Abstract
Purpose:
The aim of this study is to examine the potential effect of cilostazol and inflammation on cognitive impairment after stroke in an Asian population.
Methods:
Forty-five patients with cognitive impairment after ischemic stroke using cilostazol were enrolled as the study group and 45 patients using aspirin or clopidogrel were enrolled as the control group. Neuropsychiatric assessments were administered at the start of the study and after 6 months. Multiple logistic regression analysis was used to estimate the association between the cognitive change and cilostazol use. Macrophage polarization were assessed using flow cytometry in 7 patients.
Results:
There were a significantly higher number of patients with peripheral arterial occlusive disease in the cilostazol group. No significant differences were observed in the cognitive change between the cilostazol and control groups. M1 macrophage subset increment were observed in the patient having a declined cognitive change.
Conclusion:
Cilostazol did not make a significant difference in cognitive change after ischemic stroke. M1 macrophage subset increment may indicate post stroke cognitive decline. Due to limited number of subjects, these findings should be examined further in large-scale randomized clinical trials.
Keywords
Introduction
Cognitive impairment and dementia represent a substantial burden on society. 1 After Alzheimer’s Disease (AD), vascular dementia (VaD) is the second most common type of dementia. 2 After a stroke, as many as two-thirds of patients experience cognitive impairment or decline. The presence of cognitive impairment is associated with a substantial increase in the risk of developing dementia. 3 A 15 year follow up study found that the prevalence of post stroke cognitive impairment was 22% and it remained persistently high over time. 4 Methods for stopping or delaying the onset of cognitive impairment following a stroke have become an important issue.
The relationship between stroke and cognitive impairment has attracted significant attention in recent decades. Stroke may induce or exacerbate neurodegenerative disease, 5 or neurodegenerative disease may amplify brain injury and cognitive deficits after stroke. 6 Amyloid load in the brain and chronic inflammation have been previously reported as the most important factors associated with post stroke cognitive decline. 7 Vascular mechanisms and neuro-inflammation is a key component to the pathological findings that contributed to subsequent cognitive decline. 7,8
Microglia/macrophages are the major immune cells involved in the pathophysiological processes of brain damage induced by stroke. 9 In peripheral blood, macrophages display remarkable plasticity that enables them to perform distinct and even opposing function, such as release of either proinflammatory and anti-inflammatory cytokines and growth factors, in response to different environmental cues. 10 Depending on the activation state, macrophages can be designed as either classical activated (M1), with pro-inflammatory properties, or alternatively activated (M2) cells, which mediate anti-inflammatory response. 11 Through M2 phonotype induction in macrophages, neurological outcomes improve in well-established stroke models. 12
Cilostazol is a phosphodiesterase III inhibitor and an antiplatelet with fewer bleeding risks than aspirin in patients with small-vessel disease. 13 It can increase cyclic adenosine monophosphate levels in cells, which can lead to inhibition of platelet aggregation, protective effects in endothelial cells, vasodilation in smooth muscle cells and neuroprotective effects as an antioxidant and anti-apoptosis agent. 14 In a rat model of chronic cerebral hypoperfusion, cilostazol improved performance in neurobehavior tests, attenuated neuroinflammation and reduced white matter disintegration. 15 Saito and Ihara supposed that cilostazol exhibits pleiotropic capabilities against dementia by suppressing amyloid-β (Aβ) production in neurons, enhancing Aβ clearance through the perivascular drainage system and inhibiting platelet aggregation. 16 Our previous study found that cilostazol slowed cognitive decline when added to the drug regimen of patients with mild dementia who were already receiving donepezil. 17 Another study using the Taiwan Health Insurance Database, reported that patients using cilostazol had a significantly decreased incidence of all causes of dementia with dose-dependent effect. 18 Sakurai et al investigated the effects of cilostazol in patients with Alzheimer’s Disease and cerebrovascular disease. Compared to control group, cilostazol might have a preventive effect on cognitive decline and increase regional cerebral blood flow in the right anterior cingulate lobe. 19 Therefore, cilostazol is potentially useful for the treatment of post stroke cognitive impairment and a prospective clinical trial is needed. For these reasons, the current study was developed as a pilot study to examine the effects of cilostazol on post stroke cognitive impairment beyond stroke prevention and the prediction of cognitive change by macrophage polarization.
