Abstract
Objective
The present study aims to investigate the underlying neurochemical mechanism of physical exercise on striatum synapsis and memory function in vascular dementia model.
Methods
32 Sprague-Dawley (SD) rats were randomly divided into 4 groups: control group (C group, n = 6), vascular dementia group (Vascular dementia group, n = 7), physical exercise and vascular dementia group (Exe-VD group, n = 6), physical exercise and black group (Exe group, n = 6). 4 weeks of voluntary wheel running were used as pre-exercise training. Vascular dementia model was established by bilateral common carotid arteries occlusion (BCCAo) for 1 week. Passive avoidance test (PAT) were used to test memory function. The level of striatum catecholamine in the microdialysate were detected by enzyme linked immunosorbent assy (ELISA). Golgi staining was used to analyze striatum neuronal spine density.
Results
Behavioral data indicated that 4 weeks of physical exercise ameliorated memory impairment in vascular dementia model. Striatum catecholamine level significantly decreased in VD group when compared with C group (P < .001). But this phenomenon can be rescue by physical exercise (P < .001). In addition, compared with C group, neuronal spine density significantly decreased in VD group (P < .01), but 4 weeks of physical exercise can rescue this phenomenon (P < .05).
Conclusion
4 weeks of physical exercise improves memory function by mitigate the decline of striatum catecholamine and spine density in VD model.
Introduction
Vascular dementia (VD) is a progressive disease that can affect cognitive abilities.1,2 It is generally believed that VD is the second leading form of dementia after Alzheimer’s disease (AD), being responsible for around 15% of all dementia patients.3-5 Diverse types of pathology have been reported in VD, including atherosclerosis, arteriolosclerosis, infarcts, white matter (WM) changes, and microbleeds. 6 Vascular dementia, a heterogeneous group of brain disorders in which cognition impairment is attributable to cerebrovascular pathologies. 7 Recent clinical-pathological studies have highlighted the role of cerebrovascular disease, not only as a primary cause of cognitive impairment, but also as an adjuvant to the expression of dementia caused by other factors, including VD and other neurodegenerative pathologies.8,9 The prevalence of VD is higher in the Asian compared to the Caucasian population, and VD patients might suffer from slowed thinking, forgetfulness, depression and anxiety, disorientation, or loss of executive functions.10,11 And this phenomenon also can be shown in animal model. In a preclinical genetic mouse model of VD with a point mutation in the NOTCH3 gene, TGF-β signaling is disrupted, and pericyte numbers are reduced leading to blood-brain-barrier breakdown, which can aggravate cognitive impairment. 12 All approved anti-AD drugs have been investigated in VD, including the acetylcholinesterase (AChE) inhibitors. 13 While all were found to be useful in some (but not all) measures, the effect size was rather small and marked heterogeneity among studies was observed.14,15
For prevention of dementia, it is important to treat aggressively all factors, even in survivors who do not show evidence of cognitive decline. Numerous studies have examined the positive effect of physical exercise intervention on improving cognition in VD model.16,17 But the mechanism is further needed to be studied. Many literature reports have shown that regular exercise induces angiogenesis and vascular adaptation.18,19 Chen et al and various groups have proven that exercise training improves cognition via selective activation of mTOR and synaptic plasticity.20-22 As a key features of striatum, the ventral striatum (VS) components of both the episodic and procedural memory system, constituting a third system integrating inputs from the hippocampus, prefrontal cortex and other areas to generate motivational (outcome-predictive) signals that act on downstream motor systems to invigorate or disinhibit goal-directed behaviors.23,24 Compared with dorsal striatum, the vs is distinguished by its role in how discrete cues and contexts come to exert pavlovian control over behaviors during learning. 25
However, the chemical mediators in the ventral striatum responsible for cognitive impairment in VD are incompletely understood. Therefore, the purpose of this article is aim to explore the neuroprotective of mechanism of physical exercise on memory function in VD model. This finding may provide a theoretical basis for the clinical prevention of VD.
