Abstract
Introduction
Hepatocellular carcinoma (HCC) is one of the major worldwide health problems. It represents the fifth most common tumor worldwide, with an increasing prevalence and mortality. Any chronic inflammatory liver disease has the potential to induce HCC; approximately 90% to 95% of tumors are the biologic consequences of persistent hepatitis B virus (HBV) and hepatitis C virus (HCV) infections. 1
Much attention has recently been directed to the role of oxidative stress and apoptosis in the pathogenesis of HCC, being widely recognized as the central features of the disease. 2 Persistent oxidative stress in cancer explains part of the characteristic tumor biology of cancer such as activated transcription factors and proto-oncogenes, genomic instability, chemotherapy resistance, invasion, and metastasis. 3 Living organisms have developed complex antioxidant systems to counteract reactive oxygen species (ROS) and to reduce their damage. Thus, antioxidants protect against oxidative stress, prevent damage to cells and thus development of cancer. 4
Nitric oxide is a vital biological messenger molecule involved in many physiological and pathological processes, which is both beneficial and detrimental. Appropriate levels of NO• production are important in protecting organs such as the liver. However, sustained levels of NO• production result in direct tissue damage and increased chronic expression of NO• is associated with various carcinomas and inflammatory conditions.5,6
Apoptosis plays a role in preventing cancer; if a cell is unable to undergo apoptosis, due to mutation or biochemical inhibition, it can continue dividing and develop into a tumor. 7 Caspase-3 is the main key executioner of apoptosis, being responsible either partially or totally for the proteolytic cleavage of many key proteins. 8 Nitric oxide has also been demonstrated to be involved in the regulation of apoptosis. 9 High levels of nitric oxide have been demonstrated to inhibit apoptosis in a number of cell types, including hepatocytes. 10
Recently, chemoprevention by the use of naturally occurring substances has been considered as a priority to reduce the ever-increasing incidence of cancer. Their intervention in multistage carcinogenesis by modulating intracellular pathways may provide a molecular basis for chemoprevention. 11 Several naturally occurring compounds were shown to inhibit different types of cancers, including cancer of the colon, in laboratory animal models. 12
Honey is a product produced by bees (BH) from the nectar and other sugary substances derived from many plants. It has been traditionally used for centuries to promote health and fight disease, but the associated biochemical mechanisms for its possible protective and therapeutic effects are not yet clarified and remain an important challenge in research. It has been reported to contain about 181 active substances. 13
Recently, much attention has been directed to its anti-inflammatory, antioxidant, antiproliferative, apoptotic, and immunomodulatory properties. 14 Some of the observed biological activities of honey may be traced to its chemical constituents,15,16 including caffeic acid, benzoic acid and esters, substituted phenolic acids and esters, flavonoid glycones, and beeswax. 17 Caffeic acid (3,4-dihydroxycinnamic acid) ester derivatives, which are present in honey at levels of 20% to 25%, are thought to exhibit a broad spectrum of activities that possibly include tumor inhibition.17,18
Black seed (Nigella sativa L; NS) is an oriental spice of the family Ranunculaceae that has long been rationally used as a natural medicine. 19 It was also cited for destroying cancer cells,20,21 as well as for raising the number of antibody-producing cells and antiproliferative activity. 22 It has been reported that black seed preparations may have a cancer chemopreventive potential and may reduce the toxicity of standard antineoplastic drugs. 20 The chemotherapeutic and chemoprotective effects of NS extract may be because of quinones that include thymoquinone and dithymoquinone that are present in the oil of this seed. Studies on the biological effects of its active principal thymoquinone, which represents 54% of the seed oil, included inhibition of cancer cell growth and induction of apoptosis.23-25 The beauty of black seed rests in its ability to restore body function and keep homeostasis. 26 But still there have been very few reports on the effects of NS in liver cancer prevention and/or therapy.
Since there are limited studies on the effects of BH and NS on liver cancer and the adverse effects of current cancer therapies are major, we designed this study to expose HepG2 liver cancer cells to a range concentration of pure BH and ethanol extracts of NS to investigate their antineoplastic activities against human liver cancer cell lines, by measuring the survival rate of cancer cells under the influence of these natural remedies. Moreover, we tried to investigate the biochemical and molecular pathways through which they could exert their preventive and therapeutic effects.
