Abstract
Introduction
Currently, there are no Food and Drug Administration-approved therapies to treat dementia-related psychosis (DRP). This study investigated the association between using antipsychotics and the anticonvulsant divalproex (sodium valproate) to manage DRP and adverse outcomes.
Methods
A retrospective case/control matching study evaluated the risk of mortality, extrapyramidal symptoms (EPS), ischemic stroke, and cardiac arrest/ventricular arrhythmia (CA/VA) with ever-use of antipsychotics/divalproex in patients with DRP vs never-use.
Results
49 509 patients were included; 76.8% used an antipsychotic/divalproex. Treatment ever-use was associated with an increased risk of all-cause mortality (odds ratio, 1.14; 95% CI, 1.10-1.18) and a smaller increase in the risk of EPS (1.10; 1.00-1.19) relative to never-use (adjusted for matching demographic variables, comorbid conditions, and disability)
Conclusions
Current agents used for DRP were associated with increased risk of death and adverse outcomes. An increased risk of death was evident within 3 months of antipsychotic/divalproex initiation and persisted with long-term use.
Significance Statement
• Unapproved and off-label therapies used to treat dementia patients with psychosis may be associated with increased risk of death/adverse outcomes. • Safe and effective therapies are needed for patients with dementia-related psychosis.
Introduction
More than 90% of people with dementia experience neuropsychiatric symptoms during the course of their illness. 1 These difficult-to-manage behavioral and psychological symptoms of dementia include hallucinations and delusions (psychosis), agitation, aggression, mood disorders, apathy, and disinhibition. 2 In the absence of any medications approved by the US Food and Drug Administration (FDA) for the treatment of dementia-related neuropsychiatric symptoms, antipsychotic agents are often prescribed to manage these distressing symptoms. 3
Meta-analyses and clinical trials suggest that antipsychotics have modest efficacy at best in the treatment of key neuropsychiatric symptoms in patients with dementia.4-6 Yet concerns have been raised about the safety of these agents, including an increased risk of cerebrovascular events (e.g., ischemic stroke),1,5 extrapyramidal symptoms (EPS),4,5 and sudden cardiac death or ventricular arrhythmias.7-9 Furthermore, several studies have suggested that the use of antipsychotics in patients with dementia is associated with an increased risk of mortality,10-12 which led to FDA-mandated black box warnings on atypical antipsychotics in 2005 13 and on conventional antipsychotics in 2008. 14
Early data on these outcomes were obtained from meta-analyses of randomized clinical trials5,10,13 that studied treatments for only a few weeks or months, with brief follow-up periods, whereas some adverse outcomes, such as EPS, may appear rapidly after treatment initiation, long-term outcomes (particularly mortality) may not occur during a short-term study, thus limiting our understanding of the association between treatment and outcomes. There is also a paucity of data regarding the influence of cumulative treatment exposure and long-term outcomes.
We conducted a case-control analysis from a retrospective cohort study using real-world claims data to determine the risk of adverse outcomes (i.e., all-cause mortality, EPS, ischemic stroke, and cardiac arrest/ventricular arrhythmia [CA/VA]) of antipsychotics and divalproex (sodium valproate; an anticonvulsant agent commonly used to treat psychotic symptoms) in patients with dementia-related psychosis (DRP) relative to no treatment. Associations between adverse outcomes and duration of treatment exposure were also investigated. We hypothesized that use of these agents would be associated with increased rates of mortality and other adverse outcomes in this large population of Medicare enrollees.
Methods
Data Source
This retrospective, longitudinal, observational cohort study was based on claims data from Medicare part A (covering hospital and nursing home stays), part B (covering outpatient services and procedures), and part D (covering prescription drugs) from 2008 to 2016. The study used anonymized data from the 20% Medicare random sample, including enrollment information, patient demographics, coded diagnoses and procedures, and outpatient prescription-dispensing records. Standard analytic files were generated by the Centers for Medicare and Medicaid Services. Approval for the study was received from the Human Subjects Research Committee of the Hennepin County Medical Center, Hennepin Healthcare System, Inc, which provided a waiver of consent because all data were deidentified. These methods were also described and used in a previous report analyzing the risk of death and use of long-term care associated with DRP in the same patient population. 15
Patient Population and Cohorts
The study population included patients 40 years of age or older with Medicare parts A, B, and D fee-for-service coverage but no health maintenance organization coverage. Patients could enter the study any time from January 1, 2008, to December 31, 2016, as long as they had 1 year of observable baseline data. The baseline period was defined as 1 year before the dementia or DRP index date.
