Abstract
This study aimed to describe the clinical journey of patients with different stages of Alzheimer disease (AD). This was a cross-sectional survey of US primary care physicians (PCPs)/specialists and patients using the Adelphi Real World AD Disease Specific Programme™ (December 2022 – September 2023). Patients were stratified by disease severity and data are presented as the mean (SD) or frequencies/percentages. In the overall sample (N = 990), mean time from symptom onset to first evaluation was 31.4 (40.6) weeks and mean time from evaluation to diagnosis was 14.2 (29.0) weeks (mild cognitive impairment due to AD, 12.0 [22.7] weeks; mild AD dementia, 15.7 [31.6] weeks; moderate AD dementia, 14.0 [29.9] weeks; severe AD dementia, 5.1 [9.6] weeks). 74.5% of the overall sample was initially evaluated by their PCP and 13.8% by a neurologist. Patients with AD experience many barriers during the diagnostic journey; however, PCPs and neurologists play key roles in early diagnosis.
Plain Language Summary
Alzheimer disease (AD) is a main cause of dementia in older adults. Early diagnosis of AD is key, as it increases opportunities for treatment that may slow cognitive decline. However, underdiagnosis and diagnostic delays are common, particularly in the early stages of the disease. This study aimed to describe the clinical journey of patients with different stages of AD and to describe the diagnostic and monitoring tests used in these patients. This study also aimed to evaluate the role of primary care physicians (PCPs) and specialists in the diagnostic process. This study used survey data from the Adelphi Real World AD Disease Specific Programme™ between December 2022 and September 2023. The survey included data from US PCPs and specialists and their patients. Patient data was separated by disease severity into 4 categories: mild cognitive impairment due to AD (MCI due to AD), mild AD dementia (MAD), moderate AD dementia (MoAD), and severe AD dementia (SevAD). In the overall sample (N = 990), the average time from the beginning of symptoms to the first doctor’s appointment was 31.4 weeks, and the average time from the first doctor’s appointment to diagnosis was 14.2 weeks. The average time from the first doctor’s appointment to diagnosis was 12.0 weeks for patients with MCI due to AD, 15.7 weeks for MAD, 14.0 weeks for MoAD, and 5.1 weeks for SevAD. In the overall sample, 74.5% of patients were first evaluated by their PCP and 13.8% were first evaluated by a neurologist. Blood tests for AD-specific biomarkers were used to diagnose 9.0% of patients. This study found that PCPs and neurologists play an important role in diagnosing AD during early disease stages and that biomarker tests were not frequently used during diagnosis. In sum, patients with AD experience many barriers that may prevent quick diagnosis and potential treatment.
Keywords
Introduction
Alzheimer disease (AD) is the main cause of dementia in older individuals, affecting approximately 50 million people worldwide 1 and 6.7 million people aged ≥65 in the United States. 2 Pathological hallmarks of AD include the presence of amyloid β and phosphorylated tau. 1 Currently, AD is the fifth leading cause of death in US adults aged ≥65. 2 AD represents a substantial cost to society, including an estimated $339.5 billion in unpaid caregiving costs in 2022 and $345 billion in health care costs in 2023. 2 Patients with advanced AD also have higher AD-related health care resource utilization compared to patients with early AD. 3
Diagnosis of AD typically begins with an assessment of cognitive status, often using a brief cognitive assessment tool such as the Mini Mental State Exam (MMSE). MMSE scores indicating cognitive impairment may lead physicians to further evaluate specific etiologies and to consider the potential contribution of AD-related pathology to the impairment.4,5 Diagnosis may involve structural neuroimaging tests, functional imaging such as fluorodeoxyglucose positron emission tomography (FDG-PET), or more rarely amyloid-positron emission tomography (amyloid-PET). 1 However, confirming AD pathology as early as possible may be difficult due to a variety of factors including difficulties distinguishing between normal aging and cognitive impairment. 6 Therefore, underdiagnosis and diagnostic delays are common, particularly in the early stages of disease.7,8 Moreover, while several tools are available to establish a biomarker-confirmed diagnosis (amyloid-PET, cerebrospinal fluid [CSF] biomarkers, and emerging blood-based biomarkers), these tools are not widely used and can be difficult to access.