Materials and Methods
Study Population
A prospective open-label study was conducted at Kaohsiung Municipal Ta-Tung Hospital and Hsiao-Kang Hospital in southern Taiwan. Patients aged ≥50 years with a diagnosis of ischemic stroke, stable neurological deficits without obvious physical limitation and reported cognitive impairment after stroke were recruited from the outpatient departments. The diagnosis of stroke was based on a clinical neurological examination together with neuroimaging. The classification of stroke subtypes was based on the TOAST criteria. The experimental group (case) in the current study were patients using cilostazol (100 mg twice daily) as an antiplatelet drug for secondary prevention of stroke, while the control group were patients using aspirin (100 mg daily) or clopidogrel (75 mg daily). The patients were assigned to cilostazol or control group by clinicians according to their clinical condition, health insurance guidelines of Taiwan or patients’ preference. Patients who had an active diagnosis of drug or alcohol abuse or dependence, delirium, or were unable to complete the neuropsychiatric assessment and receive regular follow up were excluded. Patients with a clinical diagnosis of AD, those aged >75 years with a medial temporal lobe atrophy (MTA) 20 score of ≥3 and those aged <75 years with an MTA score of ≥2 were excluded from the study. Also, patients with recurrent stroke, those who change medication or drug dosage during the follow-up period were excluded.
Evaluation
Demographic data, including age, gender and education were collected as well as the patient’s medical history, including hypertension, diabetes mellitus and peripheral arterial occlusive disease (PAOD). Brain magnetic resonance imaging (MRI) was performed at the start of the study. A set of comprehensive neuropsychological assessments, including mini-mental state examination (MMSE), Cognitive Assessment Screening Instrument (CASI), clinical dementia rating (CDR)-global score and sum of boxes (CDR-SB) were performed at the beginning of the study and at 6 months. The first neuropsychological assessments were administered after 3 months post stroke. These assessments were performed by a senior neuropsychologist and an experienced physician based on information provided by a knowledgeable collateral source (usually a spouse or adult child). Patients with a second MMSE or CASI score equal to or higher than their first MMSE or CASI score (ΔMMSE or CASI score ≥0) were considered as having an improved cognitive change, whereas those with a lower second score were considered as having a declined cognitive change. Similarly, patients with a second CDR-SB score lower than or equal to their first CDR-SB score (ΔCDR-SB score ≤0) were considered as having an improved global status change, whereas patients with a higher score were considered as having a declined global status change.
Flow Cytometry for Determining the Macrophage Polarization
In our study, we used flow cytometry for determining the macrophage polarization. We extracted peripheral blood mononuclear cells for survey from the subjects. We stained the samples with purified antimacrophage antibody PM-2 K (AbD Serotec, Kidlington, UK) followed by anti-mouse IgG-FITC, washed, and then stained with other fluorochrome-labeled antibodies, including CXCR1-Alexa 700 (R&D, Minneapolis, USA), CD14-PE/Cy7, CD3-Pacific blue (BD biosciences, San Jose, CA), CD19-Pacific blue, CD86DerCP (eBioscience, San Diego, USA), CCR2-PE (BioLegend, San Diego, CA), and CCR7-APC (MiltenyiBiotec, Bergisch Gladbach, Germany) as well as appropriate isotype controls. The whole staining procedure took about 3 hours. We ran the samples on an LSRII flow cytometer (BD biosciences, San Jose, CA) collecting data on 106 live cells. We analyzed the data using FCS Express software (BD biosciences, San Jose, Red CA). In summary, we isolated circulating monocytes from the peripheral blood mononuclear cells directly and analyzed for markers of macrophages. The work in our study was done on macrophages after staining the isolated monocytes in autologous whole blood to mimic that in vivo condition. We used the cells as circulating macrophage-like cells instead of macrophages to reflect the macrophage polarization patterns. PM-2K+ cells could be divided into 2 subsets based on their CD14 expression, namely PM-2K+ CD14+ and PM-2K+ CD14–subsets. In the PM-2K+ cell population, cells were defined as M1-like macrophages for CCR7+CD86+, while those were defined as M2a–, M2b–, and M2c–like macrophages for CCR7–CXCR1+, CCR7+CD86–, and CCR7–CCR2+ respectively (Figure 1).

Macrophage polarization.
The current study was approved by the Institutional Review Board of Kaohsiung Medical University Hospital (KMUHIRB-E(II)20160075) and written informed consent was obtained from all patients or their legal representatives prior to their inclusion within the study. The informed consent was obtained after assignment.
