Abstract
Introduction
Neurological disorder including: Alzheimer’s disease (AD), Parkinson’s disease (PD), stroke, major depressive disorder (MDD), epilepsy, etc., is a major and increasing global health challenge, which accounts for a substantial portion of the disease burden worldwide. It is the leading cause of disability and the second leading cause of death. 1 In the past 30 years, the absolute numbers of deaths and people with disabilities owing to neurological diseases have risen substantially, particularly in low-income and middle-income countries, and further increases are expected globally as a result of population growth and ageing. 2 However, medicines developed for treating brain diseases have not met the clinical expectations.3-6 Encouragingly, some physical therapy modalities for brain diseases have been established in recent a decade, such as: electroconvulsive therapy, transcranial magnetic stimulation, vagus nerve stimulation, deep brain stimulation,7-9 and low-intensity pulsed ultrasound (LIPUS). 10
LIPUS as a non-invasive treatment for clinical diseases has been developed rapidly in recent years. 11 LIPUS can exert therapeutic effects in animal models of several neurological disorders, including ischemic brain injury,12,13 PD, 14 AD,15-17 and epilepsy.18,19 This method can be carried out by using a low-intensity ultrasound with a pulsed wave output that allows the transmission of acoustic energy to the target tissue while maintaining minimal thermal effects. 20 Recently, a typical LIPUS with frequency: 1.5 MHz, spatial average temporal average(SATA) intensity: less than 1 W/cm2; and pulse duty cycle: 20%, has been used to treat depression, because it can be delivered to cells and tissues through the medium in the form of acoustic waves, and alleviate depression-like behaviors in mice. 21 Although these ultrasounds with different low-intensity have been established for clinical application in the future, the most noteworthy issue is the safety of LIPUS on the brains.
Previous studies have shown that in vitro LIPUS treatments protect cells by regulating antioxidant proteins and attenuating oxidative stress through potassium channels and stretch-activated ion channel-mediated downstream pathways. 22 The use of lipid-coated GDNF microspheres in the striatum in combination with LIPUS stimulation can increase GDNF levels, 23 and improve the levels of striatal dopamine and nigrostriatal tyrosine hydroxylase. It also promotes the neurodegenerative cascade responses in the substantia nigra and striata pathway, thereby reducing neuronal cell death. 24 In addition, LIPUS could directly increase BDNF expression. 25
In this study, we investigated whether acute and chronic LIPUS treatments affect the brain advanced functions for controlling motion, reproducibility, food and drink, mood, cognition and social interaction in healthy adult male and female mice. The results suggest that although LIPUS itself does not affect brain functions, physical restraint for LIPUS therapy induces struggle-like behaviors and impair short-time memory. Hence, a suitable and unbound helmet is proposed to apply in ultrasound therapy.
Materials and Methods
Subjects
Male and female C57BL/6 mice, 9 weeks of age, were purchased from Zhejiang Charles River Company (Zhejiang, China). The mice were maintained at a constant temperature (22 ± 2°C) and a 12-hour light/dark cycle, and allowed free access to food and water. All mice were acclimated for one week before the experiment. Food and water were given freely except for the periods of restraint and sonication and for conducting behavioral tests.
Experimental Apparatus and Equipment
Low-intensity pulsed ultrasound generator: SonaCell (Hadaca Ltd., Canada). LIPUS parameters-frequency: 1.5 MHz; pulse: 1.0 kHz; SATA intensity: 25 mW/cm2; and pulse duty cycle: 20%.
Grouping of Mice
Adult male and female wild-type mice (N = 10 per group) were subdivided into 3 groups in both acute and chronic studies, including: control group (Con), physical restraint group with 50-mL centrifuge tube for fixing mouse head (Restraint), and physical restraint combined with LIPUS-treated group in the centrifuge tube (Restraint + LIPUS), respectively. During operation, the mice were put into these restraint tube and their heads were exposed to the pre-opening position with a triangle angle at 15∼22o. LIPUS was applied to mouse head in acute test for 20 minutes, and in chronic experiment for consecutive 10 days, respectively.
Behavioral Evaluation
Body weight for assessing appetite
These mice were weighed and monitored daily for drinking and feeding.
Accelerating rotarod test (ART, assessing movement ability)
The mice were placed on the turnbuckle at 5 r/min for 10 seconds to acclimatize, and after 10 minutes, the mice were placed on the rotarod and the rotarod was accelerated uniformly at 5 r/min to 30 r/min for 5 minutes. The total time the mice were on the rotarod was recorded and analyzed to reflect mouse movement balance ability. 26
Three-chamber social interaction test (TCSI, assessing sociality ability)
Mice were placed in the 3 chamber for 10 min for familiarization, after 10 min, an unfamiliar mouse was placed on the left chamber and the familiar mouse was placed on the right chamber and the subject mice explored freely for 10 min. The spent time of the mice exploring the left, right, and middle chambers was recorded and analyzed to reflect social function. 27
Mating test (MT, assessing reproducibility)
The room environment was quiet. These male mice were placed in transparent experimental cages for 5 min before being placed in female mice of the same month of age C57BL/6J and observed for 30 min. The incubation period and several riding behaviors were recorded. 28
Nest building test (NBT, assessing maternal behavior)
Tissues weres evenly distributed across the bottom of the cage opposite the mouth of the water bottle, and the nests were scored after 24 hours. 29 Elevated plus maze test (EPM, assessing anxiety-like behaviors)
Mice were put into head-first the closed arm of the elevated cross maze at the end of the closed arm and explored freely for 10 minutes. Recording the latency and number of these mice entered the open arm and the total time in the open arm. Only when the mouse limbs were fully in the open arm, started counting the time in the open arm, and ended counting the time when the mouse limbs were fully out of the open arm. This experiment reflects the anxiety state of the mice. 30
Tail suspension test (TST, assessing depression-like behaviors)
The mice were suspended from the end of the tail 1 cm for 10 minutes. The total time the mice remained immobile was recorded. If the mice stop struggling, their bodies remain in a vertical overhang, motionless, these behaviors are considered to be the immobile depressive state. 31
Inhibitory avoidance test (IAT, assessing short-time memory behaviors)
On the first day of electroshock: mice were put into the light box of the electroshock dark and light box (the dark box had a shock bar), and the experiment was conducted for 5 min. After the mice entered the dark box, the door was closed and the electroshock (.5 mA, 5 s) was applied, and the mice were taken out at the end of the electroshock. Test on the second day: mice were placed into the apparatus (the dark box had a shock bar) for 5 minutes of the experiment, and the latency period of the mice entering the dark box was recorded. 32
After each behavioral test, feces were cleaned up and lab equipment was cleaned with 75% alcohol solutions.