Early diagnosis can expand opportunities for patients to access treatments, such as anti-amyloid monoclonal antibodies, indicated for early stages of AD. 9 A diagnostic delay could prevent patients from receiving treatment, should cognitive impairment progress during the delay or while awaiting biomarker confirmation. 6 In order to obtain timely AD diagnoses, it is important to identify factors that may delay diagnoses.
AD may be treated with therapies that address symptoms or medications that remove amyloid. US Food and Drug Administration (FDA)-approved symptomatic treatments include cholinesterase inhibitors and N-methyl-D-aspartate receptor antagonists, which are intended to enhance cognitive function.1,10 More recently, the FDA approved the disease-modifying therapies donanemab and lecanemab.10-12 First approvals for lecanemab and donanemab were received in January 2023 13 and July 2024, 12 respectively. While emerging therapies have potential benefits for patients with early stages of AD, current diagnostic and monitoring processes remain poorly understood and the roles of primary care physicians (PCPs) and specialists in these processes are unclear. The data from this study serve as a benchmark for the field as more biomarkers and therapies enter the market.
The objectives of this study were to describe the clinical journey of patients with mild cognitive impairment (MCI) due to AD, mild AD dementia (MAD), moderate AD dementia (MoAD), and severe AD dementia (SevAD) at diagnosis, understand which tests are used to diagnose and monitor patients, and understand the roles of PCPs and specialists in the diagnostic process.
Methods
Data were drawn from the Adelphi Real World AD Disease Specific Programme™ (DSP), a cross-sectional survey with elements of retrospective data collection which captured data from PCPs, neurologists, psychiatrists, geriatricians, and their patients and was conducted in the United States between December 2022 and September 2023. The DSP methodology has been previously described,14,15 validated, 16 and demonstrated to be representative and consistent over time. 17
Physicians were instructed to complete patient record forms for their next 9 consecutive patients with AD or MCI due to AD. Each patient record form captured details regarding patient demographics, cognitive test results at the time of diagnosis and at the time of survey completion, comorbidities, physician specialties consulted, time to diagnosis, and testing conducted. Each patient for whom the physician completed a patient record form was invited to fill out a patient self-completion form; these were completed independently immediately following evaluation and were returned in a sealed envelope, ensuring that responses were kept confidential.
Patients included in the survey were aged ≥50 years with a current diagnosis of AD dementia or MCI due to AD based on physician assessment. MCI due to AD included prodromal AD, predementia AD, and amnestic/nonamnestic MCI due to suspected AD. AD dementia included AD and mixed dementia. Patient- or physician-reported biomarker testing was not required in this population. For all diagnosis-related results, patients were classified based on their MMSE score at diagnosis. For all other results, including demographics, comorbidities, and patient experiences, patients were classified based on their MMSE score at the time of survey completion, as their MMSE score and AD stage may have changed since their initial diagnosis. Patients with MMSE scores of 26 to 29, 21 to 25, 11 to 20, and 0 to 10 were classified as having MCI due to AD, MAD, MoAD, and SevAD, respectively. 18
Post hoc analyses were performed to assess the differences between patients who had a shorter or longer than median time duration from symptom onset to first evaluation (≤21.9 or >21.9 weeks) and first evaluation to diagnosis (≤12.0 weeks or >12.0 weeks), including variables such as age, sex, comorbidities, and types of medications. The statistical testing method is indicated with footnotes in each table. Additionally, a Cox proportional hazards model was used to explore the relationship between the hazard function and a set of covariates. This model assumes that covariates multiplicatively shift the hazard function. This assumption was evaluated using Schoenfeld residuals after fitting the Cox proportional hazards model. In cases of ties, the Breslow method was applied.
An oversample of patients with a biomarker-confirmed diagnosis of MCI due to AD (eg, based on a positive CSF test or amyloid-PET scan) was also collected. These patients were included in the analyses to boost the number of patients with milder disease to better understand this patient cohort and reduce variability from individual patients. These patients were removed from analyses where their inclusion would influence sizing (eg, analyses of severity and diagnostic testing).
This article was developed in accordance with STROBE cross-sectional reporting guidelines. 19
Results
Patient Demographics, Comorbidities, and Treatment Regimens Stratified by Disease Severity at Survey Completion a

aData included in this table were obtained from the physician-completed patient record form. An oversample of patients with a biomarker-confirmed diagnosis of MCI due to AD was included in these analyses to boost the number of patients with milder disease and to reduce variability from individual patients.
bStatistical significance was assessed using ANOVA.
cStatistical significance was assessed using Fisher’s exact test.
dData for ‘American Indian, Indigenous American, or Alaska Native,’ ‘Native Hawaiian or Pacific Islander,’ and ‘Other’ groups were not included in the table due to a low sample size.
eShown are the top 10 comorbidities in the overall sample.
fMedications shown are the top 3 medication types reported in the overall sample.