Materials and Methods
Animals
Adult male SD rats (3 months of age, weighting 280-320 g) were bred in-house. All rats were housed under 12-h light/dark cycle and had a sufficient amount of food and water. In addition, animals with significant abnormalities in development, motility or mental status were excluded from all the experiments.
In our study, the animals were divided into C group, VD group, Exe-VD group, Exe group. The rats in Exe-VD and Exe group were received 4 weeks of voluntary wheel running intervention. Then, the rats in VD and Exe-VD group were received bilateral common carotid artery occlusion (BCCAO) to establish VD model. After 1 week, all rats received neurological evaluation by “Zea-Longa” 5-point scale to determine VD model are successful or not. Last, passive avoidance test were used to test memory function. Striatal catecholamine (dopamine, epinephrine and noradrenaline) level in the microdialysate samples were detected by ELISA. Golgi staining was used to analyze striatum neuronal spine density (Figure 1). Study design diagram. “+” represents rats received exercise intervention or/and VD operation and subsequent test; “-” represents no.
Physical Exercise Intervention
The rats in Exe-VD and Exe group were placed in the voluntary running wheel and had free running wheel access for a consecutive 4 week period, respectively. Daily wheel revolutions were recorded digitally and distance was calculated by multiplying wheel circumference (1 m) by the number of wheel revolutions.
26
The Figure 2 showed that the average running distance per day between Exe-VD and Exe group. Average running distance per day.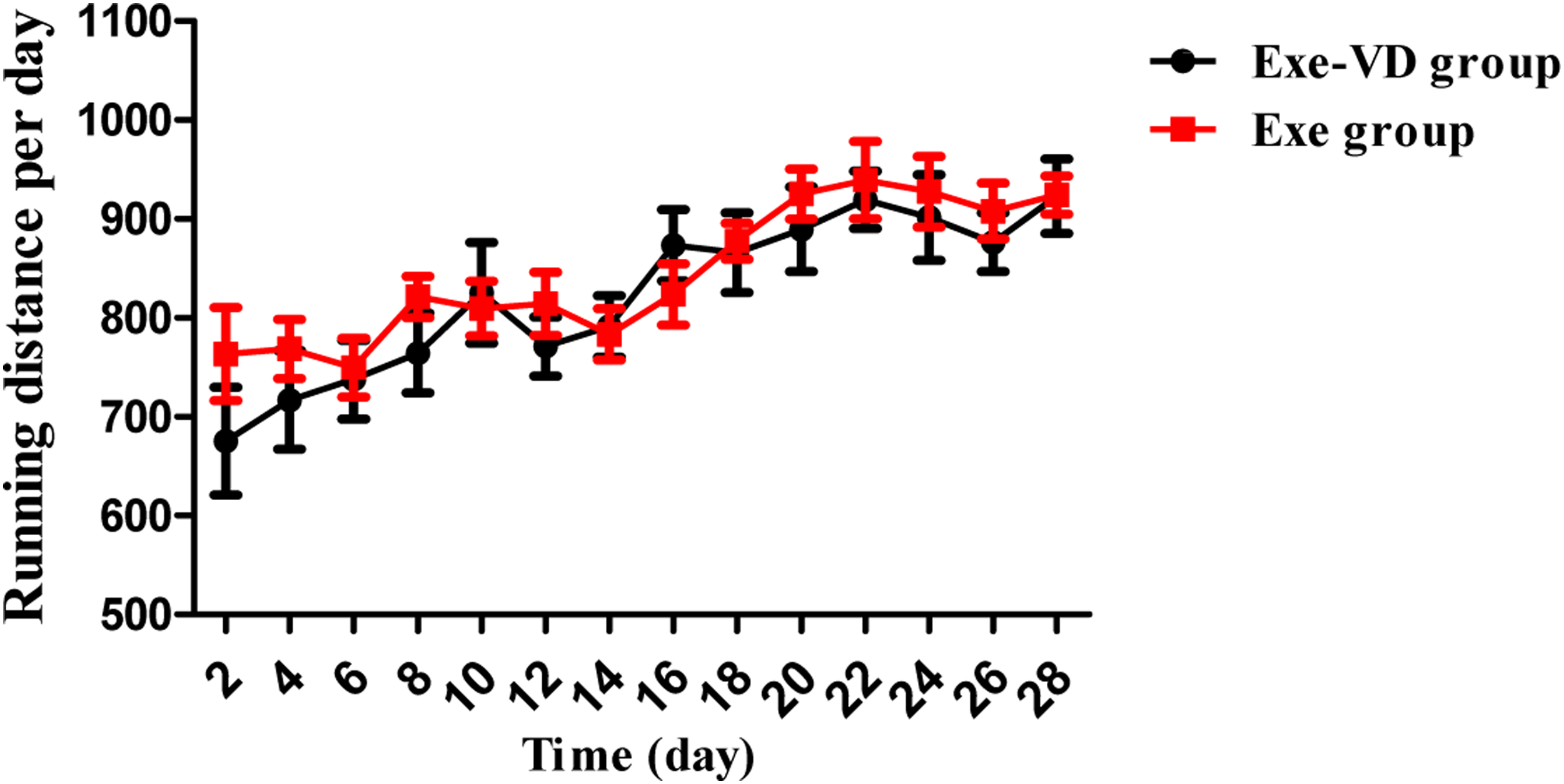
Establishment of VD Model
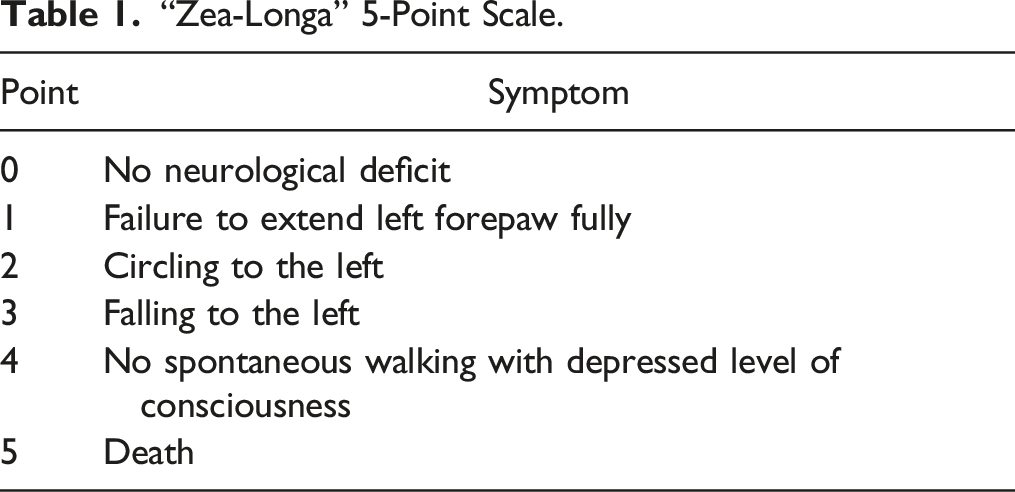
“Zea-Longa” 5-Point Scale.
