Abstract
In older adults with cognitive decline and epilepsy, diagnosing the etiology of cognitive decline is challenging. We identified 6 subjects enrolled in the Imaging Dementia-Evidence of Amyloid Imaging Scanning (IDEAS) study and nonlesional epilepsy. Three cognitive neurologists reviewed each case to determine the likelihood of underlying Alzheimer’s disease (AD) pathology. Their impressions were compared to amyloid PET findings. In 3 cases the impression was concordant with PET findings. In 2 cases “possibly suggestive,” the PET reduced diagnostic uncertainty, with 1 having a PET without elevated amyloid and the other PET with intermediate amyloid. In the remaining case with lack of reviewer concordance, the significance of PET with elevated amyloid remains uncertain. This case series highlights that in individuals with a history of epilepsy and cognitive decline, amyloid PET can be a useful tool in evaluating the etiology of cognitive decline when used in an appropriate context.
Significance
We identified 6 individuals with late onset epilepsy and cognitive decline. In 2 cases highly suggestive of AD, and in 1 not suggestive, the PET was concordant. In the 2 cases possibly suggestive of AD, amyloid PET reduced diagnostic uncertainty.
Introduction
The incidence of epilepsy has a bimodal distribution with the first peak occurring under the age of 1, and another prominent peak later in life after the age of 65.1,2 The etiologies behind this second peak include cerebrovascular disease, tumors, neurodegenerative diseases, and a significant proportion with no identifiable cause.2,3 An emerging hypothesis is that new-onset “unexplained” epilepsy may represent the symptomatic expression of an underlying neurodegenerative disease such as Alzheimer’s disease(AD).4-6 In the clinic, physicians caring for older patients with epilepsy face two common challenging clinical scenarios: patients with new-onset epilepsy and complaints of memory decline, and patients with longstanding epilepsy and complaints of memory decline. While the burden of seizures, the burden of epileptiform abnormalities, and drug side effects may explain cognitive symptoms in these patients, 7 it is often difficult to rule out underlying neurodegeneration. Identifying the driver of the memory complaints impacts management, prognosis, and counseling. In this case series of patients prospectively enrolled in the Imaging Dementia-Evidence of Amyloid Imaging Scanning (IDEAS) study 8 we explore the clinical utility of amyloid PET imaging in the evaluation of these patients with epilepsy.
Materials and Methods
Subjects
We included subjects who had a history of nonlesional epilepsy defined as the absence of cortical lesions on MRI (excluding atrophy patterns) with onset of epilepsy later in life (>40 years), and who were prospectively enrolled in the IDEAS study at Brigham and Women’s Hospital. Patients were evaluated either in a sub-specialty epilepsy or cognitive neurology clinic.
Inclusion criteria in the IDEAS study included: (1) Medicare as the primary health insurance (2) Age older than 65 (3) Diagnosis of mild cognitive impairment or dementia within 24 months verified by a dementia specialist (4) Cognitive complaint with objective impairment (5) Mild cognitive impairment or dementia etiology is uncertain after a comprehensive evaluation including cognitive testing, laboratory testing for toxic-metabolic causes, and structural neuroimaging (6) AD is a diagnostic consideration (7) Knowledge of amyloid PET status is expected to alter diagnosis and management.
Exclusion criteria for IDEAS: normal cognition, knowledge of amyloid status may cause significant psychological harm, amyloid status already known, enrollment in anti-amyloid therapeutic trial, cancer requiring active therapy, scan being ordered solely based on a family history of dementia or for nonmedical purposes, hip/pelvic fracture within the 12 months prior to enrollment, body weight exceeds PET scanner weight limit, life expectancy less than 24 months, residence in a skilled nursing facility (assisted living facility was not an exclusion criterion). We also excluded patients who had a history of alcohol abuse and severe traumatic brain injury.
Amyloid PET was performed with F-18 florbetaben (NeuraceqTM) within 30 days of enrollment. Scans were read locally by radiology/nuclear medicine physicians according to consensus practice guidelines and interpreted as “elevated”, “not elevated”, or “intermediate” for cerebral amyloid pathology. 9 The study was approved by the Mass General Brigham IRB P002690 and performed in accordance with its ethical standards as well as the declaration of Helsinki.
