Abstract
Lower extremity traumatic wounds pose unique challenges in pediatric patients, including vessel caliber, compliance with postoperative instructions, parental concerns about multiple operations, and long-term function. An autologous heterogeneous skin construct (AHSC) has demonstrated the ability to cover avascular structures and regenerate full-thickness functional skin. The objective of this study is to report our experience using AHSC in a cohort of pediatric trauma patients. This study is a noncontrolled, retrospective cohort analysis of all pediatric patients (<19 years of age) treated with AHSC for lower extremity traumatic wounds with at least one exposed deep structure (tendon, bone, and/or joint) at a single institution between May 1, 2018, and April 1, 2019. Seven patients with 10 traumatic wounds met inclusion criteria. The median follow-up time was 11.8 months. Five patients were male (71%); the median age was 7 years (range = 2-15 years). Average wound size was 105 cm2. All wounds achieved coverage of exposed structures and epithelial closure in a median of 13 and 69 days, respectively. There were no donor site complications and no reoperations required. All patients returned to normal activity, ambulate without limp, can wear shoes normally, and have normal tendon gliding. AHSC covered exposed structures and achieved closure within a single application in complex traumatic lower extremity wounds in a pediatric cohort.
Keywords
Introduction
Pediatric traumatic wounds present unique challenges including small caliber vessels for microsurgery, parental concerns over multiple procedures, and social challenges related to prolonged hospital stays. 1 The first goal in reconstructing complex trauma wounds is to achieve adequate coverage of exposed structures such as tendon and bone. This coverage is often accomplished with dermal substitutes or free tissue transfer. Dermal substitutes typically require multiple procedures and the donor site morbidity associated with split-thickness skin grafts.2-4 Free flaps are technically challenging, require prolonged operations and hospital stays, impart even greater donor site morbidity, and can impose challenges with pediatric patients related to postoperative protocols. Large series of free flap reconstruction have reported failure rates (partial and complete) approaching 19% and 1.7 reoperations per flap.1,5
An autologous heterogeneous skin construct (AHSC) has become available in recent years that has demonstrated the regeneration of full-thickness skin with follicles and glands.6-13 AHSC utilizes a small, elliptical full-thickness skin harvest from the patient that can be closed primarily. The autologous donor skin is shipped to a biomedical manufacturing facility where it is manufactured into AHSC and can be returned for application to the wound bed as early as 48 hours after harvest and is viable for use for up to 14 days after harvest. Notably, AHSC has been shown to be capable of covering exposed structures such as tendon and bone.6,8,9,12
Given the lack of literature surrounding the use of AHSC in pediatric trauma, this study serves as a first series reported in this defined patient population to facilitate larger, systematic studies. Moreover, most reports concerning the treatment of pediatric lower extremity trauma are descriptive and focused on free flap reconstruction, with few reports quantifying results of dermal matrix or local flap reconstruction.1-3 Therefore, we sought to report our initial outcomes using AHSC to treat this population given the overall paucity of evidence in this defined population and to demonstrate its potential benefits in clinical practices that care for similar injuries. It was hypothesized that AHSC would be capable of closing complex traumatic wounds with adequate function, and therefore result in low resource utilization. This hypothesis was based on the low metabolic demand of the viable multicellular aggregates within AHSC that are capable of surviving within the wound bed over exposed structures and the demonstrated regeneration of full-thickness functional skin in prior reports.6-8
Methods
Study Design and Sample
This study is a noncontrolled, retrospective cohort of all pediatric patients (0 to 18 years of age) who received AHSC for treatment of traumatic lower extremity wounds with at least one exposed deep structure presenting to Children’s Hospital of New Orleans between May 1, 2018, and April 1, 2019. Exposed deep structures included tendon, bone, and/or joint. There were no further inclusion or exclusion criteria applied to the cohort studied. Patient authorization and consent was obtained for use of all photographs.
Operative Technique and AHSC Workflow
Prior to AHSC application, serial debridement and washout were performed on all wounds until all devitalized tissue and contamination was removed, analogous to wound bed preparation for any traditional graft or flap. In between serial washouts, negative pressure wound therapy (NPWT) was applied.
Skin harvest and AHSC application were performed using aseptic technique. A small, elliptical full-thickness skin harvest was taken from the patient’s groin, including a layer of hypodermal fat, using a sharp scalpel to minimize cell and tissue injury. All harvests were performed in the operating room during the patient’s final washout and debridement. The donor site was closed primarily.
The excised skin was shipped overnight at 4 °C to a Food and Drug Administration–registered biomedical manufacturing facility (PolarityTE, Inc) for processing. The full-thickness donor skin was utilized in its entirety to manufacture the AHSC, which is composed of multicellular segments. The AHSC multicellular segments retain progenitor cells and are optimized for graft take within the wound bed, where they expand and regenerate full-thickness skin. The manufactured AHSC arrives in a syringe and has the consistency of a paste. AHSC increases the surface area to volume ratio and facilitates nutrient and metabolic waste exchange through diffusion. 6 The tissue is not cultured or expanded ex vivo, and no enzymes, growth factors, or serum derivatives are utilized during manufacturing. AHSC was returned within 2 to 4 days following harvest, depending on the planned operative schedule, and was applied the same day as delivery in each case. AHSC is shipped at 4 °C and must be kept 1 to 10 °C prior to use and does not require rewarming.
