Abstract
Laser immunotherapy (LI) has been demonstrated to be a promising modality for cancer treatment. The present study was designed to further investigate the impact of LI combined with surgery. LI consists of a near-infrared laser, a light-absorbing dye (indocyanine green, ICG), and an immunostimulant (glycated chitosan, GC). ICG and GC were intratumorally injected, followed by laser irradiation. Female BALB/c mice bearing EMT6 tumor cells were divided into 4 groups: control, LI, LI followed by immediate surgery resection of residual tumor (LI + S0wk), and LI followed by surgical removal of residual tumor after 1 week (LI + S1wk). Successfully treated mice from all treatment groups were rechallenged twice with 105 and 5 × 105 EMT6 cells, respectively. The LI + S1wk group had the highest survival rate (72%) after 90 days, whereas the mice survival rates of the LI + S0wk, LI, and control groups were 50%, 46%, 0%, respectively. The median survival times of control, LI, LI + S0wk, and LI + S1wk groups were 32, 66, 74, and 90 days, respectively. Survival rates of the treated mice after the first and second tumor rechallenges, ranging from 73% to 95%, were not significantly different among the 4 groups (P > .05). The results show that LI is a useful tool for the treatment of tumor-bearing mice. Long-term antitumor effect can be induced by LI. They also indicate that combination of LI with surgery can further improve the therapeutic efficiency of LI.
Keywords
Introduction
An ideal tumor treatment modality should not only eradicate the treated primary tumors but also control untreated metastases at remote sites. The ultimate long-term control of cancer lies in the host immune surveillance and defense system. 1 Much has been learned about the potential of the immune system to control cancer and the various ways that immunotherapy can boost the host immune system in cancer treatment. Many new strategies have been proposed, including cytokine therapy, dendritic cell–based vaccines, and immune-activating antibodies, which are starting to be used in clinical studies, either alone or in combination with other treatment modalities.2-4
These new methods are expected to result in improved cancer treatment outcomes and, eventually, the cure of cancer. However, these immunotherapy strategies have so far had limited success with response rates usually in the range of 5% to 10% among treated patients. 5 Most studies indicated that immunotherapy could be an effective tool in treating cancer, but much needs to be learned and explored in order to improve its therapeutic effect. In the studies using immunotherapy for cancer treatment, immunoadjuvants have been shown to play an important role. 6
Laser immunotherapy (LI), a combination of local selective photothermal therapy and immunological stimulation using immunoadjuvant, was proposed in 1997. 7 The strategy of LI is to directly destroy the tumors at the treatment site and to induce tumor-specific host immune responses. It consists of 3 components: (a) a near-infrared laser, (b) a light-absorbing dye—indocyanine green (ICG), and (c) an immunoadjuvant—glycated chitosan (GC).8,9 It involves in situ administration of the dye and the adjuvant and a local laser irradiation. Preclinical studies and preliminary clinical trials for late-stage melanoma patients have revealed promising outcomes.10,11 The standard procedure of LI leaves the treated tumor at the primary site. It is hypothesized that treated tumor cells can provide an antigen load, along with the adjuvant-assisted immunological stimulation, generating a long-term antitumor immunity. However, these treated tumors may have residual, viable tumor cells that could continue to multiply and metastasize. This can be problematic, especially when LI is used in clinical applications. Thus, residual tumors become an issue that needs to be addressed. Is it possible to strike an optimal balance between using the residual tumors for immunological stimulation and removing the residual tumors to prevent tumor invasion and metastasis after LI treatment?
The present study was designed to investigate the effects of LI in combination with surgical removal of LI treated tumors in order to understand the balance and relationship between the presence of treated tumors for stimulation of tumor-specific immune response and surgical removal of residual tumors for prevention of potential tumor local invasion and distant metastasis. Moreover, it is of clinical importance to investigate whether surgery, possibly as a new LI component, can further improve the therapeutic efficiency of LI.