ACE, angiotensin-converting enzyme; AD, Alzheimer disease; ADHD, attention deficit hyperactivity disorder; ALS, amyotrophic lateral sclerosis; ANOVA, Analysis of Variance; BMI, body mass index; CBS, corticobasal syndrome; IBS, irritable bowel syndrome; IQR, interquartile range; MAD, mild Alzheimer disease dementia; MCI, mild cognitive impairment; MND, motor neuron disease; MoAD, moderate Alzheimer disease dementia; NASH, nonalcoholic steatohepatitis; SevAD, severe Alzheimer disease dementia.
According to physician-completed patient record forms, patients experienced a mean of 3.2 comorbidities at the time of data collection (Table 1). Frequently occurring comorbidities in the overall sample included arterial hypertension (36.0%), anxiety (35.8%), depression (28.6%), and hyperlipidemia (25.3%; Table 1). The mean (SD) number of comorbidities in patients with SevAD, MoAD, MAD, and MCI due to AD was 3.5 (2.4), 3.7 (2.9), 2.8 (2.3), and 3.0 (2.4), respectively; disease severity was significantly associated with the number of comorbidities (P < 0.001; Table 1).
Physicians reported that patients in all 4 cohorts commonly received treatments for comorbidities. In the overall sample, the most frequently received comorbidity treatments were antidepressants (37.1%), angiotensin-converting enzyme (ACE) inhibitors (28.4%), and statins (28.2%; Table 1). Antidepressants and ACE inhibitors were used by 39.9% and 37.5% of patients with MoAD and 47.6% and 31.0% of patients with SevAD, respectively. These medications were used by 34.8% and 23.0% of patients with MAD and 32.7% and 19.1% of patients with MCI due to AD, respectively. Conversely, 35.5% of patients with MCI due to AD were using statins compared to 26.0% of patients with MAD, 29.2% of patients with MoAD, and 19.0% of patients with SevAD (Table 1).
Top Ten Symptoms Stratified by Disease Severity at Survey Completion a

aData included in this table were obtained from the physician-completed patient record form. An oversample of patients with a biomarker-confirmed diagnosis of MCI due to AD was included in these analyses to boost the number of patients with milder disease and to reduce variability from individual patients.
bStatistical significance was assessed using the Kruskal-Wallis test.
AD, Alzheimer disease; MAD, mild Alzheimer disease dementia; MCI, mild cognitive impairment; MoAD, moderate Alzheimer disease dementia; SevAD, severe Alzheimer disease dementia.
Diagnostic Journey Stratified by Disease Severity at Diagnosis a

aData included in this table were obtained from the physician-completed patient record form. An oversample of patients with a biomarker-confirmed diagnosis of MCI due to AD was included in these analyses to boost the number of patients with milder disease and to reduce variability from individual patients.
AD, Alzheimer disease; IQR, interquartile range; MAD, mild Alzheimer disease dementia; MCI, mild cognitive impairment; MMSE, Mini Mental State Exam; MoAD, moderate Alzheimer disease dementia; PCP, primary care physician; SevAD, severe Alzheimer disease dementia.
According to physician-completed patient record forms, symptoms prompting a patient to seek an initial evaluation included loss of short-term memory (83.2%), difficulties recalling names/words (39.5%), and difficulties in concentration/attention (37.6%; Table 3). In the overall sample, mean (SD) time from symptom onset to first evaluation was 31.4 (40.6) weeks (Table 3). Most patients (74.5% of the overall group) initially consulted with their PCP, while 13.8% initially consulted a neurologist (Table 3). A total of 76.8% of patients with MCI due to AD and 76.3% with MAD consulted their PCP before any other physician compared to 69.9% of patients with MoAD and 66.7% with SevAD.
Approximately half (48.5%) of the overall sample was referred to a secondary provider prior to diagnosis, including 51.1% of patients with MCI due to AD, 46.1% of patients with MAD, 52.0% of patients with MoAD, and 37.5% of patients with SevAD (Table 3).