Clinical Predictions of AD Neuropathology
Three cognitive neurologists (SMM, HSY, SAG) independently reviewed selected clinical information for each subject, up to the timepoint at which amyloid PET imaging was obtained, to determine the likelihood of underlying AD neuropathology. Criteria are summarized in Figure 1. Three factors were considered: history, neuropsychological evaluations, and MR imaging, including qualitative reads by the neurologists and MR reports by a clinical neuroradiologist. Each subject had at least one brain MR obtained in the clinical setting, on 1.5 or 3T, and within ∼1 year of the initial evaluation for cognitive decline. The MRI images were reviewed by each reviewer. Criteria used to evaluate each case.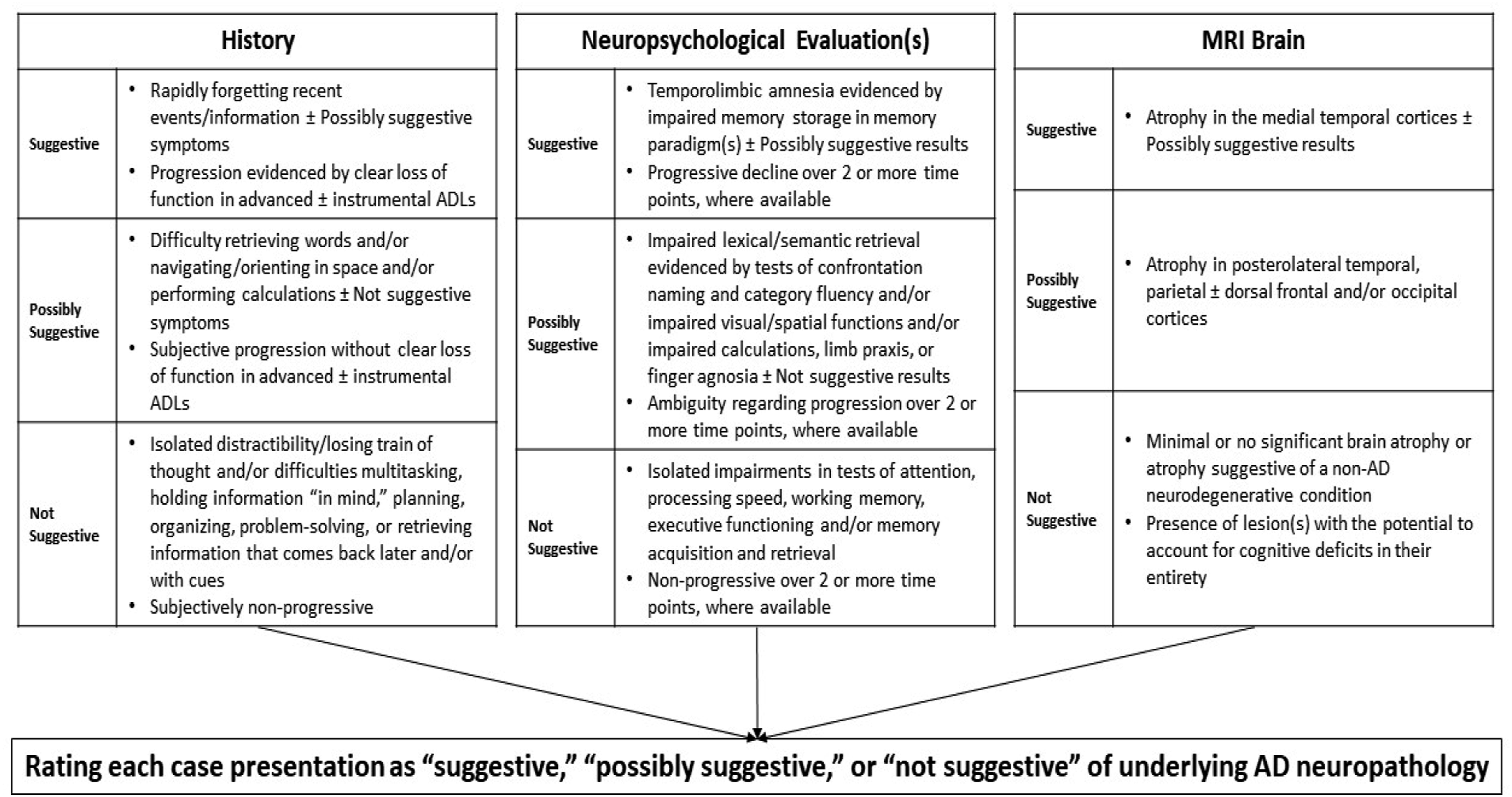
Criteria for evaluating histories and neuropsychological data were based on established cognitive/neuropathological relationships in AD,10-12 scores were normalized to generate Z scores and were considered impaired if ≤ -1. Criteria for evaluating brain MRIs qualitatively, as clinicians commonly do in practice, were based on established relationships between atrophy and the likelihood of AD neuropathology.13,14
Results
Clinical Demographics of the Cohort.