Statistical Analysis
All data were tested for normality with the Shapiro-Wilk test. When data were normally distributed, the statistical significance of differences was assessed with the one-way or two-way ANOVA, analyzed by Dunnett's multiple comparisons test, and Bonferroni's correction. When the data were not normally distributed, the statistical significance of differences was assessed on the basis of P values with the Kruskal-Wallis test. P < .05 was considered statistically significant. Analyses were performed in GraphPad Prism 9 software.
Results
In this study, wild-type male and female mice were respectively grouped as 3 group: Con, Restraint, and Restraint + LIPUS; and their brain functions-related behaviors were assessed by ART, EPM, MT or NBT, IAT, TST and TCSI, respectively, as showed in Figure 1. Schematic diagram of acute LIPUS experiment. (A) Acute LIPUS exposure schedule and behavioural test methods. (B) Con group; (C) Restraint group; (D) Restraint + LIPUS group. (E) Ultrasound transducer. (F) Ultrasonic operation diagram.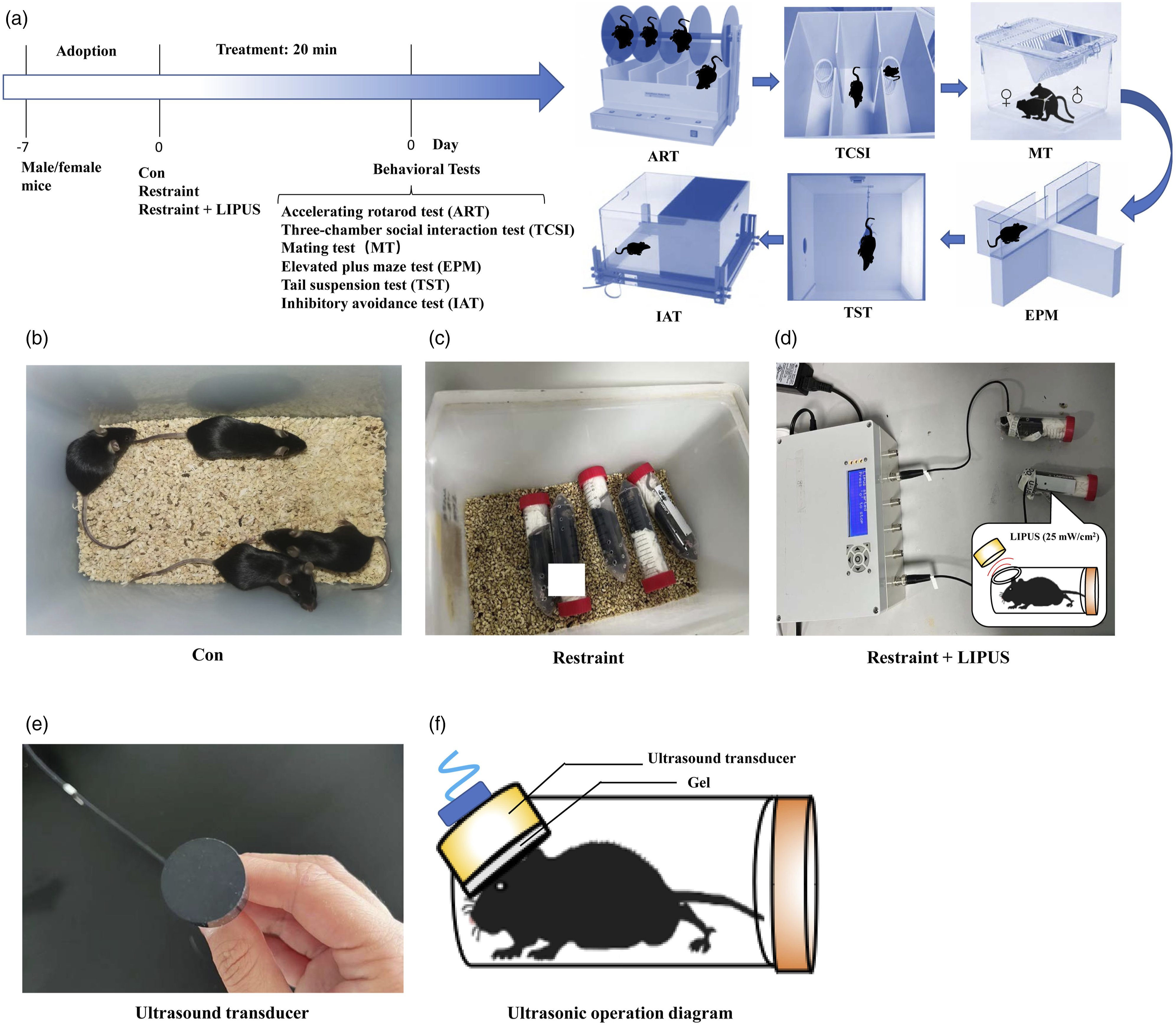
Acute safety tests (Male mice)
In ART experiment, after 20 minutes of acute LIPUs stimulation, both the Restraint group and Restraint + LIPUS group didn’t show a statistical significant difference in staying time on the rotarod compared to Control