In the overall sample, 39.3% of initial diagnoses were made by a PCP, while 43.7% were made by a neurologist (Table 3). PCPs made 34.7%, 41.2%, 39.5%, and 41.7% of diagnoses in patients with MCI due to AD, MAD, MoAD, and SevAD (Table 3). Neurologists made 56.0%, 42.4%, 37.9%, and 16.7% of the diagnoses in patients with MCI due to AD, MAD, MoAD, and SevAD, respectively (Table 3).
In the overall group, mean (SD) time from the first evaluation to diagnosis was 14.2 (29.0) weeks. Time to diagnosis ranged from 0.0 (0.0 years; immediate diagnosis) to 305.3 weeks (5.9 years), with a median (IQR) of 3.7 (0.0-13.6) weeks (Table 3, Figure 1). Median (IQR) time from first evaluation to diagnosis was 4.4 (0.0-14.4) weeks for MCI due to AD, 3.8 (0.0-14.7) weeks for MAD, 3.5 (0.0-13.0) weeks for MoAD, and 0.0 (0.0-9.3) weeks for SevAD. Median (IQR) time from symptom onset to first evaluation was 18.7 (8.7-52.1) weeks in the MCI due to AD group, 16.7 (4.4-35.0) weeks in the MAD group, 18.6 (5.7-44.7) weeks in the MoAD group, and 33.0 (9.6-53.3) in the SevAD group. Length of time spent at each stage of the diagnostic journey, with patients stratified by disease severity at diagnosis. Hazard curves show the time from symptom onset to the first evaluation (A) and first evaluation to diagnosis (B). Patients with MCI due to AD, MAD, MoAD, and SevAD at diagnosis are shown in blue, green, yellow, and red curves, respectively. AD, Alzheimer disease; MAD, mild Alzheimer disease dementia; MCI, mild cognitive impairment; MoAD, moderate Alzheimer disease dementia; SevAD, severe Alzheimer disease dementia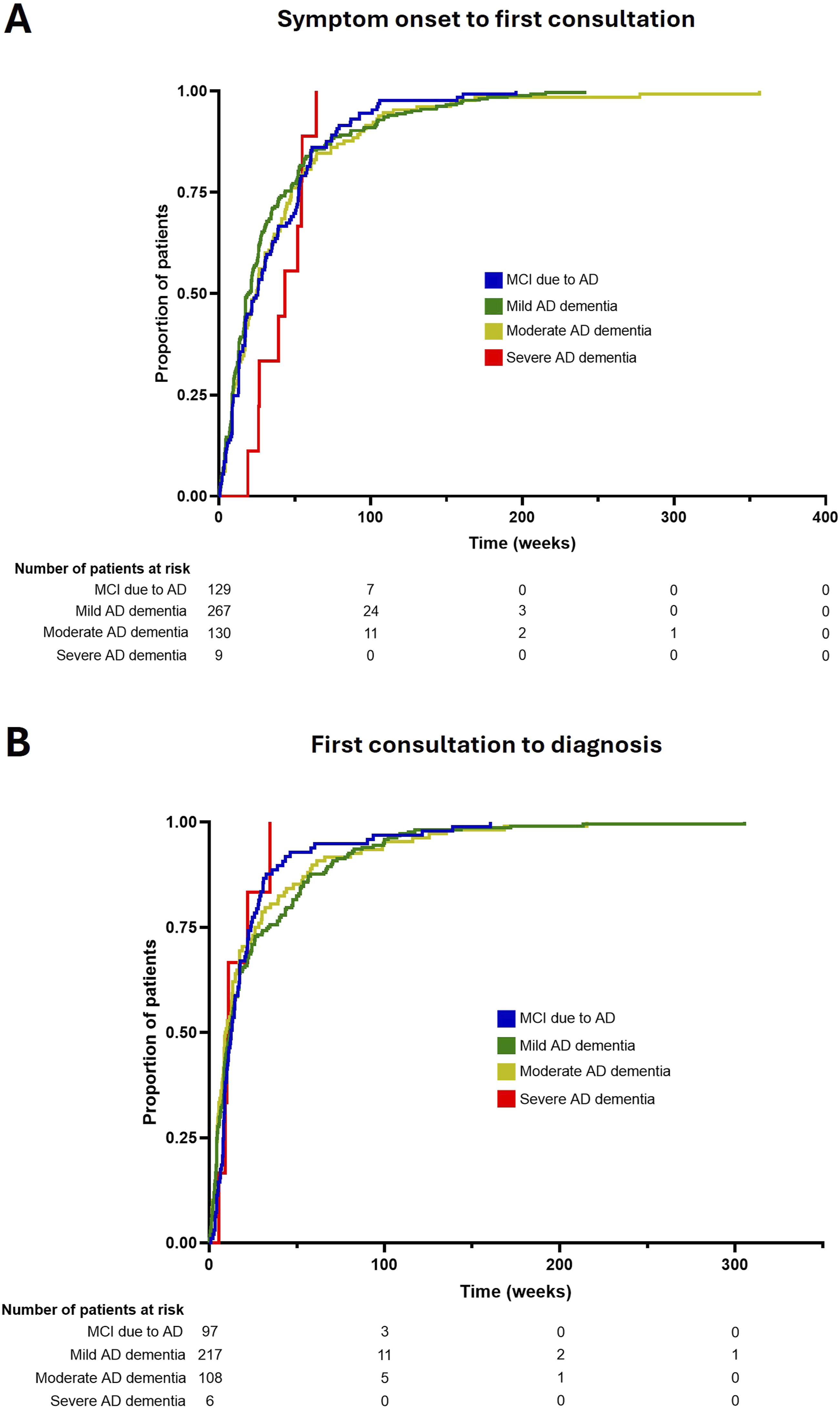
aAn oversample of patients with a biomarker-confirmed diagnosis of MCI due to AD was included in these analyses to boost the number of patients with milder disease and to reduce variability from individual patients.
bData for patients with SevAD were not included in this table due to low patient numbers. No patients with SevAD provided information on their reasons for delay in obtaining an initial evaluation and physicians provided information on reasons for delay between first evaluation and diagnosis for only 15 patients with SevAD.
cData included in this section were obtained from the patient self-completion form. The sample size in this section is smaller than samples obtained from physician-provided data, as not every patient completed the questionnaire.
dData included in this section were obtained from the physician-completed patient record form.
AD, Alzheimer disease; MAD, mild Alzheimer disease dementia; MCI, mild cognitive impairment; MoAD, moderate Alzheimer disease dementia; SevAD, severe Alzheimer disease dementia.