Materials and Methods
Human hepatocellular carcinoma cell line (HepG2) was provided from the National Cancer Institute (NCI). It was originally purchased from the American Type Culture Collection (ATCC HB8065, Manassas, VA). HepG2 cells were maintained in RPMI-1640 with 10% fetal calf serum (FCS) in the presence 1% glutamine, 2% penicillin and streptomycin, and 1% amphotericin B. Cells were seeded in 25-cm2 cell culture flasks and allowed to grow to 90% confluence under standard culture conditions (5% CO2, 37°C).
Preparation of Bee Honey Mixtures
Pure unfractionated BH fed on Egyptian clover flowers was purchased from the market and diluted with RPMI-1640 medium to give 5%, 10%, 15%, and 20% solution concentrations.
Artificial honey was prepared to exclude the effect of honey osmolarity and acidity on the HepG2 cells. The average carbohydrate content in 100 g% of honey was as follows: fructose 38.5 g, glucose 31 g, maltose 7.2 g, and sucrose 1.5 g. 27
Preparation of Alcoholic Nigella sativa Extract
NS was obtained from a commercial source in the form of black seed. Extraction followed the conventional protocols: The seeds were washed with clean water, dried, and ground with a clean electric grinder. Ten grams of the seed powder were added to clean vial where 100 mL of 95% ethanol were added. The vial was kept at 4°C overnight. The ethanol extract was separated and placed on a clean wide pan and allowed to dry under a laminar flow hood. From the completely dried products of the extract, 1 g was resuspended in 1 mL dimethyl sulfoxide.
After complete suspension, the extract was filtered using a 0.22 μm sterile syringe filter and stored in sterile vials at 4°C. Stock solution of NS 10 000 μg/mL were prepared with RPMI-1640 medium. Further dilutions were made to prepare concentrations of 1000, 2500, and 5000 μg/mL.
Treatment of HepG2 Cells With Natural Compounds
Immediately prior to the experiment, HepG2 cells (2 × 106) were washed twice with phosphate-buffered saline (PBS) and then the experimental medium (RPMI-1640 containing BH or NS at the indicated concentrations was added). Cells were treated in triplets and incubated at 37°C for 6, 24, 48, and 72 hours. Untreated cells and HepG2 cells treated with artificial honey were used as controls for the experiments.
Experimental Approach
The antiproliferative activity of BH and NS was assessed by trypan blue exclusion followed by counting the surviving cells and calculating the cell survival percentage of treated HepG2 cells.
The antioxidant activities of BH and NS were assessed by measuring the total antioxidant status in HepG2 cell culture lysates by the commercial total antioxidant assay kit 709001 (Cayman Chemical Company, Ann Arbor, MI). Steps of the assay followed the manufacturer’s instructions. The assay relied on the ability of antioxidants in the sample to inhibit the oxidation of ABTS (2,2′-azino-di-[3-ethylbenzthiazoline sulfonate]) to ABTS• + by metmyoglobin. The amount of ABTS• produced was monitored by reading the absorbance at 405 nm.
NO• levels were measured by nitrate/nitrite colorimetric assay kit 23479 (Fluka, Buchs, Switzerland), applying the Griess assay. The assay mechanism is summarized as the azo coupling between diazonium species, which are produced from sulfanilamide with NO2, and naphthylethylenediamine. Absorbance was measured at 540 nm with a microplate reader.
The apoptotic effects of BH and NS were evaluated by measuring changes in the levels of caspase-3 activities in the cell lysates of HepG2 cell line by ApoTaget colorimetric assay reagents (Invitrogen, Carlsbad, CA). The assay was performed according to the manufacturer’s instructions. The method depends on the recognition of an amino acid sequence, DEVD present in the substrate supplied in the kit, which is the upstream amino acid sequence of the caspase-3 cleavage site in poly(ADP ribose) polymerase (PARP). On cleavage of the substrate by caspase-3, free pNA is liberated; light absorbance was quantified using a microplate reader at 405 nm. Comparison of the absorbance of pNA from treated apoptotic samples with an uninduced control allowed determination of the fold increase in caspase-3 activity.
Total protein was measured in cell lysates by the Bradford method (Bio-Rad, Protein assay, dye reagent).
Statistical Analysis
Statistical analyses were performed using the Statistical Package for Social Science (SPSS, version 16.0). Percentage change was calculated for the highest (best) dose of treatment at different intervals of exposure. Percentage coefficient of variation was calculated to show the imprecision of the assays. Student’s t test was used to detect the statistical significance between treated and untreated HepG2 cells. One-way analysis of variance (ANOVA) was used to compare different time intervals of exposure to same dose of treatment. Linear regression analysis was performed to estimate the significance of the linear relationship between the dependent variable (HepG2 cells viability) and the independent variables (NO• level, total antioxidant status [TAS], and caspase-3 activities).