group (F2,27 = 3.251, P = .054) (shown in Figure 2(a)). Assessment the abilities of move, social interaction, reproducibility, short-time memory and anxiety-like and struggle behaviors in male mice after acute LIPUS exposure. (A) Staying time on the rotarod of ART. (B, C, D) Time spent in the middle, left and right chamber in TCSI. Data analyzed by one-way ANOVA with Dunnett's multiple comparisons test. (E, F) Mount latency and mount frequency of MT. (G, H, I) Spent time in open arms of EPM, the latency of entering the open arm of EPM and open arm entries in EPM. Data analyzed by one-way ANOVA with Dunnett's multiple comparisons test. (J) Immobility time in TST. (K). Latency into dark box in IAT. Data analyzed by the Kruskal-Wallis test. ART: accelerating rotarod test; EPM: elevated plus maze test; MT: mating test; IAT: Inhibitory avoidance test; TCSI: three-chamber social interaction test; TST: tail suspension test. N = 10. Data represent mean ± S.D. *P < .05, **P < .01, ****P < .0001 NS P > .05. No statistical significance.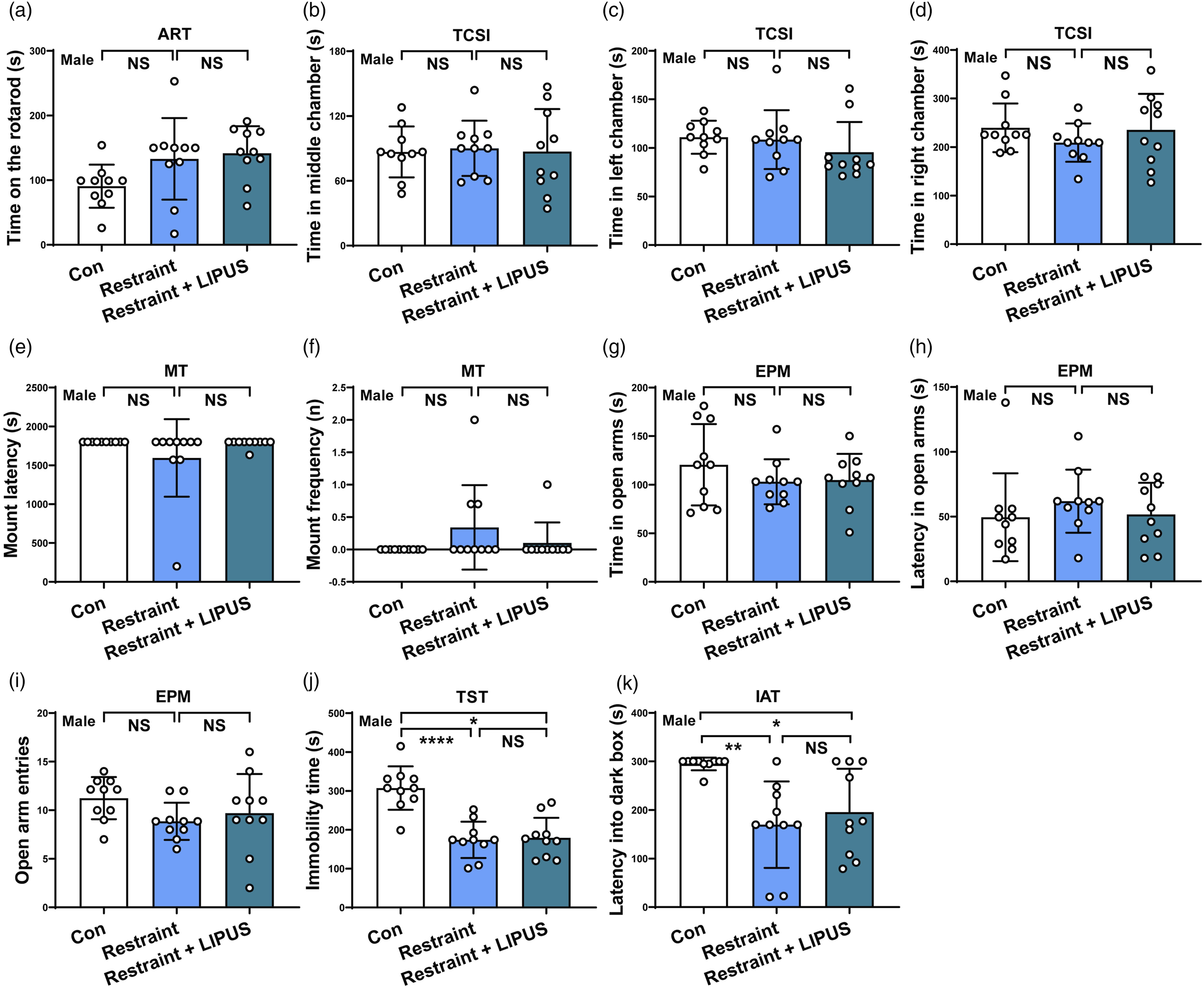
In TCSI study, there also were no significant difference in staying time in the middle chamber (F2,27 = .0359, P = .965), the left chamber (F2,27 = .854, P = .437) and the right chamber (F2,27 = .955, P = .397) among these 3 groups (shown in Figure 2(b)–(d)).
