Abstract
Saraca asoca (Family Caesalpiniaceae) has been widely used in traditional Indian medicine especially due to its wound-healing property. The present study investigates the chemopreventive property of flavonoids from Saraca asoca (flowers) on 2-stage skin carcinogenesis in mice models. Skin cancer was induced in Swiss albino mice by single topical application of 7,12-dimethyl benzanthracene (100 µg/50 µL of acetone) followed by thrice a week treatment of croton oil for 20 weeks. The topical pretreatment of the flavonoid fraction from S asoca (FF S asoca) was 30 minutes prior to the application of croton oil thrice weekly for 20 weeks. At the end of the experimental period the animals were sacrificed, and the tumor statistics and various marker parameters were studied (enzyme assays, Western blotting). The pretreatment of the FF of S asoca caused significant reduction in the number of tumors per mouse and the percentage of tumor-bearing mice. Also, the latency period for the appearance of the first tumor was delayed by S asoca pretreatment. In plant-treated animals there was a significant increase in the levels of reduced glutathione, catalase, and protein in skin when compared with the untreated animals. Conversely, there was a significant decrease in the lipid peroxidation levels. A significant reduction in the expression of ornithine decarboxylase, a key enzyme in the promotion stage of 2-stage skin cancer, in the plant-treated group was also observed. These findings suggest the chemopreventive activity of flavonoids from S asoca on 2-stage skin carcinogenesis.
Introduction
The role of plants in the prevention and treatment of disease has been known from the dawn of civilization. Plants maintain the health and vitality of individuals and also cure diseases, including cancer without causing toxicity. More than 50% of all modern drugs in clinical use are of natural products, many of which have the ability to control cancer cells. 1 According to the estimates of the World Health Organization, more than 80% of people in developing countries depend on traditional medicine for their primary health needs.
Over the past decade, herbal medicines have been accepted universally, and they have an impact on both world health and international trade. Hence, medicinal plants continue to play an important role in the health care system of a large number of the world’s population. 2 The use of plants and phytomedicines has increased dramatically worldwide in the past 2 decades. Herbal products have been classified under “dietary supplements” and are included with vitamins, minerals, amino acids, and “other products intended to supplement the diet.” 3 In fact, there are several medicinal plants all over the world, including India, that are being used traditionally for the prevention and treatment of cancer. Now scientific validation of the same is gaining prominence. Natural plant products such as curcumin have inhibitory effects on tumor initiation. 4 Ellagic acid in fruits, nuts, and vegetables inhibits chemically induced cancer in the lungs, liver, skin, and esophagus of rodents. 5 The antioxidant effects of the flavonoids contribute to their anticarcinogenic ability by reducing the bioavailability of the carcinogens and by interfering with their biotransformation in the liver. 6
Developing novel strategies to prevent skin cancer represents a desirable goal due to increasing rise in the incidence of skin cancer patients throughout the world.7,8 It has been estimated that of all new cancers diagnosed annually in the world almost one third originates in the skin. 9 Skin carcinogenesis is a multistep process comprising initiation, promotion, and progression, where environmental toxins play a major role in initiation and their clinical appearance. 10
Cancer chemoprevention is a means of cancer control in which the occurrence of the disease can be entirely prevented, slowed, or reversed by the administration of 1 or more naturally occurring or synthetic compounds. 11 The supplementation or topical application of synthetic agents such as retinoids or vitamins and natural agents such as flavonoids or polyphenols have shown promise for their development as chemopreventive agents. The recent trend worldwide has been in favor of phytochemical therapeutics as they are economical and largely free from adverse side effects.