Patients were designated as having dementia if they fulfilled 1 of the following 2 criteria: relevant diagnosis codes on 2 medical claims for dementia according to International Classification of Diseases (9th and 10th revisions) Clinical Modification codes (Supplemental Table 1) ≥30 days and ≤3 years apart, for which the date of the second medical claim became the index date; or 1 claim for a dementia diagnosis plus 1 prescription for an antidementia medication (donepezil hydrochloride, galantamine hydrobromide, rivastigmine, tacrine hydrochloride, or memantine hydrochloride) within 1 year of the dementia diagnosis claim, for which the later of these dates became the index date. For patients who satisfied both definitions, the earlier of the 2 index dates were used. Patients with dementia were excluded from analyses if they had a chronic psychiatric disorder, a history of seizures, other possible etiologies of dementia (including chronic alcohol-induced dementia and drug-induced persisting dementia), behavioral claims, or psychosis or had received antipsychotic medication prior to the dementia index date. Patients with a history of stroke within 6 months before the dementia index date were also excluded (Supplemental Table 1).
Incident psychosis among patients with dementia was identified by the following criteria: 2 or more diagnosis codes for psychosis or a behavioral disturbance (Supplemental Table 1) ≥7 days and ≤3 years apart; or 1 diagnosis code for psychosis or a behavioral disturbance plus 1 prescription for an antipsychotic drug or divalproex within 1 year of the diagnosis claim date; or 2 or more prescriptions for an antipsychotic drug or divalproex ≥180 days apart. Of note, divalproex was included within this analysis because of its approved use in the treatment of psychotic episodes of bipolar disorder and its reported role in the treatment of behavioral and psychological symptoms of dementia.16,17 The later date for each individual criterion was used to define the DRP index date. For patients who met multiple inclusion criteria, the earliest index date following the dementia diagnosis was selected.
Adverse Outcomes and Incidence Rates
The following adverse outcomes were considered in the present analyses: all-cause mortality, EPS, ischemic stroke, and CA/VA. Deaths were identified from Medicare beneficiary enrollment records, and other outcomes were identified from diagnosis codes on medical claims during the follow-up period (Supplemental Table 2
Incidence rates and 95% confidence intervals (CIs) per 100 person-years at risk during the follow-up period were calculated for each adverse outcome for all patients with DRP (regardless of treatment). Analyses were repeated according to the specific drug prescribed, that is, antipsychotic agent (quetiapine, risperidone, aripiprazole, olanzapine, or haloperidol) or divalproex. Among all patients with DRP, the first adverse outcome that occurred between the DRP index date and the end of follow-up was identified. Among patients who were prescribed an antipsychotic agent or divalproex, the first adverse outcome between the date of first prescription for an antipsychotic or divalproex and the end of follow-up was identified. Patients with an adverse outcome occurring prior to the DRP index date or date of first prescription were excluded from analyses to allow for a proper estimation of incidence rates.
Analytical and Statistical Approach
A series of nested case-control analyses was performed to estimate the risk of an adverse outcome in those who were ever treated with an antipsychotic or divalproex (cases) relative to those who had never used any of these treatments (controls). Patients with DRP and an adverse outcome were matched to controls at a 1:1 ratio for all-cause mortality and at a 1:4 ratio for other adverse outcomes. Case-control matching was based on age (±1 year), sex, race, and DRP index year (±2 years) by using an incidence-density sample with replacement approach. Pseudo-outcome (counterfactual) dates were created for controls by adding the case patient’s time to outcome to the control patient’s index date. Baseline characteristics for cases and controls were summarized separately for each outcome.
