Abstract
Background. The concept of combination of chemoprevention holds great potential for cancer management as lower, clinically tolerable doses of individual agents could be achieved through therapeutic synergy. However, elucidation of their possible interactions—additive, synergistic, or antagonistic—must be thoroughly studied before considering for clinical use. Methods. To evaluate the effect of combination treatment of sulforaphane (SFN) and eugenol on HeLa cells, the authors performed cell viability assay, apoptosis assay, and reverse transcription polymerase chain reaction for gene expression analysis. Calculations of combination effects were expressed as a combination index (CI) with CI < 1, CI = 1, or CI > 1 representing synergism, additivity, or antagonism, respectively. Results. Simultaneous treatment with variable dose combinations of SFN and eugenol resulted in differential effects with an antagonistic effect at lower and synergistic at higher sub-lethal doses as reflected in cell cytotoxicity and apoptosis induction. Importantly, gemcitabine used in conjunction with the low- and high-dose combinations showed no significant cell death at lower doses suggesting that cell cytotoxicity is proportional to gemcitabine alone, whereas at higher sublethal doses of SFN and eugenol, it was found to act in a synergistic manner with gemcitabine. Furthermore, SFN and eugenol combinations at synergistic dose significantly downregulated the expression of Bcl-2, COX-2 and IL-β but not the antagonistic combinations. Conclusion. This study clearly indicates that 2 (or more) chemopreventive agents can act antagonistically or synergistically necessitating elucidation of possible mechanistic interactions for favorable and reliable outcomes of dietary components in the field of cancer prevention.
Keywords
Introduction
The potential of chemoprevention has been widely recognized as a strategy to block or delay the process of carcinogenesis to reduce the incidence, morbidity, and mortality of cancer. The potential chemopreventive agents (such as curcumin, genistein, gingerol, etc), are several nutrients and nonnutritive phytochemicals derived from many plants and have been shown to possess anticancer and anti-inflammatory properties.1-3 These agents might act through multiple biological mechanisms and by targeting different stages of carcinogenesis.4-6 Currently, a series of human intervention trials are being considered with individual phytochemicals or their combination with known synthetic chemopreventive agents.2,3
Single modalities or monotherapy (such as chemotherapy, radiotherapy, chemoprevention) has limited success resulting from bypass mechanisms leading to an increasing interest in the use of combinations of low doses of individual agents that interfere with multiple cell-signaling pathways to achieve a therapeutic synergy thereby increasing their efficacy and minimize toxicity.7-11 Indeed, chemotherapy is associated with several cytotoxic effects not only toward cancer cells but also normal cells leading to the manifested side effects.12,13 Accumulating data indicate that concurrent use of chemotherapy with chemopreventive agents may potentiate the efficacy of chemotherapy at lower doses, thus minimizing chemotherapy-induced toxicity.10,14 For example, curcumin, a chemopreventive agent with no discernable toxicity on normal cells, when used in combination with bortezomib and gemcitabine, synergistically inhibited the growth of multiple myeloma cells and pancreatic cells, respectively, as compared with controls.10,11 Thus, combinational drug modality may be used to improve cancer treatment outcomes.
The chemopreventive agents have been studied extensively owing to their safe toxicity profile. However, these agents when used singly need to be administered at considerably high doses to achieve an effective anticancer response, which may have undesirable toxic effects if used clinically. Moreover, vegetables and fruits contain several combinations of phytochemicals which interact by affecting superimposed and/or complementary mechanisms to influence cancer development. 15 In contrast, isolated pure compounds from natural diet may lose their biological function or may not act in the same way as in the original complex milieu.15,16 For instance, vitamin E when used singly was ineffective but in combination with selenite potentiated the anticarcinogenic potency. 17 Thus, a combination of chemopreventive agents may be more effective than the single agents in inhibiting carcinogenesis by different mechanisms.18,19 For this reason, studies on synergistic effects of different chemopreventive agents might contribute to the cancer modality management. Interestingly, the combination of chemopreventive agents may fail to significantly modulate the various molecular targets that were achieved by individual agents, indicating that combinations may antagonize the individual activities.15,20 Thus, development of cancer treatment modalities using chemopreventive agents in combination must be preceded by determining whether their interactions are synergistic, additive, or antagonistic.
