Abstract
Because dementia with Lewy bodies (DLB) has various psychiatric symptoms, early diagnosis in patients without parkinsonism is difficult. To reveal associations between regional brain perfusion and psychiatric symptoms in DLB patients without parkinsonism, we quantified brain perfusion using an automated brain perfusion single-photon emission computed tomography analysis program, FineSRT. We statistically analyzed the differences in brain perfusion between groups, divided by the presence or absence of psychiatric symptoms. In DLB patients with depression, there were significant brain perfusion increases in the left angular gyrus and right upper precuneus. In DLB patients with visual hallucinations, there were significant decreases in the left inferior parietal lobule, left superior temporal gyrus, and right primary visual cortex. In DLB patients with auditory hallucinations, there were significant increases in the right middle occipital and right inferior occipital gyri. Our findings provide clues about the pathomechanisms of psychiatric symptoms and may enable early diagnosis of DLB in the future.
Keywords
Significance Statement
We revealed associations between regional brain perfusion and diverse psychiatric symptoms, which included depression and visual and auditory hallucinations in DLB patients without parkinsonism. Our findings provide clues about the pathomechanisms of psychiatric symptoms and may help in the early diagnosis and treatment of DLB in the future.
Introduction
Dementia with Lewy bodies (DLB) has attracted attention in recent years as the second most common cause of degenerative dementia, after Alzheimer’s disease. 1 Dementia with Lewy bodies shares clinical and pathological features with other dementia subtypes, such as Alzheimer’s disease and Parkinson’s disease (PD).
Dementia with Lewy bodies has various clinical characteristics, including cognitive fluctuations, 2 recurrent visual and auditory hallucinations, depression, systematized delusions, and idiopathic parkinsonism, as well as progressive cognitive decline. 3 According to DLB diagnostic criteria, 3 visual hallucinations are included as core clinical features, and auditory hallucinations and depression are included as supportive clinical features. Depression is an important evaluation item that affects caregiver burden. 4
Dementia with Lewy bodies is difficult to diagnose, especially in the early clinical stages; moreover, patients without parkinsonism are more likely to be misdiagnosed with mental disorders. 5 Furthermore, the diversity of psychiatric symptoms in DLB without parkinsonism is not well understood in relation to the pathomechanisms and biological markers of the disease. If the relationship between psychiatric symptoms and regional cerebral blood flow (rCBF) in DLB can be clarified, it may be possible to improve the diagnostic accuracy of DLB without parkinsonism in the early stages of the disease.
The aim of this study was to reveal associations between cerebral perfusion and a range of psychiatric symptoms (depression, visual hallucinations and auditory hallucinations) in DLB patients without parkinsonism using computer-assisted statistical image analysis techniques, such as FineSRT. 6 To the best of our knowledge, no previous studies have investigated the mechanisms of DLB using automated rCBF quantification program software, such as FineSRT.
Materials and Methods
Participants
Participants included in this retrospective chart review were 95 Japanese patients who were diagnosed with probable DLB without parkinsonism at Sunagawa City Medical Center from 2008 to 2017.
A clinical diagnosis of probable DLB was made with the agreement of experienced neurologists and psychiatrists, based on the Consortium on DLB International Workshop Criteria. 1 Patients underwent detailed neurological and psychiatric examinations, routine laboratory tests, and diagnostic brain magnetic resonance imaging (MRI) to exclude patients with intracranial lesions. For the clinical diagnosis of DLB, patients also underwent 99mtechnetium-ethyl-cysteinate-dimer brain perfusion single-photon emission computed tomography (SPECT). Dementia severity was assessed using the Mini-Mental State Examination (MMSE). 7 The presence or absence of cognitive fluctuations was carefully assessed using information obtained from reliable caregivers. To confirm a clinical diagnosis of DLB based on these findings, all new cases were discussed at a weekly multidisciplinary meeting that was attended by neurologists and psychiatrists. No patients had a history of cerebrovascular diseases, other degenerative neurological diseases, or intracranial lesions on brain MRI. Before SPECT imaging none of the patients had received any anti-dementia drugs, including donepezil, that have been reported to affect rCBF. 8
Procedures
Brain Perfusion SPECT
All participants were placed in a supine position with their eyes closed. After 600 MBq 99mtechnetium-ethyl-cysteinate-dimer (Fujifilm RI Pharma, Tokyo, Japan) was injected, the transit from the heart to the brain was monitored using a rectangular large-field gamma camera (E. Cam Signature; Toshiba Medical, Tokyo, Japan). Ten minutes after the angiography, SPECT images were obtained using a rotating dual-head gamma camera (E. Cam Signature) for 20 minutes. Scan conditions were as follows: 64 x 64 matrix with 1.78 zoom (pixel size 5.4 mm); 4 rotations by continuous mode (each 5-minute rotation was a 360° collection, with 4° per step and 90 views). All images were reconstructed using ramp-filtered back-projection and were then three-dimensionally smoothed using a Butterworth filter (order = 8 and cutoff = .70). The reconstructed images were corrected for gamma-ray attenuation using the Chang method, and the SPECT images were viewed with voxel dimensions of 3.56 mm. CBF was quantified using the Patlak plot method. 9 Quantitative flow-mapping images were obtained from the qualitative perfusion images using Patlak plot graphical analysis and Lassen’s correction. 10 Next, rCBF was quantified using an automated brain perfusion SPECT analysis program, FineSRT (Fujifilm RI Pharma Co., Ltd.). 6 This program performed anatomical standardization of the images, employed statistical parametric mapping to standardize the images, quantified rCBF with a three-dimensional stereotactic region-of-interest (ROI) template, calculated CBF, and displayed the results. The ROIs were categorized into 52 segments in the template. We evaluated the absolute value of rCBF in each cerebral segment.
