Abstract
This study compared the outcome of an innovative in-shoe pressure and temperature measuring device as an adjunct to standard clinical care for diabetic foot versus standard clinical care alone. It included 88 participants with Type 2 diabetes mellitus with a history of one or more plantar foot ulceration who were already using prescription orthoses. These were randomly divided into the control group (n = 44, standard care only) and the experimental group (n = 44, standard care plus the innovative device). Both groups were monitored for re-ulceration for one year. Overall, the control group exhibited a higher number of re-ulcerations (n = 14) with 2 amputations in comparison with the experimental group (only 2 ulcerations and no amputations) at the end of the study. In conclusion, this innovative in-shoe pressure and temperature measuring device appears to reduce re-ulcerations by offering objective data for clinical decision making in the management of the diabetic high-risk foot.
Keywords
Lower-extremity complications of diabetes constitute a substantial burden for people with diabetes as once healed, foot ulcerations frequently recur. Notwithstanding the advances in medicine, amputation rates are still very high and have not reduced significantly. 1 This may suggest that current management of the diabetic foot may not be as effective as desired, clearly demonstrating the need for the implementation of new and effective strategies aimed primarily at prevention of ulceration. The time to referral will determine the number of ulcerations and re-ulcerations or alternatively ulcer free days—in other words “Time is Tissue! We need to act now without further delay!”. 7
The overall prevalence of diabetic foot ulcerations and their recurrence differs from one country to another as apart from variation in population characteristics and general definitions, there is a variation in diagnostic methods and patient management. 2 In most counties diabetic foot care management focuses on the monitoring of the vascular and/or dermatological complications. Moreover, the effectiveness of devices prescribed to off-load areas at-risk of ulceration or re-ulceration, is often determined through visual observation and clinical experience of the clinician . 3
To date, in-shoe technologies can only be used to measure either pressures or temperatures separately and are known to be very costly and time-consuming to use.4,5 Furthermore, the current subjective method of asserting the effectiveness of an off-loading device may not be sufficient. In view of this, an innovative, low-cost, single-sensor, in-shoe pressure and temperature measuring device was developed and validated in a healthy population. 6
In this context, the aim of the study was to evaluate if this innovative device could be used as an adjunct objective measurement of temperature and pressure to standard foot care in the prevention and management of diabetic foot ulceration/re-ulceration.
Materials and methods
This study, conducted between June 2021 and August 2022, obtained institutional ethical approval and followed all principles of the Declaration of Helsinki (Registration Number: V_15062020 5784). Participants gave their informed consent were recruited via an intermediary from the Diabetic Foot Clinic at the Out Patients Department of the Local Hospital. The data collection was then conducted at the Biomechanics and Gait Lab at the Faculty of Health Sciences, University of Malta.
The study population consisted of 88 participants with Type 2 diabetes mellitus with a history of one or more plantar foot ulceration who were already using prescription orthoses intended to offload previously ulcerated sites. Participants living with musculoskeletal pathologies such as Rheumatoid arthritis, osteoarthritis, history of major amputations and neuromuscular disease were excluded from the study as these conditions are known to influence gait.
Participants were divided into Group 1 (n = 44, standard care only) and Group 2 (n = 44, additional use of the innovative in-shoe pressure and temperature measuring device). Participants were matched according to age, gender, duration of diabetes, presence of neuropathy, ischaemia or neuro-ischaemia. 7 Precision matching was used to match participants based on gender, presence of neuropathy, presence of ischaemia and presence of neuro-ischaemia. Neurological and vascular diagnosis were pre-determine by the participants podiatrist. Results were obtained from the participant's medical record. Frequency distribution matching was used to match participants according to age range. Once matched pairs were identified, one participant was randomly assigned to the control group (Group 1) while the matched participant was assigned to the experimental group (Group 2).