Sulforaphane (SFN) and eugenol are widely studied potential chemopreventive agents.21-25 SFN is a phytochemical that displays both anticarcinogenic and anticancer activity and has been found to modulate many cancer-related events, including susceptibility to carcinogens—by inhibiting phase 1 detoxification enzymes and increasing the activity of phase 2 detoxification enzymes, apoptosis, inhibiting histone deacetylase (HDAC) activity, inducing cell cycle arrest, and inhibiting metastasis and angiogenesis by modulating the matrix metalloproteinases (MMPs) and vascular endothelial growth factor (VEGF).21,22 Also, eugenol has been shown to induce apoptosis and cell cycle arrest in cancer cells to and inhibit prostaglandin biosynthesis and cyclo-oxygenase-2 (COX-2) activity.23-25
Considering the chemopreventive potential of SFN and eugenol, the present study was designed to examine the possible interactions between the combination of sulforaphane and eugenol on human cervical cancer cells by evaluating their effects on cell viability, apoptosis, and various molecular targets. Furthermore, we wanted to investigate whether these combinations of chemopreventive agents could be used in conjunction with chemotherapeutic agents such as gemcitabine to enhance its therapeutic index by reducing cellular proliferative signals while enhancing apoptotic function.
Materials and Methods
Cell Culture
Human cervical carcinoma cell line, HeLa cell line used in this study was generously provided by Dr Ahmed H. Hassan, UAE University, Al-Ain, UAE. It was maintained in Dulbecco’s modified Eagle’s medium (Sigma, St Louis, MO) supplemented with 10% fetal bovine serum (Sigma), and 100X Pen-strep (Sigma) in a humidified atmosphere of 5% CO2 in air at 37°C.
Preparation of Drug Solutions
Sulforaphane was purchased from Sigma. A stock solution of 10 mM was prepared in dimethyl sulfoxide (DMSO; Sigma). The solution was stored in aliquots at −20°C. Further dilutions were made in complete medium to required concentrations for the treatment of HeLa cells.
Eugenol (purity 99%) was procured from Sigma, and a stock solution of 100 mM was prepared in ethanol. Further dilutions were made in complete medium to required concentrations for the treatment of HeLa cells.
A stock solution of 133 mM (40 mg/mL) of gemcitabine (Intas Biopharmaceuticals, Ahmedabad, India) was prepared in DMSO, and further dilutions were made in complete medium at concentrations of 15 and 25 mM.
Cell Viability Assay
The antiproliferative activity of combination of SFN (S1, 2.5 µM; S2, 5 µM; S3, 6.5 µM; and S4, 8 µM) and eugenol (E1, 100 µM; E2, 150 µM; E3, 200 µM; and E4, 350 µM) or in combination with gemcitabine (G1, 15 mM and G2, 25 mM) on HeLa cells was evaluated using the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay, as previously described.26,27 Cells (~7000 cells/well; in triplicates) were plated in 96-well plates and incubated overnight in complete medium at 37°C to obtain semiconfluent cultures. This was followed by treatment with varying combinations of SFN and gemcitabine (or in combination with different concentrations of gemcitabine) in complete medium for 24 hours. MTT (Sigma; final concentration 0.5 mg/mL) was added to each well at appropriate time and incubated for 2 to 4 hours at 37°C. The formazan crystals thus formed were dissolved in 100 µL DMSO. The absorbance was read at 570 nm using an Absorbance Microplate Reader (BioTek, Winooski, VT). Cell viability was calculated as percent of control (untreated cells) and averaged from 3 independent experiments.
Calculation of Combination Effects of Sulforaphane and Eugenol
Calculations of combination effects were based on the method of Chou and Talalay 28 and were expressed as a combination index (CI). CI analysis provides qualitative information on the nature of drug interaction, and CI, a numerical value, was calculated according to the following equation:
where CA,x and CB,x are, respectively, the concentrations of drugs A and B used in combination to achieve x% drug effect. ICx,A and ICx,B are the concentrations for single agents to achieve the same effect. A CI value <1, =1, or >1 represents, respectively, synergy, additivity, and antagonism of SFN and eugenol, respectively.
Apoptosis Assay
Caspase-3 colorimetric assay kit (Sigma, St Louis, MO) was used as per the manufacturer’s instructions for detection of apoptosis on treatment with SFN and eugenol or in combination with gemcitabine. Briefly, HeLa cells (~107) were seeded in a 25-cm2 flask and incubated overnight before being treated with a combination of SFN (S1, S2, S3, and S4) and eugenol (E1, E2, E3, and E4) or in combination with gemcitabine (G1 and G2) for 4 hours. Following incubation, the cells were lysed in the lysis buffer and the cell lysates were collected for caspase-3 assay as per the instructions. The optical density of the reaction was quantified spectrophotometrically at a wavelength of 405 nm. The concentration of the pNA released from the substrate by caspase-3 was then calculated from the absorbance values at 405 nm or from a calibration curve prepared with defined pNA solutions. Normalized caspase-3 levels were expressed as percentage of control. All data were derived from 3 independent experiments.