Results
The Effect of BH and NS on HepG2 Cells Survival
This study revealed a significant inhibition of the proliferation of HepG2 cells treated with NS (1000 to 5000 μg/mL) and BH (5% to 20%). The percentages of survival rate were significantly decreased by the highest concentrations of BH and NS (20% and 37.78%, respectively), at 72 hours (P = .000), compared with the untreated control cells and cells treated with artificial honey, as their survival rates increased progressively with time of exposure (Figures 1 and 2).
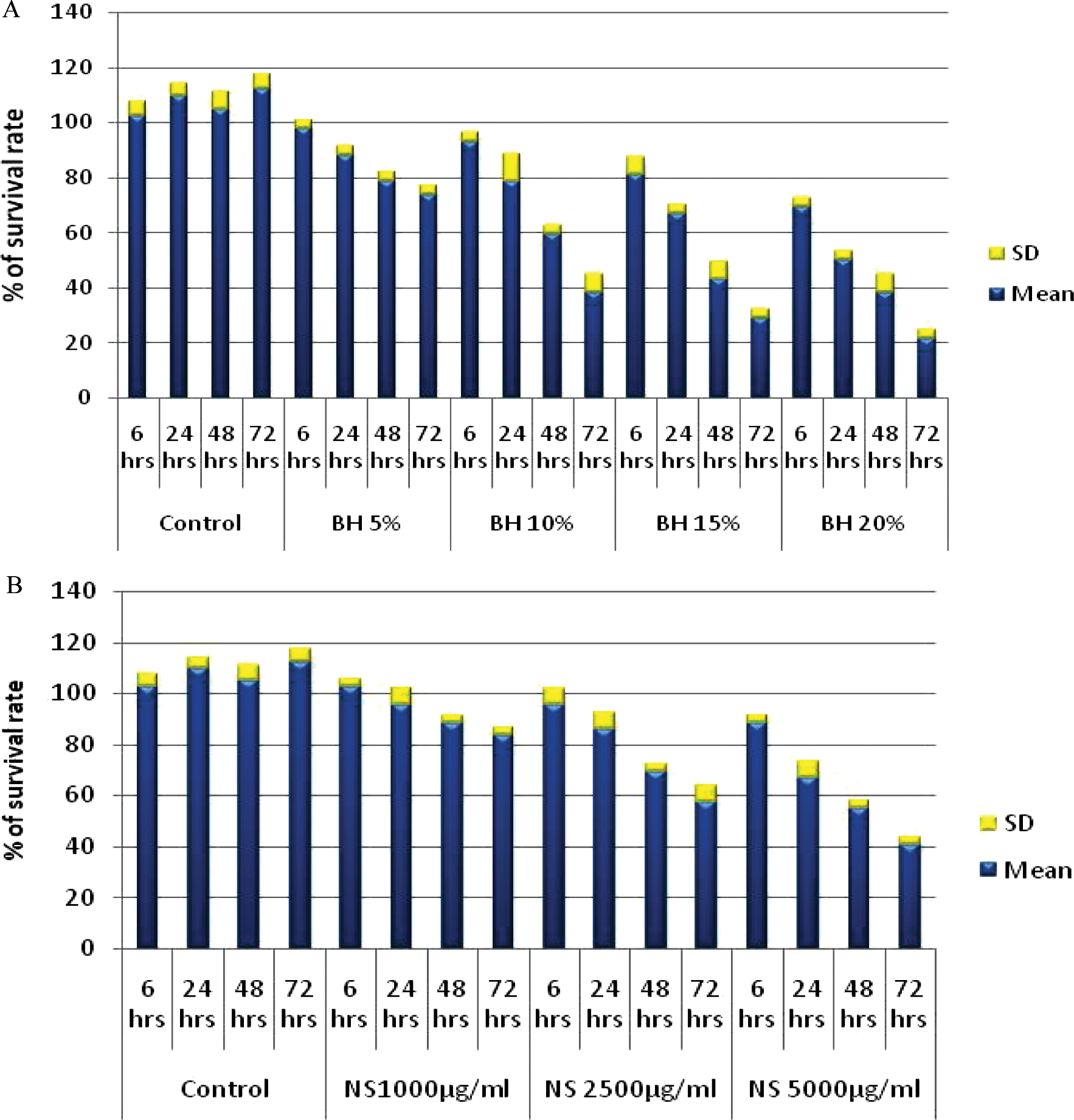
Dose and time course responses of (A) bee honey (BH) and (B) Nigella sativa (NS) on HepG2 cells survival rate
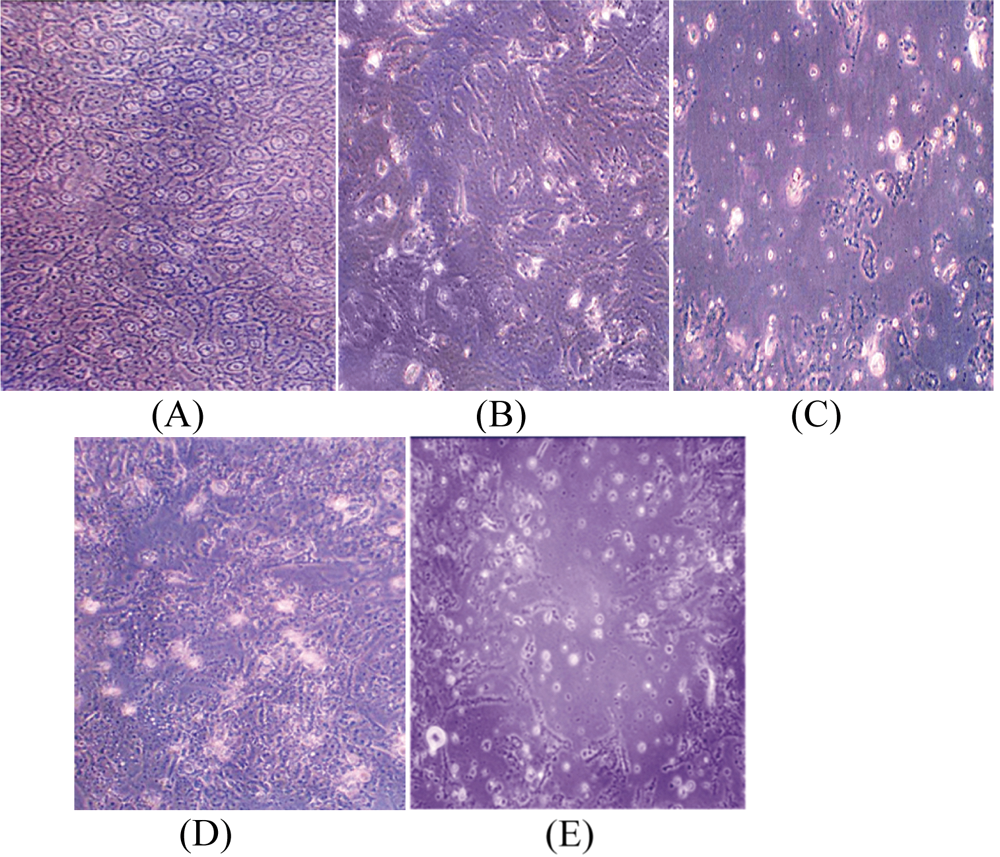
(A) Untreated HepG2 cell line (90% confluence). HepG2 cells treated by (B) bee honey (BH) 20% for 24 hours; (C) BH 20% for 72 hours. HepG2 cells treated by (D) Nigella sativa (NS) 5000 μg/mL for 24 hours; (E) NS 5000 μg/mL for 72 hours
ANOVA was used to compare the effect of BH 20% and NS 5000 μg/mL on HepG2 cell viability at different time intervals. The F values were 182.25 and 121, respectively, and the P values were .00, indicating a significant decrease in HepG2 cell viability on treatment with BH and NS at different time intervals.
The Effect of BH and NS on HepG2 Nitric Oxide Level
On measuring NO• levels in HepG2 cell culture supernatants treated by 20% BH and by 5000 μg/mL NS, the mean levels of NO• were found to be significantly (P = .001) decreased after 72 hours exposure. The percentages of changes were −49.89 and −37.7, respectively (Figure 3).