In MT experiment, no significant difference could be observed in the mount latency (P = .152) and mount frequency (P = .121) among these 3 groups (shown in Figure 2(e) and (f)).
In EPM study, these 3 groups’ mice did not show a significant difference in staying time in open arms (F2,27 = .924, P = .409), time latency in open arms (F2,27 = .5591, P = .578) and entries of open arms (F2,27 = 1.780) (shown in Figure 2(g)–(i)).
Above data indicated that acute LIPUS exposure does not affect the abilities of move, social interaction, mating and anxiety-like mood.
Notably, in TST study, compared with Control, both Restraint group and Restraint + LIPUS group showed a marked decrease in immobility time (P < .0001); however, there were no significant difference in immobility time between these 2 groups (P = .974) (shown in Figure 2(j)). This result demonstrated that LIPUS itself does not induce struggle-like behaviors; however, physical restraint for LIPUS therapy certainly results in struggle-like phenomenon.
Similarly, in IAT experiment, compared with Control, there was a significant decrease in latency time into the dark box in both Restraint group (P = .003) and Restraint +LIPUS group (P = .040), but no significant difference was observed between these 2 groups (P > .9999) (shown in Figure 2(k)). This finding indicated that LIPUS itself does not induce short-time memory deficits; however, physical restraint for LIPUS therapy indeed impairs memory.
Acute Safety Tests (Female Mice)
In ART experiment, after 20 minutes of acute LIPUS stimulation, both the Restraint group and Restraint + LIPUS group didn’t show a statistical significant difference in staying time on the rotarod compared to Control group (F2,27 = 2.127, P = .1387) (shown in Figure 3(a)). Assessment the abilities of move, social interaction, reproducibility, short-time memory and anxiety-like and struggle behaviors in female mice after acute LIPUS exposure. (A) Staying time on the rotarod of ART. (B, C, D) Time spent in the middle, left and right chamber in TCSI. Data analyzed by one-way ANOVA with Dunnett's multiple comparisons test. (E, F) Mount latency and mount frequency of MT. (G, H, I) Spent time in open arms of EPM, the latency of entering the open arm of EPM and open arm entries in EPM. Data analyzed by one-way ANOVA with Dunnett's multiple comparisons test. (J) Immobility time in TST. (K). Latency into dark box in IAT. Data analyzed by the Kruskal-Wallis test. ART: accelerating rotarod test; EPM: elevated plus maze test; MT: mating test; IAT: Inhibitory avoidance test; TCSI: three-chamber social interaction test; TST: tail suspension test. N = 10. Data represent mean ± S.D. *P < .05, **P < .01, ****P < .0001 NS P > .05. No statistical significance.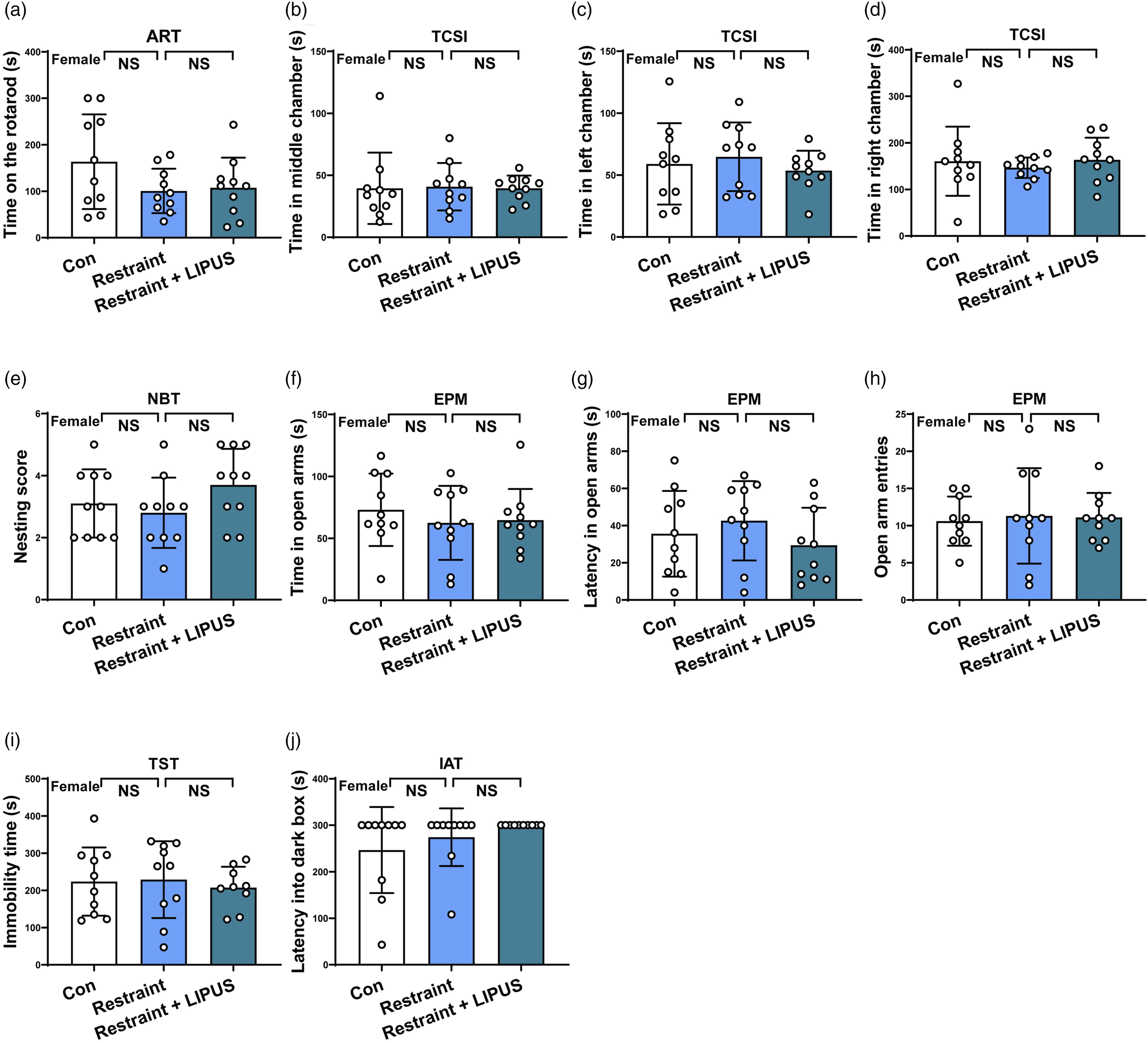
In TCSI study, there also were no significant difference in staying time in the middle chamber (F2,27 = .01200, P = .9881), the left chamber (F2,27 = .4441, P = .6460) and the right chamber (F2,27 = .2946, P = .7472) among these 3 groups (shown in Figure 3(b)–(d)).