Expression Analysis of Bcl-2, COX-2, and IL-1β by RT-PCR
Total RNA extraction from untreated HeLa cells, SFN-treated cells (S1 and S3), eugenol-treated (E1 and E3), and combination of SFN and eugenol (S1 + E1 and S3 + E3) was carried out as per the manufacturer’s protocol using GenElute Mammalian Genomic Total RNA Kit (Sigma, St Louis, MO). ProtoScript M-MuLV Taq RT-PCR Kit (New England Biolabs, Ipswich, MA) was used to synthesize cDNA strand by reverse transcription from 5 µg of total RNA (at 42°C for 60 minutes) followed by reverse transcription polymerase chain reaction (RT-PCR) using gene-specific primers. The PCR cycle was as follows: initial denaturation at 95°C for 5 minutes, followed by 35 amplification cycles (denaturation at 94°C for 30 seconds, annealing Tm [β-actin, 56°C; Bcl-2, 55°C; COX-2, 57°C; IL-1β, 51°C] for 30 seconds, and extension at 72°C for 45 seconds), with final extension at 72°C for 7 minutes. Primer sequences used were as previously described.29-32 Amplified products were visualized on a 2% agarose gel containing ethidium bromide.
Statistical Analysis
All the results are expressed as means ± SD of at least 3 independent experiments. Fisher’s exact test was adopted for statistical evaluation of the results. Values of P < .05 were considered as statistically significant.
Results
Low-Dose Combinations of Sulforaphane and Eugenol Antagonize Their Effects on Cell Proliferation in HeLa Cells
Previous studies in our lab indicate the potential chemopreventive effects of sulforaphane and eugenol on the growth inhibition of HeLa cells. The LD50 of eugenol and sulforaphane on HeLa cells was found to be 500 and 12 µM, respectively (unpublished data). Because of the heterogeneous pattern of cervical cancer, one can hypothesize that a combination of different chemopreventive agents affecting various pathways involved in cervical cancer growth and survival may be advantageous. In this study, we explored the in vitro antiproliferative effects of various combinations of SFN and eugenol on HeLa cells to determine whether this combination results in a synergistic, additive, or antagonistic effect on the growth inhibition of these cells. HeLa cells were treated with different concentrations of SFN and eugenol alone (S1 or S2, and E1 or E2) and in combination (S1 + E1, S1 + E2, S2 + E1, and S2 + E2; Figure 1).

Low-dose combinational effect of sulforaphane (S1 and S2) and eugenol (E1 and E2) on HeLa cells over a duration of 24 hours
Our results demonstrated 16% and 26% cell death with S1 and S2 alone, respectively, and cells treated with E1 and E2 individually showed 19% and 24% decrease in cell viability, respectively. But when used in combination, we found at doses S1 + E1, there was only 4% decrease in the cell viability (Figure 1). Similar results were found at doses S1 + E2, S2 + E1, and S2 + E2 with only 6%, 10%, and 8% decrease in the cell viability, respectively (Figure 1). Interestingly, the combination of SFN and eugenol at these doses did not result in significant change in cell viability as compared with the same doses when used alone. At these low-dose combinations of SFN and eugenol, CI values were greater than 1, indicating that cell proliferation was affected in an antagonistic manner. Thus, it can be inferred that these combinations of SFN and eugenol result in antagonistic effects on cell viability of HeLa cells.
Cotreatments of Sulforaphane and Eugenol at Higher Doses Act in a Synergistic Manner to Inhibit Cell Proliferation in HeLa Cells
HeLa cells responded differently at higher dose combinations of SFN and eugenol with decreasing CI values at these dose combinations. When higher sublethal doses of both SFN (S3 and S4) and eugenol (E3 and E4) were used, our results demonstrated 30% and 37% cell death with S3 and S4 alone, respectively, and cells treated with E3 and E4 individually showed an 21% and 32% decrease in cell viability, respectively (Figure 2). But when the cells were treated with a combination of S3 + E3, we observed a 55% decrease in the cell viability (Figure 2). Similar results were observed with the other combination doses of S3 + E4, S4 + E3, and S4 + E4 with a 67%, 60%, and 75% decrease in the cell viability of HeLa cells, respectively (Figure 2). As is evident from Figure 2, significant synergy was noted between these 2 chemopreventive agents when used in combination at higher sublethal doses demonstrating an even higher cell death ranging from 60% to 75%.

High-dose combinational effect of sulforaphane (S3 and S4) and eugenol (E3 and E4) on HeLa cells over a duration of 24 hours
Synergism was strongest when the cells were treated with a combinational dose of SFN (S4) and eugenol (E4) for 24 hours with CI = 0.7. Taken together, low concentrations of SFN and eugenol appear to inhibit proliferation of HeLa cells in an antagonistic manner (CI > 1), whereas at higher dose combinations of SFN and eugenol, the compounds act in a synergistic manner.