Materials and Methods
Tumor Cell Line
EMT6, a BALB/c breast sarcoma line, 12 was used in this study. Cells were cultured in Waymouth’s MB 752/1 medium (Invitrogen, Carlsbad, CA) with 15% fetal bovine serum, 100 U/mL penicillin, and 100 U/mL streptomycin (Sigma, St. Louis, MO) at 37°C in a humidified atmosphere of 5% CO2/95% air. The cells were harvested and prepared in the medium (1 million cells per mL) for injection.
Animal Model
Female BALB/c mice, aged 6 to 8 weeks, were used. Mice were anesthetized with a gas mixture of isoflurane (2%) and oxygen before any invasive procedures (laser irradiation or surgery). After the completion of laser irradiation and/or surgical resection, mice were allowed to recover. All animal experiments were approved by the Institutional Animal Care and Use Committee and were in compliance with National Institutes of Health guidelines. All BALB/c mice were depilated on the back; they were then injected subcutaneously with 105 EMT6 cells suspended in 100 μL of phosphate-buffered saline. Tumors grew predictably in all mice and reached a size of 5 to 10 mm in diameter 8 to 10 days after injection. Tumor growth was assessed 2 times a week throughout the entire experiment. The orthogonal tumor dimensions (a and b) were measured with a Vernier caliper. The tumor volume was calculated according to the formula, V = (4π/3)[(a + b)/4]3. Mice were monitored carefully throughout the study and were preemptively euthanized when they became moribund. Mice were considered cured when they were healthy and tumor free 90 days after treatment.
Laser Immunotherapy
GC, the immunoadjuvant used in our study, was prepared in our lab by incubating an aqueous suspension of chitosan with a 3-fold excess of galactose and subsequent stabilization by borohydride reduction of a mixture of Schiff bases and Amadori products. GC of 1% concentration was injected in the center of the tumor for immunological stimulation 1 day before laser irradiation. ICG, obtained from Akorn Inc (Buffalo Grove, IL), is a light-absorbing dye with an absorption peak at 800 ± 5 nm. It was injected into the center of the tumor 30 minutes before laser irradiation to enhance laser light absorption with a concentration of 0.25% and a volume approximately equal to half of the tumor volume. Laser irradiation was conducted using a diode laser emitting 805-nm light (Diomed 25, Diomedics, The Woodlands, TX). The laser energy was directed to the treatment site through an optical fiber. The output power density used was 1 W/cm2, and the irradiation duration was 10 minutes.
Surgical Removal of Tumors
Mice were anesthetized and the skin above the tumor was shaved and aseptically prepared. An elliptical skin incision was made, and tumor borders were established with blunt and sharp dissection. The entire tumor was removed along with wide margins of normal tissue on all sides. Skin was then closed using 5-0 prolene. Tumor removal was performed immediately after LI or 1 week after LI.
Tumor Rechallenges
All cured mice went through two consecutive rechallenges with EMT6 cells. The first rechallenge was performed 90 days after LI with a tumor dose of 1 × 105 cells/injection. All the surviving mice from the first rechallenge were rechallenged again 60 days later with a tumor dose of 5 × 105 cells/injection. After each EMT6 rechallenge, tumor growth and animal survival were monitored as described earlier.
Study Design
Mice were randomly assigned to one of the following groups: (a) control group without any treatment (15 mice), (b) LI treatment group (LI, 37 mice), (c) LI treatment followed by immediate surgical removal of tumor (LI + S0wk, 30 mice), and (d) LI treatment followed by surgical removal of tumor after 1 week (LI + S1wk, 36 mice). Figure 1 shows the time point of each procedure of the study.