Memory Function Test
The passive avoidance test was carried out as previously described. 31 In this study, all rats were trained to enter within 300 seconds. The passive avoidance test is performed in a cage composed of 2 parts: lighted and dark chambers. The task consisted of a semi-automated system with a shuttle chamber. An animal may initially freely explore the light and dark compartments. Normal rat have a natural tendency to enter dark environments. The animal learns to avoid the compartment in which an aversive stimulus (such as electrical stimulus) was previously delivered. The latency to pass the gate in order to avoid the shock is used as an indicator of learning and memory. 32
In the acquisition trial, the rats were placed in the lightened chamber and when they entered the dark chamber, the door was closed manually and an electrical foot shock (2 mA) was delivered for 3 seconds through the grid floor. And the time taken to enter the dark compartment was noted as “acquire time”. The retention test was measured 24 hours after the acquisition trial, the rats were again placed in the lightened chamber without electric foot-shock, and the time to enter the dark chamber was measured for 5 minute that “latency time”.33,34
Neurotransmitter Measurement
The level of striatal catecholamine were measured by microdialysis coupled with ELISA. First, the animals were anaesthetized with chloral hydrate (350 mg/kg, i. p.) and placed in a stereotaxic frame with the incisor-bar set at 3.3 mm below the interaural line for the flat skull position. A microdialysis guide cannula was implanted into the ventral striatum (AP = 0 mm, L = 3.0 mm from bregma, V = 4.5 mm from the surface of the skull), according to standard stereotaxic procedures. Next, the perfusion syringes were loaded with fresh aCSF and probes were allowed to equilibrate for an additional 1 h at a flow rate of 1 μl/min. Then, extracellular fluid was collected every 15 minutes in a EP tube (with 15 μL of 10 mmol HCL). Last, striatal catecholamines in the microdialysate samples obtained by microdialysis were measured by ELISA. 35
Golgi Staining
Golgi staining kit was used to analyze striatum neuronal spine density. The whole brain tissues were rinsed with normal saline, and placed in a EP tube with impregnation solution. After 16 h, the impregnation solution was replaced at room temperature in the dark. The tissues were then transferred to solution 3 and replaced for 12 h in the dark. After 24 h, the tissues were subsequently frozen and coronal sections (150 mm) were obtained. Coronal sections were subsequently performed in a series of alcohol solutions prior to completion with xylene. Last, panoramic images of coronal sections were obtained by microscopic and analyzed by Imag J software. 36
Statistical Analysis
All data in this study are presented as the mean ± SEMs. Differences in the data were analyzed using one-way ANOVA. All of the statistical tests were performed with SPSS 20.0, Imag J and GraphPad Prism 5. All differences were considered significant at P < .05.
Results
VD Model’s Neurological Evaluation
Figure 3 shows that the neurological defect score in each group. Our results showed that: the grade score in VD group were significantly higher than C group (P < .001). But there are no significant difference between VD and Exe-VD groups (P > .05). It indicated that vascular dementia model was successfully established in our experiment. The neurological defect score in each group.

Exercise Prevents Memory Impairment Caused by Vascular Dementia
Memory function were evaluated by passive avoidance test. Figure 4 shows that the time of passive avoidance test in each group. On the 24 h retention trial of the passive avoidance test, using a maximum cut-off time of 300s. In the retention trial, the time of VD group were significantly shorter than C group (P < .001); the time of Exe-VD group were significantly longer than VD group (P < .001). However, the retention time has no significant difference between Exe-VD and Exe groups in the retention trial (P > .05). These data indicated that 4 weeks of physical exercise can improved memory function in VD model. Latency time in each group.

Effect of Exercise on Striatal Catecholamine Level in Each Group
Striatal catecholamine level in each group were shown in Figure 5. Catecholamine usually include dopamine (DA), norepinephrine (NE)and epinephrine(E). Compared with C group, The level of DA, NE, E decreased significantly in VD group (P < .001). We also found elevated DA, NE, E level in the striatum from Exe-VD group when compare with VD group (P < .01, P < .001). However, there are no significant difference between Exe-VD and Exe groups (P > .05). Our results revealed that 4 weeks of physical exercise can mitigate the decline of striatum catecholamine in VD model. Dynamic changes of striatal catecholamines in each group. ###P < .001 vs C group; ***P < .001 vs VD group.