Cytotoxicity of Gemcitabine With the Combinations of Sulforaphane and Eugenol at Various Doses: No Significant Growth Inhibition on HeLa Cells at Lower Doses Whereas Acts Synergistically at Higher Doses
As previously studied in our lab, the combination of gemcitabine with a chemopreventive agent (eugenol or sulforaphane) resulted in dose-dependent growth inhibition of HeLa cells in a synergistic manner (unpublished data). This suggests that depending on the combination ratio, the doses of chemotherapeutic drug, for a given degree of effect in combination, may be reduced.
Previous results from our lab found the LD50 of gemcitabine to be 35 mM on HeLa cells, 33 and the LD50 of eugenol and SFN was found to be 500 and 12 µM, respectively (unpublished data). At lower doses of eugenol and SFN (S1 + E1, S1 + E2, S2 + E1, and S2 + E2) in combination with sublethal doses of gemcitabine (G1 and G2), we observed no significant change in viability of HeLa cells in comparison with gemcitabine alone (Figure 3). The decrease in cell viability on treatment with a S1 + E1 + G1 combination was found to be ~70%, which was almost similar to that with gemcitabine alone (G1 = 75%). Similar results were obtained with the other combinations, that is, S1 + E1 + G2, S1 + E2 + G1, S1 + E2 + G2, S2 + E1 + G1, and S2 + E2 + G2, suggesting that at these concentrations SFN and eugenol are nullifying each other’s effect (Figure 3).
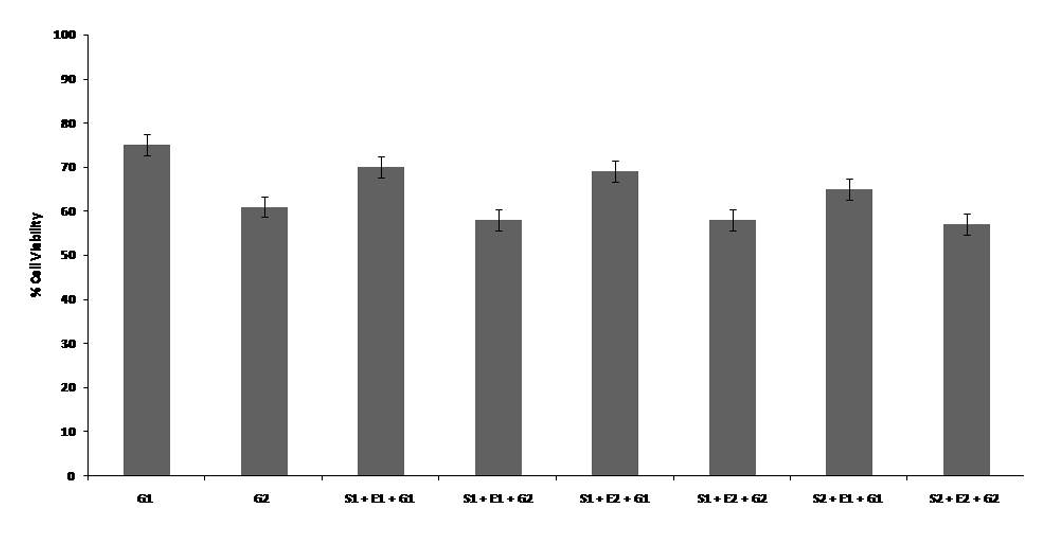
Combinational treatment of sulforaphane (SFN) and eugenol with gemcitabine at lower doses
Furthermore, the effect of combination of same sublethal doses of gemcitabine (G1 and G2) with higher sublethal doses of SFN (S3 and S4) and eugenol (E3) was studied. Our results demonstrated 25%, 30%, 34%, and 30% decrease in cell viability with G1, S3, S4, and E3 alone, respectively (Figure 4). On treatment with the combinations S3 + E3 + G1 and S4 + E3 + G1, there was a synergistic decrease in cell viability (18% and 12%, respectively) as compared with either of the compound alone (Figure 4). Thus, these results indicate that combinations of SFN and eugenol at higher sublethal doses with sublethal doses of gemcitabine could be used to potentiate the efficacy of gemcitabine in synergistic manner.