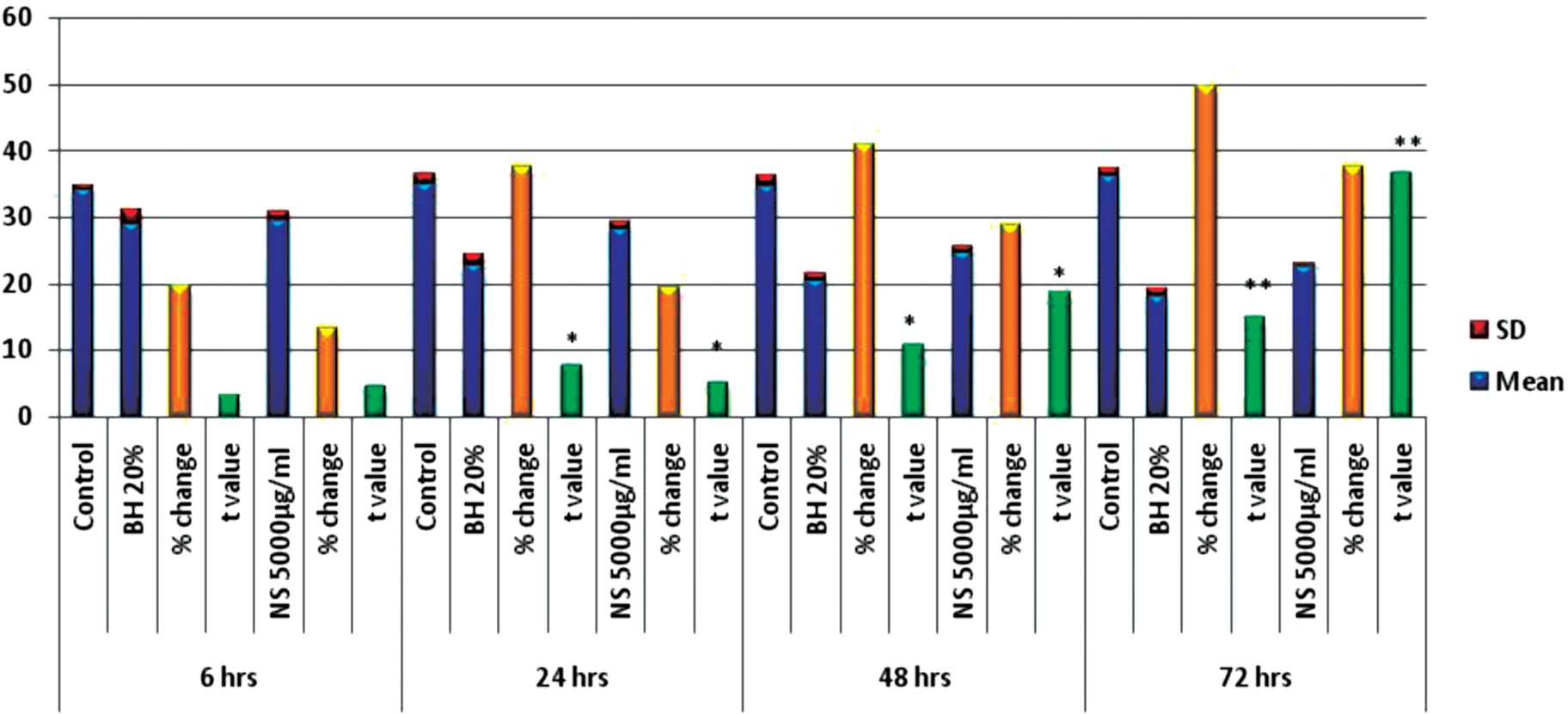
The effects of bee honey (BH) 20% and Nigella sativa (NS) 5000 μg/mL on NO (levels (μM/mL) measured in culture supernatant of HepG2 cell line)
ANOVA test was used to compare the effects of BH and NS at different time intervals; the F values were 35.502 and 40.561, respectively and the P values were 0.001, indicating a significant decrease in NO• levels on treatment with BH and NS at different time intervals.
Linear regression analysis estimated how much a change in NO• could evoke a change in cell viability; it was clear that the decrease in the surviving viable HepG2 cells was proportional to the decrease in the NO• levels (Table 1).
Linear Regression Analysis of Dependent and Independent Factors Used in This Study

The effect of BH and NS on HepG2 Total Antioxidant Status
TAS measured in treated HepG2 cell lysates showed a significant improvement in comparison with the untreated cell lysates. Moreover, this improvement was proportional to the increase in the treatment dose with BH and NS as well as with the increase of the time of exposure to reach the maximum levels at 72 hours, giving +90.1% and +64.8% of change, respectively (Figure 4).
