Abstract
Individuals with attention-deficit/hyperactivity disorder (ADHD) are at increased risk of developing eating disorders (EDs) compared to the general population. Methylphenidate is a common first-line pharmacological treatment for ADHD and has been explored for symptom interruption in bulimia nervosa (BN); however, its use and efficacy in individuals with comorbid ADHD and restrictive eating pathology, such as anorexia nervosa (AN), remain underexplored. This case study presents a 28-year-old female who initially presented to eating disorder services with BN, was later diagnosed with ADHD, and subsequently re-diagnosed with AN. The patient was treated with a combination of methylphenidate alongside outpatient psychological therapy and participation in a Day Services group programme. While in Day Services, the patient restored 2.2 kg (4lb 9oz); however, the patient remained medically underweight at discharge. EDE-Q scores demonstrated a downward trend in cognitive eating disorder symptoms; however, the patient felt this was not reflective of her mental state. The case examines the interaction between ADHD and co-occurring ED symptomatology, illustrating how pharmacological management of one condition may inadvertently maintain or exacerbate the other. The case highlights a divergence between psychological improvement on self-report measures and persistent underweight status and disordered behaviours, raising questions about the interaction between ADHD pharmacotherapy and eating disorder outcomes.
Keywords
Theoretical and Research Basis for Treatment
Attention-deficit/hyperactivity disorder (ADHD) and eating disorders (EDs) such as bulimia nervosa (BN) and anorexia nervosa (AN) present significant public health concerns due to physical risks, psychosocial impairment, and adverse academic and occupational outcomes. Evidence indicates that individuals with ADHD are at increased risk to experience clinical EDs, binge and/or purge eating behaviors, and restrictive eating (Bleck et al., 2015; Makin, Zesch, et al., 2025). Epidemiological studies further suggest that ADHD is a prevelant comorbidity within ED populations, with estimates suggesting prevalence rates of 17% in BN and 23% in binge eating disorder (BED) (Hay et al., 2017).
Stimulant medications, particularly methylphenidate, constitute first-line pharmacological treatment for ADHD in adults in the United Kingdom (NICE, 2009). Case reports and case series have documented improvements in both ADHD symptoms and bulimic behaviours during treatment with methylphenidate (Bhat et al., 2023; Drimmer, 2003; Guerdjikova & McElroy, 2013; Ioannidis et al., 2014; Schweickert et al., 1997; Sokol et al., 1999). However, stimulant treatments are associated with appetite suppression and weight loss, raising clinical concerns in individuals with restrictive pathology (Finn et al., 2023; Keshen et al., 2021; Vickers et al., 2024). Despite these potential risks, systematic investigation of stimulant use in comorbid ADHD and ED presentations remain limited.
A review the literature by Makin, Meyer, et al., 2025, identified that ADHD traits were significantly elevated in individuals with BN compared to BED and anorexia nervosa–restrictive subtype (AN-R), while emphasising that the impact of ADHD pharmacotherapy in ED populations remains largely underexplored. Within the literature there is a view that eating disorders are transdiagnostic, meaning that the processes maintain one eating disorder pathology, are likely similar across other eating disorder diagnoses (Fairburn et al., 2003). Makin and colleagues review identified six case studies (Bhat et al., 2023; Drimmer, 2003; Guerdjikova & McElroy, 2013; Ioannidis et al., 2014; Schweickert et al., 1997; Sokol et al., 1999), involving seven participants prescribed methylphenidate for ADHD with a co-occurring BN diagnosis. Across, the cases, reductions in ADHD symptomology and improvements in EDE-Q scores were observed (Makin, Zesch, et al., 2025). Additional stimulants reported in the treatment of ADHD and BN include: Lisdexamfetamine, Adderall, and Dextroamphetamine (Ioannidis et al., 2014; Jeremias et al., 2024; Keshen et al., 2022).
Nonetheless, concerns persist regarding the use of stimulants for treatment of BN due to their potential for misuse regarding appetite suppressant (Herzog et al., 2006). Compared with BED populations, individuals with BN demonstrate higher levels of restrictive eating and an increased drive for thinness (Sullivan, 2001), which may be further exacerbated by stimulant medication (Hoopes et al., 2003). A feasibility trial by (Keshen et al., 2021) examined the effects of stimulant treatment for adults with BN, reporting an average weight reduction of 2.1 kg over the eight-week period, with one of the 23 participants withdrawn due to clinically significant weight loss. Although the study provides some reassurance regarding weight maintenance, its small sample size, and the exclusion of individuals with severe restrictive eating or a history of AN-R limit the generalisability of its findings.
According to the British National Formulary, a diagnosis of AN constitutes a contraindication to prescribing methylphenidate (British Medical Association, 2025). Clinical consensus further advises that stimulants such as methylphenidate should not be prescribed in people with BMI <17.5 (the ICD-10 diagnostic cut-off for an AN diagnosis), and should be used cautiously in those with electrolyte abnormalities due to the risk of arrhythmias (Keshen et al., 2022). Notably, two published clinical case reports describe weight restoration in individuals with comorbid AN and concomitant stimulant use (dextroamphetamine/amphetamine and methylphenidate); however, BMI at treatment initiation was 19.1 and 17.5 in the respective cases (Shear et al., 2021).