Autologous heterogeneous skin construct was applied in its entirety by dispensing it from the syringe in small aliquots throughout the wound bed and spreading it as evenly as possible using the back of a forceps. In this series, each wound received a single application of AHSC. AHSC was fixated with a nonadherent, nonabsorbent fenestrated silicone dressing that was secured directly to the wound margin with staples or sutures, which was then bolstered with a compression dressing or NPWT. Preference is to use NPWT as a bolster when feasible because it fixates AHSC to the wound bed with less risk of micro-motion and shear, and removes excess fluid through the fenestrations in the silicone. However, NPWT is not always practical, may not hold reliable suction/seal in certain wounds due to contour and location, and compression dressing may be more reliable in such circumstances (ie, NPWT frequently losing suction is at risk for motion and shear). This initial dressing was left in place for 1 week.
Following the initial dressing, the surrounding skin was cleaned with chlorhexidine gluconate soap, followed by application of fenestrated silicone, sterile bulky gauze bandage (Kerlix, Covidien/Kendall), and an elastic bandage wrap (ACE Elastic Bandage, 3M). This dressing was left in place for 1 additional week. Following the initial 2-week period of weekly silicone dressings, the primary dressing layer was switched to daily bacitracin and petrolatum gauze (Xeroform, Covidien/Kendall) with Kerlix and ACE wrap overlay. If any signs of hypergranulation developed, the primary dressing layer was changed to an antimicrobial silver dressing (ACTICOAT, Smith and Nephew), which was changed every 3 days until hypergranulation tissue resolved, at which point dressings were switched to daily bacitracin and petrolatum gauze.
Postoperative precautions and immobilization depended on the wound location and exposed structures. When not precluded by other injuries, patients could bear full weight immediately postoperatively. Pressure-offloading walking boots were used as indicated for immobilization of foot, ankle, and calf sites, and were typically continued until exposed structures were completely covered.
Demographics and Wound Characteristics
Demographics collected included age, gender, mechanism of injury, associated injuries, and comorbidities. Wound characteristics and AHSC treatment variables collected included total number of wounds per patient, wound size, donor skin harvest size, type of exposed structure within the wound, time from presentation to AHSC treatment, and number of washouts prior to AHSC treatment. For the purposes of this analysis, when a patient had more than one wound, the total donor harvest size was divided proportionally between each wound. Wound size was measured by ruler and calculated based on dimensions.
Outcomes
Outcomes collected included complete coverage of exposed deep structures with vascular tissue (percent of wounds and time), complete epithelial closure (percent of wounds and time), function (ambulation, ability to wear shoe, and tendon gliding), length of stay (overall and intensive care unit), reoperation rate, and complications (overall, wound-specific, and donor site). Coverage of exposed structures was defined as complete coverage with vascularized tissue capable of accepting a graft, and without any portion of the structure remaining visible. Complete closure was defined as complete epithelial closure without drainage. Functional outcomes were defined as follows: (1) ambulation: without assistance or a limp, with a limp, with assistance of crutches/device, or nonambulatory; (2) ability to wear shoe: wearing shoe of same size of uninjured foot or same size previous to injury (normal), wearing shoe of larger size than uninjured foot or previous to injury, wearing orthotic shoe, or wearing walking boot; (3) tendon gliding: full range of motion (ROM) without observed tethering/restriction, partial ROM without tethering, partial ROM with observed tethering/restriction, no ROM; and (4) contracture: no contracture limiting ROM, contracture resulting in partial ROM restriction, and contracture resulting in no ROM. Complications were further stratified as minor and major if they were nonoperative or operative, respectively.
Results
Demographics and Wound Characteristics
During the study period, 7 pediatric patients with 10 traumatic lower extremity wounds that contained exposed deep structures were treated with AHSC at the senior author’s (GSM) institution and met inclusion criteria. Descriptive patient demographics and wound characteristics are listed in Table 1. The median follow-up time was 11.8 months (range = 4.6-14.5 months). The median age was 7 years (range = 2-15 years).
Patient Demographics and Wound Characteristics.