Case-control analyses to examine the association between exposure to an antipsychotic or divalproex and the risk of an adverse outcome were conducted using conditional logistic regression. For each outcome, 2 sets of models were fit on the basis of the following categorizations of medication use before the event or pseudo-event date: ever-use (ie, according to whether or not a medication was prescribed before the outcome) and cumulative duration of exposure (i.e., medication use for <3 months, 3 to <6 months, 6 to <12 months, and ≥12 months). Patients who never used an antipsychotic or divalproex were set as the reference with an odds ratio (OR) of 1.00. Models were adjusted for matching variables (i.e., age, sex, race, and DRP index year), the comorbid conditions of the Charlson Comorbidity Index (i.e., myocardial infarction, congestive heart failure, peripheral vascular disease, cerebrovascular disease, chronic obstructive pulmonary disease (COPD), rheumatologic disease, peptic ulcer disease, mild liver disease, moderate or severe liver disease, diabetes with or without chronic complication, hemiplegia or paraplegia, renal disease, any malignancy, including leukemia and lymphoma, metastatic solid tumor, and human immunodeficiency virus/acquired immunodeficiency virus), 18 and a claims-based disability index that considers use of durable medical equipment and other factors to provide additional covariate control.19,20
Results
Sample Demographics and Baseline Characteristics
Overall, 256 408 patients were identified with dementia, 49 509 of whom met the eligibility criteria for DRP and were included in the analyses (Figure 1). Of these, 38 004 patients (76.8%) had prescriptions for either an antipsychotic drug or divalproex; 11 505 patients (23.2%) did not. The agents most commonly first prescribed were quetiapine (n = 15 090; 39.7%), risperidone (n = 9630; 25.3%), and divalproex (n = 5539; 14.6%) (Figure 2). Patient selection. Antipsychotic agent or divalproex first prescribed in patients with dementia-related psychosis (n = 38 004).

Baseline Characteristics in All Patients With Dementia-Related Psychosis, and in Cases and Controls According to Adverse Outcome.

Except where specified, values are n (%).
CA/VA, cardiac arrest/ventricular arrhythmia; COPD, chronic obstructive pulmonary disease; HIV/AIDS, human immunodeficiency virus/acquired immunodeficiency; SD, standard deviation.
aComorbidity categories are determined according to the Charlson Comorbidity Index.
bData are not reportable when cell sizes represent <11 individuals, per Data Use Agreement with Centers for Medicare and Medicaid Services.
Incidence of Adverse Outcomes in Patients With DRP
Crude Incidence of Adverse Outcomes in Patients With Dementia-Related Psychosis Overall and by Specific Treatment After First Prescription.

CI, confidence interval; PY, person-years.
aIncluded all events occurring after the dementia-related psychosis index date. For all other columns in this table, only the events occurring after the medication start date were included.
bExcludes patients with Parkinson’s disease-type dementia.
Extrapyramidal symptoms occurred in 7.9% of patients with DRP, with an incidence rate of 5.0 events (95% CI, 4.8–5.1) per 100 person-years. Aripiprazole (8.7%; incidence rate, 4.8 events per 100 person-years) and divalproex (8.6%; incidence rate, 6.1 events per 100 person-years) were associated with the highest occurrence of EPS.
Ischemic stroke and CA/VA events occurred in 2.0% (1.2 event per 100 person-years) and 1.5% (.9 event per 100 person-years) of DRP patients, respectively. The incidence of ischemic stroke was slightly higher with haloperidol (2.4%; 1.7 event per 100 person-years) than with other medications (1.6%–2.1%; 1.1 event per 100 person-years). CA/VA occurred slightly more frequently with aripiprazole and risperidone (1.6% each) than with other medications (1.3%–1.4%), but incidence rates were similar across all medications (.8–1.0 event per 100 person-years).
Association Between DRP Treatment and Adverse Outcomes
For each adverse outcome in the case-control analyses, the numbers of patients included were 34 986 cases and 34 986 controls for all-cause mortality, 3610 cases and 14 389 controls for EPS, 993 cases and 3952 controls for ischemic stroke, and 741 cases and 2951 controls for CA/VA. Demographics, baseline characteristics (Table 1), and disability index scores (Supplemental Table 3) were similar between cases and controls.