Methylphenidate acts by inhibiting the reuptake of noradrenaline and dopamine reuptake back into the presynaptic terminal, thereby allowing increased onward noradrenaline and dopamine transmission as well as direct agonism on 5-HT1A and 5-HT2B receptors (Docherty & Alsufyani, 2021). These mechanisms are associated with increased alertness, productivity, and cause cognitive enhancement, which may explain the high rates of non-prescription stimulant use observed in non-clinical populations (Jeffers et al., 2013). It could be postulated that such increased productivity may amplify evolutionarily conserved foraging responses in a state of starvation, symptomatically manifesting as high levels of activity in EDs (Sodersten et al., 2019). Through a compassion-focused neuroevolutionary lens, this would equate to a stimulation of the ‘activation pathway’ or ‘drive’ system associated with the pursuit of goals, resource acquisition and achievement, as opposed to the compassionate self-soothing/self-acceptance brain pathway, the latter of which is vital to recovery from an ED (Goss & Allan, 2010).
Taken together, these findings highlight the complex and often paradoxical interplay between neurobiological mechanisms, motivational systems, and eating disorder psychopathology in individuals with comorbid ADHD. Whilst stimulant medications may alleviate inattention, impulsivity, and executive function deficits (Huss et al., 2017), which are factors known to contribute to binge–purge cycles, appetite suppression can reduce loss-of-control eating through reductions in impulsivity symptoms and increased executive control (Manasse et al., 2015). Concurrently, stimulants may simultaneously exacerbate restrictive or compulsive tendencies through appetite suppression (Finn et al., 2023; Vickers et al., 2024). This dual action underscores the importance of nuanced, case-by-case clinical decision-making, where potential therapeutic gains must be weighed carefully against physical health risks and the reinforcement of maladaptive eating-related cognitions and behaviours.
The present case illustrates the clinical complexities encountered in treating a patient with co-occurring ADHD and BN. It examines the precipitating and maintaining factors of the eating disorder and highlights the challenges associated with initiating stimulant treatment, while also undergoing eating disorder treatment. With particular attention to therapeutic benefits, clinical response, physical health and psychological measures, and potential adverse effects. Physical health was monitored through regular weight measurements. Psychological change across treatment was assessed using the Eating Disorder Examination Questionnaire (EDE-Q), which captured changes in eating disorder–related cognitions over time.
Case Introduction
A 28-year-old female (referred to as Anna for anonymity) provided consent to having her case published. Anna met the international classification of diseased and related health problems (10th ed., ICD-10, World Health Organization, 2019), diagnostic criteria for AN, binge-eating/purging type. On admission to the eating disorders Day Services programme, Anna’s weight was 44.1 kg (97lb 2oz), with a body mass index (BMI kg/m sq) of 17.2 (normal range for BMI is between 19 and 25).
Presenting Complaints
Anna was accepted to the Day Services treatment programme within the Maudsley Eating Disorders Service in the South London and Maudsley (SLaM) NHS Trust, following persistent difficulties maintaining an adequate weight in the community. She had initially been treated in an outpatient setting, attending weekly therapeutic sessions; however, continued weight loss necessitated referral to an intensive, multidisciplinary programme to support weight restoration and reduce symptoms.
Anna demonstrated a marked overvaluation of weight and shape as central determinants of self-worth, accompanied by identity disturbance, alexithymia, and low self-esteem. She attributed her difficulties with emotional regulation and reward processing to her ADHD diagnosis, conceptualising her binge–purge episodes as secondary to these impairments. Although she presented with autistic traits, including cognitive inflexibility, adherence to ritualised behaviours, numerical preferences, and aversion to change (Cahalan et al., 2025; Chen et al., 2009; Petty et al., 2025), she did not identify with a diagnosis of autism. Instead, she interpreted these features as extensions of her ADHD symptomatology.
History
Personal History
Anna reported an awareness of her body from age 5, reporting negative thoughts and feelings regarding body image due to societal pressures and comments made by both classmates and her family. At age 12, the Anna reports her teacher questioning traits of neurodivergence, later being assessed, and diagnosed with ADHD by the national health care service at 12 years old. GP records of the patient at age 16 documented a weight of 74.6 kg (164 lb 5oz), corresponding to a BMI of 29.9 (99th age percentile). Despite persistent challenges around weight and Neurodivergence, she was high functioning and able to complete her A-levels, undergraduate, and master’s studies.
Family
Anna was raised in Europe, alongside her family consisting of her younger brother, adopted older sister, mother, and father. Anna described being “close” to her brother, but the dynamic with her parents as being “complicated” due to not feeling cared for by them. Anna was 10 years old when her older sister was adopted. She recalls feeling a need to compete for attention with her adopted sister, who was four years her senior. As a child, Anna witnessed emotional volatility at home, where physical altercations could break out amongst family members or her father would isolate himself, and at times when Anna attempted to seek support for herself, her feelings were dismissed. Anna disclosed that her family moved to different areas of their country of origin, often throughout childhood due to financial insecurity. The family then relocated to the United Kingdom (UK) when Anna was 16 years old, she described this move as “abrupt”. Anna began carrying the financially responsibility for her family’s settling into the UK by working a job and carrying out household tasks at the same time. Anna now lives with her wife and reports intermittently seeing her family.