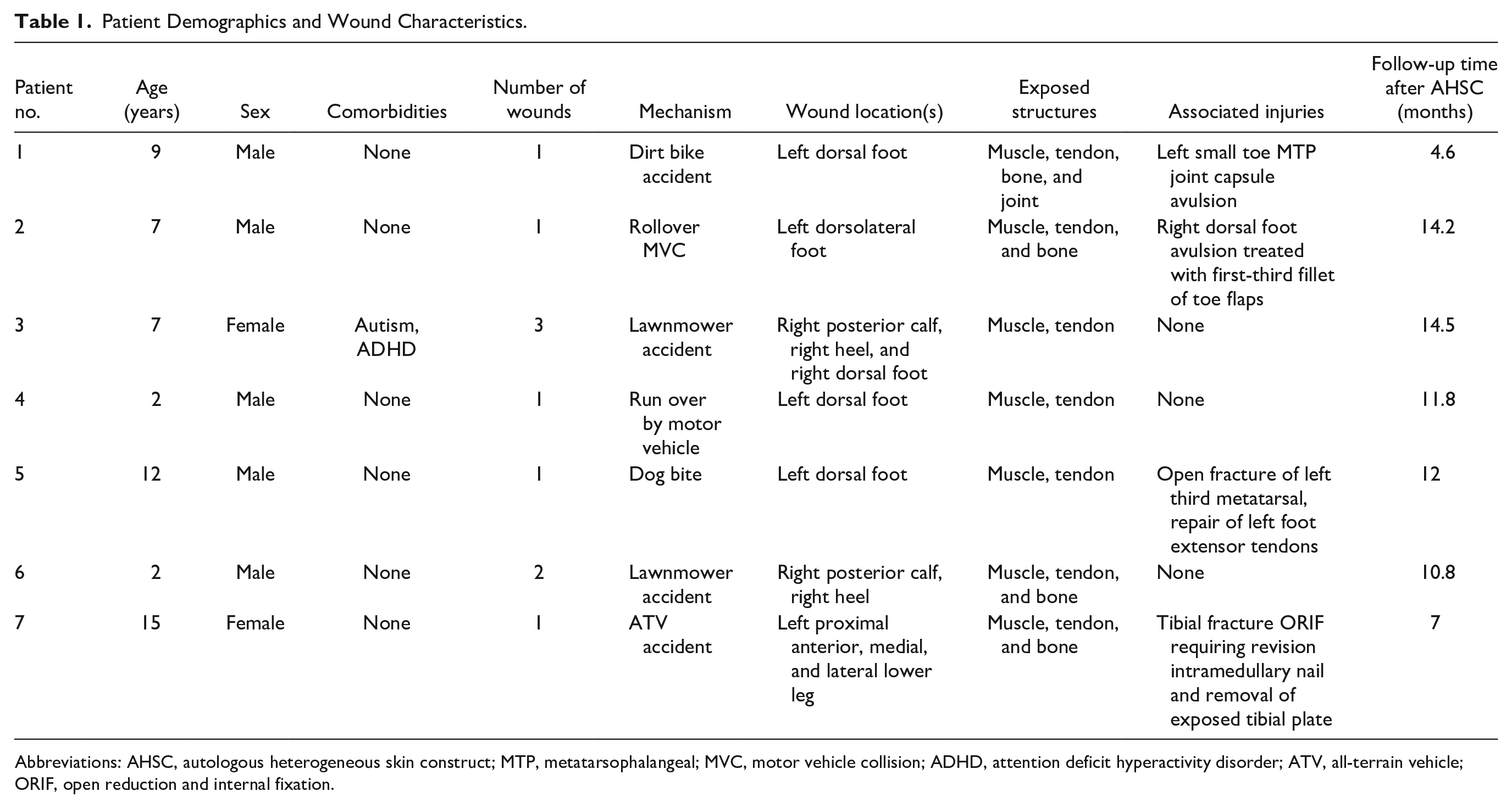
Abbreviations: AHSC, autologous heterogeneous skin construct; MTP, metatarsophalangeal; MVC, motor vehicle collision; ADHD, attention deficit hyperactivity disorder; ATV, all-terrain vehicle; ORIF, open reduction and internal fixation.
Wound and harvest sizes with treatment timelines are listed in Table 2. Each patient received 3 to 4 washouts prior to AHSC treatment, was admitted to the hospital for an average of 9 days (range = 5-11 days) prior to AHSC application, and the median number of days between harvest and AHSC application was 2 days (range = 2-3 days). Each patient received a single application of AHSC.
AHSC Treatment Details, Wound Healing Times, and Patient Outcomes.
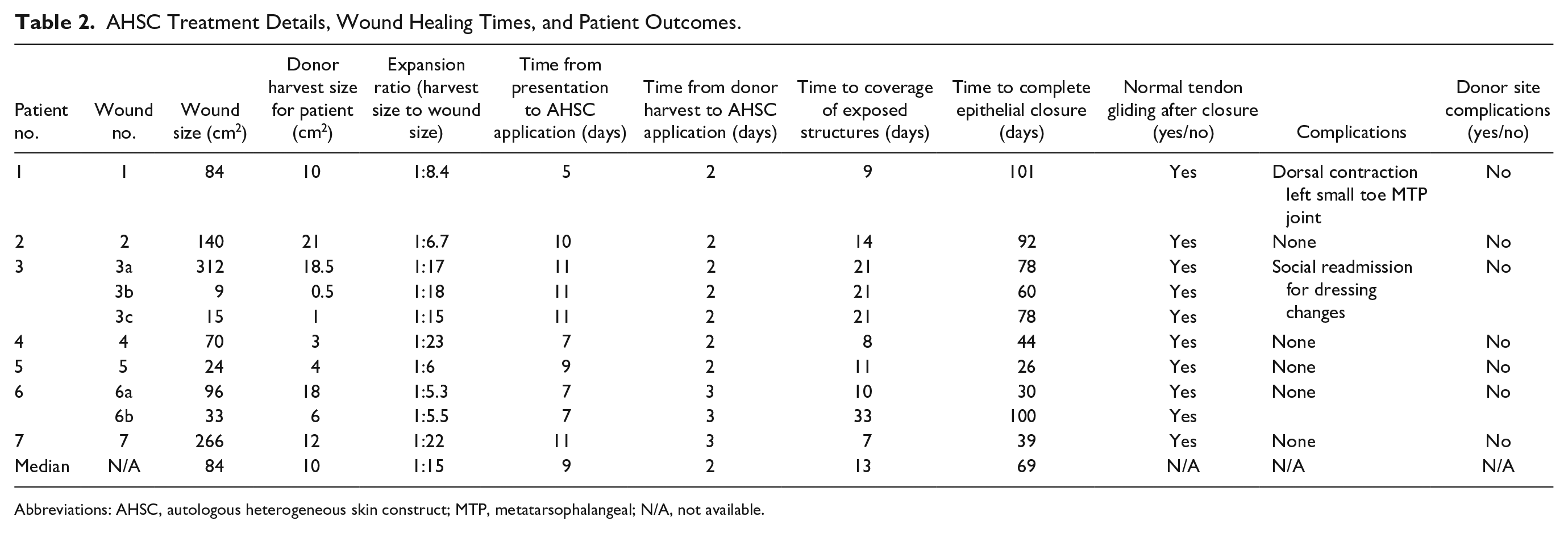
Abbreviations: AHSC, autologous heterogeneous skin construct; MTP, metatarsophalangeal; N/A, not available.