Statistical Analysis
Data was presented as the mean ± standard deviation (SD) or proportion. The chi-squared test was used for categorical data, and the Student’s t-test was used for continuous data. Multiple logistic regression models were conducted to calculate the odds ratios (ORs) and 95% confidence intervals for the association between cognitive or global status change and cilostazol use. This model was adjusted for age, gender and educational level. Analyses were performed using SPSS (SPSS Inc., Chicago, IL, USA). A 2-tailed P-value <0.05 was considered to indicate a statistically significant difference.
Results
One hundred and thirty-six patients were the candidates for this study and we explained and introduced this study to all of them. One hundred and twenty-four patients agreed with the participation. During the follow-up period, 34 patients were excluded due to change of medications or dosage in 12 patients, loss of follow up in 13 patients, refuse regular follow up in 6 patients and recurrent stroke in 3 patients.
A total of 90 patients who completed the full 6 months follow up were included in the statistical analysis; there were 45 patients in each group (cilostazol and control). The mean age of all participants was 69.8±11.8 years (range 50-94) and 71.9% of the participants were male. The average number of years education the patients had received was 7.8±4.9 (range 0-19). The proportion of patients with PAOD, hypertension and diabetes mellitus were 28.7%, 73% and 39.3%, respectively. Grossly, there was no significant difference in the stroke subtypes in these 2 groups. The frequency of stroke subtypes (large artery atherosclerosis vs small vessel occlusion) were 40 vs 60% and 46.4 vs 53.6% in the cilostazol and control group, respectively (p = 0.658). At baseline, the mean CASI score of all participants was 72.0±21.2; MMSE was 20.9±6.7; and CDR-SB was 2.7±3.2. The distribution of CDR-global score of 0, 0.5, 1 and 2 were 24.1%, 57.5%, 11.5%, and 6.9%, respectively. The follow up CASI score was 73.4±21.5; MMSE was 21.4±6.9; and CDR-SB was 2.5±3.3. There were no significant differences in age, gender, education level, proportion of diabetes or hypertension between the cilostazol and control groups at baseline (Table 1). However, there were a significantly higher number of patients with PAOD in the cilostazol group. This group also had a higher number of elderly patients and a worse cognitive performance, although these differences