Anna’s father has diabetes, consequentially, she learned how to measure blood glucose levels using Boehringer Mannheim (BMs). While living in the family home she began measuring her own blood sugar levels after purging or when feeling dizzy. Anna’s mother reports historically struggling with an eating disorder, and significant preoccupation with weight and shape. Anna was able to recall memories of her mother in childhood crying in front of the mirror due to her weight and her mother self-describing herself as ‘fat’.
Assessment
Assessment after being referred to the eating disorders outpatient department concluded that, Anna met the ICD-10 criteria for BN. These symptoms include overeating and excessive preoccupation with body weight, and compensatory behaviours like self-induced vomiting. This diagnosis was later revised by a cognitive behavioural psychotherapist to AN when Anna began psychological therapy. Her diagnosis was revised based on symptoms, including a BMI <17.5, and behaviours such as restrictive food intake. Anna was diagnosed with ADHD externally by an ADHD clinic, she obtained the diagnosis in March 2024. Anna’s weight was measured, and BMI was calculated during her initial assessment, when she began outpatients psychological therapy, upon acceptance to the Day Services programme, weekly while in the programme, and when discharged (see Figure 1). Case formulation
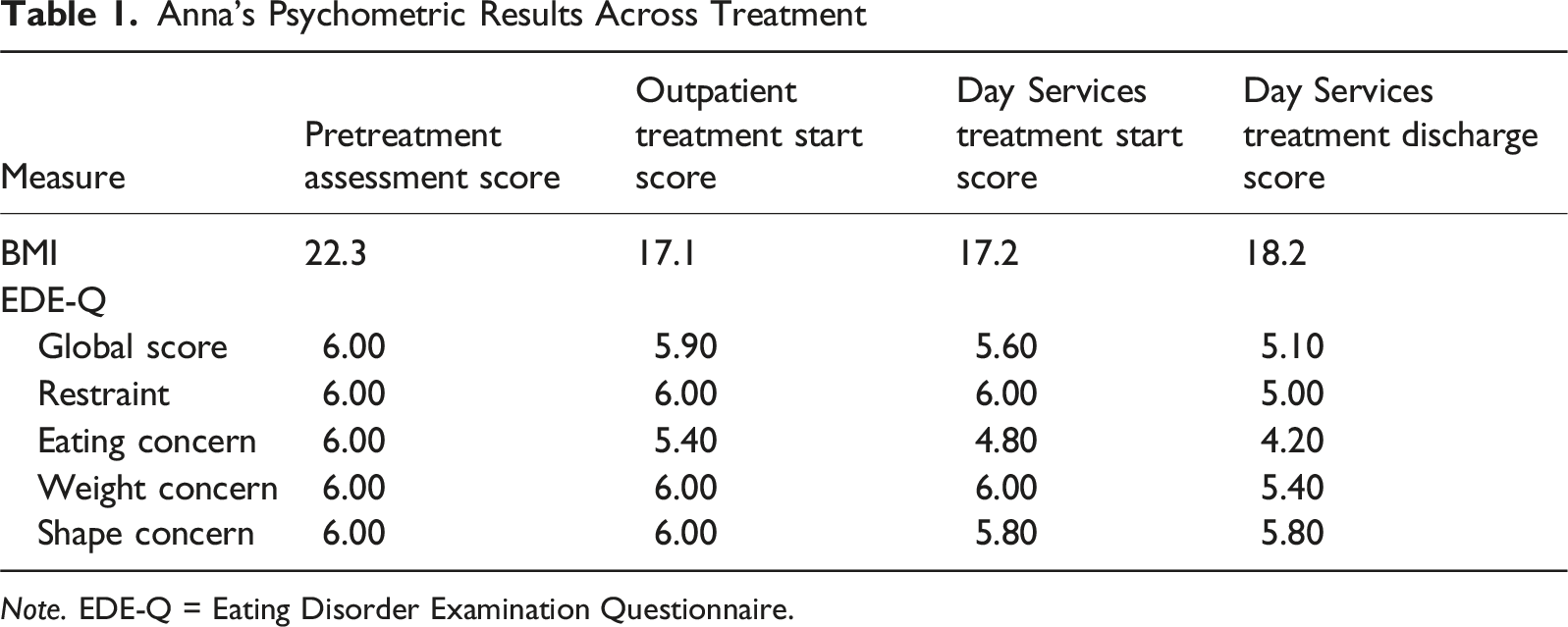
The Eating Disorder Examination Questionnaire (EDE-Q) self-report measure was administered at treatment intake and discharge points. The EDE-Q (Fairburn & Beglin, 1994) is a widely used self-report measure that assesses the core attitudinal and behavioural