Exercise Prevents Striatum Synaptic Impairment Caused by Vascular Dementia
We observed each group’s Golgi staining results, which are given in Figure 6. Our results showed that: when compared with C group, neuronal spine density significantly decreased in VD group (P < .01), but the neuron from Exe-VD group rat exhibited a more density than those from VD rat (P < .05). Thus, we concluded that 4 weeks of physical exercise can ameliorated synaptic impairment caused by vascular dementia. Representative microphotographs of the Golgi-stained striatum in each group. ###P < .01 vs C group; *P < .05 vs VD group.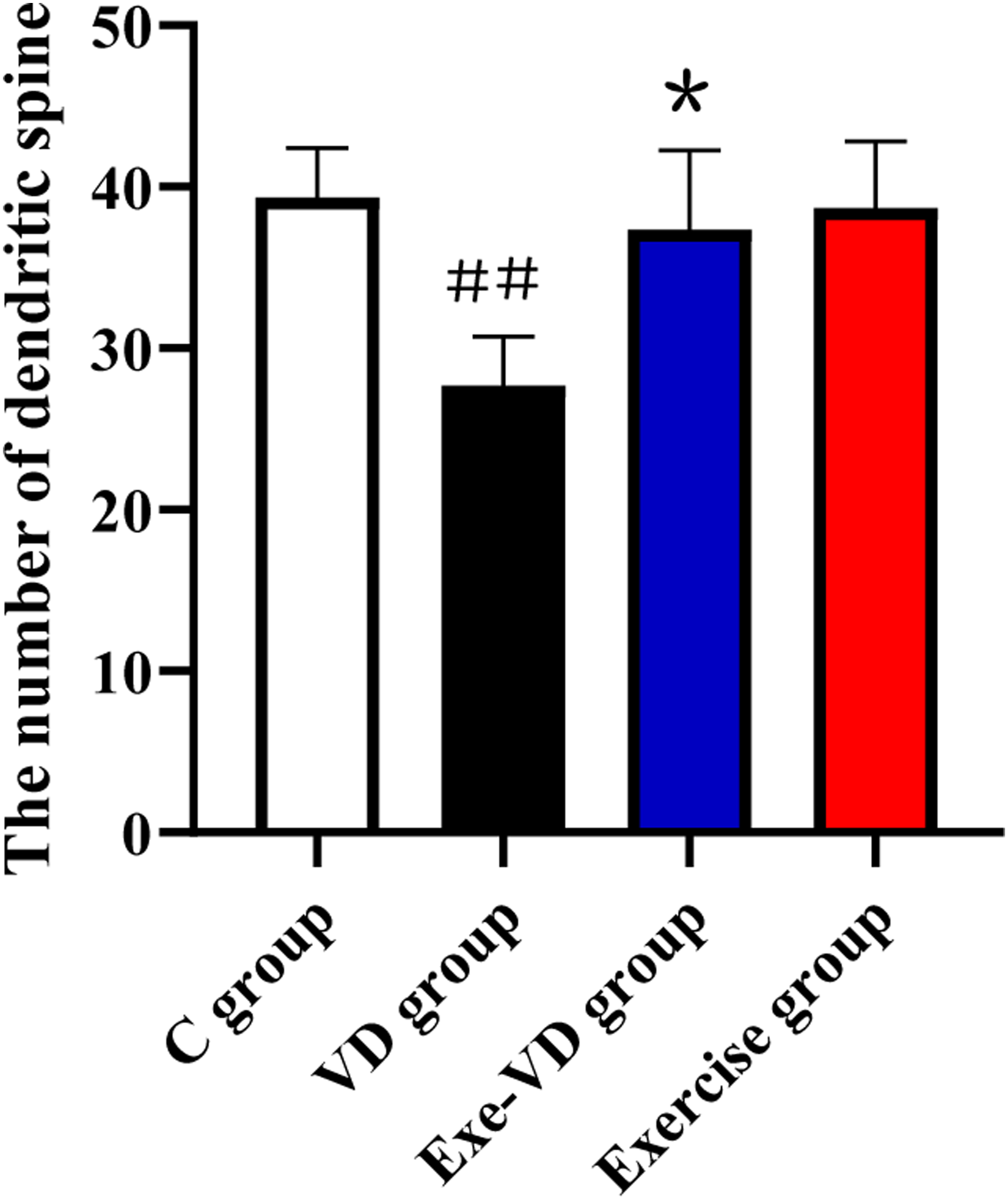
Discussion
Vascular dementia is considered 1 of the most common causes of dementia after Alzheimer’s disease (AD), which performance memory impairment in clinic and in vivo.37-39 Previous studies often used passive avoid test to evaluated memory function in rat of vascular dementia model. Consistently with previous studies, our results also indicated that vascular dementia rat showed memory impairment in passive avoid test. Neuropathology studies have revealed that the abnormal of striatal catecholamine is closely related to the memory impairment in various disease.40,41 Our study also demonstrated that striatal catecholamine (DA, NE, E) level significantly decrease after vascular dementia. In addition, several investigations also have been reported that memory function may involve changes in the density and morphology of dendritic spines in the striatum, hippocampus, and cerebral cortex. Golgi staining in our study also demonstrated that neuronal spine density was significantly reduction after vascular dementia.42-44 Golgi staining in our study also demonstrated that neuronal spine density was significantly reduction after vascular dementia. Thus, we can concluded that the memory impairment after vascular dementia is closely related to striatum synapsis damage and catecholamine loss.
Therefore, it is very important to find a practical and effective way to improve the memory function after vascular dementia. Neurobiology researcher reported that exercise can ameliorate memory by improve synaptic plasticity and BDNF45-47 In vivo study has revealed that exercise can regulated neurotransmitters system, thus to improve the information transmission and cognitive function.48-50 A number of papers emphasize the beneficial effects of exercise on cognition, including (but