Outcomes
Detailed outcomes for each wound can be found in Table 2, and aggregated outcomes for the entire cohort can be found in Table 3. Following a single application of AHSC, 100% of wounds achieved complete coverage of exposed structures including tendons, bones, and joints with vascularized tissue in a median of 13 days (range = 7-33 days), 100% of wounds achieved epithelial closure in a median of 69 days (range = 26-101 days), and all wounds achieved normal unrestricted tendon gliding. Figures 1 to 4 show serial photographs of example wounds over time. All patients were able to ambulate without assistance or limp and wear normal shoes.
Cohort Aggregated Outcomes and Complications.
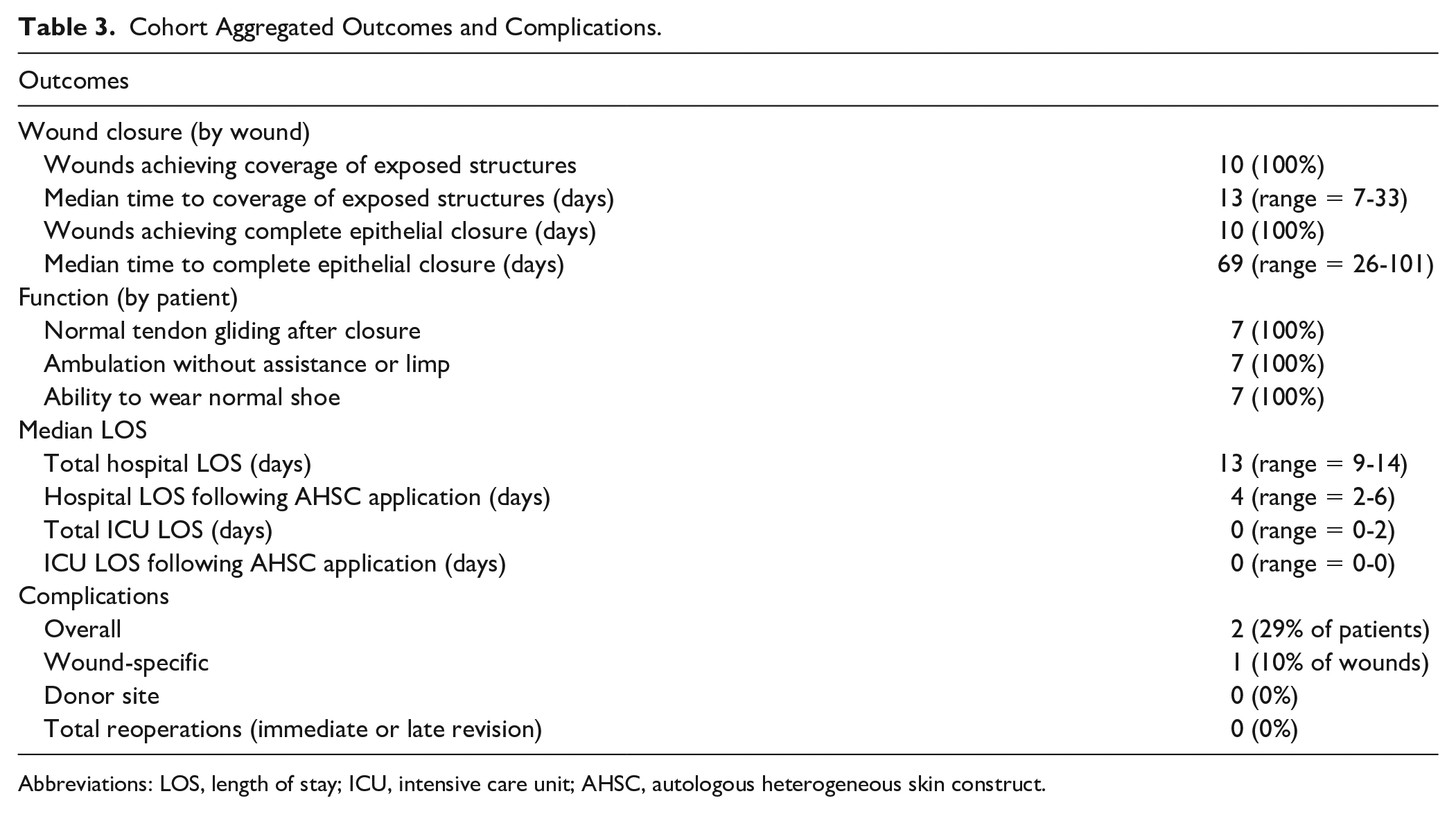
Abbreviations: LOS, length of stay; ICU, intensive care unit; AHSC, autologous heterogeneous skin construct.