features of eating disorders over the past 28 days. The EDE-Q is comprised of a global score and four subscales measuring dietary restraint, eating concern, weight concern, and shape concern. Global and subscale scores range from 0 to 6, with higher scores indicating greater pathology. For the purpose of this case study, we reported the global score and four subscales at four time points throughout Anna’s community treatment (see Figure 2 and Table 1). Anna’s weight data during treatment in Outpatients, MANTRA, and Day Services. Anna’s Psychometric Results Across Treatment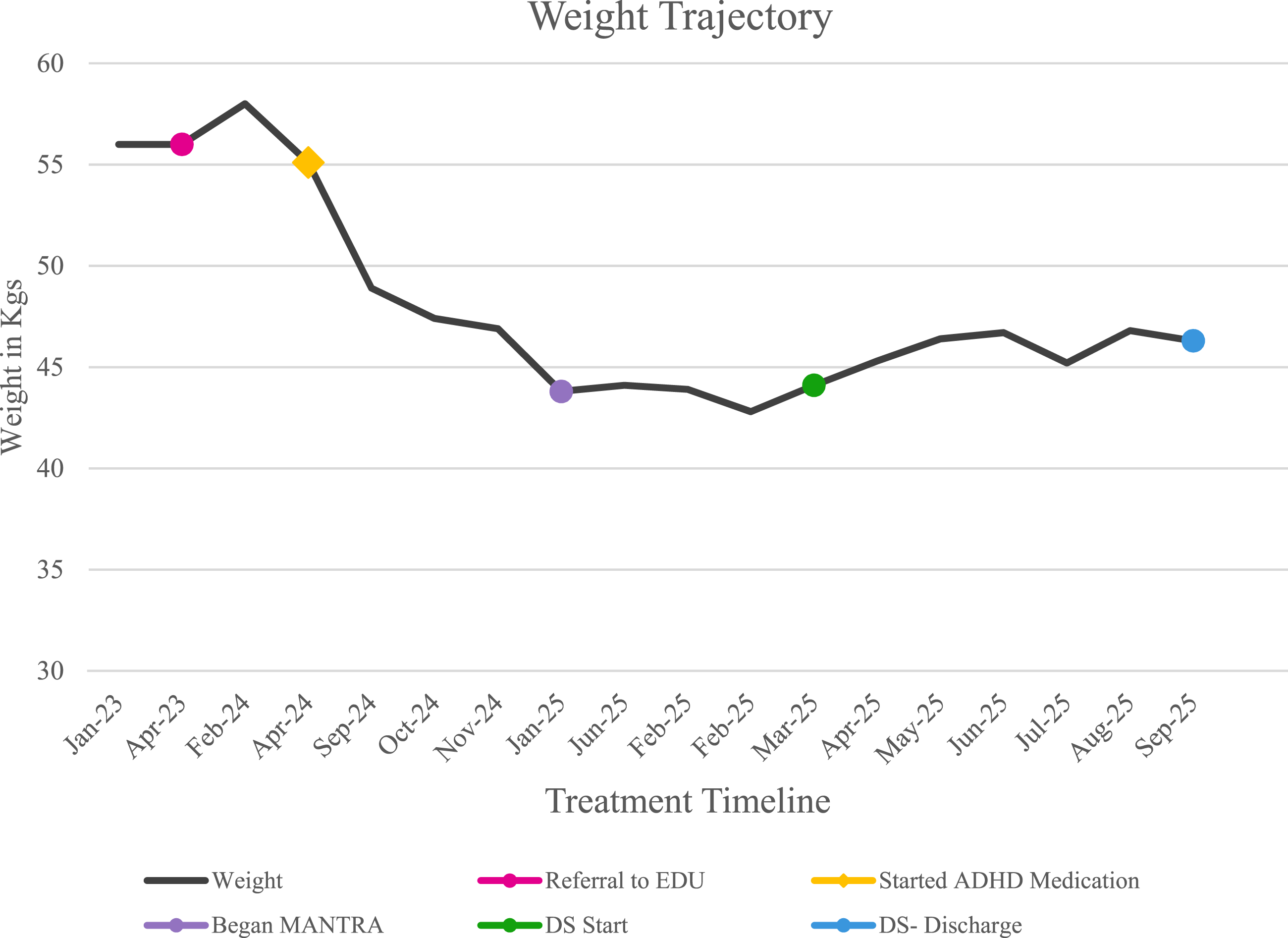
Case Conceptualisation
The cognitive behavioural model informs the following case formulation. The formulation is based on information provided by Anna, her assessment data, clinical notes, and her formulation (vicious flower - a tool used in MANTRA (Maudsley model of Anorexia Nervosa Treatment for Adults)). The predisposing factors stem from Anna’s childhood due to a high BMI from a young age, her South European upbringing, and their migration to the UK at the age of 16. Anna reported numerous challenges due to her weight and size during childhood; she disclosed being unable to find clothes in her size, resulting in her wearing her father’s clothing. Additionally, Anna spoke about her European upbringing and the cultural beauty standards of how individuals are expected to appear. This is tied to difficulties at school and “relentless bullying” due to her body composition. At the age of 16, Anna’s family migrated to the UK. The ensuing financial instability led Anna to feel the need to assume a responsible role by supporting the family’s needs above her own needs.