Statistical Analysis
We performed statistical analyses of the differences in rCBF between groups, which were divided by the presence or absence of each of the following symptoms: depression and visual and auditory hallucinations.
We chose these 3 psychiatric symptoms because they are included in the diagnostic criteria for DLB 3 and are relatively common in clinical settings. All subgroup comparisons were performed using t-tests. All calculations were performed using StatFlex version 6 for Windows (Artech Co., Ltd., Osaka, Japan). For multiple comparisons of the 3 psychiatric symptoms, we performed Bonferroni’s method. P < .05/3 was accepted as statistically significant. Because the distribution of blood flow to each segment was performed mechanically using software, there was no room for researcher bias. We treated each segment as an independent variable and explored the differences in blood flow in the segment. According to the advice of a statistician, we did not need to control for multiple comparisons of each segment.
Ethics
The provided data were anonymized. The ethical review board of Sunagawa City Medical Center approved this study in accordance with the ethical standards stated in the Declaration of Helsinki. Because the present study was a retrospective chart review, we used the opt-out method via the hospital bulletin board, in accordance with the Act on the Protection of Personal Information.
Results
Demographic data of the patients.

aMMSE: Mini-Mental State Examination.
bHDS-R: revised Hasegawa Dementia Scale.
Associations between regional cerebral blood flow and psychiatric symptoms in patients with dementia with Lewy bodies without parkinsonism.

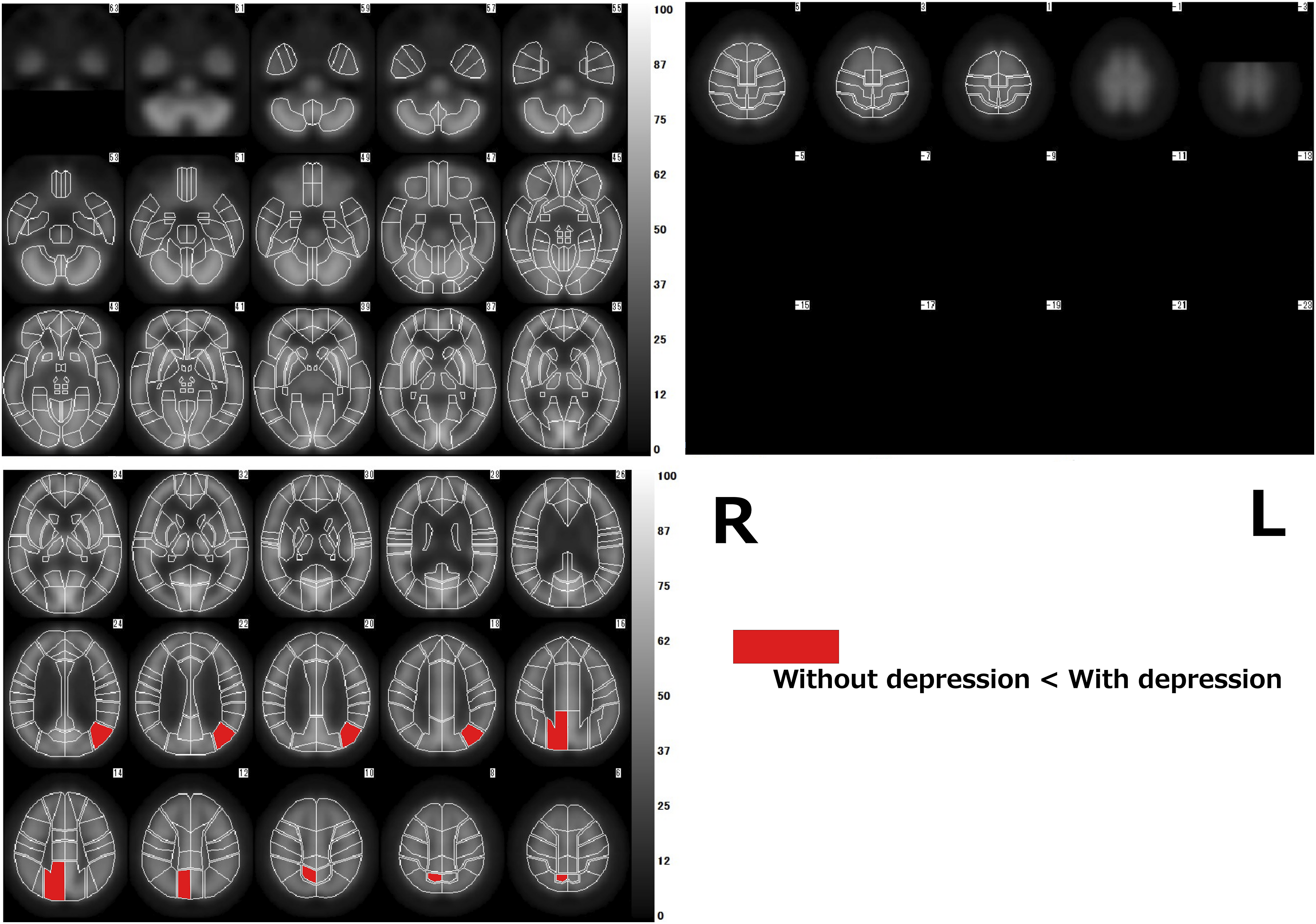
Comparison of regional cerebral blood flow between the group with depression and the group without depression. Red areas indicate increased regional cerebral blood flow in the group with depression compared with that in the group without depression.
Compared with the group without visual hallucinations, the group with visual hallucinations had a significant increase in brain perfusion in the left putamen (P < .05/3).
This group also had a significant decrease in brain perfusion in the left inferior parietal lobule, and the left superior temporal gyrus, as well as in the right primary visual cortex (P < .05/3; Figure 2). Comparison of regional cerebral blood flow between the group with visual hallucinations and the group without visual hallucinations. Red areas indicate increased regional cerebral blood flow, and blue areas indicate decreased regional cerebral blood flow in the group with visual hallucinations compared with that in the group without visual hallucinations.