were not significant (Table 1). After 6 months, 67.1% of all patients had an improved CASI score, 69.7% had an improved MMSE score and 72.9% had an improved CDR-SB score. No significant differences were observed in the cognitive (MMSE, CASI) and global status (CDR-SB) changes between the cilostazol group and the control group (Table 2). However, age was a significant predictor for cognitive change. The risk of having cognitive decline was 1.05-fold each year (adjusted p = 0.048 for ΔMMSE).
Demographic Characteristics of Patients Using Cilostazol, Aspirin or Clopidogrel.
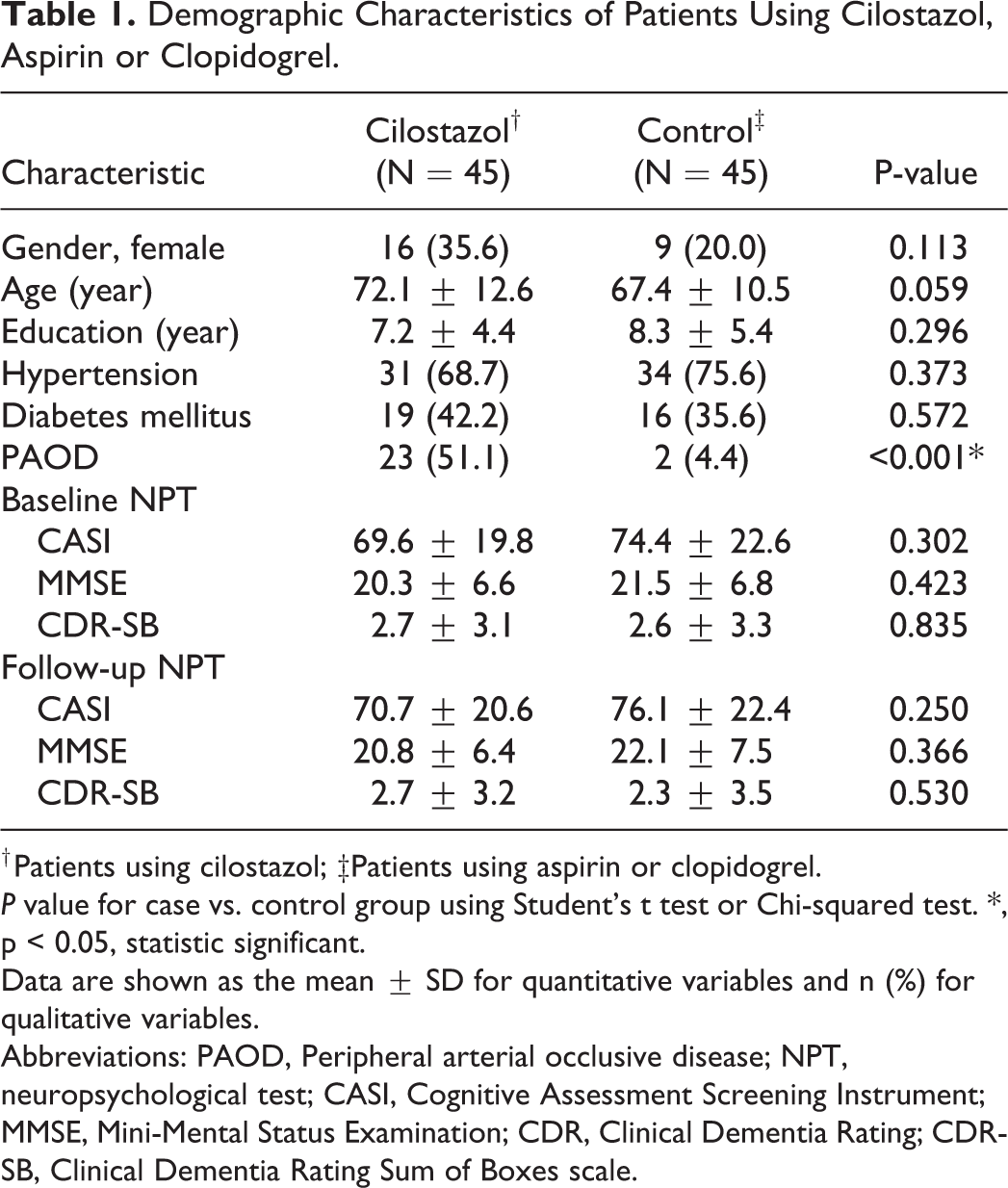
† Patients using cilostazol; ‡Patients using aspirin or clopidogrel.
P value for case vs. control group using Student’s t test or Chi-squared test. *, p < 0.05, statistic significant.
Data are shown as the mean ± SD for quantitative variables and n (%) for qualitative variables.
Abbreviations: PAOD, Peripheral arterial occlusive disease; NPT, neuropsychological test; CASI, Cognitive Assessment Screening Instrument; MMSE, Mini-Mental Status Examination; CDR, Clinical Dementia Rating; CDR-SB, Clinical Dementia Rating Sum of Boxes scale.
Factors to Cognitive and Global Status Change After 6 Months Follow Up by Logistic Regression.

ΔCASI = 2nd CASI—1st CASI; ΔMMSE = 2nd MMSE—1st MMSE; ΔCDR-SB = 2nd CDR-SB—1st CDR-SB.
ORs are based on comparing odds of decline (ΔCASI < 0, ΔMMSE < 0, ΔCDR-SB > 0) vs. improve (ΔCASI ≥ 0, ΔMMSE ≥ 0, ΔCDR-SB ≤ 0).
*, p < 0.05, statistic significant.
Abbreviations: OR, Odds Ratio; CI, Confidence Interval; CASI, Cognitive Assessment Screening Instrument; MMSE, Mini-Mental Status Examination; CDR-SB, Clinical Dementia Rating Sum of Boxes scale.
Since there are different cognitive domains, factors for CASI subdomain change were further examined by multivariate regression analysis (Table 3). Again, this revealed no significant differences between the cilostazol treated group and the control group. Age was a statistically significant predictor of remote memory, orientation and language domain decline (adjusted p = 0.008, 0.002 and 0.004, respectively). Sex was also associated with cognitive subdomain changes. Males had a significantly lower risk of decline in remote memory and abstract domain compared with females (OR = 0.06 and 0.21; adjusted p = 0.021 and 0.019, respectively).
Factors to CASI Subdomain Change After 6 Months Follow Up by Logistic Regression.

ΔCASI subdomain = 2nd CASI subdomain—1st CASI subdomain.