These factors in Anna’s childhood subsequently informed Anna’s self-perception, her relationships and her core beliefs. Anna established the belief that being overweight leads to rejection and anger in others, with low weight becoming central to her self-view and something that aids in eliciting care from others.
A significant precipitant to the onset of Anna’s eating disorder behaviours was her preoccupation with avoiding a return to the higher body weight she experienced in childhood. Anna described her younger self as “fat” and attributed experiences of bullying to her weight at that time. She now closely associates her identity with her underweight body and finds it difficult to envision happiness at a higher weight. Anna recalled that food was consistently tied to emotion throughout her life, describing it as a source of “joy.” Due to longstanding difficulties with restricted emotional range, she used food and overeating as a means of eliciting positive emotions. Following the prescription of methylphenidate for her ADHD, Anna reported an increased capacity for emotional depth, experiencing what she described as “a full emotional spectrum”. Despite this improvement, she continued to experience obsessive tendencies, a trait that predated methylphenidate treatment. These obsessions manifested in various ways, such as repeatedly preparing her lunch until it met her internal standards or through persistent preoccupation with body image, leading to ongoing discomfort and dissatisfaction with her physical appearance. Anna noted that this obsessive quality had been present since childhood; however, prior to ADHD treatment, her drive for emotional fulfilment through food had taken precedence.
A maintaining factor for Anna’s eating disorder included ‘control’, whereby following set rules around her eating it created a sense of control in her life. Anna had a preference for routine and is resistant to change. Her ADHD manifests in a way that reinforced her strict food rules through inflexible thinking, and a preference for predictability. The same poor flexibility that was associated with her ADHD, maintains, and perpetuates Anna’s eating disorder behaviours. Furthermore, Anna reported anorexia makes her feel “powerful”, a perception that intensified restrictive behaviours and contributed to the ongoing cycle of illness. Anna’s low weight led to feelings of “pride”; however, while in treatment with services, Anna spoke about comparisons to others and feeling bigger than other patients. On an interpersonal level, Anna’s eating disorder also functioned as a means of communication, allowing her to elicit care and support from others while simultaneously relieving her of certain personal responsibilities.
Protective factors for Anna included being well enough to continue her studies and attend a doctorate programme she had worked for several years to be admitted into. Anna describes this educational opportunity as a significant motivator for her recovery, as her career aspirations are central to her self-view, allowing her to feel financially secure, which was missing during her childhood. Additionally, protective factors for Anna were her supportive partner, family and friends who were involved in her care and supported her throughout treatment. Anna’s traits of being hard-working, resilient and solution focused were vital to her recovery as they contributed to her motivation to engage in treatment and with psychological work.
Course of Treatment and Assessment of Progress
Upon referral to the eating disorder service Anna was admitted, patients are comprehensively assessed to evaluate clinical risk and determine the most appropriate treatment pathway. Consistent with a stepped-care framework, the service aims to provide the least restrictive evidence-based intervention before escalating to more intensive treatment if required. In Anna’s case, individual psychological therapy was initiated as the first-line intervention for AN; however, due to ongoing weight loss, she was subsequently transitioned to the Day Services programme for a higher level of support.