Saraca asoca
Flowers of Saraca asoca (Roxb.) De Wilde (Family Caesalpiniaceae) has been widely used in the Ayurvedic system of medicine especially due to its wound healing property. 12 S asoca is highly regarded as a universal panacea in Ayurvedic medicine. This versatile plant is the source of various types of compounds. In the present scenario, S asoca is an ancient and reliable source of medicine. 13 The flowers of S asoca mixed with coconut oil have been used traditionally as medicated oil for the treatment of scabies. The bark has been used as a chemopreventive in the 7,12-dimethyl benzanthracene (DMBA) induced skin cancer models. 14 Antibacterial activity of both the leaves and flowers of S asoca have been reported. 15 Oxytocic activity of the plant has also been reported. 16
Phytochemical studies have reported the presence of oleic, linoleic, palmitic, and stearic acids, P-sitosterol, quercetin, kaempferol-3-0-P-D-glucoside, quercetin-3-0-P-D-glucoside, apigenin-7-0-p-D-glucoside, pelargonidin-3,5-diglucoside, cyanidin-3,5-diglucoside, palmitic, stearic, linolenic, linoleic, p and y sitosterols, gallic acid, and leucocyanidin in the flower of S asoca. 13
As skin is the most widely exposed tissue to environmental carcinogens and the progression/cure of skin cancer can be visualized by the naked eye, we selected skin cancer as a model system for the present study. The objective of the study was to determine the role of bioactive component of S asoca as a chemopreventive agent. Furthermore, its effect on croton oil–induced activation of ornithine decarboxylase (ODC), a biochemical hallmark of tumor promotion, is also explored.
Materials and Methods
Extraction, Isolation, and Estimation of Flavonoids From Saraca asoca Flower
Fresh plant materials were collected, authenticated by an expert, and a specimen deposited at the Kerala University Botany Herbarium, Trivandrum, India (No. KUBH-5789-Cibin). The plant material was washed thoroughly and dried in shade. The dried plant material (1 kg) was then crushed and extracted in a soxhlet apparatus using 85% ethanol. The extract was decanted, filtered, and distilled on a water bath to remove the solvent. The extract was cleared of low-polarity contaminants such as fats, terpenes, chlorophyll, and xanthophyll by repeated extraction with petroleum ether (60-80°C) and ethyl acetate. The ethyl acetate fraction contained the bulk of polyphenols, which contain the flavonoid fraction. This was evaporated in vacuum, and the flavonoid content in this fraction (254 mg/kg) was determined by the method of Eskin et al 17 using quercetin as reference.
Animal Models
The animal models for the in vivo study (Swiss albino mice, male, 6-8 weeks old, weighing 20-25 g) were from the departmental animal house. For maintaining the experimental animals, the institutional ethical guidelines were strictly followed as per CPCSEA (Committee for the Purpose of Control and Supervision of Experiments on Animals) rules, Government of India (Sanction No.: IAEC-KU-15/05-06-BC-AA (7) (ii)). The animals were weighed weekly and food and water consumption rates were recorded. The animals were observed regularly for the detection of any lesion and tumor development.
In vivo 2-Stage Carcinogenesis Using DMBA
The mice were divided into 5 groups consisting of 10 mice in each group as follows:
Group I: Normal mice. Mice were on normal diet with no treatment.
Group II: Solvent alone. Acetone was applied topically over the shaven area of the skin of mice for 20 weeks.
Group III: Flavonoid fraction alone. The flavonoid fraction of SA (10 mg/kg body weight in sterile distilled water) was topically applied over the shaven area of the skin for 20 weeks.
Group IV: DMBA + croton oil. Single topical application of DMBA (100 µg/50 µL of acetone) over the shaven area of the skin. Two weeks later, croton oil (the promoter; 1% in acetone) was applied topically 3 times a week until the end of the experimental period.
Group V: DMBA + FF of SA + croton oil. Single topical application of DMBA (100 µg/50 µL of acetone) over the shaven area of the skin. Two weeks later, croton oil (the promoter; 1% in acetone) was applied topically 3 times a week till the end of the experimental period. The flavonoid fraction of SA (10 mg/kg body weight in sterile distilled water) was topically applied 30 minutes prior to croton oil application.