Study Tool
The sensor components of the device consist of one flexible, piezoresistive Force Sensing Resistor (FSR) pressure sensor [A401 FlexiForce (Tekscan, USA)] and one 10-Kohm thin-film Negative Temperature Correlation temperature sensor (DS18B20) interfaced to electronic components that include an operational amplifier and a microcontroller. The FSR used for this device features linearity of ±3%, reproducibility of ±2.5% of the full-scale drift of 5% per logarithmic time scale, and a response time of less than five micro-sec. The temperature sensor features a tolerance of 1%, an operation range between −50 and 90°C, a Beta value of 3435 K with a small form factor having a height of 0.5 mm which makes it ideal for such embedded applications. Both pressure and temperature sensors extend from the main components of the device so that only the sensors are placed under the desired anatomical landmark on the plantar aspect of the foot (Figure 1). The device is connected to a mobile application for capturing of data, which is then sent to a server for processing and display through a secured website. The innovative in-shoe pressure and temperature measuring device is designed to be attached to the uppers of footwear (Figure 2).

Photograph of the impulse device.

Photograph of the device as should be worn on exterior of footwear.
Methods
Both groups received standard diabetic foot care: offloading insole supplied to each participant by the Orthotics and Prosthetics Department of a General Hospital, together with regular podiatric monitoring. Additionally, if an ulcer developed, standard care was applied as per hospital standard operating procedures. Effectiveness of offloading devices relied solely on visual observation and clinical experience throughout this initial stage. Both the experimental group and control group presented with no ulcerations at the recruitment stage. All participants were followed every 4 months.
In the experimental group, the site of their previously ulcerated area was assessed for peak pressure and skin temperature using the innovative in-shoe pressure and temperature measuring device. At each visit, participants in this group were asked to walk on a treadmill for 10 min to represent mild physical exertion during which in-shoe peak plantar pressures and in-shoe peak temperatures were measured, first without the orthoses then with the orthoses. This was done to evaluate whether there was a significant (>30%) plantar pressure reduction with the hospital-prescribed orthoses. 8 All hospital-prescribed insoles were made custom to fit the needs of the participants. Off-loading devices not providing the required amount of pressure reduction were modified at each visit so that the desired pressures were maintained throughout the intervention period of 12 months. The experimental group was also reviewed every 4 months and was monitored for any signs of re-ulceration, similarly to the control group. Temperature and pressure data readings obtained from the experimental group were also used to investigate the correlation between the two parameters.
Statistical Analysis
Statistical analysis was conducted through SPSS (Version 28; Statistical package for social sciences, IBM Corp., Armonk, NY, USA). Data were investigated using Cochran's Q-test to confirm or otherwise, statistically significant differences in yield of ulceration between the 2 groups. Spearman's Rank Order Correlation was used to determine statistically significant correlation between in-shoe pressure and temperature parameters. Significance was defined at the 5% level (P < .05).
Results
Among the 88 participants, 70 were males (80%) and 18 were females (20%). Mean HbA1c was 7.39 mmol/mol in the experimental group and 9.43 mmol/mol in the control group. Type 2 diabetes duration was 19 years for both groups. The commonest site of previous ulceration (34%) was the hallux for both groups.
Intervention With the Innovative in-Shoe Pressure and Temperature Measuring Device
Overall, the control group exhibited a higher number of re-ulcerations. Moreover, at the end of the trial, there were 4 amputations in the control group and none in the experimental group (Table 1).
Table Providing a Visual Representation of Cases of Re-Ulceration Presented in Both Groups During the Clinical Study.