In April 2023, Anna sought support for her ED from her GP and was referred to ED Outpatients at a weight of 56 kg (123lb 5oz; BMI = 21.9) (See Figure 2 for weight trajectory and Figure 3 for BMI trajectory), due to escalating binge–purge behaviours. At assessment, Anna weighed 58 kg (127lb 9oz; BMI = 22.3) and received a formal diagnosis of BN in line with ICD-10 criteria. She had not previously received ED-specific treatment and was therefore placed on the waitlist for CBT, a first-line treatment for BN. Anna’s BMI data during treatment in Outpatients, MANTRA, and Day Services. 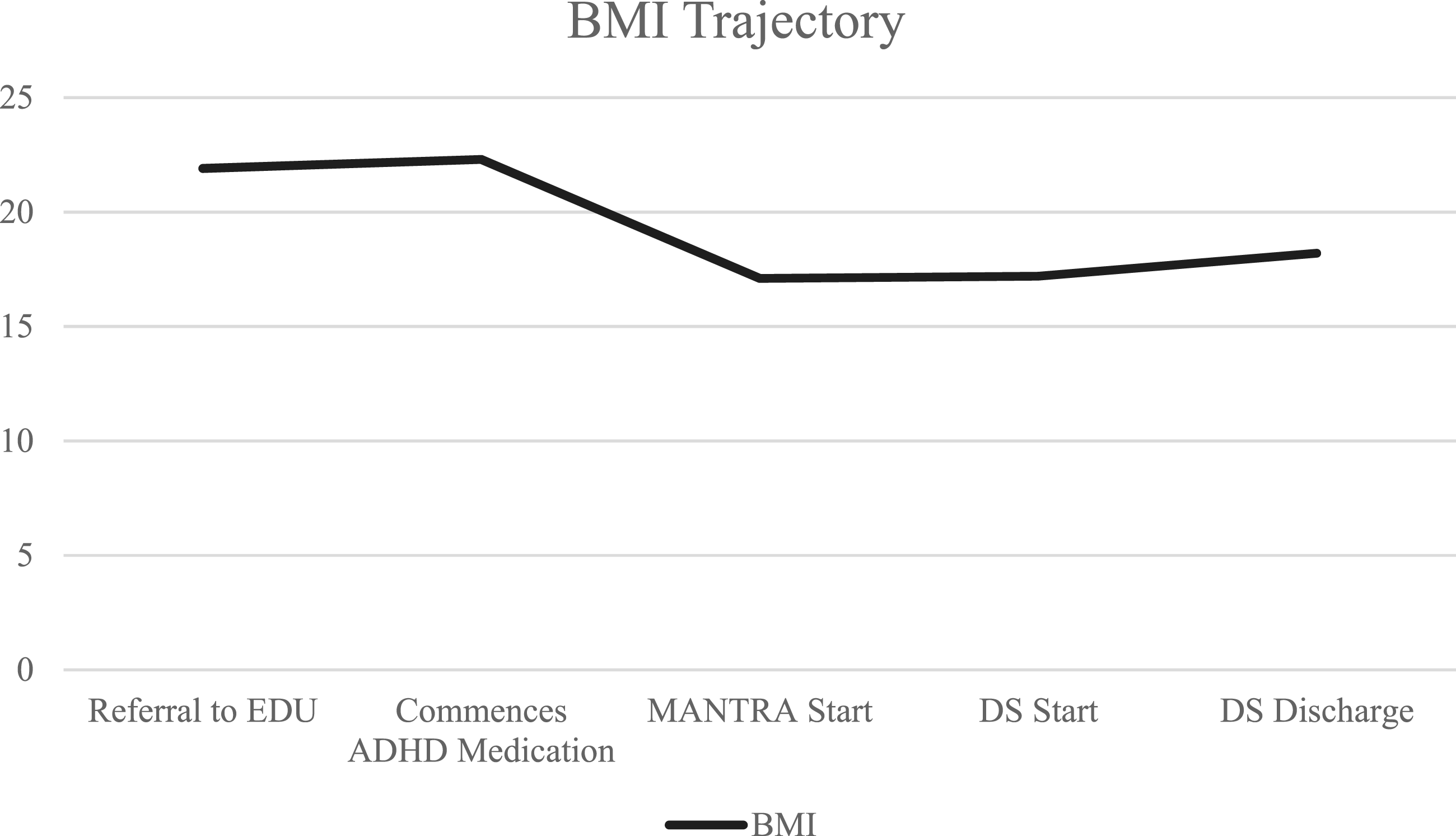
Anna underwent an adult ADHD assessment in March 2024 and was subsequently diagnosed with ADHD. She was commenced on methylphenidate in April of 2024, beginning on 5 mg and being titrated up to 20 mg three times daily. Anna reported this dosage enabled symptom management and she began taking a dose at 08:00 AM, 12:00 PM, and 16:00 PM, daily. Anna reported increased emotional regulation and motivation, decreased aggression and obsession around food, but a maintenance in negative thoughts relating to body image. Initially, Anna reported a reduction in binge-purge episodes; however, by November 2024, she described increased purging and difficulty retaining food. At that time, her weight had fallen to 46.9 kg (103lb 4oz; BMI = 18.2), and she reported restrictive intake with purging equal to or above four times per week.
Due to rapid weight loss, Anna was prioritised for psychological intervention, and was moved to the rapid waitlist. In January 2025, she began working with a Cognitive Behavioural Psychotherapist who revised the treatment plan and began MANTRA (Mountford et al., 2021), a cognitive-interpersonal treatment for adults with AN that typically consists of 20-40 individual sessions, through exploration of intra- and interpersonal illness-maintaining factors (Schmidt et al., 2021). When beginning MANTRA treatment Anna’s weight was 43.8 kg (96lb 6oz; BMI = 17.1), and her diagnosis was revised from BN to AN due to low BMI, in line with the ICD-10. Despite psychological input, her weight declined further to 42.8 kg (94lb 4oz; BMI = 16.7). As a result, she was referred to SLaM’s ED Day Services programme in February 2025 for more intensive treatment. The ED Day Services programme is an intensive outpatient service that focuses on routine, symptom interruption, and practical support, in tandem with psychological support. The programme requires daily attendance for an initial three months, followed by gradual reduction in attendance over an additional three months, the maximum period of treatment is nine months.
Anna participated in the Day Service group programme for six months, during which she engaged with the multidisciplinary team. Her treatment included individual sessions with an occupational therapist, development of a meal plan with a dietician specialising in eating disorders, and weekly medical monitoring by a registered mental health nurse. She also received support from the wider team, including an assistant psychologist and a cognitive behavioural therapist. Anna’s care was overseen by a consultant psychiatrist specialising in eating disorders (CD). Throughout her participation in the programme, Anna had treatment reviews with the multidisciplinary team every four to six weeks; however, the team reviewed her treatment progression weekly. While Anna attended the group programme, she continued attending her weekly MANTRA sessions with her outpatient psychotherapist.