For the induction of skin tumors, dorsal hair between the cervical and caudal portions of group IV and V animals was removed using a surgical clipper, 2 days prior to the initiation of the experiment. DMBA (100 µg dissolved in 50 µL acetone) was used as a tumor initiator and applied to the skin of mice in groups IV and V. After 14 days, the tumor initiation by DMBA was promoted by the topical application of croton seed oil (1% v/v in acetone) thrice a week for the next 20 weeks. 18
The body weight of each animal and papillomas appearing on the shaved area of the skin were recorded at weekly intervals. Tumor-promoting activity was evaluated by determining both the proportion of tumor-bearing mice and the number of tumors (>2.00 mm in diameter) per mouse. 19 Tumor volume and burden were calculated as described by Subapriya and Nagini. 20
Treatment of the flavonoid fraction
The application of flavonoid fraction of S asoca (10 mg/kg body weight) was 30 minutes prior to the application of croton oil thrice weekly for 20 weeks. In the FF alone treated group (group III), S asoca was applied topically for 20 weeks.
After the treatment period, mice from all groups were euthanized by sodium pentothal injection. Liver and kidney were collected, homogenized in appropriate buffers, and used for biochemical estimations. Skin flaps and tumor tissues from the back of mice were excised. Skin/tumorous tissues for biochemical studies were immediately frozen at −80°C until used for estimations.
Biochemical Parameters
Various biochemical parameters were assayed according to standard procedures.
Activity of catalase
Catalase activity was measured by the method of Aebi. 21 The reaction mixture contained enzyme preparation (in 50 mM phosphate buffer, pH 7) and 30 mM H2O2 (in 50 mM phosphate buffer, pH 7). The reaction was started by the addition of the substrate and decrease in absorbance monitored at 240 nm for 30 seconds at 25°C. The difference in absorbance per unit time was expressed as the activity.
Levels of reduced glutathione (GSH)
Reduced GSH content of tissues were estimated by the method of Sedlak and Lindsay. 22 Tissues were homogenized in cold 20 mM EDTA solution on ice. After deproteinization with 5% TCA, an aliquot of the supernatant was allowed to react with 150 µM DTNB. The product was detected and quantified spectrophotometrically at 416 nm. Pure GSH was used as standard for establishing the calibration curve.
Levels of thiobarbituric acid reactive substances (TBARS)
Levels of TBARS were estimated by the method of Niehaus and Samuelsson. 23 Tissue homogenate was prepared in 0.1 M Tris–HCl buffer. The homogenate was combined with TCA–TBA–HCl reagent and mixed thoroughly. The tubes were boiled for 15 minutes and the precipitate removed after cooling by centrifugation at 1000 × g for 10 minutes. Absorbance of the samples was read at 535 nm against a blank (without tissue homogenate). Results are expressed as micromoles per gram tissue and calculated from the extinction coefficient of MDA.
Levels of conjugated diene
Levels of conjugated diene were estimated by the method of Recknage and Ghoshal. 24 Tissue homogenate prepared in Tris–HCl (0.1 M, pH 7.5) was centrifuged and the supernatant was mixed with chloroform–ethanol mixture (2:1). The content was centrifuged at 1000 × g for 5 minutes and the upper layer removed by aspiration. The lower chloroform layer was taken in a test tube and evaporated to dryness at 45°C in a water bath. The residue was dissolved in cyclohexane and absorbance noted at 233 nm against a cyclohexane blank. The concentration of conjugated dienes was calculated from its extinction coefficient (2.52 × 10 m−1 cm−1).
Estimation of protein content
Protein content of the sample was estimated by the method of Lowry et al. 25 To the sample, distilled water and alkaline copper sulfate reagent were added and incubated for 10 minutes at room temperature. To this, Folin’s phenol reagent was added, incubated for 20 minutes at room temperature, and absorbance measured at 670 nm. Protein concentration was calculated from a standard curve (BSA) and measured against a reagent blank.