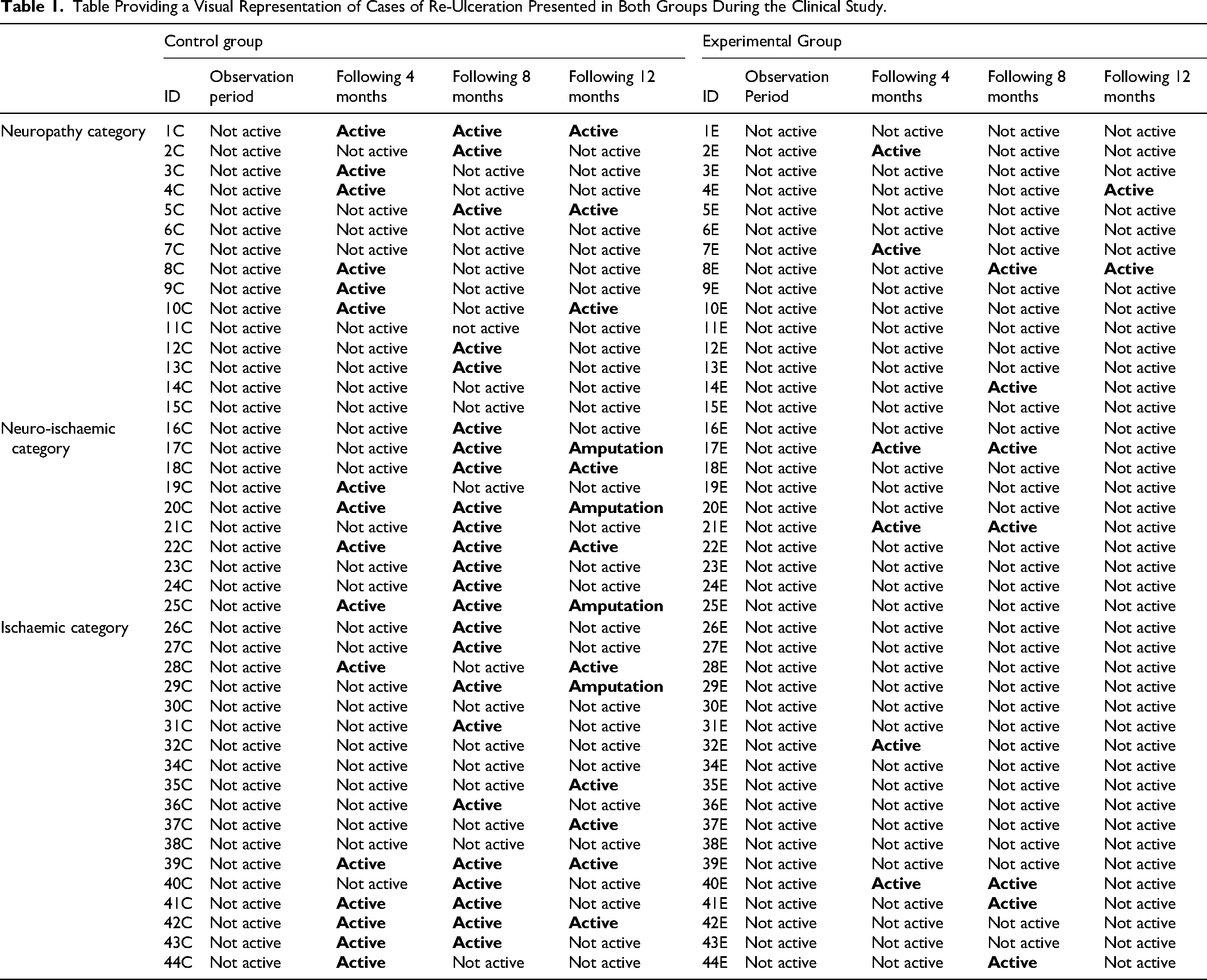
The Cochran's Q-test confirmed a significant difference (P ≤ .014) in the proportion of healed re-ulcerated lesions within the experimental group across 3 time points: (1) at 4 months, (2) at 8 months and (3) at 12 months (Table 2).
Re-ulcerations in the Two Groups During the Study.