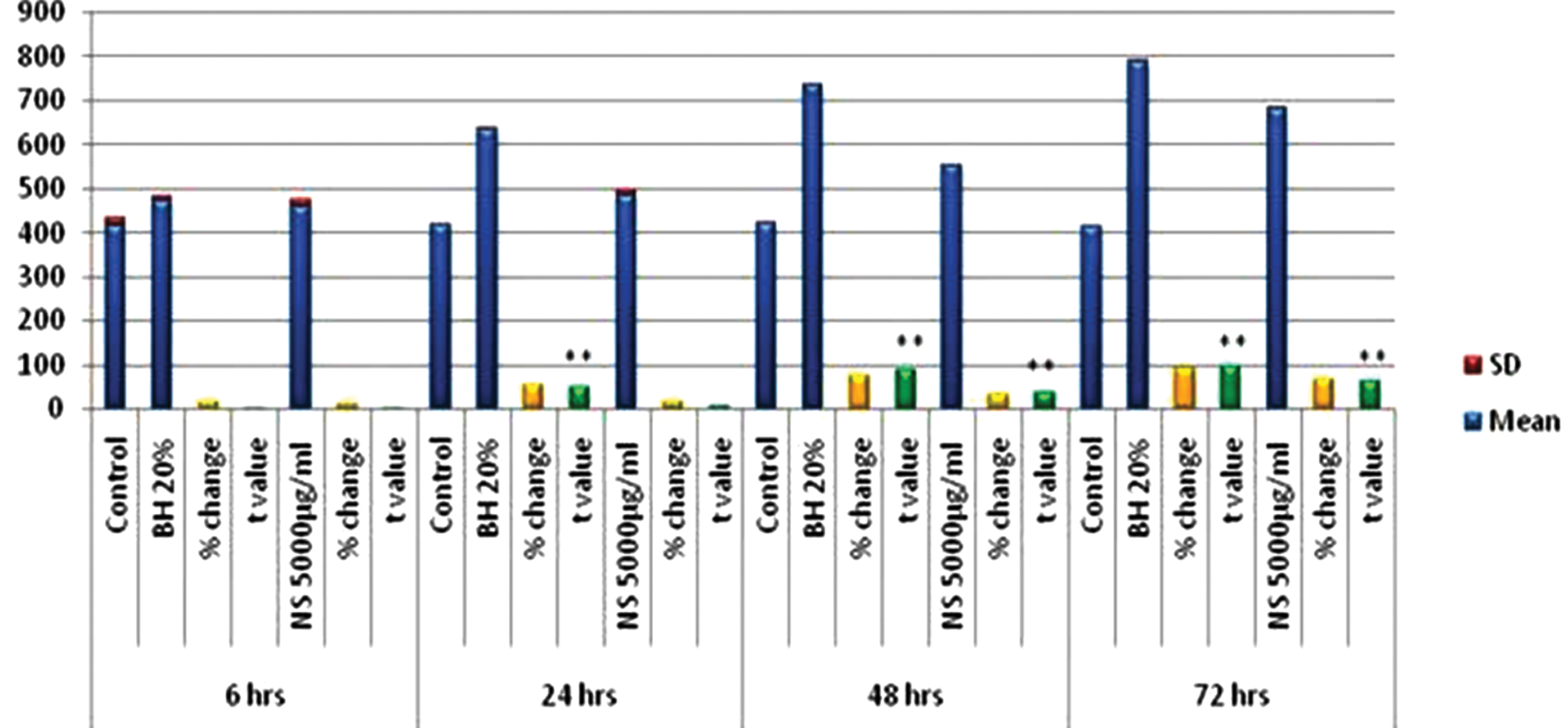
The effects of bee honey (BH) 20% and Nigella sativa (NS) 5000 μg/mL on total antioxidant status (TAS) levels (nmol/mg protein) measured in cell lysate of HepG2 cell line
ANOVA revealed a significant increase in TAS levels (nmol/mg protein) on using BH and NS. The F values were 411.639 and 122.287, respectively; the P values were all .00.
Linear regression analysis between the increase in TAS in treated HepG2 cells and the drop in the number of viable cells showed no significant association between the levels of the 2 variables (Table 1).
The Effect of BH and NS on HepG2 Apoptosis
Exposure of HepG2 cells to an increasing dose of BH and NS led to a significant increase (P = .000) in the activity of caspase-3. The percentage changes in the caspase-3 activity at 72 hours using BH (20%) and NS (5000 μg/ml) were +12.69% and +9.61%, respectively (Figure 5).

