Abstract
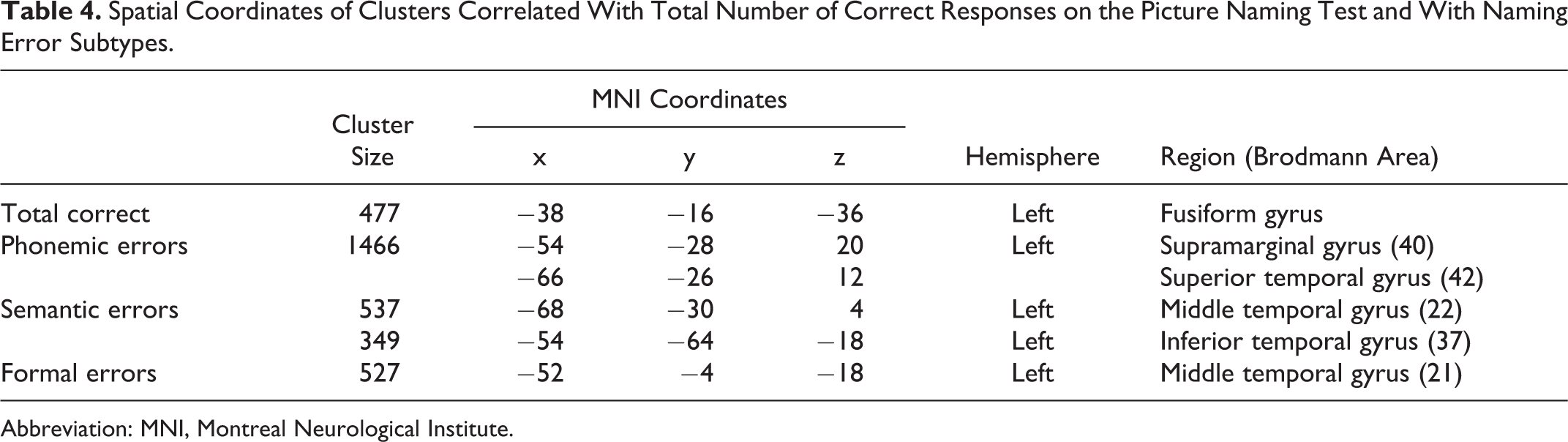
Patients with Alzheimer disease (AD) produce a variety of errors on confrontation naming that indicate multiple loci of impairment along the naming process in this disease. We correlated brain hypometabolism, measured with 18fluoro-deoxy-glucose positron emission tomography, with semantic and formal errors, as well as nonwords deriving from phonological errors produced in a picture-naming test by 63 patients with AD. Findings suggest that neurodegeneration leads to: (1) phonemic errors, by interfering with phonological short-term memory, or with control over retrieval of phonological or prearticulatory representations, within the left supramarginal gyrus; (2) semantic errors, by disrupting general semantic or visual-semantic representations at the level of the left posterior middle and inferior occipitotemporal cortex, respectively; (3) formal errors, by damaging the lexical–phonological output interface in the left mid–anterior segment of middle and superior temporal gyri. This topography of semantic–lexical–phonological steps of naming is in substantial agreement with dual-stream neurocognitive models of word generation.
Keywords
Introduction
Models of word production posit a process composed by multiple representational/processing levels and sublevels. 1 -6 Essential steps following activation of the concept from the semantic system are retrieval of the corresponding lexical entry and phonological word form and activation and implementation of the associated articulatory program. The lexical and phonological stages of this process have been further fractionated into a lexical–semantic, a lexical–phonological, and a postlexical–phonological level, whose content is putatively related to, respectively, meaning, abstract lexical–phonological structure and phonological articulatory features of the word to be generated. 7 -9 Three types of word production errors are known to arise from dysfunction of stages between conceptual activation and articulation: semantic errors, formal errors, and nonwords deriving from phonological errors. Semantic errors (eg, dog for cat) are real words with a (purely) conceptual relationship with the target word, entailing a link with the lexicon and awareness of identity of the stimulus. Along the multistep process of word generation, they may therefore be mapped at the lexical–semantic level, but impairment of conceptual representations, or of control over their retrieval, is also known to cause semantic errors. 5,7,10 Formal errors (mat for cat) are also real words, whose (purely) phonological resemblance with the target word denotes awareness of its phonological form. These errors can therefore be associated with the lexical–phonological processing 2,9,11 -15 as well as with a postlexical–phonological level. 2,11,12 Also phonemic errors (cag for cat) are phonologically similar to the target word, implying some access to the phonological word form but are not represented within the lexicon. They are thus best linked to a postlexical–phonological level of word generation.