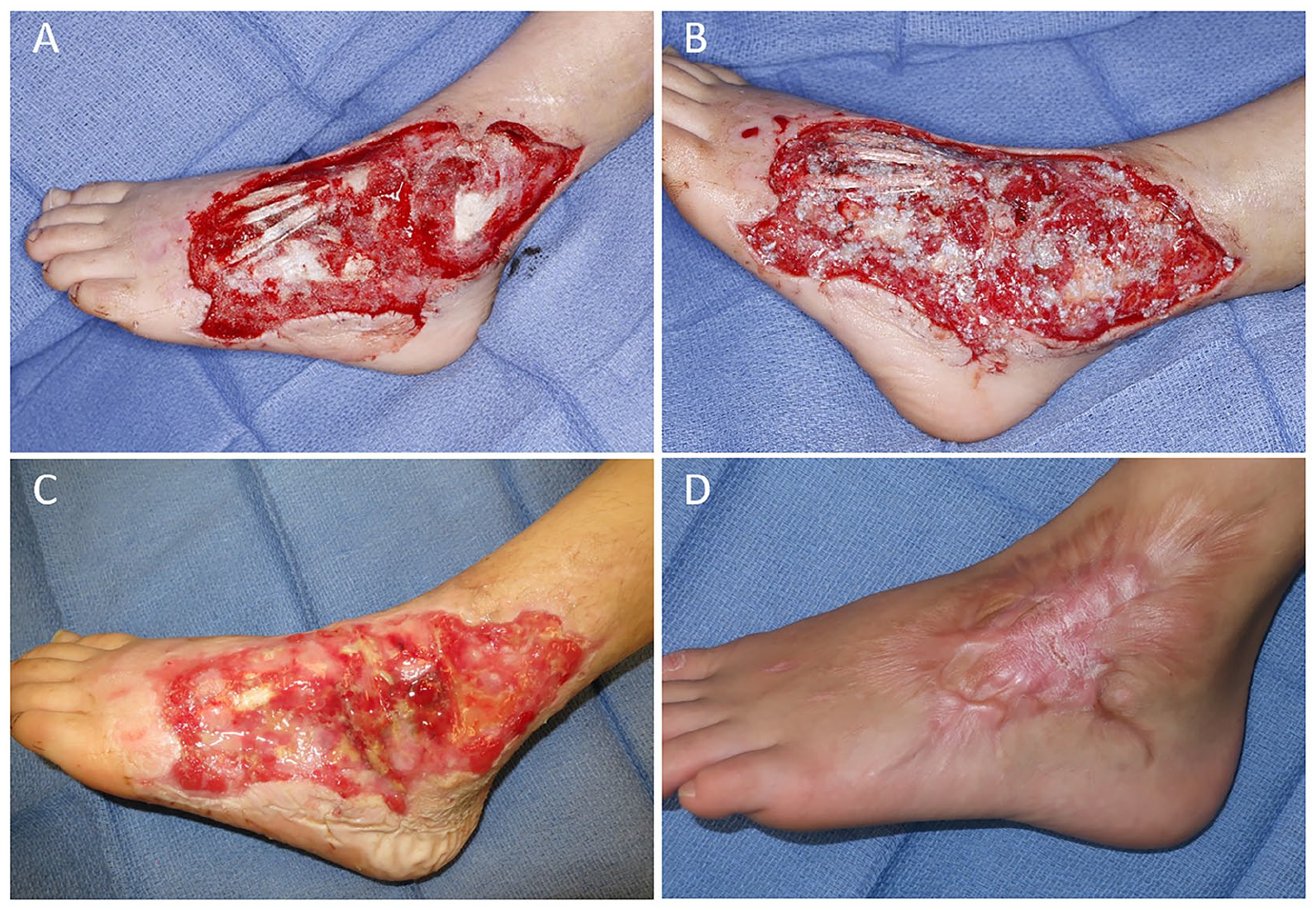
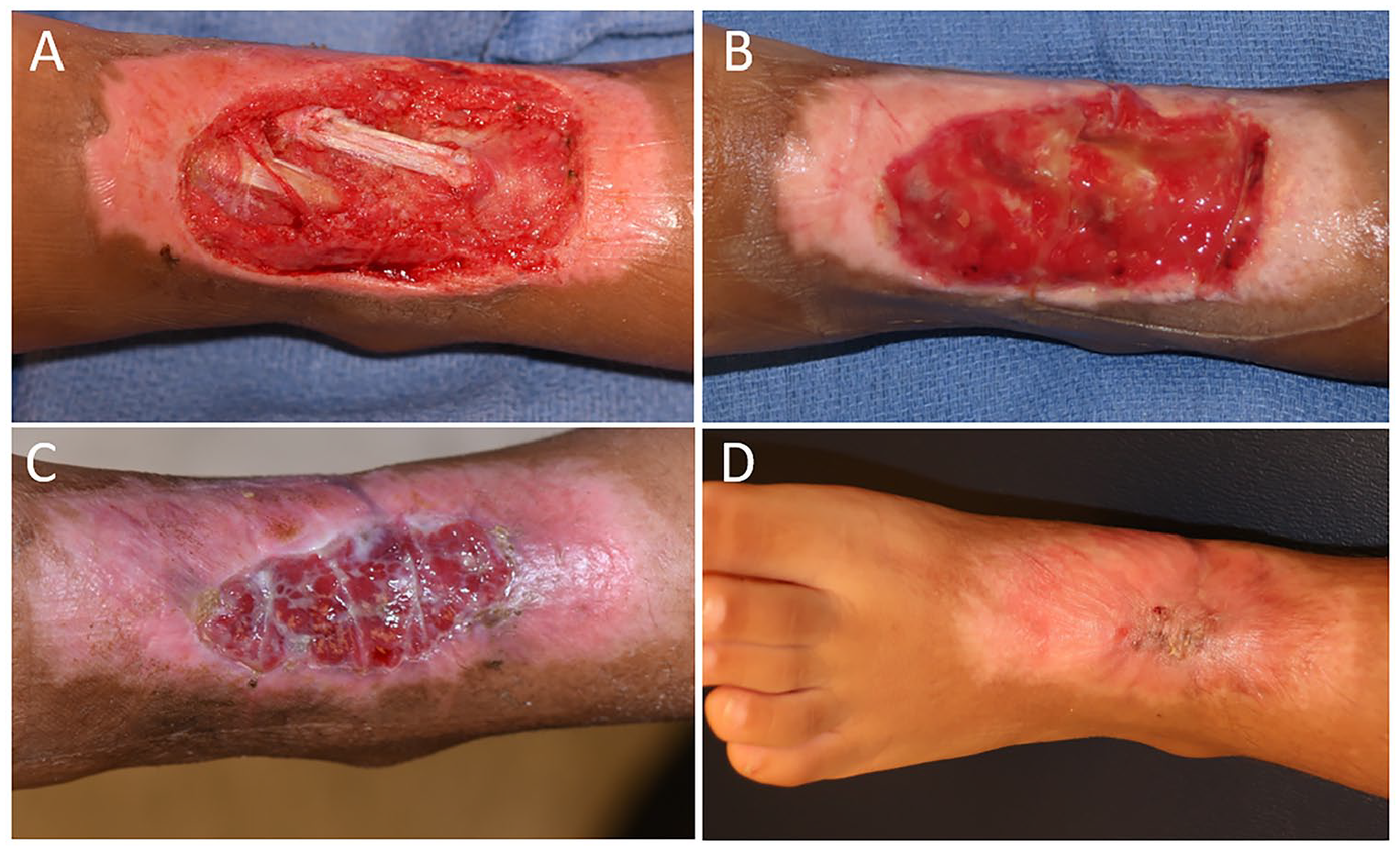
Serial photographs of Patient 2, Wound 2 (140 cm2), who sustained a left dorsolateral foot wound from a rollover motor vehicle collision with exposed extensor tendons and lateral malleolus. Intraoperative photographs following wound debridement showing exposed structures (A) and following application of autologous heterogeneous skin construct (AHSC) (B). Wound progression 3 weeks after AHSC application (C) demonstrating regenerated vascular tissue covering all exposed tendons and bone and 14 months after AHSC application (D) showing durable closure. Of