In NBT experiment, no significant difference could be observed in the nesting score (F2,27 = 1.639, P = .2130) among these 3 groups (shown in Figure 3(e)).
In EPM study, these 3 groups’ mice did not show a significant difference in staying time in open arms (F2,27 = .3958, P = .6770), time latency in open arms (F2,27 = .9394, P = .4033) and entries of open arms (F2,27 = .06208, P = .9399) (shown in Figure 3(f)–(h)).
In TST study, compared with Control, both Restraint group and Restraint + LIPUS group didn’t show a significant difference in immobility time (F2,27 = .1714, P = .8434) (shown in Figure 3(i)).
Similarly, in IAT experiment, compared with Control, there was no significant difference in latency time into the dark box in both Restraint group and Restraint + LIPUS group (F2,27 = 1.733, P = .1958) (shown in Figure 3(j)). This finding indicated that LIPUS itself does not induce short-time memory deficits.
Above data indicated that acute LIPUS exposure does not affect the abilities of move, social interaction, maternal behaviors, anxiety-like mood, depression-like behaviors and short-time memory behaviors.
Chronic Safety Experiments (Male Mice)
After 20 minutes per day of LIPUS exposure for consecutive 10 days, the body weight of Control mice showed a significant and gradual increase; while both Restraint group and Restraint + LIPUS group had no this trend with group factor: F (9, 270) = .723, P = .688; time factor: F (2, 270) = 106.1, P < .0001, interaction: F (18, 270) = .817, P = .680) (shown in Figure 4(a)). This result indicated that physical restraint impairs appetite. Assessment body weight and the abilities of move, social interaction, reproducibility short-time memory and anxiety-like and struggle behaviors in male mice after chronic LIPUS exposure. (A) Chronic LIPUS experiment Treatment program and illustration of ultrasound apparatus and mice weight. Data analyzed by two-way ANOVA with Bonferroni's correction. (B) Staying time on the rotarod in ART. (C, D, E) Time spent in the middle, left and right chambers in TCSI. Data analyzed by one-way ANOVA with Dunnett's multiple comparisons test. (F, G) Mount latency and mount frequency in MT. Data