Compared with the group without auditory hallucinations, the group with auditory hallucinations had a significant increase in brain perfusion in areas, such as the right middle occipital and right inferior occipital gyri (P < .05/3). This group also had a significant decrease in brain perfusion in the left rectal gyrus, left transverse temporal gyrus, and left primary auditory cortex (P < .05/3; Figure 3). Comparison of regional cerebral blood flow between the group with auditory hallucinations and the group without auditory hallucinations. Red areas indicate increased regional cerebral blood flow, and the blue areas indicate decreased regional cerebral blood flow in the group with auditory hallucinations compared with that in the group without auditory hallucinations.
Discussion
The FineSRT program uses 1394 constant ROIs, which can be divided into 52 segments on each hemisphere after anatomical standardization using the SPM99 algorithm. FineSRT is useful for analyzing perfusion conditions because it can display more detailed areas than other analytical techniques. It also allows for the objective assessment of rCBF by setting the ROIs identically on anatomically standardized SPECT images. As described in the introduction, no previous studies of DLB have been performed using FineSRT.
In DLB patients with depression, we found increased brain perfusion in the right upper precuneus and left angular gyrus. The angular gyrus is thought to be involved in language, cognition, and the understanding of metaphors. 11 The parietal cortex, which includes the angular gyrus, is an area in which multiple forms of sensory information, such as somatosensory, visual, and auditory information, are integrated.
There have been previous studies of CBF in PD with depression. Kim et al. 12 revealed that brain perfusion was increased in the left cuneus in depressed PD patients using SPECT, but they also observed that it was decreased in the right superior temporal gyrus and medial orbitofrontal cortex, as well as in the amygdala, anterior cingulate cortex, hippocampus, and parahippocampal gyrus. In addition, Matsui et al. 13 reported hypoperfusion of the left superior and inferior frontal gyri in PD patients with depression. These findings are inconsistent with the results of the present study. The differences in results between these previous studies and ours may be caused by differences in diagnostic and analytical methods.