ODC expression (Western blotting)
Swiss Albino mice (male, 6 weeks of age) were topically treated on their shaven back with the indicated doses of flavonoid fraction of S asoca in 0.2 mL vehicle (DMSO–acetone: 15-85 v/v) 30 minutes prior to the application of 10 nmol croton oil in 0.2 mL vehicle. Mice in the induction group were topically treated with 10 nmol croton oil in 0.2 mL vehicle. Control mice were treated with vehicle alone. Four hours later, the mice were sacrificed and their dorsal skin was excised for isolation of protein. After the fat was removed on ice, the remaining skin tissues were immediately pulverized in liquid nitrogen. The pulverized skins were homogenized and lysed in ice-cold lysis buffer (20 mM Tris–HCl, pH 7.5, 150 mM NaCl, 1 mM Na2 EDTA, 1 mM EGTA, 1% Triton, 2.5 mM sodium pyrophosphate, 1 mM β-glycerophosphate, 1 mM Na3VO4, 1 µg/mL leupeptin) containing protease inhibitors for 1 hour. The lysates were centrifuged at 14 000 rpm for 30 minutes at 4°C. The supernatant was collected and the protein concentration was determined. The aliquot of total protein containing 30 µg protein was boiled in sodium dodesylsulfate (SDS) sample buffer for 5 minutes and resolved in 12% SDS–PAGE gel. After electrophoresis, the proteins in the gel were transferred to a nitrocellulose membrane (Biorad, Hercules, CA). The blots were blocked with 5% nonfat dry milk in phosphate-buffered saline containing 0.1% Tween-20 (PBST) for 2 hours at room temperature and then washed in PBST buffer. The membranes were incubated with a 1:1000 dilution of primary antibodies for ODC (Santa Cruz Biotechnology, Santa Cruz, CA) for 4 hours at 4°C. Blots were washed 3 times with PBST at 5-minute intervals followed by incubation with a 1:5000 dilution of horseradish peroxidase conjugated secondary antibody (Santa Cruz Biotechnology) for 1 hour and again washed in PBST 3 times. The transferred proteins were developed using NBT/BCIP solution.
Statistical Analysis
All statistical calculations were carried out with Statistical Package for the Social Sciences software program (version 10.0 for Windows). The values are expressed as the mean ± standard deviation. The data were analyzed using analysis of variance, and significant difference of means was determined using Duncan’s multiple range tests at the level of P < .05. 26
Results
The major aim of the present study was to evaluate the efficacy of flavonoids from S asoca against DMBA-induced skin carcinogenesis. The results of the present study are represented in Table 1 and Figures 1 to 5. The topical application of S asoca extracts on mouse skin prior to croton oil (promoter) application resulted in a strong protection against DMBA-induced tumor initiation, tumor promotion, and complete carcinogenesis. The protective effect of the flavonoid fraction is shown in terms of number of tumors per mice, percentage of tumor bearing mice, and tumor burden (Table 1).