ORs are based on comparing odds of decline (ΔCASI subdomain <0) vs. improve (ΔCASI subdomain ≥0).
*, p < 0.05, statistic significant.
Abbreviations: CASI, Cognitive Assessment Screening Instrument; OR, Odds Ratio; CI, Confidence Interval; Remote, Remote memory; Recent, Recent memory; Fluency, Verbal fluency; Abstract, Abstract thinking.
Among the patients included at baseline, macrophage polarization with related subsets were assessed using flow cytometry in 7 patients (Table 4). Six patients (patient A.B.D.E.F.G) had an improved CASI score and 1 patient (patient C) had a declined score after 6 months. Six patients (patient B-G) used aspirin for stroke prevention and 1 patient (patient A) used cilostazol. PM2K+CD14+ (G3) M2b macrophage subset decrement and M1 macrophage subset increment were observed in the patient (patient C) having a declined cognitive change compared with those having an improved cognitive change (Figure 2).
Demographic Information, Cognitive Change and Percentage of Macrophage Polarization Subsets of 7 Patients.

Cognitive change: improve (ΔMMSE or CASI score ≥0); decline (ΔMMSE or CASI score <0).
Macrophage polarization subsets: PM-2K+ CD14+ (Gate 3; G3), PM-2K+ CD14–(Gate 4; G4), M1–like macrophages for CCR7+CD86+, M2a–like macrophages for CCR7–CXCR1+, M2b–like macrophages for CCR7+CD86–, M2c–like macrophages for CCR7–CCR2.+

Percentage of macrophage polarization subsets in 7 patients.
Discussion
In the current study, the clinical course of post stroke cognitive changes was described. A total of 67.1% of patients improved their CASI score, 69.7% improved their MMSE score and 72.9% improved their CDR-SB score at 6 months follow up. There was no significant difference in the cognitive and global status change between the cilostazol and control groups. M1 macrophage subset increment were observed in the patient having a declined cognitive change. To the best of our knowledge, this is the first study to provide an objective neuropsychological assessment of post stroke cognitive change in Taiwanese patients and to explore the role of cilostazol, as well as macrophage polarization, in cognitive change after ischemic stroke.
In the current study, more than 60% of patients improved their cognitive performance and more than 70% improved their global status after 6 months. This was comparable to the previous study. A study conducted by Britta et al also demonstrated that cognition improved significantly between 2 and 6 months after stroke. 21 Therefore, even a cognitive decline may develop post stroke, a proportion of patients with cognitive impairment improve in a period in which acute changes have stabilized. Although cognitive improvement is seen ≤6 months post stroke, long-term cognitive decline are prevalent. 22,23 There are some possible determinants of post stroke cognitive impairment. One of them is the stroke characteristic. The stroke severity was reported to be associated with cognitive impairment 3 months post stroke. 4 Cognitive impairment rates were also high in anterior circulation infarct and large artery atherosclerosis subtypes up to 5 years after stroke. 4 Cognitive impairment in lacunar infarct and small vessel occlusion strokes was increasingly prevalent during the first 3 to 4 years after stroke. 4 In a meta-analysis a similar proportion of patients with lacunar and non-lacunar stroke had mild cognitive impairment or dementia up to 4 years after stroke. 24
Although no significant differences in the overall outcome of cognitive change were noted between the cilostazol and control groups after 6 months follow up, cilostazol may be considered as “non-inferior” for post stroke cognitive change due to inherent bias. There was higher rate of PAOD in the cilostazol group; according to the national health insurance guidelines of Taiwan, cilostazol is suggested for the reduction of symptoms of intermittent claudication in individuals with PAOD. Therefore, a higher number of PAOD patients were recruited into the cilostazol group. Low ankle-brachial index or PAOD are associated with worse cognitive function. 25 Nonsignificant poorer cognitive function and older age at baseline were also noted in the cilostazol group. To combat this, a randomized case control study should be conducted in the future.
The negative finding of cilostazol on post stoke cognitive impairment in this study was similar to those for previous studies reporting that potential therapeutic agents did not exhibit beneficial effects on patients with developed and progressed AD. 26 To explain the disappointing results of several randomized controlled trials, many design problems in the researches for AD were reported, including target group selection (various types of neuropathology and mixed causes of dementia), disease stage (too advanced stage for a disease-modifying effect of a specific drug), optimization of drug dosage and treatment duration and the outcome measurements. 26 These issues should also be addressed to examine the effect of cilostazol on cognitive impairment after stroke.