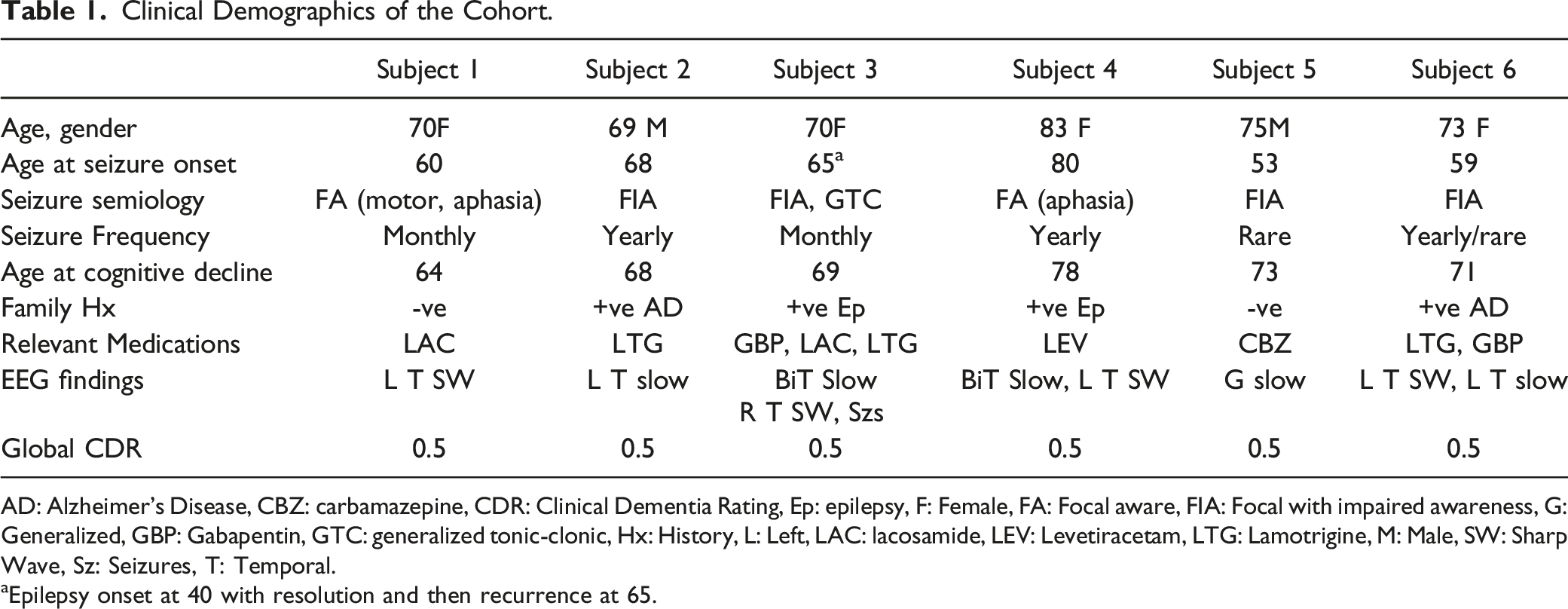
AD: Alzheimer’s Disease, CBZ: carbamazepine, CDR: Clinical Dementia Rating, Ep: epilepsy, F: Female, FA: Focal aware, FIA: Focal with impaired awareness, G: Generalized, GBP: Gabapentin, GTC: generalized tonic-clonic, Hx: History, L: Left, LAC: lacosamide, LEV: Levetiracetam, LTG: Lamotrigine, M: Male, SW: Sharp Wave, Sz: Seizures, T: Temporal.
aEpilepsy onset at 40 with resolution and then recurrence at 65.
