toxicity as evidenced by body weight monitoring (Figure 7D) throughout the experiment. As PUE feeding to mice led to a decrease in lung metastases, we next examined whether the effect in tumors are associated with in vivo inhibition of MMP-9. We assessed the expression of MMP-9 in the tumors collected from control (water-treated group) and PUE-fed mice at the termination of the experiment. PUE showed a profound in vivo inhibitory effect on MMP-9 expression by Western blot analysis (Figure 8).
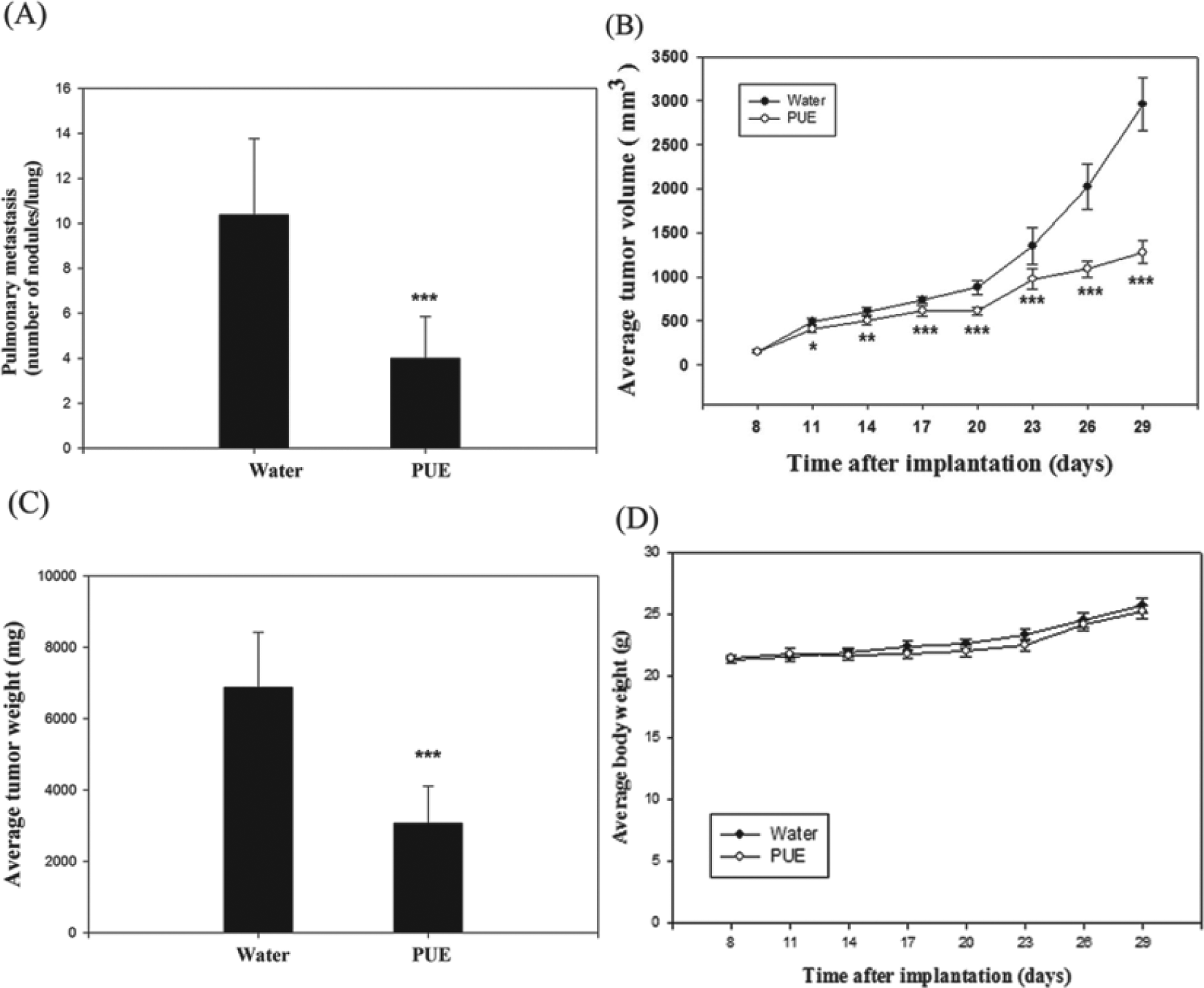
The in vivo antimetastasis effect of Phyllanthus urinaria L extracts (PUE). After subcutaneous implantation of Lewis lung carcinoma (LLC) cells, C57BL/6 mice were treated with PUE as described in the Materials and Methods section and then analyzed for the number of lung metastasis (A), the growth of tumor (B), the weight of primary tumor (C), and the body weight of mice (D). The values are presented as mean ± SD. Comparisons were performed by using one-way analysis of variance with post hoc Dunnett’s test (*P < .05; **P < .01; ***P < .001)

Inhibitory effect of Phyllanthus urinaria L extracts (PUE) on the expression of MMP-9 in tumor of C57BL/6 mice. At the end of experiment, tumor tissues were homogenized and cell lysates prepared as detailed in the Materials and Methods section. Cell lysates were subjected to SDS-PAGE followed by Western blotting. The membrane was probed with anti-MMP-9 antibody with β-actin as an internal control. The quantitative data shown are mean ± SD of 3 tumor samples from an individual mouse in each group (*P < .05)
Discussion
Lung cancer is the most commonly diagnosed malignancy and the main cause of cancer-related deaths in Asian and Western populations. Most deaths from lung cancer are related to metastasis, which is a complicated and currently uncontrolled process. The 5-year survival rate of all stages of lung cancer is estimated to be only approximately 15%, and non–small cell lung cancer accounts for approximately 80% of lung cancer. 32 Thus, inhibition of metastasis of lung cancer is thought to be one of the important therapeutic strategies to improve patient outcome. In the present study, we provide evidence for the antimetastatic potentials of PUE against the invasion of A549 and LLC cells, a highly metastatic non–small cell lung cancer cell line. In these cell lines, PUE could exert inhibitory actions on cell invasive capability with reduced cellular motility and activation of matrix-degrading proteases, MMPs and u-PA.