During the first three months of the Day Service programme, Anna gained 2.6 kg (5lb 7oz), restoring from her intake weight of 44.1 kg (97lb 2 oz) to 46.7 kg (102lb 9oz; BMI = 18.2). Despite this progress, she continued to exhibit restrictive cognitions and expressed a desire to return to her lowest recorded weight (42.8 kg; 94lb 4oz). She also presented with mild hypokalemia (serum potassium 3.0–3.5 mmol/L) and was prescribed Sando-K (potassium supplement).
In May 2025, the clinical team raised concerns that Anna’s use of methylphenidate might be acting as an appetite suppressant and perpetuating her eating disorder behaviours. Anna declined both dose reduction and discontinuation. Anna stated that the methylphenidate treatment enabled her to better regulate her emotions and increased her emotional depth and motivation. Anna’s case was discussed at a medical huddle, where the decision was made to maintain the current dosage of methylphenidate, as it was considered beneficial to her overall functioning. Although her anorexic cognitions persisted, the medication was deemed beneficial in whole.
Over the subsequent three months in the group programme, her weight decreased marginally by 0.5 kg (1lb 1oz). Despite consistent engagement in treatment, she was unable to restore to a healthy BMI and continued to display restrictive eating behaviours. After six months, she was discharged from the Eating Disorder Day Services programme at 46.3 kg (102lb 1oz; BMI = 18.1), continuing to report persistent weight and shape concerns, daily purging (∼3 times/day), and a pronounced drive for thinness. Across the six-month period of Day Services and outpatient therapy, Anna achieved a weight restoration of 2.2 kg (4lb 9oz; See Figure 1). However, her Eating Disorder Examination Questionnaire (EDE-Q) scores demonstrated a downward trend in cognitive eating disorder symptoms, with reductions across the Shape Concern, Weight Concern, Eating Concern, Restraint, and Global scales (Figure 4). When these measures were reviewed with Anna, she described inner conflict, acknowledging a minimal shift in her mindset, yet feeling that these changes were not reflected in her overall mental state or physical presentation. EDE-Q scores over the course of treatment. 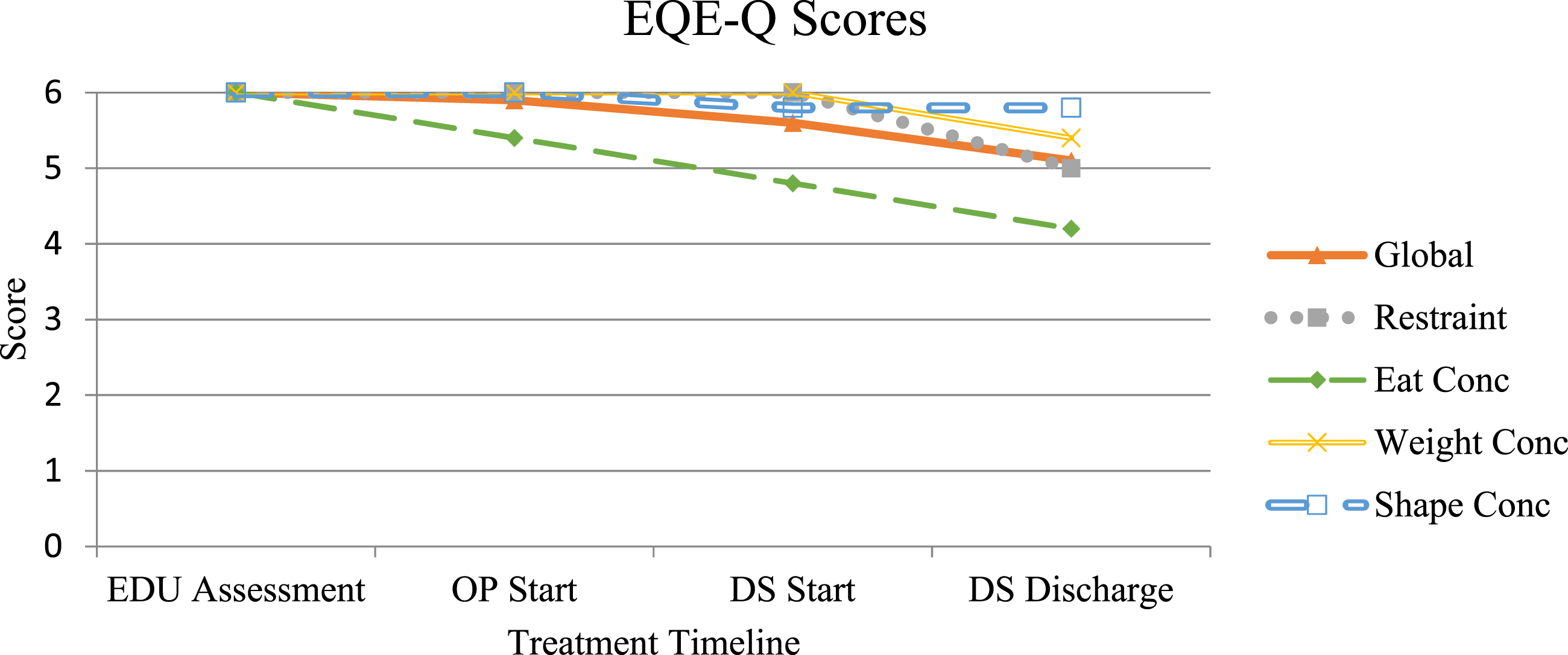
Complicating Factors
This case highlights the complexities between ADHD and a co-occurring ED, and the potential interplay of stimulant pharmacotherapy. While methylphenidate helped to decrease ADHD symptoms initially, it could be acting as a moderating factor of the Anna’s eating disorder. The case reflects the broader difficulty of addressing one psychiatric condition without exacerbating another, particularly when treatment mechanisms have opposing effects on appetite and weight.