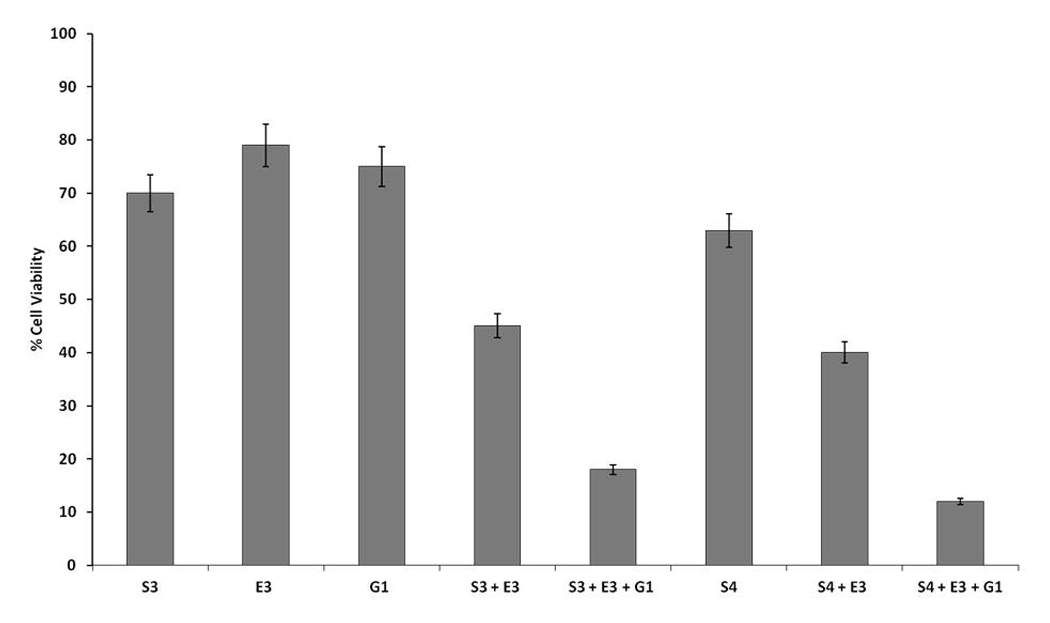
Synergistic growth inhibition of higher dose combination of sulforaphane (SFN) and eugenol with gemcitabine on HeLa cells
Low-Dose Combinations of Sulforaphane and Eugenol Fail to Induce Apoptosis in HeLa Cells
Caspase-3 is essential for propagation of the apoptotic signal after exposure to many DNA-damaging agents and anticancer drugs. Our results clearly indicate that at lower dose combinations of SFN and eugenol (S1 + E1, S1 + E2, S2 + E1, and S2 + E2), there was no significant effect on caspase-3 activity, demonstrating an antagonistic interaction between these 2 agents (Figure 5). Furthermore, when the combination of SFN and eugenol (S1 + E1) was combined with gemcitabine (G1), it showed caspase-3 activity proportional to gemcitabine alone (G1; Figure 5). This strengthens our hypothesis that at lower doses, SFN and eugenol antagonize each other’s effect.

Effect of low-dose combinations of sulforaphane (SFN) and eugenol (together and in combination with gemcitabine) on caspase 3 activity in HeLa cells
Cotreatments of Sulforaphane and Eugenol at Higher Doses Act in a Synergistic Manner to Increase Apoptosis in HeLa Cells
SFN and eugenol combinations at higher sublethal doses (S3 + E3, S3 + E4, S4 + E3, and S4 + E4) were found to increase caspase-3 activity in a synergistic manner compared with the drugs alone, indicating an increased apoptotic cell death induced by these combinations (Figure 6). Interestingly, gemcitabine (G1) in combination with SFN and eugenol (S3 + E3) also showed further increase in caspase-3 activity compared with gemcitabine alone (G1) or the said combination (S3 + E3) indicating a synergistic relationship between these drugs (Figure 6). These findings confirm that the combination of SFN and eugenol at higher doses can be used for therapeutic purposes alone or in combination with chemotherapeutic drugs to enhance the efficiency of treatment.

Activation of caspase-3 as a result of treatment with higher sublethal dose combinations of sulforaphane (SFN) and eugenol (or in combination with gemcitabine) in HeLa cells
Concurrent Treatment of Sulforaphane and Eugenol Downregulates Bcl-2, COX-2, and IL-1β Gene Expression in HeLa Cells
Bcl-2 has been established as a key player in the control of apoptosis. The aim of this study was to investigate the expression of Bcl-2 in HeLa cells before treatment and after treatment with a combination of SFN and eugenol at 2 different doses (S1 + E1 and S3 + E3) by RT-PCR analysis. As shown in our previous studies, HeLa cells express a high level of Bcl-2 (unpublished data). SFN (S1/S3) and eugenol (E1/E3) alone significantly reduced the expression of Bcl-2 (Figure 7). But when the cells were treated with the combination S1 + E1, there was no significant decrease in Bcl-2 expression compared to untreated HeLa cells (Figure 7). This correlates with the previous results showing no change in apoptotic index of HeLa cells treated with this combination. However, the other combination S3 + E3 showed significant downregulation of Bcl-2 as compared with either of the compounds alone (S3 and E3; Figure 7). This is consistent with the previous results that higher dose combinations of SFN and eugenol results in synergistic decrease in cell viability as well as increased apoptotic induction as compared with either compounds alone (Figures 2 and 6).