The neuroanatomical substrate of these 3 categories of errors and underlying cognitive processes are not completely defined. Three left hemisphere regions contend for locus of damage associated with semantic errors: posterior inferior–middle 5,8,16,17 temporal cortex, posterior middle temporal gyrus, 18 -21 and mid 3,22,23 or mid–anterior 7,24 -26 portion of the same gyrus (other areas correlated with semantic errors have been attributed a role in general semantic knowledge left angular gyrus and anterior ventral temporal cortex or in control of semantic retrieval left inferior parietal–frontal connections rather than in the lexical–semantic interplay). 5,7,10 Knowledge about the neural correlates of formal errors is relatively scarce, being limited to results of 2 recent studies that have applied a lesion symptom mapping approach to heterogeneous computational parameters of word production impairment. 7,27 Overall, these findings confirm the dual, lexical, and phonological, nature of formal errors and accordingly suggest a dual lesional pattern: Lexical-based formal errors would be associated with damage to the mid–anterior segments of the left middle and superior temporal gyri, and phonological-based errors with damage along a left superior temporal–inferior frontal path. Phonemic errors have extremely complex anatomical correlates, involving a large array of areas within the left hemisphere: mid and posterior superior temporal gyrus and sulcus, 3,8,18,23,27,28 the so called Sylvian–parietal–temporal (Spt) area, 5,6,8,27,29,30 supramarginal and post- and precentral gyri, 7,20,26,27,31 -33 as well as inferior frontal cortex and insula. 3,7,8,26,32,33 Within the framework of the dual stream model of speech, 5 semantic errors are allocated to the ventral stream, which mediates conceptual–lexical interactions, phonemic errors to the dorsal stream, which acts as a lexical–phonological–articulatory interface, and formal errors to both streams.
Patients with Alzheimer disease (AD) often have word finding difficulties in spontaneous speech, tend to score poorly on formal tests of confrontation naming since early dementia stages, and deteriorate with disease progression. 34,35 Qualitative scrutiny of picture naming performance in these patients reveals a great variety of incorrect responses including omissions (no answer or “don’t know” responses), circumlocutions (the fruit associated with Adam and Eve for apple), superordinates (fruit), visual errors (circle), and also semantic phonemic and formal errors. 36 -47 Although impairment of semantic memory or of access to semantic knowledge is considered the main cause of naming deficits in this form of dementia, 48 -52 such different types of incorrect responses suggest dysfunction at other levels of word production. In fact, naming performance of patients with AD has been shown to vary with the visual complexity of the pictorial stimuli, 53 -55 or to be influenced by phonological cues or phonological priming. 43,47,55,56 In AD populations, correlation between performance on picture naming and distribution of atrophy on magnetic resonance imaging (MRI) or of hypometabolism on 18fluorodeoxyglucose positron emission tomography (FDG-PET) has invariably shown the involvement of a locus classically associated with semantic memory, that is, the left anterior ventral temporal cortex, 57 -65 but has also highlighted the contribution of other nodes of the left hemisphere naming network that intervene in other steps of the naming process: the dorsolateral 58,63 or mesial 59,61 temporal surface, the occipito-temporal junction, 57,62,64 the inferior parietal lobule, 58,63 as well as the posterior frontal cortex. 57 -59,61,65
All of these studies, however, have considered only total number of correct answers as index of naming performance, whereas in the current study we focused on lexical and phonological error subtypes in order to obtain a more fine-grained topography of postsemantic and prearticulatory levels of the naming process. We explored the specific metabolic correlates of semantic, phonemic, and formal errors produced by patients with AD in a picture-naming task, with the final aim to confirm and possibly better define neural maps of lexical–phonological deficits in word generation. We hypothesized that semantic and formal errors would be associated with hypometabolism in the posterior and anterior regions of the left lateral temporal cortex, respectively, due to their semantic–lexical and lexical nature and that phonemic errors could be associated with dysfunction of the more posterior segment of the left dorsal pathway, since temporo–parietal regions are typical loci of degeneration in AD, whereas frontal or fronto–parietal areas tend to be involved only in a very advanced disease stage.