Use of an antipsychotic or divalproex at any point for DRP was associated with an increased risk of all-cause mortality in comparison with never-use (OR, 1.14; 95% CI, 1.10–1.18) (Figure 3A). When considering duration of exposure, an increased risk of all-cause mortality was evident within 3 months of treatment (OR, 1.08; 95% CI, 1.04–1.13). The risk of all-cause mortality increased with treatment durations of 3–6 months (OR, 1.21; 95% CI, 1.14–1.28) or longer (6 to <12 months: OR, 1.15; ≥12 months: OR, 1.17). Case-control analyses of adverse outcomes for patients with dementia-related psychosis according to treatment exposure: (A) all-cause mortality; (B) EPS; (C) ischemic stroke; and (D) CA/VA. ORs are adjusted for matching variables (age, sex, race, and DRP index year), myocardial infarction, congestive heart failure, peripheral vascular disease, cerebrovascular disease, chronic obstructive pulmonary disease, rheumatologic disease, peptic ulcer disease, mild liver disease, moderate or severe liver disease, diabetes without chronic complication, diabetes with chronic complication, hemiplegia or paraplegia, renal disease, any malignancy including leukemia and lymphoma, metastatic solid tumor, human immunodeficiency virus/acquired immunodeficiency virus, and disability index. CA/VA, cardiac arrest/ventricular arrhythmia; CI, confidence interval; DRP, dementia-related psychosis; EPS, extrapyramidal symptoms; OR, odds ratio.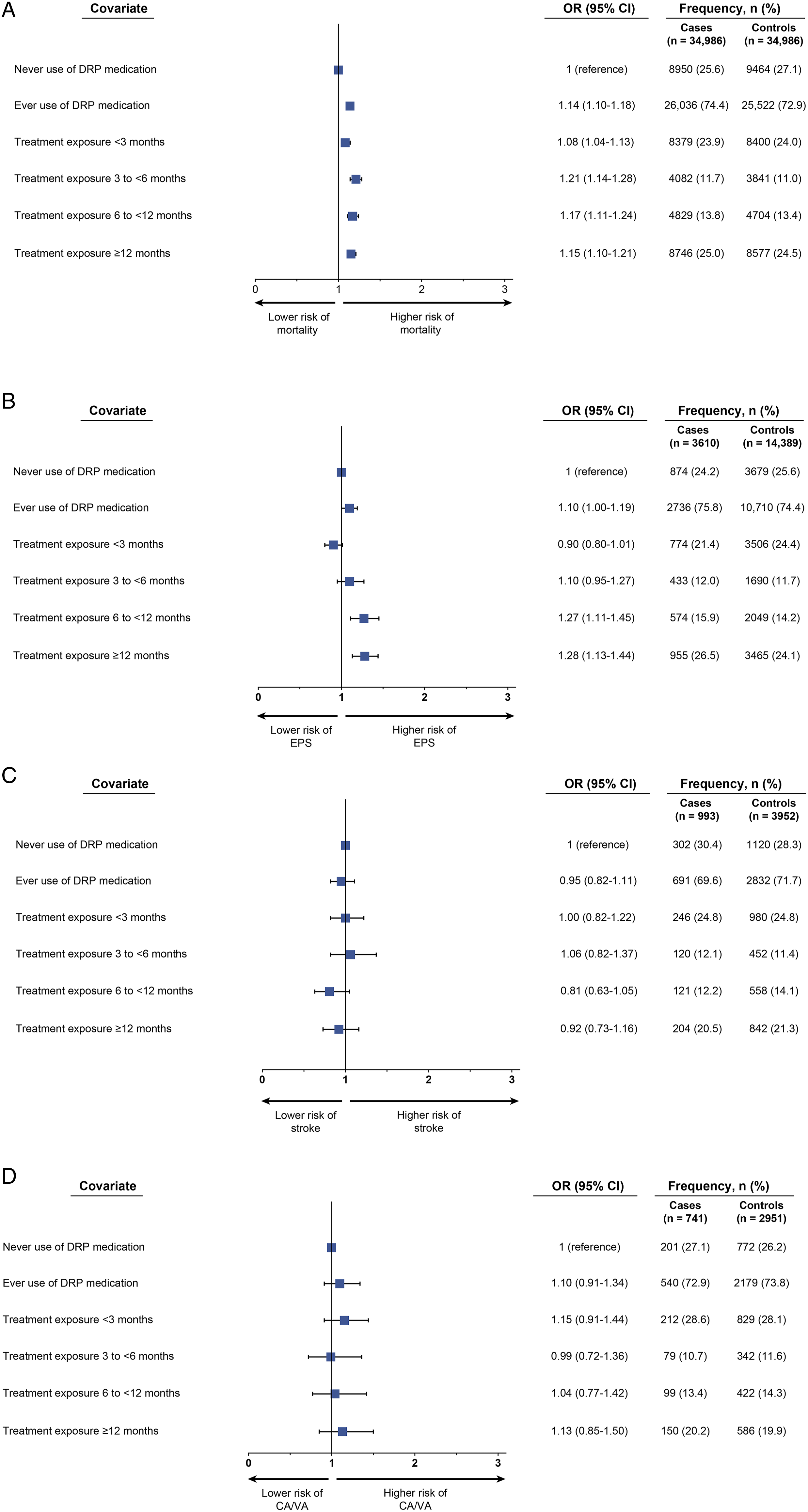
Ever-use of treatment was also associated with increased risk of EPS (OR, 1.10; 95% CI, 1.00–1.19) (Figure 3B). Longer exposure to treatment increased the risk of EPS in an exposure-dependent manner, with ORs showing a progressive increase with longer duration of treatment (<3 months: OR, .90; to ≥12 months: OR, 1.28).