No previous studies have examined patients with depression using FineSRT. However, consistent with our findings of increased brain perfusion in the left angular gyrus, some studies have reported increased parietal 14 or frontoparietal 15 perfusion in depressed patients using arterial spin labeling MRI. The brain’s default mode network, which is active during relaxed non-task states, consists of areas, which include the parietal, temporal, and medial prefrontal cortices. 16 Our results are compatible with the hypothesis that depression is associated with increased activity in regions of the default mode network, of which the posterior hub includes connections across the parietal and temporal lobes. 17
In our study, decreased brain perfusion in DLB patients with visual hallucinations was identified in the left inferior parietal lobule and right primary visual cortex. Visual information first enters the primary visual cortex and is then output to the higher visual cortices. 18 Neurons in the higher levels of the visual cortices are responsible for the response of visual stimuli to movement.19,20 There have been several reports21,22 that the function of the primary visual cortex is associated with visual hallucinations; the results of our study are consistent with these previous reports. Our results are also compatible with previous SPECT studies.13,23 For example, Matsui et al. 13 reported that hallucinatory PD patients have significant reductions in perfusion in the bilateral inferior parietal lobule and occipital cortex. Moreover, Nagahama et al. 23 reported that visual hallucinations in DLB are related to reductions in perfusion in the bilateral parietal area. In addition, Taylor et al. 24 suggested that functional MRI abnormalities in the lateral occipito-temporal cortex may contribute to visual hallucinations. Our results suggest that the lesion responsible for visual hallucinations may occur in the inferior parietal lobule in addition to the primary visual cortex. Other studies have shown that visual hallucinations are associated with the bilateral dorsolateral-frontal cortex and posterior cingulate metabolism.25,26 In our study, there was no significant difference in rCBF in the superior and medial frontal cortex, which corresponds to the dorsolateral-frontal cortex.
In DLB patients with auditory hallucinations, we identified an increase in perfusion in the right middle occipital and right inferior occipital gyri. In addition, there was a decrease in perfusion in the left rectal gyrus, left transverse temporal gyrus, and left primary auditory cortex. The temporal lobe is involved in language and hearing, whereas the occipital lobe is specialized for various visual formation tasks. A previous study has reported decreased brain perfusion in the left ventral occipital gyrus and bilateral parietal region in DLB patients with auditory hallucinations. 23 These findings are inconsistent with the results of our study. However, using functional MRI, Woodruff et al. 27 suggested that auditory hallucinations in schizophrenia are associated with reduced left and increased right temporal cortical responses to the auditory perception of speech.
We have described the relationship between a range of psychiatric symptoms and rCBF; however, the most notable relationship was between rCBF and visual hallucinations. This is likely because visual hallucinations are a core clinical feature among the diagnostic criteria of DLB and appear frequently in patients. Our findings that decreased rCBF in the right primary visual cortex and left inferior parietal lobule are associated with visual hallucinations in DLB may be useful for differentiating DLB from other diseases in its early stages.
Dementia with Lewy bodies patients are vulnerable to antipsychotics, and incorrect treatment of such individuals can cause serious problems in real-world settings. If the onset mechanisms of psychiatric symptoms are clarified in the early stages of DLB, it may be possible to distinguish DLB from other psychiatric disorders, such as depression and schizophrenia. Appropriate and safe treatments and care will then be possible for these patients.
Associations between depression and tauopathy and amyloidogenesis in DLB have been reported in a previous study. 28 The distribution of Lewy bodies in the amygdala and parahippocampal gyri reported in neuropathological studies has been shown to be associated with a higher frequency of visual hallucinations. 29 Our results are not consistent with such findings. Changes in rCBF may not be the direct result of pathological changes. For example, the primary visual cortex of the occipital lobe is not a common site of Lewy body distribution; however, this region is frequently reported to have a decrease in rCBF. 30 Thus, it is necessary to further explore the precise relationships among clinical symptoms, neurophysiological findings, neuroimaging findings, and pathological findings.
In conclusion, significant differences in perfusion were identified in various brain regions of DLB patients with depression and visual and auditory hallucinations. In the future, our findings may provide clues about the pathomechanisms of these psychiatric symptoms and may enable appropriate diagnosis and treatment of DLB in the early stages.
Limitations
Our study had several limitations. Autopsy was not performed on any of the participants to validate their clinical diagnosis, and our study was relatively small. Larger multicenter studies, preferably in patients with neuropathological confirmation of DLB, are needed in the future. In addition, our study was cross-sectional; thus, a longitudinal study should be conducted. Furthermore, our study participants were only Japanese. Future studies should be conducted in populations with a range of ethnicities.
Footnotes
Acknowledgments
We would like to express our appreciation to Dr. Shiro Hinotsu for statistical advice.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Japan's Ministry of Education, Culture, Sports, Science and Technology (21K07549).