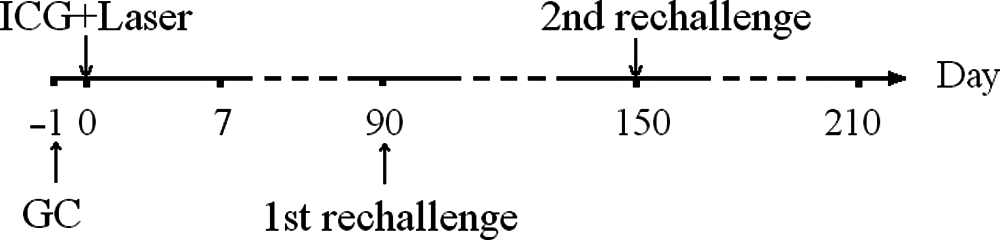
Study design of the experiment
Statistical Analysis
Survival analysis was performed using the Kaplan–Meier method. Differences in survival rates among different treatment groups were determined using log-rank multiple comparison tests (SAS 9). Differences in survival rates among each rechallenge groups were determined by Fisher’s exact test. P < .05 was considered significant.
Results
Animal Survival After Treatment
The Kaplan–Meier survival curves of mice in all treated groups are shown in Figure 2. The mice in the LI + S1wk group had the highest survival rate (72%), significantly higher than that of other groups (P < .05). The mice treated with LI alone had the lowest survival rate (46%) except for the control group. There is no significant difference between the LI and LI + S0wk groups.

Kaplan–Meier survival curves of BALB/C mice bearing EMT6 tumor
The median survival time of the control group was 32 days. At the conclusion of the survival study (90 days after laser treatment), the median survival times of the LI and LI + S0wk groups were 66 and 74 days, respectively. A total of 26 out of 36 mice in the LI + S1wk group were still alive 90 days after LI. A box plot of the survival time of mice in different groups is shown in Figure 3.

Box plot of the survival time of mice in different groups (LI = laserimmunotherapy)
Tumor Rechallenges
To test the immunity induced by the LI treatment, successfully treated mice were rechallenged with EMT6 tumor cells 90 days after laser treatment. Within a week of the rechallenge, palpable tumors were detected on most of the mice. Although some mice developed tumors of large size, most of the detected tumors started to decrease in size and eventually completely disappeared within 2 weeks after the tumor rechallenge without any intervention. The survival rates after the first rechallenge were 77% in the LI group, 80% in the LI + S0wk group, and 73% in LI + S1wk group (Figure 4). There was no statistical difference between the groups (P > .05). Although the survival rates after the second rechallenge in each experimental group increased by 16% to 22%, as shown in Figure 4, there were again no statistically significant differences between the 3 groups (P > .05).

Survival rates after the first and second rechallenge for different groups (LI = laser immunotherapy)
Discussion
Photothermal interaction using an 805-nm laser and ICG can induce a high temperature increase in the target tissue, which creates a selective tissue destruction zone covering the target tumor mass. Although this thermal reaction usually does not result in complete destruction or total acute eradication of target tumors, tumor cells swell and break into pieces, allowing the release of antigens with the increase of temperature. 13 These antigens include tumor-associated antigens, thermally induced heat shock proteins, and a large number of self-antigens. Antigen presenting cells, particularly dendritic cells, can capture these antigens and migrate to lymph nodes. They present the antigens to T cells to induce an immune response that can be effective against specific tumor cells.
LI has been using the thermal effect in the treatment of metastatic tumors. Previous histochemical results showed that sera from LI-cured tumor-bearing rats contained antibodies that bound strongly to the plasma membrane of both living and preserved tumor cells. 14 Western blot analysis of tumor cell proteins using sera from successfully treated rats as the source of primary antibodies also showed distinct bands, indicating induction of tumor-selective antibodies. 14 Further investigation found that the adoptive immunity could be transferred using the spleen cells from successfully treated rats. 15 Although the detailed immunological mechanism of LI is currently under investigation, it is believed that immune responses from both humoral and cellular arms are contributing factors.
It is hypothesized that the induction of tumor immunity observed in LI is the result of the combined photothermal and immunological interactions. The treated tumors at the treatment sites serve as the sources of tumor-specific antigens to induce tumor-specific immune responses. Therefore, LI on local tumors could provide a tumor vaccine. Eventually, the immune system would produce enough tumor-specific antibodies and initiate a systemic assault against the residual tumor cells and metastases. Furthermore, the general immune response could lead to a tumor-specific immunity against future tumor exposure.