The most important characteristic of malignant tumors is their ability to invade the surrounding tissues and then metastasize. The spread of malignant cells from the primary tumor and subsequent growth of secondary tumors in distant tissues and organs may complicate the clinical management and may even lead to death. The progression of a tumor from being in situ to invasive is a major prerequisite for cancer metastasis, 33 and this comprises attachment and proteolytic degradation of the basement membrane followed by migration through the ECM. Invasion of cancer cells also involves various cytophysiological changes, including loss of cell–cell adhesion along with gain of cell–matrix adhesion and increased activation and expression of extracellular proteases to degrade the ECM and allow cell invasion and metastasis.33,34 Basically, the progression of tumor cells from in situ to invasive is correlated with increased cellular motility and proteolytic activities.
In previous studies, silibinin, a flavonoid from silybum marianum, could inhibit the invasion of human lung carcinoma A549 cells via decreased MMP-2 and u-PA expression. 35 Earlier reports have indicated that sesquiterpenes exhibit effective inhibition of cell migration of melanoma cancer B16 cells. 36 Epigallocatechin-3-gallate has been reported to be able to inhibit the invasion of human breast cancer MCF-7 cells via suppression of MMP activities. 37 New evidence indicated that MMPs can maintain osteoblast survival by activation of latent transforming growth factor-beta. 38 The expression and activity of MMPs are increased in almost every type of human cancer, and this correlates with advanced tumor stage, increased invasion, and metastasis. Hence, the decrease of MMPs is a critical target for antimetastasis. In the present study, we have demonstrated that PUE could inhibit MMPs and u-PA expression of lung cancer A549 and LLC cells and further inhibit invasion property. Since several studies on different cell types have indicated that MAPKs, for example, ERK1/2, JNK1/2, and p38MAPK, seem to play a central role in regulating the expression of u-PA or MMPs,23,39 inhibition of the MAPK pathway might have the potential to prevent angiogenesis, proliferation, invasion, and metastasis for a wide range of tumors. Our present data have revealed that PUE treatment inhibited phosphorylation of JNK1/2 and concurrent reduction in the levels of MMP-2 and u-PA, indicating a possible mechanism of inhibition of MMP-2 or u-PA synthesis by PUE. Additionally, the PI3K-Akt signaling pathway also played a crucial role in MMPs or u-PA gene regulation, cell survival, and tumor cell invasion.39,40 In our studies, treatment of PUE inhibited phosphorylation of Akt. Our findings suggest that PUE-caused reduction of MMP-2 and u-PA activities in the culture media could possibly be through a suppression of phosphorylation of Akt. It is well known that the activation of NF-κB and AP-1, the downstream of the PI3K-Akt pathways, is associated with inflammation, angiogenesis, and cell invasion, 41 and that downregulation of any of these transcription factors is potentially an effective means to block tumor invasion and metastasis, as well as blocking the factors that bind to these regulatory elements. Therefore, this represents an appropriate approach to inhibit the synthesis of MMPs or u-PA. In our studies, PUE was found to effectively suppress the expression and DNA-binding activity of NF-κB and AP-1.
The major drawbacks of many effective cancer chemotherapeutic agents are systemic toxicity and drug resistance. In this regard, dietary supplement and phytotherapeutic agents with high anticancer activity and less toxicity to normal tissues have been suggested as possible candidates for their capability to improve the efficacy of anticancer drugs. 42 In the present study, MRC-5, a normal human fetal lung fibroblast cell, were used to demonstrate that PUE exerted no cytotoxicity on normal lung cells. In conclusion, P urinaria potently inhibits lung carcinoma invasion and metastatic potential; therefore, it might be a potentially beneficial natural component for the prevention and treatment of lung cancer.
Footnotes
Authors’ Note
Pei-Ni Chen and the first author contributed equally.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants of National Science Council, Republic of China (NSC 98-2313-B-166-004-MY3 and NSC 98-2313-B-040-004-MY3).