Sulforaphane (SFN) and eugenol combination at higher and lower sublethal doses differentially regulate Bcl-2, COX-2, and IL-1β expression in HeLa cells
COX-2 has been shown to be involved in inflammation and its upregulation has been reported in various cancers. As previously reported, COX-2 is highly expressed in HeLa cells (unpublished data). COX-2 expression was significantly reduced in response to treatment with SFN (S1/S3) and eugenol (E1/E3) alone (Figure 7). However, the combination S1 + E1 could not significantly alter COX-2 expression compared with untreated HeLa cells or either compound alone (S1 or E1; Figure 7). This compares with the above results showing no significant change in cell viability and caspase-3 activity of HeLa cells treated with this combination. Interestingly, the S3 + E3 combination showed significant abrogation of COX-2 expression compared with either of the compounds alone (S3 or E3; Figure 7). This is parallel with the previous results showing a synergistic effect of higher dose combinations of SFN and eugenol on cell viability and apoptosis (Figure 7). Thus, higher dose combinations of SFN and eugenol can be used as COX-2 inhibitors but not the lower dose combinations.
IL-1β, a pro-inflammatory cytokine, is involved in the proliferation, differentiation, and apoptosis of cells. As shown previously, HeLa cells were found to have high expression of IL-1β (unpublished data), which was significantly downregulated on treatment with either SFN (S1/S3) or eugenol (E1/E3) alone (Figure 7). The lower dose combination S1 + E1 was not able to modulate IL-1β expression significantly compared with untreated HeLa cells, whereas the higher dose combination S3 + E3 showed significant abrogation of expression of IL-1β (Figure 7). This is consistent with the previous results that higher dose combinations of SFN and eugenol results in synergistic decrease in cell viability as well as increased apoptotic induction as compared to either compounds alone (Figure 7). Thus, at lower doses, SFN and eugenol antagonized effects of each other demonstrating potential interference within signaling pathways, whereas at higher doses they potentiated each other’s effect of IL-1β.
Discussion
Treatment options for early-stage cervical cancer are well defined, and localized cervical cancer can be cured by several strategies. Yet in advanced cervical cancer, the therapeutic outcome is still disappointing. Conventional therapies have not proven to be sufficiently effective and are often limited by toxicity.34,35 Hence, there is considerable need to identify well-tolerated and easily applicable substances either for chemoprevention of cervical cancer in high-risk patients or for adjuvant chemotherapy in advanced stages. 36 Over the past decade, as molecular and biochemical mechanisms of the carcinogenesis process have been elucidated, the rationale of combining chemopreventive agents to target multiple pathways has strengthened.37-39
Several epidemiological studies indicate that consumption of dietary chemopreventive agents is strongly associated with reduced risk of chronic diseases, especially cancer.16,40 However, data from clinical trials suggest that the administration of pure chemopreventive agents obtained from diet does not explain the chemopreventive effects of whole food. This phenomenon may be accounted for by the complex mixture of phytochemicals present in the fruits and vegetables and additive and synergistic effects of these phytochemicals may be responsible for these potent antioxidant and anticancer activities.41-44 Moreover, the isolated pure compound may either lose its biological activity or may not act in the same way as in whole foods. Thus, an appropriate strategy for significantly reducing the risk of cancer would be through whole-food consumption.
Considering the above-mentioned facts, we proposed to quantitate the combination effects of SFN and eugenol using the method of Chou and Talalay 28 in terms of CI for their capability to inhibit cell growth and viability of cervical cancer cells. To our knowledge, this is the first time that the effects of these 2 chemopreventive agents have been determined in terms of CIs. As single agents, both were found to be effective in suppressing growth of HeLa cells in a dose-dependent manner (unpublished data). The results are means of 3 independent experiments (P < .05). Combination treatment resulted in differential effects on HeLa cells at different doses, being more effective in inhibiting HeLa cell proliferation at higher doses (S3 + E3, S3 + E4, S4 + E3, and S4 + E4) than at lower doses (S1 + E1, S1 + E2, S2 + E1, and S2 + E2; Figures 1 and 2).
At lower dose combinations (S1 + E1, S1 + E2, S2 + E1, and S2 + E2), no significant effect on growth inhibition of HeLa cells was observed, pointing to an antagonistic effect (CI > 1) of these combinations at the doses used (Figure 1). Khafif et al 20 also demonstrated an antagonistic interaction between (−)-epigallocatechin-3-gallate (EGCG) and curcumin at lower doses in normal, premalignant, and malignant human oral epithelial cells. Also, Pappa et al 45 found an antagonistic relationship between SFN and 3,3′-diindolylmethane (DIM) at low total concentrations (below 20 mM), which are physiologically more relevant. These studies highlight the need for elucidating mechanistic interactions for better drug designing using bioactive food components.