Age was found to be a predictor of cognitive change, which was in line with prior reports. 27 Compared with males, females had increased decline of remote and abstract thinking function after stroke. According to a review study, 28 sex difference does exist in dementia of a vascular origin. Males are at greater risk of developing VaD, but strokes tend to be more severe in females. 29 Risk factors for VaD are more common in males, but they have a more severe impact in females. 30 In addition, female sex is a stronger predictor of pre-stroke dementia, which may be a sign or cause of a primary degenerative pathology. 31 Females are also more likely to experience poststroke depression, which is another risk factor for dementia. 32
In this study, macrophage polarization with related subsets were assessed in 7 patients. M2b macrophage subset decrement and M1 macrophage subset increment were observed in the patient having a declined cognitive change compared with those having an improved cognitive change. The neuroinflammation, an important factor of post stroke cognitive decline, is demonstrated by M1 microglial activation, often associated with loss of neurons due to the production of damaging reactive oxygen species and toxic intermediates. Alternative M2 microglial activation, on the other hand, is important for resolution of this inflammation. 33 Consistent M1 macrophage subset decrement were observed in the 6 patients having an improved cognitive change, which means that resolution of neuroinflammation might improve post stroke cognitive outcomes. Our another work on AD shows similar result, in which various neuroinflammatory processes also have the impact on the pathology. 34
The current study had some limitations that should be addressed. First, the number of participants was small and macrophage polarization were only assessed in 7 patients. This pilot study with preliminary findings should be clarified by randomized clinical trials with large case number in the future. Second, a 6 months study period may be insufficient for assessing cognitive changes. Third, the diagnosis of AD or mixed type cognitive impairment cannot be totally excluded, however the MTA score was used to try and avoid this. A high MTA-score is very sensitive for the diagnosis of AD and is present in the vast majority of patients with AD. 20 Patients with a high MTA score were not included in the current study. Fourth, we did not record stroke severity by National Institute of Health Stroke Scale (NIHSS), however these patients were recruited from the outpatient departments with mild stroke without obvious physical limitation. Fifth, we did not present the interval of the index stroke and the first neuropsychological assessments in these 2 groups. However, the participants were recruited from the outpatient departments with stable neurological deficits and those who are in the acute stage of stroke were excluded. The first neuropsychological assessments were administered after 3 months post stroke, therefore the performance of first assessments may be less affected by the interval of the index stroke and the first assessments and we reported the intra-individual changes. Another limitation is that the present study did not include data on other potential risks for cognitive impairment (e.g. medicine or lifestyle factors), and no adjustments were made for medical history. Also, intracranial and extracranial arterial stenosis were not evaluated. However, there is no consistent data for its association with progressive decline. 35 Finally, at present there is no consensus on the definition of “improved” or “declined” post stroke cognitive change, which can make comparison and reporting difficult in longitudinal studies.
Conclusion
The results of the present study indicate that cilostazol did not make a significant difference in cognitive and global status change after ischemic stroke compared with other antiplatelet drugs. M1 macrophage subset increment may indicate post stroke cognitive decline. These results highlight the need for large-scale, double-blind, long-term studies to clarify the role of cilostazol and macrophage polarization on cognitive impairment after ischemic stroke. Further investigations of macrophage subsets should be discussed in detail for their therapeutic potential.
Footnotes
Acknowledgments
This study was supported by the clinical psychologists and assistant of the Department of Neurology, Kaohsiung Municipal Ta-Tung Hospital and Kaohsiung Municipal Hsiao-Kang Hospital, Kaohsiung, Taiwan and partially by Kaohsiung Medical University Research Center Grant (KMU-TC109B03).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Kaohsiung Municipal Ta-Tung Hospital (kmtth-108-009).
Significance Statement
The first study to provide an objective neuropsychological assessment of post stroke cognitive change in Taiwanese patients. Overall, 60-70% of the patients improved their cognition after 6 months follow up. A pilot study to examine the effects of cilostazol on post stroke cognitive impairment beyond stroke prevention and the prediction of cognitive change by macrophage polarization. Although no significant differences in the overall outcome of cognitive change were noted between the cilostazol and control groups after 6 months follow up, cilostazol may be considered as “non-inferior” for post stroke cognitive change. M1 macrophage subset increment may indicate post stroke cognitive decline. Further investigations of macrophage subsets should be discussed in detail for their therapeutic potential.