Neuropsychological Profile of the Cohort.

BNT: Boston naming test, CERAD: Consortium to establish a registry for Alzheimer’s disease, DSB: Digit span backwards, DSF: Digit span forward, FAS: Letter fluency with F/A/S, MMSE: Mini Mental Status Examination, N/A: Not available, RAVLT: Rey Auditory Verbal Learning Test, TMTA: trails making test part A, TMTB: Trails making test part B.

Amyloid PET findings for each individual case.
Case 1
70-year-old right-handed female who developed new onset unexplained seizures at age 60 characterized by aphasia or right facial clonic movements. When evaluated she was still experiencing monthly seizures and endorsed a several year history of difficulties recalling names of people and objects, difficulty memorizing new courses for dog agility competitions, and difficulties with focus and concentration. She was unsure about whether her symptoms were progressive. She was largely independent in instrumental activities of daily living (iADLs), though stopped managing the family’s finances when they transitioned to electronic systems. Neuropsychological testing revealed mild impairments in attention/executive functions (set shifting), with mild decline from evaluation 4 years prior in attention (more tasks low average) and visuomotor processing speed (from superior to average), and no decline on tests of memory and learning (Table 2). A prior routine EEG captured left temporal sharp waves while an ambulatory EEG did not capture epileptiform abnormalities. 3T MRI brain with volumetric sequences was read by a neuroradiologist (clinical read) as demonstrating “subtle volume loss of the left hippocampus without associated [T2] signal abnormality.”
Impression of the 3 Raters Regarding the Presence of Alzheimer’s Disease pathology.

N: Not suggestive, P: Possibly Suggestive, S: Suggestive.
aCase 6 only had 2 raters due to one rater’s direct knowledge of the case.
Case 2
69-year-old right-handed male with a history of 2 spells of altered awareness followed by confusion. That same year he and his wife had noticed a ∼6-month history of difficulties on his part remembering recent events and information, retrieving words, sustaining attention, navigating routes, and performing mental arithmetic. He continued to work as an accountant part-time (taking longer to complete tasks) and to function independently in iADLs. Neuropsychological testing highlighted impairments in attention/executive functioning (processing speed, set-shifting, motor sequencing, response inhibition), language (comprehension of complex ideational material), and higher somatosensory/motor functioning (limb praxis, finger gnosis). An ambulatory EEG captured left temporal slowing. 1.5 T MRI brain was read to show “mild nonspecific white matter change.”
This case presentation was considered by all 3 raters to be “possibly suggestive” of underlying AD neuropathology. Amyloid PET was “negative for beta-amyloid deposition.” At the most recent follow-up 4 years later, seizures remained medically controlled on a single ASM. Cognitive symptoms remained stable; he was able to continue working. The exact etiology of seizures and cognitive decline remained unclear, but progressive neurodegeneration due to AD was ruled out.
Case 3
70-year-old right-handed female with a history of new onset seizures at age 40 with resolution by age 55 and recurrence at age 65. Then, she began having monthly focal seizures with impaired awareness and was admitted to the epilepsy monitoring unit (EMU) where right temporal seizures were captured. A year prior she had noted cognitive decline characterized by difficulties retrieving words and remembering recent events and information. These difficulties prompted her to retire from work several years after her EMU admission; she remained independent in iADLs. Neuropsychological testing showed deficits in executive functioning (set shifting), language (lexical/semantic retrieval), and memory (acquisition and retrieval), mostly stable-to-improved over serial examinations 1 and 3 years later. 3T MRI brain with volumetric sequences was read to show “slight asymmetric small size of the left hippocampus without definite asymmetric abnormal signal intensity” and “mild burden of multifocal nonspecific white matter disease with multiple small old bilateral posterior inferior cerebellar artery territory infarcts.”