The effects of bee honey (BH) 20% and Nigella sativa (NS) 5000 μg/mL on caspase-3 activity (units/mg protein) measured in cell lysate of HepG2 cell line
ANOVA was used to compare the effect of BH 20% and NS 5000 μg/ml on caspase-3 activity (unit/mg protein) at different time intervals. The F values of caspase-3 activity were 173.16 and 52.49, respectively. The P values were .00 indicating a significant increase in caspase-3 activity on treatment with BH and NS at different time intervals.
Moreover, linear regression analysis showed that increase in caspase-3 activity had a highly significant relationship with HepG2 viability, P = .000 (Table 1).
Discussion
Considerable efforts have been made to identify naturally occurring substances that could prevent the development and recurrence of cancer. Cancer chemoprevention, therefore, has emerged as an important subject that, in addition to providing a practical approach to identifying potentially useful novel agents as inhibitors of cancer development, offers opportunities to study the mechanisms of carcinogenesis. 28
In the present study, the antineoplastic effects of pure unfractionated BH and alcoholic extracts of NS were tested against HepG2 cell line, and some of the mechanisms underlying their antineoplastic effects were tested with special focus on their antioxidant and apoptotic activities.
This study revealed a significant inhibition of the proliferation of HepG2 cell lines by BH and NS compared with the untreated control cells; and their effects were evident with increasing the dose and time of exposure.
The hyperosmolarity effect of BH on the viability of HepG2 was corrected by dilution so that the low concentrations were used (5% to 20%). The same principle was applied in choosing RPMI-1640 containing sodium bicarbonate as a solvent because of its ability to buffer much of the acidity of BH. Thus, the acidity and hyperosmolarity of BH alone cannot explain the drastic inhibitory effect on tumors. 28 In this study, artificial honey was used as a control to exclude the hyperosmolarity and acidity effects of BH on HepG2 cells. No effects of artificial honey on the survival rate of HepG2 was detected, as the number of viable HepG2 cells remained the same as that of untreated HepG2 cells under the influence of all concentrations of artificial honey and at all time intervals used.
Pure unfractionated honey was chosen for 2 reasons: (a) the synergism between the different chemical constituents in honey has been reported and (b) there are a number of volatile compounds reported to be abundant in honey products that might be lost during simple fractionation methods. 28
Alcoholic extracted oil portion of the NS seed was chosen to be tested for its antitumor effects, as many previous studies showed that the most potent component of NS for cytotoxicity to cancer cells is that portion of the seed. Thymoquinone, which represents 54% of seed oil, plays a major role in the inhibition of cancer cell growth and induction of apoptosis.19,21,22,29-32 The results agree with previous ones in which BH and the ethyl acetate fraction of NS were found to exhibit a strong growth inhibitory effect on all malignant cells tested.28,33
The toxicity of BH and NS on normal primary cells has been tested previously. It was reported that their active component induced a significant selective growth suppressive and toxic effects only toward transformed malignant cells.20,34,35
Farah 36 showed that NS alone or in combination with oxidative stress was found to be effective (in vitro) in influencing the survival of MCF-7 breast cancer cells, revealing promising opportunities in the field of cancer chemoprevention and/or treatment. 36 Furthermore, in other systems, Rao et al 37 have shown that in vitro treatment of HT-29 human colon adenocarcinoma cells with caffeic acid esters present in propolis significantly inhibited the cell growth and synthesis of RNA, DNA, and protein
Previous studies conducted on NS reported that topical application of black seed extracts inhibited 2 stages: initiation and promotion of skin carcinogenesis in mice by dimethylbenz[a]anthracene (croton oil). 35 Moreover, Salomi et al 20 have shown that a crude methanol extract of black seed exhibited a strong cytotoxic action on Erlich ascites carcinoma, Dalton’s ascites lymphoma, and sarcoma 180 cells, with minimal cytotoxicity to normal lymphocytes. Furthermore, a new triterpene saponin (α-hederin) recently isolated from black seed showed significant dose-dependent tumor inhibition when given intraperitoneally for 7 days at 5 and 10 mg/kg to mice with formed tumors. 38
Moreover, in the study conducted by Iddamaldeniya et al, 31 oral administration of NS extracts was found to protect mice from diethylnitrosamine-induced liver cancer in comparison with the control group. Chemopreventive potential of oil extracts of black cumin (NS) seeds against rat colon, kidney, and liver carcinogenesis induced by chemical carcinogens was also reported. 32
Although the antiproliferative effects of BH and NS were clear, the underlying mechanisms still need to be elucidated. Trying to test the endogenous mechanisms by which BH and NS exerted their antitumor effects, and relying on the assumption that oxidative stress is a condition that could underlie HCC and that the defect in the apoptosis process is implicated in carcinogenesis, led us to measure biomarkers of oxidative stress and apoptosis in both treated and untreated HepG2 cells using the same protocols to test the dose–response and time course responses.