Effect of Saraca asoca on Tumor Statistics

Abbreviation: DMBA, 7,12-dimethyl benzanthracene.
Tumor incidence in group iv was converted to 100% for normalization.

Concentration of MDA and conjugated dienes in the skin of mice
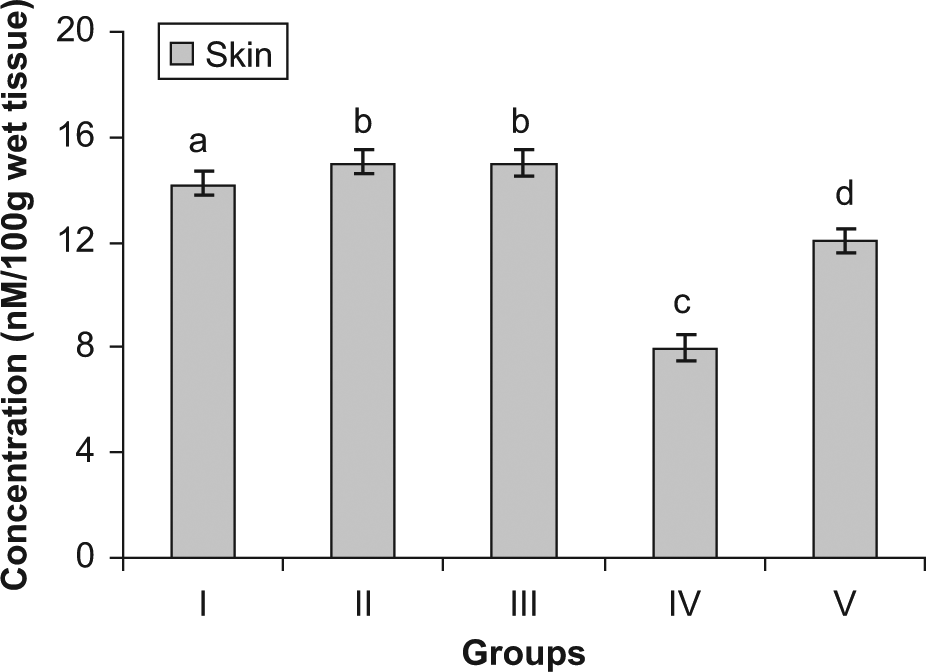
Concentration of GSH in skin

Activity of catalase in skin
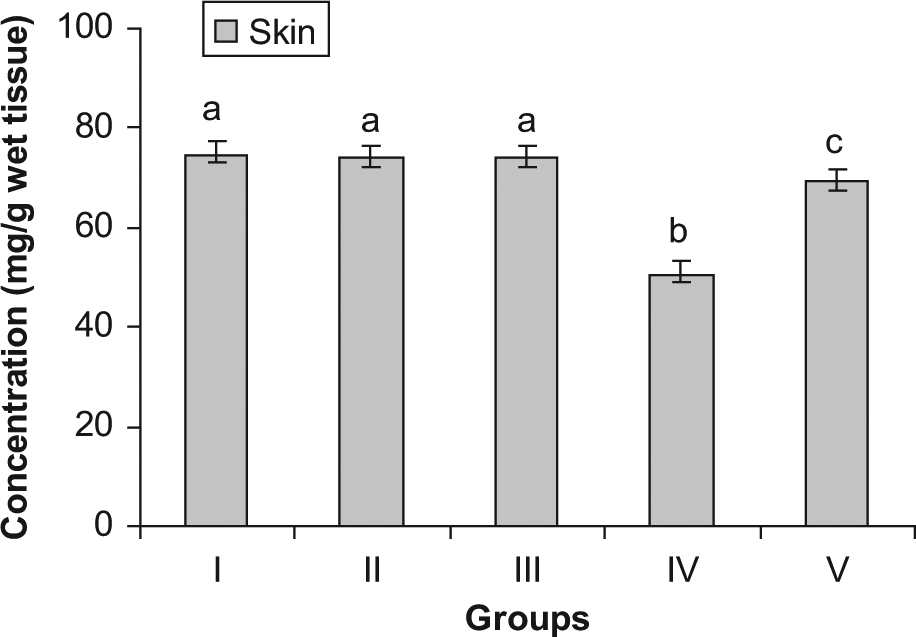
Concentration of protein in skin

Western blot analysis of ODC
Single topical application of DMBA followed by croton oil resulted in tumor formation, which started appearing from the eighth week. The tumor incidence in the DMBA/croton oil–treated mice reached 100% at the end of the experiment (20 weeks). The cumulative number of tumors in group IV mice was found to be 20, whereas in the S asoca–treated group (Group V) it was only 6. Similarly, the tumor burden was significantly reduced in the plant-treated group to 2. This suggests that the S asoca extract may be absorbed into the skin and activates a protective signaling cascade.
There was a significant decrease in the final body weight of animals in the tumor-induced group. The animals of all the other groups showed a gradual increase in body weight during the course of the experiment. No significant changes were observed in the diet and water consumption by the mice in all the groups when compared with the control animals.
The levels of various biochemical marker enzymes were also studied. The levels of glutathione peroxide, catalase, and protein were significantly reduced in the skin of the group IV (DMBA + croton oil) animals when compared with S asoca–treated animals (group V). Treatment with S asoca resulted in enhanced levels of GSH, catalase, and protein. A considerable elevation in the levels of MDA and conjugated dienes was noted in the skin after DMBA and croton oil treatment, whereas administration with S asoca significantly reduced the levels of MDA and conjugated dienes in the plant-treated group (group V). No significant change in these parameters was noticed in the FF alone treated group (group III) when compared with the normal mice.
We assessed the effect of topical application of S asoca on 12-O-tetradecanoylphorbol-13-acetate (TPA—a major constituent in croton oil)–enhanced expression of ODC protein in the epidermis. Western blotting analysis revealed that the application of S asoca significantly decreased the ODC expression when compared with the croton oil–treated group.