This case presentation was considered “possibly suggestive” of underlying AD neuropathology by all 3 raters. Amyloid PET imaging demonstrated “moderate beta-amyloid deposition” with “most affected areas… the bilateral frontal lobes, posterior cingulate gyri, and precuneal regions,” leading to a diagnosis of non-amnesic MCI due to suspected AD, and initiation of donepezil. At the most recent follow up 2 years later she continued to have rare seizures despite 3 ASMs, and she had transitioned to a clinical diagnosis of mild AD dementia.
Case 4
83-year-old right-handed female with rare focal aware seizures characterized by aphasia starting age 80 and a 5-year history of progressive cognitive difficulties, initially involving focusing, and spelling, and later involving memory for recent events and information. She remained independent in iADLs. Neuropsychological testing revealed initial impairments in attention/executive functions (processing speed, working memory) and memory (acquisition and retrieval), progressive over a period of 5 years to also include impairments in language (lexical/semantic retrieval) and memory storage. An EMU unit admission and then ambulatory EEG captured bitemporal slowing. 3T MRI brain with volumetric sequences was read to be “normal for age.”
This case presentation was considered “suggestive” of underlying AD neuropathology by all 3 raters. Amyloid PET imaging revealed “pronounced beta-amyloid deposition,” leading to a diagnosis of amnesic MCI due to suspected AD, and initiation of donepezil. At most recent follow up 2 years later, seizures remained medically controlled on a single ASM. Cognitive symptoms continued to progress, and she transitioned to a clinical diagnosis of mild AD dementia.
Case 5
75-year-old right-handed male who presented with a 3-to-5-year history of progressive difficulties maintaining focus, holding information “in mind,” solving problems, retrieving words, recalling recent events and information, and navigating routes. He had a history of focal seizures starting at age 53 characterized by impaired awareness which responded to carbamazepine. At presentation, he reported no recent discrete events suggesting seizure. He endorsed depressed mood and related having “very little interest or motivation to do anything.” Neuropsychological testing revealed impairments in attention/executive functioning (set shifting, response inhibition, planning/organization, abstraction/reasoning) and visual scanning. An EMU admission showed background slowing and his carbamazepine was discontinued. 1.5 T MRI brain was read to be “normal.”
This case presentation was considered “not suggestive” of underlying AD neuropathology by all three raters. Amyloid PET imaging was read to be “without beta-amyloid deposition.” An 18-fluorodeoxyglucose (FDG) brain PET was likewise normal. At the most recent follow-up 2 years later, he reported a slight progression in cognitive symptoms, but remained independent in iADLs. There was no recurrence of seizures off medications. The etiology of seizures remained unclear. Factors potentially contributing to cognitive impairment were determined to include effects of mood, obstructive sleep apnea, and chronic medical illness (ischemic cardiomyopathy, heart failure) superimposed on normal aging.
Case 6
73-year-old right-handed female with a ∼12-year history of forgetfulness punctuated by clearly worsening difficulties remembering recent events and information and retrieving words over the 2 years prior to presentation. She had developed unexplained epilepsy at age 59 with focal seizures with impaired awareness. These were medically controlled with 2 ASMs (lamotrigine and gabapentin). She remained independent in iADLs. Neuropsychological testing showed impairments in memory (retrieval, storage) and language (lexical/semantic retrieval), with additional decline in attention/executive functioning (set shifting, phonemic fluency) from evaluation 2 years prior. A prior ambulatory EEG revealed left temporal sharp waves and slowing. 1.5 T MRI brain was read to show “age-related tissue loss” and “prolonged T2 and FLAIR signal in the periventricular and subcortical white matter compatible with the sequela of microangiopathic white matter changes.”
The two raters reviewing this case considered it suggestive of underlying AD neuropathology. Amyloid PET imaging was read to be “positive… consistent with moderate and frequent amyloid neuritic plaques.” Unfortunately, the patient passed away from unclear causes 5 months after enrolling in the study.