analyzed by the Kruskal-Wallis test. (H, I, J) Spent time in the open arms of EPM, the latency in the open arms of EPM and the open arm entries in EPM. Data analyzed by one-way ANOVA with Dunnett's multiple comparisons test. (K) Immobility time in TST. (L) Latency entering into the dark box in IAT. Data analyzed by the Kruskal-Wallis test. (A) ART: accelerating rotarod test; EPM: elevated plus maze test; MT: mating test; IAT: Inhibitory avoidance test; TCSI: three-chamber social interaction test; TST: tail suspension test. N = 10. Data represent mean ± S.D. **P < .01 NS P > .05. No statistical significance. N = 10. Data represent mean ± S.D. *P < .05, **P < .01; NS, P > .05. No statistical significance.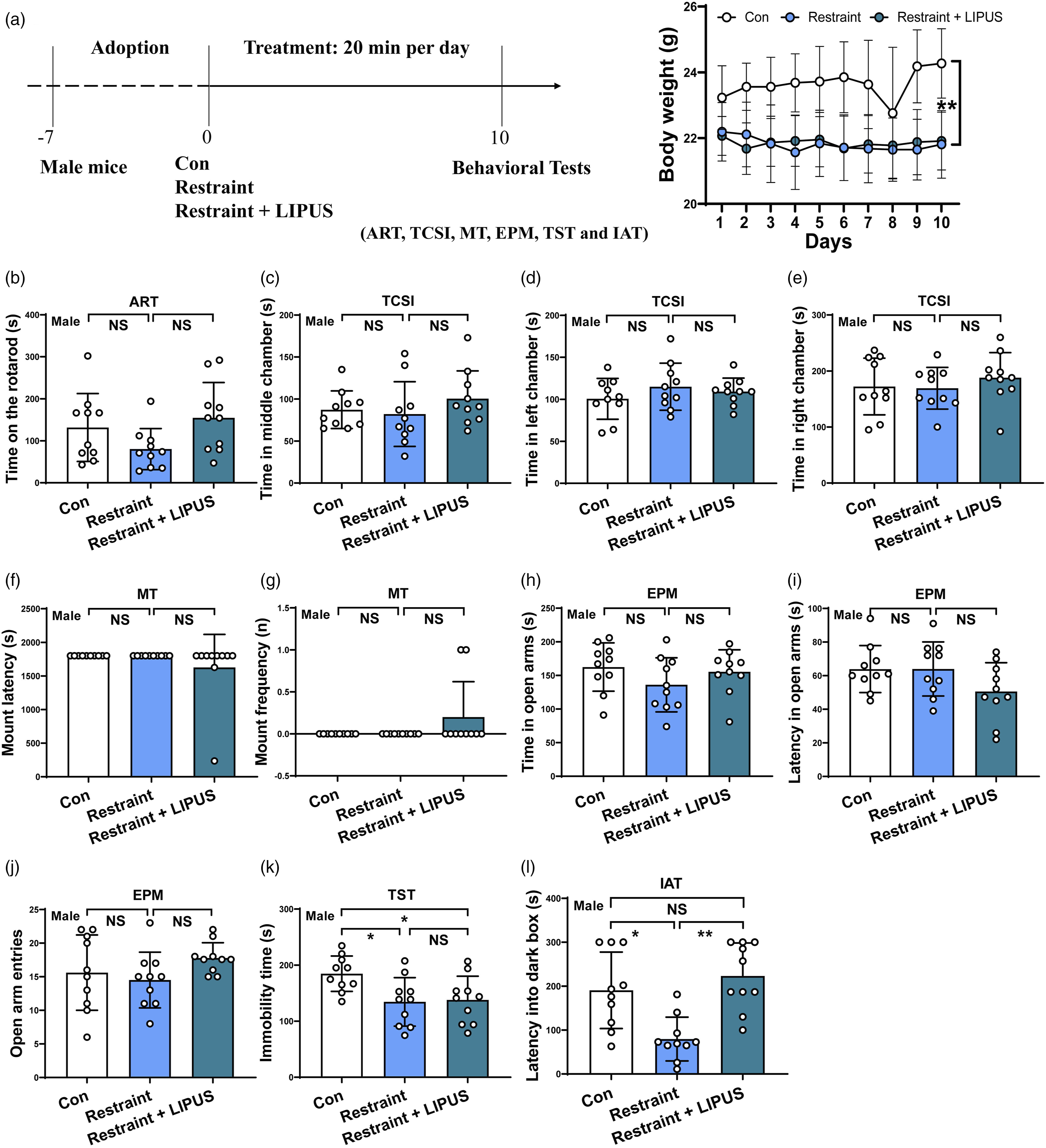
In ART experiment, after 20 minutes LIPUs stimulation for consecutive 10 days, both Restraint group and Restraint + LIPUS group didn’t show a significant increase in staying time on the rotarod compared to the Control group (P = .100) (shown in Figure 4(b)).
In TCSI study, there were no significant difference in spent time in the middle chamber (F2,27 = .877, P = .428), the left chamber (F2,27 = .965, P = .394) and the right chamber (F2,27 = .512, P = .605) among these 3 groups (shown in Figure 4(c)–(e)).
In MT experiment, no significant difference was observed in the mount latency (P = .126) and mount frequency (P = .126) among these 3 groups (shown in Figure 4(f) and (g)).
In EPM study, there was no difference in staying time in open arms (F2,27 = 1.41, P = .262), time latency in open arms (F2,27 = 2.389, P = .111) and entries of open arms (F2,27 = 1.543, P = .232) among these 3 groups’ mice (shown in Figure 4(h)–(j)).
Above results indicated that chronic LIPUS exposure does not affect the abilities of move, social interaction, mating and anxiety-like behaviors.
In TST experiment, compared with Control group, both Restraint group and Restraint + LIPUS group showed a significant decrease in the immobility time (P = .022; P = .034; respectively); however, there was no significant difference in time between these 2 groups (P = .980) (shown in Figure 4(k)). This result of chronic LIPUS was similar to the data of acute LIPUS.