Discussion
Chemical carcinogenesis is a multifactorial and multi-step process of at least 3 distinct stages: initiation, promotion, and progression. 27 The initiation stage is essentially an irreversible step in which genetic change(s) occur in gene(s) controlling differentiation. 28 Initiation is achieved by a single treatment on the skin with a chemical carcinogen, such as DMBA. This is considered to result in a population of initiated cells that remain dormant until treatment with the promoter (TPA, or croton seed oil) stimulates clonal expansion, which leads to the formation of benign well-differentiated papillomas. A portion of these papillomas will progress to squamous carcinomas and eventually invasive undifferentiated carcinomas. 29 The promotion stage is reversible and tumor progression is an irreversible step and probably requires additional insult to the cellular genome of the papilloma. 28 In this study, skin carcinogenesis using DMBA and croton oil follows the same pattern. Lesions in the form of hardened and thickened skin were observed at the fifth week of the experiment. The appearance of tumor for the first time was noted at the eights week after DMBA application.
Reactive oxygen species (ROS) are constantly generated and eliminated in the biological system and play an important role in a variety of normal biochemical functions and abnormal pathological processes. There is substantial evidence indicating the involvement of ROS in the initiation, promotion, and progression stages of chemical carcinogenesis. 30 Free radicals are considered as key factors contributing to skin tumor promotion.31,32 Lipid peroxidation, a tactile process arising from the reaction of free radicals with lipids, is considered an important feature of the cellular injury leading to the deterioration of cellular constituents including lipids, proteins, and nucleic acids.33,34 The products of lipid peroxidation include malondialdehyde (TBARS), hydroperoxides, conjugated dienes, and so on. An increase in lipid peroxidation indicates serious damage to cell membranes and their fluidity, inhibition of several enzymes, and oxidation of proteins.
In the tumor-bearing animals, the levels of lipid peroxidation products MDA and conjugated dienes showed a significant increase when compared with the control animals (Figure 1). On treatment with the FF of S asoca (group V), the levels of lipid peroxidation products decreased significantly and approached almost normal values.
The DMBA treatment led to a decrease in the activity of the antioxidant enzyme catalase. Decreased catalase activity has been reported in a wide range of tumors earlier.35,36 However, on treatment with S asoca, the activity of this enzyme showed a significant increase when compared with the tumor-bearing animals. The concentration of GSH showed a significant decrease in the mice treated with DMBA. The constituents of the glutathione system including GSH play a major role in the deactivation of free radicals produced in the body. In the plant-treated group, there was a significant elevation of the aforementioned parameters when compared with the tumor-bearing animals. The application of tumor promoters causes a significant reduction in antioxidant defenses,37,38 and our results also show a similar pattern.
Ornithine decarboxylase is a rate-limiting enzyme in the biosynthesis of polyamines, spermidine, and spermine. An elevated level of ODC activity has been consistently detected in transformed cell lines 39 and plays a significant role in tumor promotion. 40 Application of TPA to certain cells elevates the level of ODC activity and incorporation of thymidine in promotion stage. 41 Thus, inhibition of ODC induction is a promising tool for screening inhibitors of tumorigenesis. Our data indicated that pretreatment with S asoca significantly inhibited the induction of ODC caused by croton oil treatment in mouse skin. Therefore, these findings confirmed that S asoca may inhibit cellular proliferation during the tumor-promotion stage.
From this study, it can be concluded that the flavonoid fraction of S asoca possess antioxidant activity and a potent chemopreventive property against DMBA-induced skin carcinogenesis. These activities can be attributed to the presence of compounds such as quercetin and gallic acid. Also, further studies are in progress in our laboratory to understand the various mechanisms regulating the chemopreventive action of S asoca and to ascertain the particular flavonoid component(s) responsible for these actions. The use of the flowers of the plant material (S asoca) can be recommended due to its edible nature, easy availability, and cost effectiveness. This work assumes significance since the recent trend worldwide is to identify therapeutics from natural sources mainly because most of the plants and plant products are largely free from adverse effects and also to translate traditional wisdom to a scientific platform.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
Financial assistance was received in the form of a senior research fellowship to T. R. Cibin from the Indian Council of Medical Research, Government of India.