No association was identified between ever-use of an antipsychotic or divalproex and risk of ischemic stroke (OR, .95; 95% CI, .82-1.11) (Figure 3C) or the risk of CA/VA with ever-use of treatment (OR, 1.10; 95% CI, .91–1.34) (Figure 3D).
Discussion
This large, real-world database study conducted over an 8-year period revealed that elderly patients with DRP who used antipsychotics or the anticonvulsant divalproex were at a 14% increased risk of all-cause mortality relative to patients who had not received these treatments. An increased risk of death was evident within 3 months of starting an antipsychotic or divalproex and persisted in those receiving treatment for longer. The risk of EPS was also increased by 10% each in patients who had ever used antipsychotics or divalproex in comparison with those who had not used these agents.
Management of psychosis in dementia patients is an area fraught with multiple, unanswered clinical questions. Some of the factors that have contributed to the use of antipsychotics despite limited evidence are lack of evidence to guide an effective pharmacologic management plan or the selection of agents suitable for particular types of dementia, high-profile adverse effects, quality of life impacted by psychosis, and caregiver and economic burden of institutionalization. In 2016, the American Psychiatric Association issued guidelines on the judicious use of antipsychotics to improve the care of patients with dementia exhibiting psychosis or agitation. 21 The recommendations suggest implementation of a comprehensive treatment plan, including both non-pharmacological and pharmacological approaches, evaluation of response to pharmacologic therapy after a 4-week period, and tapering and withdrawal of therapy within 4 months of initiation if possible. The recommendations were based on the understanding that antipsychotic use can improve patient quality of life and reduce the risk of patient distress, violence, and caregiver burden, particularly in patients with dangerous symptoms, while recognizing that the benefits of therapy are minor at best and that antipsychotics are associated with clinically significant adverse effects, including mortality.
Prior retrospective studies11,12,22,23 have shown increased mortality with antipsychotic use including divalproex,23,24 but with a limited duration of follow-up of 180 days. Our study, which examines more contemporaneous treatment practices in the United States, had a follow-up of nearly 2 years and is the first study of US patients using the large Medicare database to address this topic. By evaluating adverse outcomes in this cohort, our work extends the findings of previous meta-analyses 10 and observational studies11,12,22,23 that have reported an increased risk of mortality with the use of antipsychotic drugs in patients with dementia. Our observations of a persistent elevated risk of mortality with longer exposure are also consistent with the findings of others. 12 Furthermore, our results align with the boxed warnings issued by the FDA cautioning about mortality risk with antipsychotics in patients with dementia.13,14
The mechanisms by which antipsychotic agents increase the risk of death are unknown. Intermediate medical events, such as EPS, may be involved, and the positive associations between treatment and EPS in our study support this possibility. EPS are well-documented side effects of antipsychotic agents that inhibit D2 receptors in the nigrostriatal system 25 and of divalproex, though the exact mechanism for divalproex is unknown. 26 Other established adverse events of antipsychotics that may contribute as potential underlying causes of death include cerebrovascular adverse events, sedation, weight gain, diabetes, metabolic syndrome, cognitive worsening, and in the case of conventional antipsychotics, anticholinergic effects, postural hypotension, and prolonged QT interval. 2 Divalproex has a mechanism of action that is distinct from that of most antipsychotics; however, associations between divalproex and adverse effects such as EPS, gastrointestinal complications, urinary tract infections, thrombocytopenia, sedation, or weight gain could similarly contribute to potential underlying causes of death.16,26,27 Although we were unable to examine the causes of death in our study, data from randomized clinical trials indicate that most deaths in patients with dementia are accounted for by cardiac events (e.g., heart failure and sudden death) and infections (mostly pneumonia). 13
The association of treatment with all-cause mortality in our study (OR, 1.14), though more modest than that reported in previous meta-analyses of short-term randomized placebo-controlled trials (OR, 1.5 10 or 1.6–1.7 13 or observational studies (hazard ratio, 1.23–1.55) 12 was nevertheless substantial and clinically relevant. These differences are likely driven by methodologic differences between studies. Observational studies based on healthcare claims data, such as ours, have several recognized methodologic limitations, including incomplete control of confounders and the potential for misclassifying diagnoses. Although we used matching to attempt to control for a range of demographic factors, comorbid conditions, and disability, and used multivariable modeling, the possibility of residual confounding factors cannot be excluded. In addition, some studies have reported a positive association between neuropsychiatric symptoms and mortality after adjusting for antipsychotic medication,28,29 suggesting that a portion of the mortality risk may be related to the presence of symptoms. Another noteworthy finding in our study is the lack of clear association between antipsychotic use and ischemic stroke. Some prior studies have suggested a link between antipsychotic use and cerebrovascular events,30,31 whereas other groups were not able to identify this association. 32 Our analyses support the latter, but further evidence in this area is needed.