In IAT study, compared to Restraint group, a significant increase in latency into the dark box were shown in both Control group and Restraint + LIPUS group (P = .005 P = .0001, respectively), but no significant difference was observed between Control group and Restraint + LIPUS group (P = .485) (shown in Figure 4(l)). This finding indicated that chronic LIPUS itself may not induce short-time memory deficits; however, physical restraint for LIPUS therapy certainly impairs memory. In addition, it is possible that physical restraint impairs memory, and LIPUS treatment compensates for the impairment. Hence, an unbound helmet for ultrasound therapy is proposed in clinical application (shown in Figure 5(l)). Assessment body weight and the abilities of move, social interaction, reproducibility short-time memory and anxiety-like and struggle behaviors in female mice after chronic LIPUS exposure. (A) Chronic LIPUS experiment Treatment program and illustration of ultrasound apparatus and mice weight. Data analyzed by two-way ANOVA with Bonferroni's correction. (B) Staying time on the rotarod in ART. (C, D, E) Time spent in the middle, left and right chambers in TCSI. (F) Nesting score in NBT. Data analyzed by one-way ANOVA with Dunnett's multiple comparisons test. (G, H, I,) Spent time in the open arms of EPM, the latency in the open arms of EPM and the open arm entries in EPM. Data analyzed by one-way ANOVA with Dunnett's multiple comparisons test. (J) Immobility time in TST. (K) Latency entering into the dark box in IAT. Data analyzed by the Kruskal-Wallis test. (L) Illustration of ultrasound apparatus for mice and human. ART: accelerating rotarod test; EPM: elevated plus maze test; MT: mating test; IAT: Inhibitory avoidance test; TCSI: three-chamber social interaction test; TST: tail suspension test. N = 10. Data represent mean ± S.D. **P < .01 NS P > .05. No statistical significance. N = 10. Data represent mean ± S.D. *P < .05, **P < .01; NS, P > .05. No statistical significance.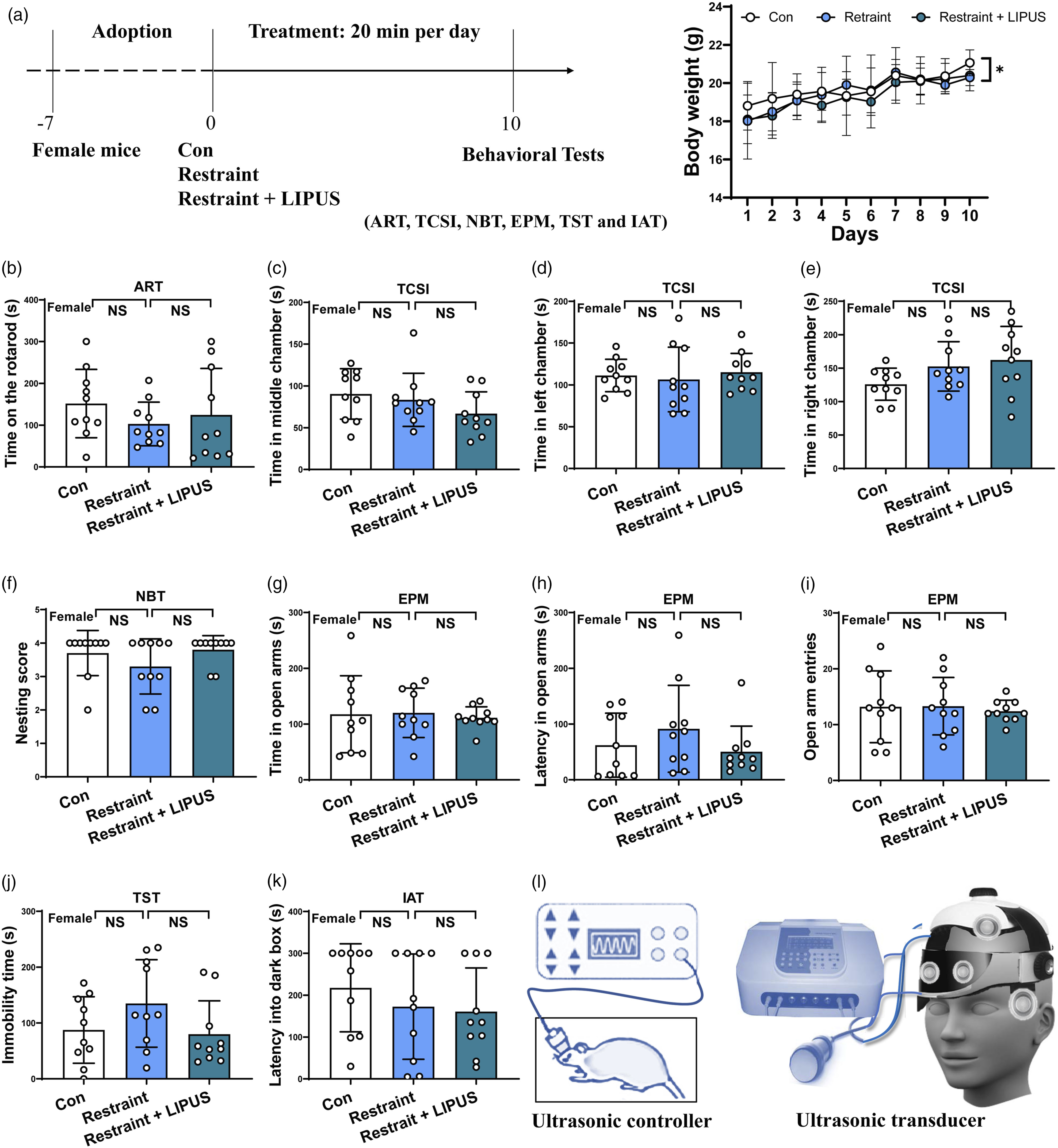
Chronic Safety Experiments (Female Mice)
After 20 minutes per day of LIPUS exposure for consecutive 10 days, the body weight of Control mice showed a significant and gradual increase; while both Restraint group and Restraint + LIPUS group had no this trend with group factor: F (2, 270) = 3.688, P = .0263; time factor: F (9, 270) = 12.50, P < .0001, interaction: F (18, 270) = .5292, P = .9432) (shown in Figure 5(a)).
In ART experiment, after 20 minutes LIPUs stimulation for consecutive 10 days, both Restraint group and Restraint + LIPUS group didn’t show a significant increase in staying time on the rotarod compared to the Control group (F2,27 = .8248, P = .4491) (shown in Figure 5(b)).
In TCSI study, there were no significant difference in spent time in the middle chamber (F2,27 = 1.686, P = .2041), the left chamber (F2,27 = .2360, P = .7914) and the right chamber (F2,27 = 2.382, P = .1115) among these 3 groups (shown in Figure 5(c)–(e)).
In NBT experiment, no significant difference was observed in the nesting score (F2,27 = 1.602, P = .2201) among these 3 groups (shown in Figure 5(f)).