not limited to), increased neurogenesis and synaptic plasticity, decreases neuroinflammation.51-54 Thus, we also choose exercise as an intervention in vascular dementia model. Behavioral data in our study indicated that 4 weeks of physical exercise significantly improved memory function after vascular dementia. Emerging evidence demonstrated that exercise can relieved memory impairment via NF-κB/miR-503/BDNF in vascular dementia model. 55 Our results present evidence that 4 weeks of physical exercise can reduce striatal catecholamine (DA, NE, E) loss in vascular dementia rat. Molecular biology studies show that exercise-induced noradrenergic and dopaminergic activation enhance cognitive improvement.56-58 Those studies also directly confirmed our result. Moreover, changes in memory function produced by physical exercise are associated with spine density. 59 Our study also suggest that striatum neuron exhibited a more complex branching pattern after exercise intervention, such as neuronal spine density.
Therefore, we can concluded that 4 weeks of physical exercise can improve the memory function by increase striatum synapsis density and catecholamine level in vascular dementia model. Although further studies are needed to elucidate the mechanisms between dendritic spine density and catecholamine to memory function, it should be emphasized that physical exercises represent a natural and useful strategy to improve memory function.
Conclusion
4 weeks of physical exercise improve the memory function in vascular dementia model. It is presumed that the mechanism may be related to the neuroprotective effect of physical exercise, which can mitigate the decline of striatum catecholamine and synapsis density after vascular dementia in rats.
Supplemental Material
Supplemental Material - Physical Exercise Exerts Neuroprotective Effect on Memory Impairment by Mitigate the Decline of Striatum Catecholamine and Spine Density in a Vascular Dementia Rat Model
Supplemental Material for Physical Exercise Exerts Neuroprotective Effect on Memory Impairment by Mitigate the Decline of Striatum Catecholamine and Spine Density in a Vascular Dementia Rat Model by Hangzhou Ren, Zhongyuan Zhang, and Jianwei Zhang in American Journal of Alzheimer's Disease & Other Dementias®
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material for this article is available on the online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