note, the area of scarring in (D) correlates with an area of repeated wound opening due to a pressure point beneath his walking boot.
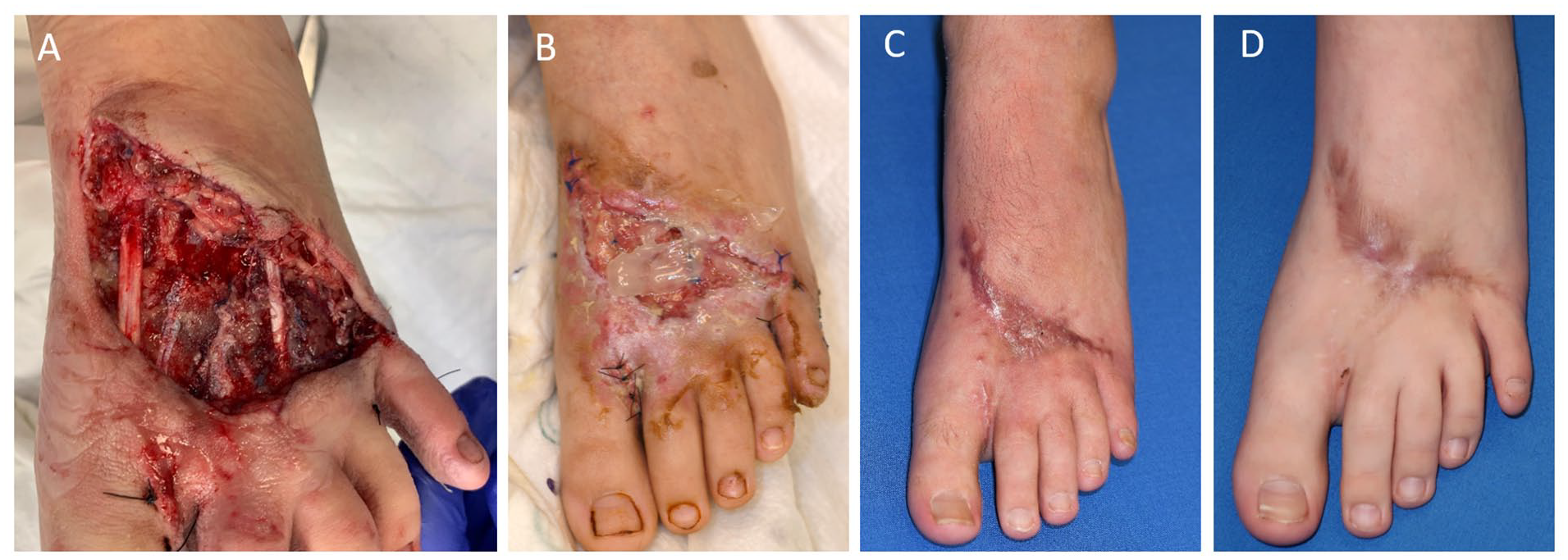
Serial photographs of Patient 4, Wound 4 (70 cm2), with a left dorsal foot wound due to a pedestrian versus motor vehicle accident with exposed extensor tendons. The wound following surgical debridement (A) shows the exposed extensor tendons. Two weeks after autologous heterogeneous skin construct (AHSC) (B), the tendons are covered in vascular tissue, with increased coverage by 3 weeks (C). The closed wound is shown 3 months after AHSC application in (D).