When asked about factors that contributed to a delay between the first evaluation appointment and diagnosis, physicians identified difficulties related to appointment availability, including, “time taken to schedule the next consultation” (43.2% of patients), “time taken for patient’s referral to a specialist” (35.0%), and “time taken to schedule a diagnostic test” (27.5%; Table 4).
Several post hoc analyses were performed to assess differences between patients who had a shorter or longer duration than the median for aspects of the diagnostic timeline (Supplemental Table 2). These analyses revealed that patients who experienced longer times than the median from symptom onset to first evaluation and from first evaluation to diagnosis had a statistically significantly higher number of comorbidities (Supplemental Table 2 and Supplemental Table 3). Results from the Cox proportional hazards models indicate that patients in the early stage of AD have a higher likelihood of receiving their first clinical evaluation sooner after the onset of initial symptoms. They are also more likely to receive a final diagnosis sooner after the initial evaluation, when controlling for all other covariates. However, this effect is not consistent across all model specifications (Supplemental Table 4).
Monitoring and Diagnostic Tests a

aData included in this table were obtained from the physician-completed patient record form.
bPatients are stratified by their disease severity at diagnosis. Oversample patients were not included in these data.
cPatients are stratified by their disease severity at the time of survey completion. An oversample of patients with a biomarker-confirmed diagnosis of MCI due to AD was included in these analyses to boost the number of patients with milder disease and to reduce variability from individual patients.
dOnly those tests used in ≥5.0% of patients in the overall sample are listed in this table. Tests used in <5% of patients in the overall sample included FAQ, Florbetapir/Amyvid, ADAS-Cog, Lumipulse, FDG-PET, PrecivityAD®, fMRI, ADCS-ADL, Elecsys, CIBIC, Elecsys Amyloid Plasma Panel, neurofilament light blood test, CDR, ptau-181 CSF assay, AD8 Dementia Screening Interview, Flortaucipir/Tauvid, s-MoCA, SLUMS, ptau-181, CSF test (assay unknown), QUEST AD-Detect® Amyloid Beta 42/40 ratio, MRS, GPCOG, CAMDEX, computerized cognitive test battery, Araclon Biotech SL (Grifols) Elisa, other blood tests, DTA, Florbetaben/Neuraceq, CDR-SOB, Diadem SRL AlzoSure Predict U-p53AZ kit, Alzohis Noratest, other CSF assays, other MRI, Flutemetamol/Vizamyl, SPECT, and 123I MIBG cardiac scintigraphy.