Access and Barriers to Care
There were no significant barriers to Anna’s access to care. She received treatment through publicly funded NHS services, including both outpatient care and eating disorder Day Services, which enabled timely and sustained engagement with treatment.
Follow-Up
Anna was discharged from the Day Services programme in September of 2025 and completed 30 sessions of MANTRA in October 2025. After six months of Day Services treatment, she had a BMI of 18.1 kg/m2. She has since concluded her outpatient treatment and been discharged from the service. Anna has now returned to her education and is studying a doctorate programme in the health care sector.
Treatment Implications of the Case
This case illustrates the complex interaction between ADHD and eating disorders, particularly regarding the use of stimulant medication. ADHD is increasingly recognised as a common comorbidity in ED populations (Hay et al., 2017; Makin, Meyer, et al., 2025) and stimulant medications like methylphenidate may transiently reduce binge frequency. However, stimulants are also well known to suppress appetite and may exacerbate restrictive eating, weight loss, and compensatory behaviours in individuals with EDs (Keshen et al., 2021; Stutzman & Dopheide, 2024; Vickers et al., 2024).
In the present case, Anna initially experienced a reduction in binge episodes but subsequently demonstrated increased restrictive thoughts, progressive weight loss, and transitioned from a diagnosis of BN to AN. The persistence of methylphenidate use despite clinical concerns underscores the potential risks of stimulant pharmacotherapy in patients with an active ED pathology (Herzog et al., 2006).
Anna’s trajectory also highlights a notable divergence between psychological and behavioural outcomes. While self-reported ED cognitions (measured via EDE-Q) improved during her treatment with ED Services, this was not mirrored by sustained weight restoration or behavioural recovery. This dissociation raises questions about the sensitivity of self-report measures in populations where ego syntonic conditions, limited insight, minimisation, or cognitive rigidity may influence responses, and suggests that multi-modal assessment (including weight trends and behavioural frequency) remains essential in treatment monitoring.
Clinically, this case emphasises the importance of integrated care approaches for patients with dual diagnoses. Early multidisciplinary discussion between ED and ADHD services may have facilitated closer medication monitoring, consideration of non-stimulant pharmacological alternatives (Bastiaens, 2007), or structured behavioural interventions for ADHD. Furthermore, it highlights the challenge of balancing symptom reduction in one disorder (ADHD) with potential exacerbation of another (ED).
Recommendations to Clinicians and Students
This case study provides valuable clinical insights into the management of eating disorders co-occurring with ADHD. However, its conclusions are limited by the reliance on a single case and the potential recall bias in self-reported behaviours. Nevertheless, the case underscores the urgent need for further systematic investigation into the safety, efficacy, and clinical outcomes of stimulant pharmacotherapy in individuals with concurrent EDs, particularly those at risk of transitioning from BN to AN.
In Anna’s case, she felt that a more collaborative approach between eating disorder services and ADHD services would have enhanced her care. With this in mind, an integrated model of care may be beneficial in promoting shared care planning and reducing fragmentation across services. It is also recommended that clinicians working within eating disorder services receive training on the comorbid presentation of ADHD and eating disorders. Additionally, greater interdisciplinary collaboration is recommended to support individuals with an ADHD-ED comorbidity, particularly those presenting with AN or restrictive eating pathologies.
This case contributes to the emerging literature on ADHD–ED comorbidity and stimulant use, illustrating that while stimulant treatment may initially reduce binge-purge behaviours through enhanced impulse control, it can also simultaneously exacerbate restrictive pathology, hinder weight restoration, and blunt treatment response. These findings highlight the necessity for cautious, closely monitored, and multidisciplinary approaches when prescribing stimulant to individuals presenting with concurrent ADHD and ED symptoms.
Footnotes
Acknowledgements
The authors thank Ellie Popp for her support with the case formulation.
Ethical Considerations
This case report was concluded with the written consent of the patient and clinical governance (ID 788/17.6.25). Consent was given for publication.
Author Contributions
Author A.M gathered the data, authors A.M. and C.D shared in writing the case report and researching relevant literature, and all authors were involved in conceptualising, drafting, editing and finalizing the case study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is support by the One King’s Impact Fund and aligns with King’s Impact Priorities: Whole-life health for mind and body & Technology for good. KT would like to acknowledge the National Institute for Health and Care Research (NIHR) Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London. The views expressed are those of the author and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
Declaration of Conflicting Interests
The authors declare no potential conflicts of interests with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings for this study are available from the corresponding author upon reasonable request.
Author Biographies
![]() for details).
for details).