Serial photographs of a left dorsal foot wound with exposed extensor tendons from a dog bite in Patient 5, Wound 5 (24 cm2). The initial injury is shown (A), followed by the wound 16 days after autologous heterogeneous skin construct (AHSC) treatment (B), with complete wound closure shown at both 2 months (C) and 14 months (D) after AHSC application. This patient also sustained fracture of the third metatarsal and extensor tendon injuries requiring repair.
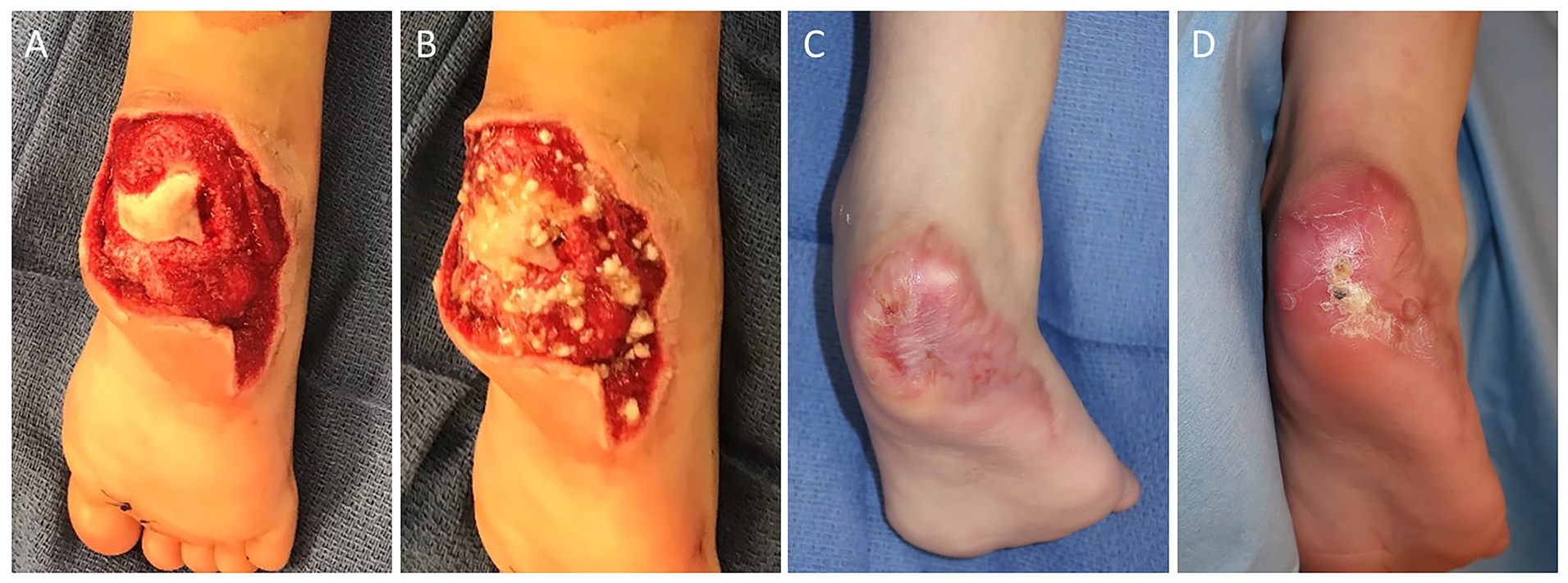
Serial photographs of a posterior right heel wound with exposed calcaneus from a lawnmower accident in Patient 6, Wound 6b (33 cm2). Intraoperative photographs after debridement (A) and after autologous heterogeneous skin construct (AHSC) application (B). Wound closure at 8 (C) and 11 (D) months after AHSC application with durable coverage over the calcaneus.
There were no donor site complications within this cohort. No wound required additional procedures. Two patients (29%) had minor complications not requiring operative management (Table 1): (1) a patient with autism required social readmission to perform dressing changes and (2) one patient with exposed extensor tendons, metatarsals, and small toe metatarsophalangeal joint developed a dorsal contracture of the small toe at the metatarsophalangeal joint but was able to wear a normal shoe without limitation of ambulation.
Discussion
This retrospective study demonstrates the ability of AHSC to achieve closure of traumatic soft tissue defects, with exposed tendon, bone, and/or joints, in a pediatric population. In 7 patients with 10 wounds, complete coverage of deep structures with newly generated vascular soft tissue was accomplished in a median of 13 days; and epithelial closure in a median of approximately 10 weeks with a single application of AHSC. No additional procedures were required following AHSC application.