AD, Alzheimer Disease; ADAS-Cog, Alzheimer Disease Assessment Scale–Cognitive Subscale; ADCS-ADL, Activities of Daily Living for Mild Cognitive Impairment; CAMDEX, Cambridge Mental Disorders of the Elderly Examination; CDR, Clinical Dementia Rating; CDR-SOB, Clinical Dementia Rating - Sum of Boxes; CIBIC, Clinician Interview-Based Impression of Change; CSF, cerebrospinal fluid; CT, computed tomography; DTI, diffusion tensor imaging tractography; EEG, electroencephalogram; FAQ, Functional Activities Questionnaire; FDG-PET, fluorodeoxyglucose-positron emission tomography; fMRI, functional magnetic resonance imaging; GPCOG, General Practitioner Assessment of Cognition; MAD, mild Alzheimer disease dementia; MCI, mild cognitive impairment; MEG, magnetoencephalogram; MIBG, metaiodobenzylguanidine; MMSE, Mini Mental State Exam; MoAD, moderate Alzheimer disease dementia; MoCA, Montreal Cognitive Assessment; MRI, magnetic resonance imaging; MRS, magnetic resonance spectroscopy; PET, positron emission tomography; ptau-181, Simoa® phospho-Tau 181; SevAD, severe Alzheimer disease dementia; SLUMS, Saint Louis Mental Status Exam; s-MoCA, Short Montreal Cognitive Assessment; SPECT, single-photon emission computed tomography.
In most patients with an AD diagnosis, disease progression was monitored using feedback from the patient and their family (87.6% of the overall sample at the time of data capture; Table 5). Several other cognitive tests were used to monitor disease progression, including the Clock Draw test (20.4%), the Montreal Cognitive Assessment (MoCA; 17.0%), and the Mini-Cog test (9.3%). Laboratory tests such as PrecivityAD® and the Elecsys® Amyloid Plasma Panel were infrequently used to monitor patients (3.2% and 1.7% of the overall sample, respectively). Imaging was not typically used for monitoring, with CT scans and FDG-PET used in 6.7% and 3.3% of the overall sample, respectively, although volumetric and diffusion MRI were used in 20.4% and 8.4% of patients. Physicians reported that 30.6% of the overall sample did not undergo any monitoring tests. The percentage of patients who did not receive monitoring tests was 33.6% in patients with MCI due to AD, 31.9% in patients with MAD, 27.9% in patients with MoAD, and 30.4% in patients with SevAD.
Some patients included in this study completed a survey describing their feelings and experiences with AD. Patients in all disease severity groups reported problems with memory, sleep, and mood (Supplemental Table 5).
Discussion
This study demonstrates that delays may be present throughout the AD diagnostic journey. On average, 31.4 weeks elapsed between symptom onset and initial evaluation, while an additional 14.2 weeks elapsed between the first evaluation and diagnosis. The maximum time from symptom onset to first evaluation was 356.4 weeks (6.8 years), while the maximum time from first evaluation to diagnosis was 305.3 weeks (5.9 years). These findings are similar to those of previous studies that also demonstrated that AD and dementia may be subject to delayed diagnosis.7,8,20 Underdiagnosis and diagnostic delays are common in patients with AD.7,8 However, it is important to diagnose patients in the early stages of AD, when individuals may be eligible for disease-modifying therapies and when therapeutic and lifestyle interventions may have the most impact on cognitive decline and functioning.2,4,6,9,21 Individuals who are diagnosed in earlier dementia stages are less likely to report engaging in potentially unsafe activities, such as driving, preparing hot meals, and managing medications. 22 Individuals with milder AD may also have lower AD-related health care resource utilization compared with those with more severe AD, demonstrating the impact of early diagnosis and access to treatment on health care burden. 3
In this study, patients with MCI due to AD reported some comorbidities and mild symptoms, while patients with severe disease reported several comorbidities including mental health-related concerns. This reinforces the importance of prompt treatment in patients with early stages of AD. Not only are patients in early disease stages eligible for disease-modifying therapies, but treatment at this stage may also help to slow the progression of symptoms.