We attempted to study individuals whose psychosis was plausibly related to dementia and used a highly specific approach to exclude potential non-dementia-related causes of psychosis; as such, it is possible that some patients may have been misclassified. Our approach to identifying patient cohorts from claims has not been validated. Diagnostic codes were used to identify EPS, ischemic stroke, and CA/VA. This may have led to underreporting of these events, which is common with other database studies. In particular, even though EPS is a common adverse event associated with antipsychotic agents, it is not well documented with diagnosis codes in administrative claims databases such as Medicare. In terms of generalizability, Medicare covers nearly all US individuals ≥65 years old and provides lengthy follow-up data, but country-level variations in healthcare usage and coverage may limit the applicability of our findings in other countries. The study was designed to assess incidence rates and risk of certain adverse outcomes associated with medications for DRP but did not evaluate treatment efficacy or other safety measures.
Conclusions
In this large Medicare database analysis, medications currently used to treat DRP were associated with an increased risk of all-cause mortality relative to never-use of these medications. An increased risk of all-cause mortality was evident within 3 months of initiating treatment, and persisted with longer-term use. There was also an increase in the risk of EPS with treatment, but no clear association between treatment and ischemic stroke. Although causal inferences cannot be made from our data, these findings support the call for caution with the use of antipsychotic agents not approved by the US FDA in this vulnerable patient population.
Supplemental Material
sj-pdf-1-aja-10.1177_15333175221081374 – Supplemental Material for Adverse Outcomes Associated With Off-Label Agents Used to Treat Dementia Patients With Psychosis: A Case-Control Medicare Database Study
Supplemental Material, sj-pdf-1-aja-10.1177_15333175221081374 for Adverse Outcomes Associated With Off-Label Agents Used to Treat Dementia Patients With Psychosis: A Case-Control Medicare Database Study by Nazia Rashid, James B. Wetmore, Muna Irfan and Victor Abler in American Journal of Alzheimer's Disease & Other Dementias®
Footnotes
Acknowledgments
Editorial and medical writing support for this manuscript were provided by Harriet Lamb, BSc, and Dena McWain of Ashfield Healthcare Communications and was funded by Acadia Pharmaceuticals Inc.
Author Contributions
Nazia Rashid: Study design, data analysis, interpretation of data, revising the manuscript for critical content.
James B. Wetmore: Study design, acquisition of data, data analysis, interpretation of data, revising the manuscript for critical content.
Muna Irfan: Study design, interpretation of data, revising the manuscript for critical content.
Victor Abler: Study design, data analysis, interpretation of data, revising the manuscript for critical content.
All authors contributed to the writing and development of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Acadia Pharmaceuticals Inc.
Disclosures
James B. Wetmore is employed by the Chronic Disease Research Group, which received research funding from Acadia Pharmaceuticals Inc, and has served on ad hoc advisory boards for the BMS-Pfizer alliance. Victor Abler and Nazia Rashid are employees of Acadia Pharmaceuticals Inc Muna Irfan has nothing to disclose.
Data Availability Statement
The data used in this analysis are available for a fee to qualified individuals and institutions from the Centers for Medicare and Medicaid Services and are subject to the terms of a Data Use Agreement.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