The lack of late reoperations in this series using AHSC may be related to achieving contour match without bulk and preserved ROM. All patients have thus far been able to ambulate without assistance or limp, wear normal shoes, and maintain normal tendon gliding. We believe that the preserved tendon gliding could be due to (1) a regenerated fat layer beneath the regenerated skin shown on biopsy in a previous case report from the first author, (2) a robust vascular soft tissue that forms over tendons and can be seen early in the healing process prior to epithelial formation (Figures 1C, 2B and C), and (3) early ambulation, weight bearing, and ROM to prevent adhesion formation.7,9,12
Alternative treatments for these complex traumatic wounds with exposed structures were discussed extensively with the patients and their families. These included free tissue transfer, tissue expansion, and dermal matrix application followed by delayed skin grafting. The patients, their families, and surgical team chose to avoid dermal matrix reconstruction in this cohort due to the frequent requirement for a skin graft donor site, and possible risk of failure in fully covering exposed structures. Although dermal matrix reconstruction does allow for granulation and coverage of exposed structures, sometimes not requiring secondary grafting, it was believed that the cases in this series would have necessitated secondary grafting due to their size. The literature is limited in regard to dermal matrix reconstruction in pediatric trauma, and is mostly composed of case reports and small case series.2,3,14 Ghazi et al reported 4 cases of pediatric trauma wounds treated with a dermal regeneration template (Integra Lifesciences), with success in obtaining coverage in all wounds, and only one late complication of hypertrophic scarring requiring revision. 2 Schiestl et al reported 4 pediatric cases of large lower extremity avulsion injuries treated with Integra Dermal Regeneration Template (IDRT, Integra Lifesciences), with 95% to 100% take of IDRT within 22 to 26 days of application and 100% take of all subsequent skin grafts. 3
Examining the pediatric trauma free flap literature, complete, partial, and total flap failure rates range from 0% to 21%, 0% to 75%, and 0% to 75%, respectively.1,5,15-40 Lee et al recently reported a series of 53 free flaps performed in 49 pediatric patients with lower extremity trauma. 1 Overall, they found a total flap failure rate of 18.9%, with complete failure in 11.3% and partial failure in 7.6%, all of which required immediate operative takeback (18.9%). On analysis of risk factors in their series, arterial injury and vessel size mismatch were found to be the only significant predictors of failure, which are common scenarios in traumatic reconstruction. In one of the largest series of pediatric free flaps, with 91 patients receiving 93 primary flaps, Lin et al reported 155 revisions (1.7 per flap) in 78 patients (86% of patients), including 13 additional free flaps. 5
Autologous heterogeneous skin construct is not without its limitations. One of the main considerations before using AHSC, especially in pediatric patients, is the overall time to achieve epithelial closure. In this series, it took a median of 69 days (range = 26-101 days) to achieve complete epithelial closure, which can be lengthy for dressing changes in children. The closure time reported in this series is slightly inflated because of its retrospective nature and lack of a formalized study protocol for precise postoperative follow-up. Of note, if time to closure is the primary goal when treating a wound with AHSC, delayed skin grafting can be pursued immediately after deep structures are covered with vascular tissue, analogous to dermal matrix reconstruction followed by delayed grafting. This approach is worth considering for certain patients, such as adult manual laborers wishing early return to work. However, in this pediatric cohort, a primary goal was to avoid skin graft donor sites and to deliver functional outcomes; therefore, it was decided to provide the wounds with adequate time for complete epithelial closure without additional procedures. Additionally, it is difficult to predict or optimize cosmetic outcomes. Areas within the wound that develop hypergranulation tissue correlated with areas that healed with increased scarring. Ultimately, the goal was return of function, which was accomplished consistently.
This study has several limitations, including its retrospective nature, small sample size, lack of matched controls, and relatively short follow-up. Despite these limitations, the results achieved in these 7 patients with 10 traumatic wounds demonstrate the potential advantages of AHSC. If AHSC was not available or not used to treat these wounds, the treatments pursued would have likely been mixed between alternative treatment options, making strict comparisons to a matched cohort difficult within our single center. Longer follow-up could result in late contractures or tendon adhesions requiring revisions. Comparison to other reconstructive modalities for exposed structures are imperfect, as many of the flap series include injuries of greater severity, contain nontraumatic etiologies of high complexity, and report varied outcome measures. The direct impacts of wound bed preparation, dressings, and postoperative management, such as the number of debridements and/or the use NPWT as a bolster on top fenestrated silicone at the time of AHSC application, are difficult to evaluate in this small retrospective series. Nevertheless, serial debridement and washout were performed until all devitalized tissue and contamination were removed, as would be done with wound bed preparation prior to any graft or flap procedure. Although NPWT may have had some direct impact on the outcomes, the silicone contact layer prevented direct contact with the wound bed, limiting its action to fluid removal and bolstering AHSC. Furthermore, the maximum period of NPWT was 2 weeks, which was a relatively short timeframe in comparison to wound closure. Further evaluation of supply, hospital, and service costs are warranted to elucidate potential cost savings from this reconstructive technique.
Conclusions
Autologous heterogeneous skin construct covered exposed structures such as tendon, bone, and joint, and achieved closure with a single application in complex traumatic lower extremity wounds in a pediatric cohort. Benefits of AHSC included a low complication rate, minimal donor site, short operation room time and length of stay, and satisfactory functional outcomes. While these early results are encouraging, the study is limited by the small cohort and additional controlled studies are needed to further validate these results.
Footnotes
Acknowledgements
The authors wish to acknowledge Karen Beach for her assistance in writing this manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Mundinger receives consulting fees from PolarityTE MD, Inc. Drs Sopko and Swanson are full-time employees of PolarityTE MD, Inc. No other author has any conflict of interest to disclose. The content of this article was expressly written by the authors listed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medical writing services provided by Karen Beach were funded by PolarityTE MD, Inc.
Ethical Approval
This retrospective study was approved by the institutional review board of the Louisiana State University Health Sciences Center (Protocol #10326).