However, a synergistic effect (CI < 1) was found at higher doses of combination (S3 + E3, S3 + E4, S4 + E3, and S4 + E4; P < .05; Figure 2). A combination of xanthorrhizol and curcumin has been shown to act in a synergistic manner to inhibit growth via apoptosis induction in human breast cancer cells. 46 Also, indole-3-carbinol and genistein were shown to synergistically induce apoptosis in human colon cancer cells. 19 Results of these analyses indicate that combinatory interactions between SFN and eugenol are strongly dose dependent.
This study is consistent with another report, which found antagonism between SFN and DIM at low-dose combinations and with increasing doses, synergism was observed. A possible explanation for this phenomenon might be that at low concentrations, mechanisms other than cell cycle arrest or apoptosis induction play a more prominent role for chemopreventive potential. 47 Also, SFN at concentrations as low as 1 to 5 µM was very effective in inducing phase 2 detoxification enzymes.47-49 Therefore, treatment with low doses of SFN might cause increased “detoxification” of the compounds themselves or of reactive intermediates involved in inhibition of cell proliferation, resulting in the observed antagonistic effects.
The additive and synergistic effects of chemopreventive agents present in fruits and vegetables are known to account for their potent antioxidant and anticancer activities.40,50 van Breda et al 51 showed that the effect of 4 different vegetables on gene expression pattern differed from the effect of the mixture of these 4 vegetables in female mice. The above combination was found to modulate the gene expression, which was not significantly affected by one of the specific vegetables present in the mixture, whereas the individual vegetables were able to alter the gene expression which was not significantly modulated by the mixture. Thus, combinations of different foods containing different complex mixtures of phytochemicals can also have an antagonistic effect on gene expression.15,51 The phytochemicals present in fruits and vegetables vary in their bioavailability and distribution in different macromolecules, subcellular organelles, cells, organs, and tissues, which may be because of differences in molecular size, polarity, and solubility and may also affect interactions between these agents. The combination of orange, apple, grape, and blueberry was found to display a synergistic effect in antioxidant activity, suggesting synergistic effects after the combination of these fruits. Therefore, before individual phytochemicals or their combinations develop into cancer treatment modality, a comprehensive understanding of their interactions, efficacy, and the long-term safety is necessary.
The concept of combination chemoprevention seeks to increase the chemopreventive effectiveness of chemotherapeutic agents, while decreasing toxicity by dose reduction.10,11,52,53 Interestingly, when SFN and eugenol combinations were used concurrently with gemcitabine, we observed that at lower doses SFN and eugenol (S1 + E1 + G1, S1 + E1 + G2, S1 + E2 + G1, S1 + E2 + G2, S2 + E1 + G1, and S2 + E2 + G2) nullified each other’s effect such that the observed decrease in cell viability could be accounted for by gemcitabine alone (G1 and G2) whereas when higher dose combinations of SFN and eugenol (S3 + E3 + G1 and S4 + E3 + G1) were simultaneously taken with gemcitabine (G1), a synergistic decrease in cell viability was observed (P < .05; Figures 3 and 4). Thus, at higher doses SFN and eugenol potentiate the efficacy of gemcitabine whereas at lower doses there is no beneficial effect of SFN and eugenol. Thus, chemopreventive agents can be used to enhance the efficacy of chemotherapeutic drugs. 54
Chemotherapeutic drugs (including gemcitabine) and chemopreventive agents (such as eugenol and SFN) exert their effects by inducing apoptosis in cancer cells.24,55-59 This is a desirable property for anticancer agents as unlike necrosis, apoptosis does not result in wide spread inflammation. Thus, we performed apoptosis assay using the various SFN and eugenol combinations. There was no significant change in caspase-3 activity, a key enzyme involved in apoptosis, on treatment with the lower dose combinations of SFN and eugenol (S1 + E1, S1 + E2, S2 + E1, S2 + E2) as compared with the untreated cells (P < .05; Figure 5). This is consistent with previous studies in which low-dose combinations of 2 chemopreventive agents failed to induce cell cycle changes or apoptosis in cancer cells.20,45 However, caspase-3 was significantly higher in HeLa cells treated with higher dose combinations of SFN and eugenol (S3 + E3, S3 + E4, S4 + E3, S4 + E4) compared with the individual drug treatment (Figure 6). Similarly, when these low- and high-dose combinations of SFN and eugenol were used in conjunction with gemcitabine (S1 + E1 + G1 and S3 + E3 + G1, respectively), we found similar results as with cytotoxicity assay (Figures 5 and 6). This confirms our hypothesis that the combination of SFN and eugenol is antagonistic at lower doses whereas it is synergistic at higher doses. These results are supported by previous studies showing chemopreventive agents when used simultaneously results in synergistic increase in apoptotic index.19,46 Thus, the strategies of cancer treatment using combined therapies or combined agents with distinct molecular mechanisms are considered more promising for higher efficacy, resulting in better survival.60-63