NO• system alterations appear to play a key role in the pathogenesis of chronic liver disease.39,40 NO• is considered as an oxidant molecule; it may mediate hepatic injury from ROS and lipid peroxidation products. Nitric oxide has also a significant role in regulating apoptosis. NO• has been reported to inhibit apoptosis by acting on a variety of targets, including mitochondrial permeability transition pore (PTP), thus decreasing the release of cytochrome C, 41 inhibition of Bcl-2 cleavage, 42 and inactivation of caspases by S-nitrosylation. 43 In this study, NO• levels in HepG2 cell culture supernatants treated by BH and NS were found to be significantly decreased after exposure for 72 hours, compared with untreated control cells. There was an obvious time course effect of BH and NS detected by comparing their effects on NO• levels at different time intervals: The maximum significant decrease in NO• levels on treatment with BH and NS was obtained at 72 hours.
Linear regression analysis revealed a statistically significant association between decrease in NO• levels and the number of viable HepG2 cells, and estimated how much a change in NO• could evoke a change in cell viability. It was clear that the decrease in the surviving viable HepG2 cells was proportional to the decrease in the NO• levels produced.
Oxidative stress is a condition that could underlie HCC. Interestingly, the TAS measured in treated HepG2 cell lysates showed a significant increase in comparison with the untreated cell lysates. Moreover, this improvement in the antioxidant status was proportional to increases in the dose of BH from 5% to 20% and NS from 1000 to 5000 μg/mL, as well as by increasing time of exposure to 72 hours. These findings clearly demonstrate that the high antioxidant capacity of BH and NS could be a possible anticancer mechanism. However, linear regression analysis revealed no statistically significant association between TAS and the number of viable HepG2 cells. Similar results were obtained by Farah 36 and Farah and Begum, 44 who studied the effects of NS and oxidative stress on the survival pattern of MCF-7 breast cancer cells.
Many investigators have suggested that honey is a strong scavenger of active oxygen species. 45 The antioxidant power of honey is attributed mainly to its phenolic compounds, which function as natural antioxidants in diet and its inclusion in the diet may be recommended to complement other polyphenol sources. 46 Phenolic compounds work by reducing and chelating ferric ions that catalyze lipid peroxidation. 47 Hence some studies have reported that honey has the capacity to decrease significantly the concentration of lipid hydroperoxides and malondialdehyde (MDA) produced during the lipid peroxidation process. 48 One of the important antioxidants in honey is caffeic acid phenethyl ester (CAPE), which has been reported to have anti-inflammatory, anticarcinogenic, and immunomodulatory properties. 49
Considering the role of BH and NS in apoptosis, exposure of HepG2 cells to increasing doses of BH and NS led to a significant increase in the activity of caspase-3, the key executioner of apoptosis, indicating the waking up of the apoptotic process, which was relatively low in untreated cancer cells. Moreover, linear regression analysis revealed a statistically significant association between the measured caspase-3 activities and the drop in the number of viable HepG2 cells: Increases in caspase-3 activity were related to significant decreases in HepG2 viability (P = .000).
It was recently reported that ethyl acetate extracts of NS triggered apoptotic cell death in human colorectal cancer cells, which was correlated with G1 phase arrest of the cell cycle and associated with an overexpression of the p53 gene, p21WAF1, and a significant inhibition of anti-apoptotic Bcl-2 protein. 50 Apoptosis of human breast cancer cell line was also triggered by bee honey and detected by TUNEL assay in the work conducted by Swellam et al. 28
Conclusions
BH and NS are effective agents, inhibiting the growth of HepG2 cells through several possible mechanisms. These could include improving the antioxidant status, which might play a role in preventing the initiation, promotion, and progression of cancer, and by inducing apoptotic death of cancer cells. These results are promising in the field of cancer chemoprevention and treatment. However, further research is needed to clarify the exact biochemical and molecular mechanisms of cell death by BH and NS and to confirm their role as candidates for cancer chemoprevention and/or treatment.
Footnotes
Acknowledgements
We would like to thank Dr Fatima Abu-Zahra at the Medical Research Unit, Faculty of Medicine, Ain Shams University, for her generous technical help in the cell culture work. We also thank Prof Dr A. Zekry, National Cancer Institute (NCI), who kindly provided this study with HepG2 cell line.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research and/or authorship of this article.