Discussion
In this case series of 6 older adults with a history of nonlesional epilepsy enrolled in the IDEAS study, we investigated the utility of amyloid PET imaging in evaluating the etiology of cognitive decline. The over-arching goal of the IDEAS study was to determine whether amyloid PET findings change clinical management in patients with mild cognitive impairment or dementia of uncertain etiology. The study recruited 11 409 patients, 61% having a positive amyloid PET, and study findings leading to the initiation of medications or counseling in 62% of the cohort. 8 The presence of amyloid-β is a core feature of Alzheimer’s disease pathology and is involved in the spread of tau pathology, the second core pathological feature. 15 Although the presence of amyloid is necessary but not sufficient in the diagnosis of AD, a negative amyloid PET is useful in excluding AD in the differential diagnosis.16,17
In our sample, amyloid PET results contributed important diagnostic information and helped reduce diagnostic uncertainty in 2 out of 6 cases. Results were less useful in 2 case presentations rated as “suggestive” of AD and one case rated “not suggestive” of AD with relatively high diagnostic confidence (reflected in consensus among raters). In the “suggestive” cases, a decline in episodic memory, the canonical course seen in AD, was apparent from history and serial neuropsychological evaluations. In the “not suggestive” case, neuropsychological data and MRI brain, taken together, were determined to be “not suggestive” of AD by all raters. Thus, amyloid PET in older adults with epilepsy and cognitive decline was mainly confirmatory in cases where history, neuropsychological profile and neuroimaging were concordant.
In 2 of the remaining 3 cases, there was a consensus among raters regarding diagnostic uncertainty. These cases lacked compelling evidence of episodic memory impairment but had other features potentially suggesting AD (ie, Case 2 with limb apraxia and finger agnosia and Case 3 with lexical/semantic retrieval impairment), and all raters agreed that brain MRIs were “possibly suggestive” of AD. The negative amyloid PET was useful in excluding AD pathology in case 2, in which the absence of clinical decline over the 4 years since the evaluation makes neurodegenerative disease in general unlikely. In case 3, the positive PET increased diagnostic confidence that AD pathology was the cause of cognitive decline, an impression also supported by subsequent symptomatic progression and transition to mild dementia a few years after testing.
The remaining case (Case 1), the only without consensus agreement amongst raters regarding the likelihood of AD, was notable for a non-amnesic cognitive profile with isolated impairments in attention/executive functions, ambiguity regarding the time course, and MRI brain considered “not suggestive” of AD by 2 out of 3 raters. This case illustrates challenges interpreting the results of a diagnostic test in the setting of a relatively lower pre-test probability, applicable with amyloid PET imaging given the 25-30% probability of elevated brain amyloid in 70-year-old individuals with normal cognition. 18 The patient’s subsequent course has yet to disambiguate the picture, as she has experienced subjective worsening of symptoms without significant loss of function in usual activities.
Proposed “appropriate use criteria” for amyloid PET include: (1) Patients with persistent or progressive unexplained MCI; (2) Patients satisfying core clinical criteria for possible AD because of unclear clinical presentation, either atypical clinical course or etiologically mixed presentation; and (3) Patients with progressive dementia and atypically early age of onset (usually defined as 65 years or less in age). 19 Not surprisingly given the overlap between these criteria and the inclusion criteria for the IDEAS study, one might consider all 6 patients in our sample to meet “appropriate use” criteria based on persistent or progressive MCI alone. Our results suggest that careful application of criterion 2 might be helpful when considering older patients with epilepsy and cognitive impairment for amyloid PET imaging, particularly noting the contributions that an atypical (non-amnesic) cognitive profile, lack of clarity surrounding the time course, and additional potential etiological factors make to diagnostic uncertainty. Older adults with epilepsy and cognitive impairment might meet these criteria more frequently than older adults with cognitive impairment without epilepsy due to confounding effects of epilepsy-related factors. Subclinical seizures, interictal discharges and medication effects can all cause or contribute to cognitive impairment.7,20 Apart from factors specific to epilepsy, AD can present atypically with principal impairment in a cognitive domain other than episodic memory,21,22 and AD patients with higher cognitive reserve might not exhibit the same pattern of neuropsychological impairment compared to their peers at the same time point of evaluation or ever. 23
Considering recent advances in our understanding of relationships between AD and seizures, confirming or excluding AD in this patient population is extremely helpful towards identifying the ultimate cause of seizures. 24 Animal models of AD exhibit network hyperactivity and spontaneous seizures. 25 Human studies looking at patients with diagnosed AD show high rates of interictal epileptiform discharges, 26 and epileptic seizures. 27 On the other hand, patients with late onset non-lesional epilepsy are at increased risk of AD dementia, 28 at the same time autoimmune and paraneoplastic etiologies are also possible etiologies, especially in the setting of frequent seizures and medication refractoriness 29 : ruling out AD in these cases could enable clinicians to focus their diagnostic work-up on the rarer autoimmune and paraneoplastic etiologies.