In EPM study, there was no difference in staying time in open arms (F2,27 = .08891, P = .9152), time latency in open arms (F2,27 = 1.178, P = .3231) and entries of open arms (F2,27 = .1020, P = .9034) among these 3 groups’ mice (shown in Figure 5(g)–(i)).
In TST experiment, compared with Control group, both Restraint group and Restraint + LIPUS group didn’t show a significant difference in the immobility time (F2,27 = 2.013, P = .1531) (shown in Figure 5(j)).
In IAT study, compared to Restraint group, there was no difference in latency into the dark box were shown in both Control group and Restraint + LIPUS group (F2,27 = .7142, P = .4986) (shown in Figure 5(k)).
Discussion
In this study, LIPUS itself do not affect brain functions; however, physical restraint for LIPUS therapy impairs appetite, mood and memory. Hence, reduction in restraint by using an unbound helmet for ultrasound therapy should be considered for clinical application in the future (shown in Figure 5(l)).
Another concern for ultrasound treatments is that ultrasound parameters may be different in accordance with the different brain diseases. For example, LIPUS at .154∼3.84 W/cm2, 200 kHz, and 20% duty cycle alleviates depression-like behavior in rats. 25 At 25 mW/cm2, 1.5 kHz, and 20% duty cycle, it has antidepressant effects in mice. 33 Ultrasound at .7-MPa peak, 1 MHz, and 10% duty cycle can reduce amyloid deposition and improve memory functions in Alzheimer’ s disease model mice.17,34,35 In the focal cerebral ischemia model mice, LIPUS at 86 mW/cm2, and 1.5 kHz have neuroprotective effects. 13 Pregnant mice exposed to LIPUS at 20 mW/cm2 and 20% duty cycle have not affected cognitive behaviors in the progeny mice 120 days after births. 36 Further, in the non-human primate-macaque, LIPUS at 11.6 W/cm2, 270 kHz, and 50% duty cycle, significantly influences decision formation. 37 Another study shows that LIPUS at 2.02 W/cm2 and 500 kHz not only improves electrophysiological activity and behaviors in a nonhuman primate model of epilepsy, but also suppresses the epileptiform activity of neurons in human epileptic slices. 38
Study Limitations
In the present study, we did not examine whether LIPUS at 25 mW/cm2, 1.5 kHz, and 20% duty cycle damages the blood-brain barrier (BBB). Although LIPUS has been found to open the blood-brain barrier (BBB), no serious clinical or imaging adverse events had been reported until now. 39
We did not detect the penetration ratio of LIPUS. It is known that intensity decay with 3.7 to 4.1-folds after transmitting LIPUS through human skull 40 considering that the average thickness of human skull is 1 cm 41 and the average mouse skull is 200 μm. 42
While our study demonstrated that LIPUS at 25, 50, and 100 mW/cm2 could penetrate the mouse brain using a needle hydrophone (Supplementary Figure 1), it is advisable to employ hydrophone scanning for more precise and accurate results. Given that physical restraint in mice impairs short-term memory and induces depression-like behaviors, there is a need for enhancing the techniques aimed at making a wearable ultrasound generator. This improvement could potentially enable the mice to move freely during LIPUS treatment in the future.
Considering its decay in the bone should be at 25∼50 mW/cm2 according to the safety intensity- 25 mW/cm2 presented in this study, the optimized ultrasound intensity for human clinical therapy still needs to be identified in the future. Notably, this predicted intensity is within the range (.72∼190 W/cm2) of USA Food and Drug Administration (FDA) requirements for safe ultrasound imaging. 25
Conclusions
Acute or chronic Low-Intensity Pulsed Ultrasound (LIPUS) itself may not directly affect brain functions; however, the physical restraint associated with LIPUS therapy can have a certain impact on brain-related behaviors, such as short-term memory deficits and struggle-like behaviors. Therefore, we propose that an unattached helmet with ultrasound at a range of 25 to 50 mW/cm2 should be considered for future clinical therapy. This helmet should be non-invasive, comfortable to wear, and allow for easy adjustment of the position of the LIPUS transducers
Supplemental Material
Supplemental Material - Neuropsychiatric Behavioral Assessments in Mice After Acute and Long-Term Treatments of Low-Intensity Pulsed Ultrasound
Supplemental Material for Neuropsychiatric Behavioral Assessments in Mice After Acute and Long-Term Treatments of Low-Intensity Pulsed Ultrasound by Ye Li, Yiqing Wu, Qi Luo, Xuanjie Ye, Jie Chen, Yuanlin Su, Ke Zhao, Xinmin Li, Jing Lin, Zhiqian Tong, Qi Wang, and Dongwu Xu in American Journal of Alzheimer’s Disease & Other Dementias®
Footnotes
Acknowledgments
We would like to thank all participants and colleagues.
Author Contributions
Conception and design: QW, ZQT, JC, XML, KZ, and JL; Acquisition, analysis, and interpretation of the data: YL, YQW, YLS, LQ; Drafting and writing: YL, ZQT, XJY; Final approval of the article: ZQT, DWX, YL,YQW.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research was supported by grants from the National Natural Science Foundation of China (82071214), Fund from Kangning Hospital (95021025) and Fund of Talent Launch Project of Oujiang Laboratory (OJQDSP2022011).
Ethical approval
Statement of EthicsAnimal manipulation was performed in accordance with the animal testing guide and approved by the Animal Ethics Committee of Wenzhou Medical University (WMU-10250).
Data Availability
The data used to support the findings of this study are included within the article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