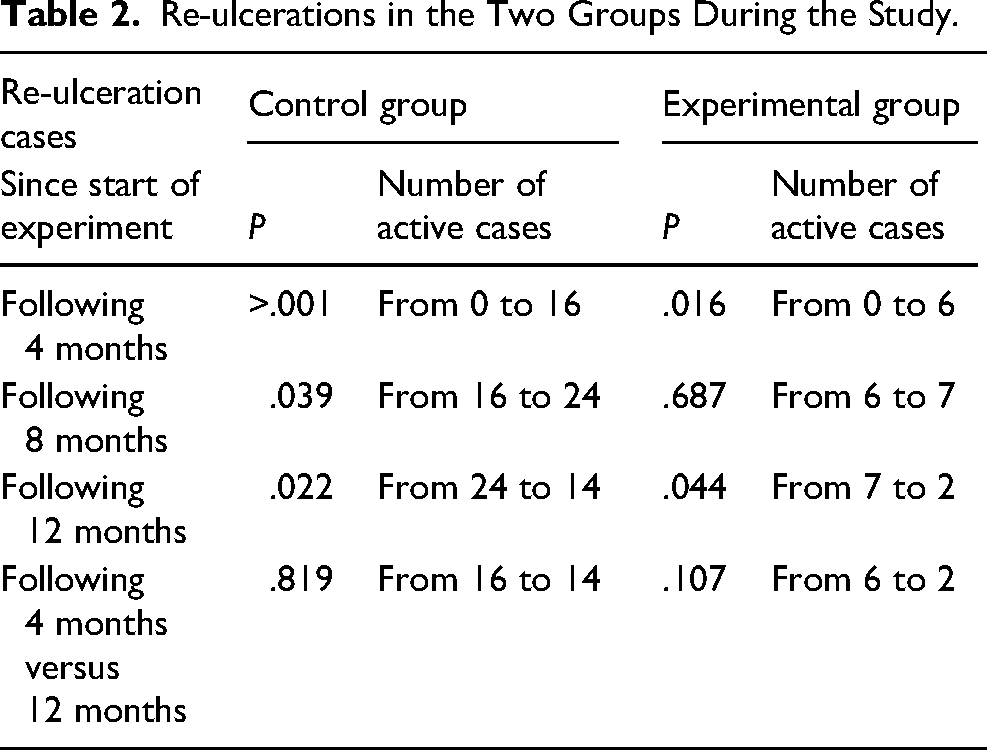
Following 8 months of observation and intervention (T2), the number of re-ulcerations in the experimental group increased by 1 case only, whereas in the control group re-ulcerations increased to 24 cases in total (P = .022). At the end of the study (T3), the number re-ulcerations in the experimental group significantly dropped to only 2 (P = .044) (Table 2). In the control group, the number of re-ulcerations remained high (n = 14) (P = .819) (Table 2) with 4 amputations, as compared with the experimental group (Table 1).
Correlation Analysis: Temperature Versus Pressure
There was a positive correlation between peak plantar pressure and peak plantar temperature when observing participants walking with their orthoses at 4 months (n = 40, r = .155, P = .339), at 8 months (n = 38, r = .191, P = .252) and at 12 months (n = 38, r = .054, P = .746). When observing the correlation analysis for peak plantar pressure and peak plantar temperature for participants walking without their orthoses, a medium to strong correlation was observed following at 4 months (n = 41, r = –.284, P = .072) and at 12 months (n = 34, r = .277, P = .113).
Discussion
This trial has shown a beneficial effect of the innovative in-shoe pressure and temperature measuring device as adjunct to standard clinical care in patients with previous diabetic foot ulcerations. This is attributable to the objective data obtained with the device and used for monitoring. Almost half of the participants in the experimental group required a modification to their offloading device since a minimum of 30% reduction of the original peak plantar pressure was not being achieved with the current offloading design. 9 Various studies have shown that inappropriate design of an off-loading device may have deleterious consequences on the morphology and function of a healthy foot, let alone on the diabetic high-risk foot.10,11
Furthermore, a positive correlation between peak plantar pressure and peak plantar temperature was observed in two scenarios, walking without orthoses and walking with orthoses. In the first scenario (walking without orthoses), a medium to large correlation between pressure and temperature was observed. In the second scenario (with orthoses), a small correlation was observed. This study was first to explore the correlation between pressure and temperature during different scenarios such as walking with/without an offloading device. Findings concur with other studies which have explored the correlation between pressure and temperature as parameters for tissue breakdown in the high-risk diabetic foot.12‐14
The major limitation of the present is the relatively small number of participants, partly due to the COVID 19 pandemic. This may have reduced the significance of results obtained from the Cochran's Q-test analysis and Spearman's rank order correlation analysis as both tests are influence by the sample size. However, despite this limitation, results still showed a significant improvement in the experimental group. Another limitation may be the failure to match subjects by Glycated hemoglobin (HbA1c), which may affect wound healing.
In conclusion, these results indicate a role for the innovative in-shoe pressure and temperature measuring device in reducing re-ulcerations by offering objective data for clinical decision making in the management of the diabetic high-risk foot. Further experience is now required before such technology is more widely used when prescribing off-loading devices.
Footnotes
Declaration of Generative AI in Scientific Writing
AI and AI-assisted technologies in the writing process of this manuscript was not used.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