Currently, CSF AD biomarkers 30 are the only clinically available option covered by some health insurance plans, but the invasive nature of the testing may deter patients from pursuing it. In our series, the seizure onset preceded cognitive decline in 4 patients by a range of 2 to 20 years, 2 of whom had positive amyloid PET, suggesting AD pathology. This raises the possibility that in some patients, seizures may be the presenting symptom of AD, and early diagnosis might represent an opportunity for intervention. 31 Although a long lag between the seizure and cognitive decline may also suggest 2 separate pathological processes related to aging.
In one case with positive amyloid PET, the recurrence of seizures after a prolonged quiescent period could reflect an impact of AD pathology to unmask a latent epileptogenic focus, in keeping with a “two-hit” model of seizure recurrence whereby individuals with lowered seizure thresholds due to a host of factors experience seizures after a second insult or “hit”. 32 In general amyloid PET tracers have been found to have high sensitivity in distinguishing healthy controls from AD; Florbetaben, the tracer used in the study, has a sensitivity of 89-97%. 33 Future research directions could include comparisons between FDG and amyloid PET in this patient population and performing amyloid imaging in a population with late onset seizures.
Relatively few studies have examined relationships between seizures and AD biomarkers.
Patients with childhood onset epilepsy showed elevated prefrontal amyloid on [11C]Pittsburgh compound B ([11C]PIB PET) compared to age-matched controls, 50 years after follow-up. 34 In a cohort of 54 patients with AD-related MCI or dementia with epilepsy or subclinical epileptiform activity, three patients were identified based a positive [11C]PIB PET scans, but the details of these particular cases were not provided. 6 Other studies of individuals with late onset epilepsy and CSF AD biomarkers4,35 have shown that CSF Aβ42 levels below normal values for age were associated with a 3.4 hazard ratio of developing AD dementia. 35 Our study thus represents one of the few evaluations of the clinical utility of this technology in this patient population. Other PET ligands including tau 36 are also being investigated and their role in the diagnostic evaluation of late onset seizures would also be helpful, especially since tau PET seems superior to amyloid PET in predicting cognitive decline. 37 Finally, the era of blood-based AD biomarkers 38 is also upon us and promises to be an additional resource in the clinician’s armamentarium. All these biomarkers need additional investigation for their utility in evaluating cognitively impaired older adults, both with and without concurrent seizures.
Conclusion
Amyloid PET Imaging is a potentially useful clinical tool in older patients with epilepsy and cognitive impairment and allows a narrowing of the differential diagnosis in selected cases of diagnostic uncertainty due to a cognitive profile less typical for AD or uncertain time course. Future studies will need to evaluate its clinical utility at a larger scale.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Funding for the IDEAS study was provided by the Alzheimer’s Association; the American College of Radiology; Avid Radiopharmaceuticals, Inc. (a wholly owned subsidiary of Eli Lilly and Company); General Electric Healthcare; and Life Molecular Imaging (formerly Piramal Imaging). PET scans in the study were reimbursed by the Centers for Medicare and Medicaid Services (CMS). Dr. Rani sarkis received grants from the National Institutes of Health (K23 NS119798) during the submitted work. Dr. Alice D. Lam received grants from the National Institutes of Health Funding K23NS101037 during the submitted work. She received grants from Sage Therapeutics; and has served as a consultant for Sage Therapeutics, Neurona Therapeutics, and Cognito Therapeutics. Dr. Hyun-Sik Yang reports grants from the National Institutes of Health (K23 AG062750) during submitted work; personal fees (honoraria) from Genentech, Inc., outside the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