Chemopreventive agents might reduce the cancer risk through various mechanisms and by targeting different stages of carcinogenesis. 4 The failure to undergo of apoptosis is associated with various diseases such as cancer, autoimmune disorders and viral infections. Bcl-2, involved in apoptosis, plays an important role in cancer and resistance of cancer to conventional therapies.64,65 As previously reported, various cancer cell lines, including HeLa cells express high levels of Bcl-2.66,67 However, HeLa cells treated with SFN (S3) and eugenol (E3) alone as well with synergistic combination (S3 + E3) showed significant downregulation of Bcl-2 after treatment as compared with the untreated cells (U; Figure 7). Importantly, when the cells were treated with lower dose combination of SFN and eugenol (S1 + E1), there was no significant decrease in Bcl-2 expression compared with untreated HeLa cells (U; Figure 7). This further confirms that low-dose combination of SFN and eugenol results in interference in the signaling pathways thereby resulting in antagonistic effects on growth inhibition and induction of apoptosis as compared with either compounds alone or higher dose combination of SFN and eugenol. Various chemopreventive agents, such as curcumin, green tea, 6-gingerol, resveratrol, SFN, and so on, alone and in combination with other agents downregulate Bcl-2.27,66,68,69 Previous studies have established that the functional blockade of Bcl-2 may drive the tumor cells toward apoptosis or sensitize them for chemotherapy and radiotherapy.70-73
Numerous preclinical studies point to the importance of regulation of COX-2 expression in the prevention and most important, in the treatment of several malignancies.74,75 Complicating matters further is the fact that conventional cancer therapies such as radiation and chemotherapy can induce COX-2 and prostaglandin biosynthesis. Thus, inhibition of this enhanced COX-2 activity in tumors clearly has a therapeutic potential. 74 Previously, it has been reported that HeLa cells express high levels of COX-2 (unpublished data). However, treatment with SFN (S1/S3) and eugenol (E1/E3) alone as well as in combination (S3 + E3) resulted in significant downregulation of COX-2 as compared with the untreated control (Figure 7). Additionally, when the cells were treated with the antagonistic combination of SFN and eugenol (S3 + E3), there was no significant decrease in COX-2 expression compared with untreated HeLa cells (U; Figure 7). Other chemopreventive agents have been shown to possess significant COX-2 inhibiting activity.27,74,76-78 Also, COX-2 inhibitors along with chemotherapeutic agents have been found to enhance therapeutic effects in various cancers.63,79
A number of clinical studies have suggested IL-1β is associated with a number of human tumors.80-82 HeLa cells express a high level of IL-1β (unpublished data). Treatment with SFN (S1/S3) and eugenol (E1/E3) alone as well in combination (S3 + E3) showed significant downregulation of IL-1β as compared with the untreated control (U; Figure 7). But when the cells were treated with the antagonistic combination of SFN and eugenol (S1 + E1), IL-1β expression was not found to change significantly compared with untreated HeLa cells (Figure 7). IL-1β is known to activate the nuclear factor-κB pathway and thus is involved in inflammation and carcinogenesis.83,84 Other studies have also reported downregulation of IL-1β expression by various chemopreventive agents.85-87 In another interesting study, a combination of indole-3-carbinol and tamoxifen resulted in enhanced effects of chemotherapy, which may be related to immunopotentiating activities through reduction of interleukin-6 (IL-6) and enhancement of lymphocyte proliferation, natural killer cell cytotoxicity, CD4+/CD8+ ratio, IL-2, and interferon-γ productions. 62 Therefore, SFN and eugenol can be used in combination at higher doses for better cancer management.
Conclusion
This study presents an insight into the possible synergistic or antagonistic relationship between 2 chemopreventive agents—SFN and eugenol (alone or in combination with chemotherapeutic agent, gemcitabine). The concept of combination chemoprevention holds great potential and needs to be carefully investigated because of potential antagonistic effects in the clinical setting despite promising results of a monotherapy. The lower doses of cancer therapy agents used in combination may allow each agent to be administered at clinically more tolerable dosages than when administered as single agents. This paradigm provides a platform to elucidate mechanistic interactions and dose-dependent differences in the outcomes of combination treatments and may serve as a model for a continuing mechanistic approach to chemoprevention.
Footnotes
Acknowledgements
The authors are grateful to Dr B. Ramjee, Director, and Dr Firdos Alam Khan, Chairperson, Department of Biotechnology, Manipal University, Dubai, for the constant support and encouragement.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article:
This work was funded by the Intramural Research Program, Department of Biotechnology, Manipal University, Dubai, United Arab Emirates.