The immune systems of cancer patients are often compromised, and tumor debris may not be sufficient in inducing a potent antitumor response. 16 Additional immunological interventions are required to enhance the immune system to achieve an effective and protective immune response against residual tumor cells.17,18 In LI, GC was administered to augment the immune effect. However, previous experiments show that GC is most effective when introduced in parallel with laser and ICG. 8
Surgery alone can cure most of stage I cancer patients. For example, the 5-year survival rate in stage I breast cancer patients is nearly 100%. Although surgical removal of primary tumors results in rebounding of antibody and cell-mediated responses, the induced immunity is very limited in curing late-stage cancer patients. 19 For stage II and late-stage breast cancer patients, combination therapy is needed to prevent recurrence and metastasis. Therefore, a combination of LI and surgery could be a new approach for an effective cancer therapy.
Although LI can kill most of the tumor cells in the treatment area, some tumor cells can inevitably survive the treatment. Thus, surgical resection of the residual tumor may become necessary to reduce and/or prevent tumor invasion in the primary area and metastasis at distant sites. However, since the residual tumors can assist in immune stimulation and at the same time also be the source for tumor recurrence, the timing of surgery after LI may affect the outcome of the cancer treatment.
In this study, we found that the combination of LI with surgery resulted in a long-term antitumor effect, thus helping the mice resist tumor rechallenges and increase their chances of survival. It seems that the balance between immunological stimulation by treated tumor cells and the removal of the residual tumors may be a significant contributing factor for LI to successfully eradicate the primary tumors and to prevent local tumor invasion and distant metastasis. It is hypothesized that LI treats local tumors and uses the product of laser irradiation, the destroyed and damaged tumor cells, to induce an immune response against future tumor invasion. When the surgery was performed immediately after LI, the presence of the residual tumor cells at the treatment site was shortened, thus reducing their ability to initiate and enhance immune response. On the other hand, if the residual tumor remained in the host for too long, the tumor cells that survived the treatment continued to grow and overwhelm the newly triggered immune response, hence negatively affecting the overall outcome.
The mice in the LI + S0wk and the LI groups achieved similar survival rates, as shown in Figure 2. This was attributed to the fact that in the LI + S0wk group, the acute inflammation induced by LI enhanced the therapeutic effect of surgery, and the immediate surgical removal of the tumors resulted in a reduced immunological function of the residual tumor. In comparison, the much higher survival rate of the LI + S1wk group (Figure 2) was attributed to the fact that time between LI treatment and the surgical resection of treated tumors (1 week) was long enough for the treated tumor cells to enhance the host immune responses, while short enough to prevent significant local invasion and distant metastasis by the surviving tumor cells. Therefore, the time between LI procedure and the surgical resection of residual tumor should be carefully planned for better treatment outcomes.
Furthermore, our results suggest that the procedure for the combination of LI and surgery requires LI to be performed 1 week before the surgery resection. The idea of LI treatment prior to surgery should be easy and comfortable for surgeons and patients to accept.
In conclusion, this study provides more insight into the mechanism of laser immunotherapy. We have shown that LI, when combined with surgery, can induce a long-term antitumor effect better than the standard treatment of LI. Therefore, the concept of LI and surgery as a new combination therapy is suggested. This study also reveals the importance of the balance between immunological stimulation by treated tumor cells and the removal of the residual tumor to prevent further local tumor invasion and distant metastasis. The optimal time interval between LI and surgery is yet to be established. Future studies are needed to determine the appropriate schedule to surgically remove LI-treated residual tumors to allow a maximal survival rate, ultimately leading to an ideal tumor treatment modality.
Footnotes
Acknowledgements
The authors thank Dr Cynthia Murray, Department of Mathematics and Statistics, University of Central Oklahoma, for assistance with the statistical analysis.
Xiaosong Li and Henry Le contributed equally to this study.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article:
This research is supported in part by the US National Institutes of Health (P20 RR016478 from the INBRE Program of the National Center for Research Resources) and by the National Natural Science Foundation of China (No. 81000994).