In this study, most patients saw a PCP or neurologist at their initial evaluation, demonstrating the key role of these health care providers in the diagnosis of AD. Notably, PCPs made few diagnoses in patients with MCI due to AD, highlighting a diagnostic gap within this provider type. This suggests that increased education and involvement of PCPs in the AD diagnostic journey, especially in early disease stages, could lead to increased diagnosis rates, more effective referrals, and shorter diagnostic journeys.20,23 Awareness of the signs and symptoms of the early stages of AD should be emphasized in educational efforts. This may help patients and providers to understand and differentiate between age-related and AD-related memory problems and help patients access critical treatments early in the disease process.7,20 Additionally, the potential emergence of blood-based biomarker tests for clinical use may reduce the need for referrals and specialized expertise in early detection among PCPs, thus reducing diagnostic delays.2,4 Finally, clinical practice guidelines should be established to help health care providers evaluate disease progression and implement follow-ups and referrals. 23
Patients in this study reported that delays in scheduling an initial evaluation were frequently due to thinking memory problems were a normal part of aging, concerns around perception from others, not wanting to face a serious diagnosis, and the potential for a loss of independence. Physicians reported that appointment availability was an important contributor to diagnostic delays. Diagnostic barriers reported in the literature, including time constraints, limited specialist availability, and lack of confidence from non-specialists in their diagnostic capabilities, are in agreement with those identified in this study.20,24,25 Insurance coverage and reimbursement issues have also been reported as significant barriers.20,24 These findings call for patient support programs to better understand AD dementia, overcome emotional burden, and support timely diagnosis.
The data from this study indicate that the number of patient comorbidities may be associated with delays in initial evaluation and diagnosis. Patients with a greater number of comorbidities may be unable to dedicate attention, money, or resources to seeking medical care for memory-related issues. Thus, the disease burden associated with certain comorbidities may hinder the diagnostic process. During our analysis, findings from the Cox proportional hazards model were not consistent. Our results suggest that patients in the early stage of AD have a higher likelihood of receiving both an initial evaluation and a final diagnosis sooner than those in later stages of the disease. However, these conclusions are limited by the small sample sizes and the nature of survey-based data. Therefore, larger studies will be necessary to confirm these findings.
The data from this study reaffirm that functional abilities may be preserved by maintaining a patient in earlier stages of AD. For example, 90.3% of patients with SevAD had severe difficulties with managing finances while mild, moderate, and severe difficulties were reported for 80.6%, 12.9%, and 6.5% of patients with MCI due to AD, respectively. In this study, the severity of multiple symptoms (eg, loss of short-term memory, difficulties in recall of names/words, and others listed in Table 2) was significantly associated with disease severity; however, it is difficult to draw firm conclusions about the data due to the small sample number. Patients self-reported the functional and emotional burden of their disease, including problems with sleeping, remembering names, and misplacing objects. These results demonstrate the importance of diagnosis and preservation of functional abilities in the early disease stages, when maximal benefit may be seen from treatment and lifestyle changes.4,26
Historically, achieving a diagnosis of AD was limited by access to appropriate cognitive testing and limited tools for biomarker confirmation. However, the landscape is changing rapidly. There is a growing infrastructure for formal cognitive testing, including the potential for digital cognitive testing. In addition, the barriers to achieving a biomarker-confirmed diagnosis seem to be diminishing. Amyloid-PET imaging27,28 is now covered by the Centers for Medicare and Medicaid Services. 29 The future of blood-based biomarkers (eg, plasma phosphorylated tau30-32) to confirm an AD diagnosis looks promising. There is also growing interest in other non-invasive tools, such as retinal imaging and other digital biomarkers.
Moreover, as more AD diagnoses are made, there will be growing interest in developing new methods to monitor disease progression beyond cognitive assessments (eg, blood-based biomarkers or imaging tools).
Several methodological details limit the generalizability of these findings. First, the DSP is based on a pseudo-random sample of physicians and patients. While minimal inclusion criteria governed the selection of participating physicians, participation was influenced by their willingness to complete the survey. The physician sample may not fully represent the proportion of PCPs and specialists treating patients with AD, and the data will likely be influenced by the relative number of each physician type recruited into the survey. Participating patients may not reflect the general AD population, since the DSP only includes patients who are consulting with their physician; therefore, patients who consult more frequently were more likely to be included in the study. Further, to minimize selection bias, physicians were asked to provide data for a consecutive series of eligible patients, thus ensuring that all patients had an equal chance of being included. Patient eligibility was based on the judgement of the responding physician and not on a formal diagnostic checklist; however, it is representative of the physician’s real-world classification of their patients. This selection bias might explain the higher likelihood of initial evaluation and diagnosis among patients with early-stage AD, as individuals who remain undiagnosed or who were not evaluated may not be represented in the sample. Recall bias, a common survey limitation, might also have affected physician and patient responses. However, physicians were able to refer to patients’ records while completing the patient record form, thus minimizing the possibility of recall bias. Furthermore, data were collected at the time of each patient’s appointment to reduce the likelihood of recall bias where the opinion of the physician was required.
Several sample size limitations exist. First, a small number of patients with SevAD at diagnosis was available, as most patients present to their health care provider before the disease progresses to this stage. MMSE tests are also less likely to be performed on patients with severe disease; therefore, these data should be interpreted with caution. Second, the sample size obtained from voluntarily completed surveys is relatively small, particularly in patients with SevAD, likely biasing some of the results; therefore, caution is required when interpreting these results. Finally, the cross-sectional nature of this study allows for identification of association but not causation.
Conclusions
Patients with AD face a complicated diagnostic journey. Diagnostic delays may be due to factors such as time constraints and availability of appointments. It is important to strive for early diagnosis of AD, as currently available disease-modifying therapies are indicated for patients with earlier disease stages. These therapies may help slow disease progression and preserve patient functional abilities. PCPs and neurologists play a key role in this process and further work should be done to provide improved tools for them to better distinguish between early AD and normal aging. Recent advancements in blood-based biomarker tests may help reduce the need for referrals and speed up the diagnostic process.
Supplemental Material
Supplemental Material - A Real-World Analysis of the Clinical Journey, Diagnosis, and Monitoring Patterns of Patients With Alzheimer Disease by Stage in the United States
Supplemental Material for A Real-World Analysis of the Clinical Journey, Diagnosis, and Monitoring Patterns of Patients With Alzheimer Disease by Stage in the United States by Elnara Fazio-Eynullayeva, MA, Paul Mystkowski, MD, Lei Lv, PhD, MPH, Abdalla Aly, PhD, Sarah Cotton, MA, Nikisha Grant, MSc, Will Johnson, BSc, Soeren Mattke, MD in American Journal of Alzheimer's Disease & Other Dementias®
Footnotes
Acknowledgements
The authors thank Adele Musicant, PhD, of Precision AQ (Bethesda, Maryland) for providing writing and editing assistance in accordance with Good Publication Practice (GPP 2022) guidelines. This assistance was financially supported by Novo Nordisk, Inc.
Ethical Considerations
Data collection was undertaken in line with European Pharmaceutical Marketing Research Association guidelines (EPHMRA. Code of conduct. September 2024. Accessed January 21, 2025. ![]() ) and, as such, it did not require ethics committee approval. Each survey was performed in full accordance with relevant legislation at the time of data collection. No patients or physicians can be directly identified in the data. Data were aggregated before being shared with the subscriber and/or for publication. This research was submitted to the Pearl Institutional Review Board, study protocol number #22-ADRW-172/3.
) and, as such, it did not require ethics committee approval. Each survey was performed in full accordance with relevant legislation at the time of data collection. No patients or physicians can be directly identified in the data. Data were aggregated before being shared with the subscriber and/or for publication. This research was submitted to the Pearl Institutional Review Board, study protocol number #22-ADRW-172/3.
Consent to Participate
All participants provided informed consent to take part in the Adelphi DSP survey.
Consent for Publication
All participant data were aggregated and deidentified before receipt by Adelphi Real World.
Author Contributions
Funding
This study was funded by Novo Nordisk Inc. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Data collection was undertaken by Adelphi Real World as part of an independent survey, entitled the Adelphi Real World AD DSP. The DSP is a wholly owned Adelphi Real World product. Novo Nordisk is one of multiple subscribers to the DSP. Publication of survey results was not contingent on the subscriber’s approval or censorship of the publication.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Data Availability Statement
All data (ie, methodology, materials, data, and data analysis) that support the findings of this survey are the intellectual property of Adelphi Real World. All requests for access should be addressed directly to Sarah Cotton at
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.