Material and Methods
Participants
Patients were recruited from the memory clinic of S. Gerardo Hospital, Monza. Criteria for inclusion were a diagnosis of probable AD according to standardized criteria by the National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer’s Disease and Related Disorders Association 66 (NINCDS-ADRDA), a Mini Mental State Examination 37 (MMSE) score ≥ 18 of 30, a minimum of 5 years of education, and Italian as mother tongue. Exclusion criteria were severe uncompensated eye diseases, large and/or multiple focal vascular lesions on brain MRI, and history of neuropsychiatric disorders, brain injury, mental insufficiency, or substance abuse.
A total of 63 patients with AD, 29 men and 34 women, were included in the study. Their individual sociodemographic and clinical features are reported in Appendix 1. Their average characteristics indicated a mild-to-moderate disease stage (Table 1).
Study Cohort’s Sociodemographic and Clinical Characteristics.

Abbreviation: MMSE, Mini Mental State Examination.
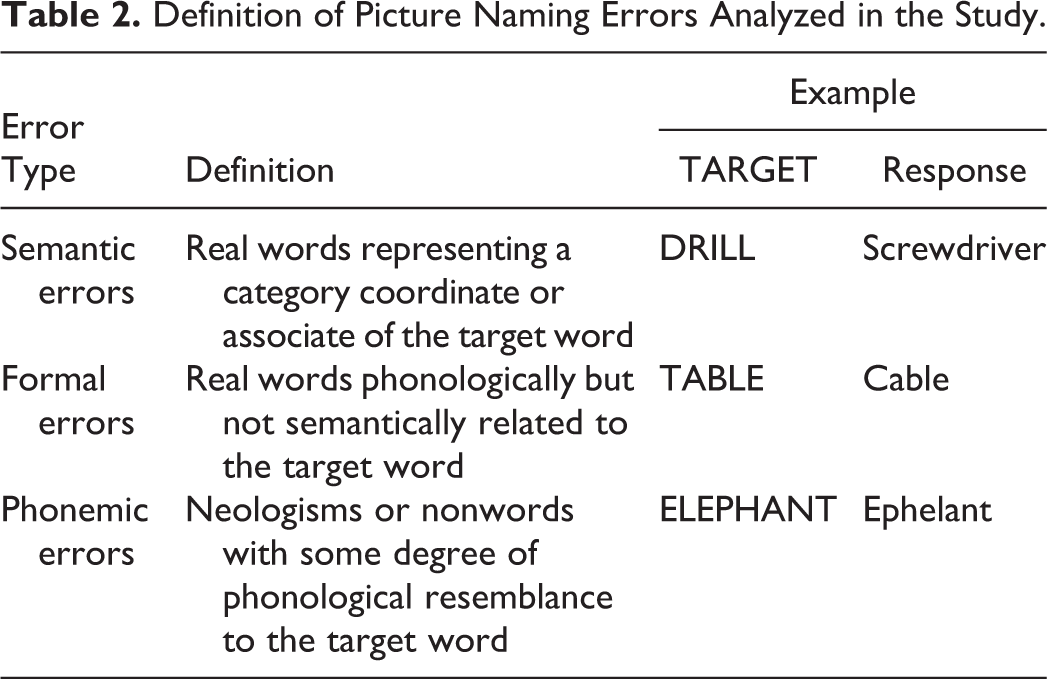
18Fluorodeoxyglucose positron emission tomography scans showed significant hypometabolism in the temporal and posterior parietal regions, with left hemisphere predominance (Figure 1). Positron emission tomography with tracer for amyloid deposits was also available for 13 (20.6%) patients and was positive for all of them.

Distribution of hypometabolism in patients with Alzheimer disease compared with healthy controls. Clusters are shown at P < .05 FWE-corrected and a minimum size of 100 voxels.
All participants were unpaid volunteers and signed a written informed consent. The study was approved by our institution ethics committee and carried out in accordance with the ethical standards of the Helsinki Declaration.
Picture Naming and Errors Categorization
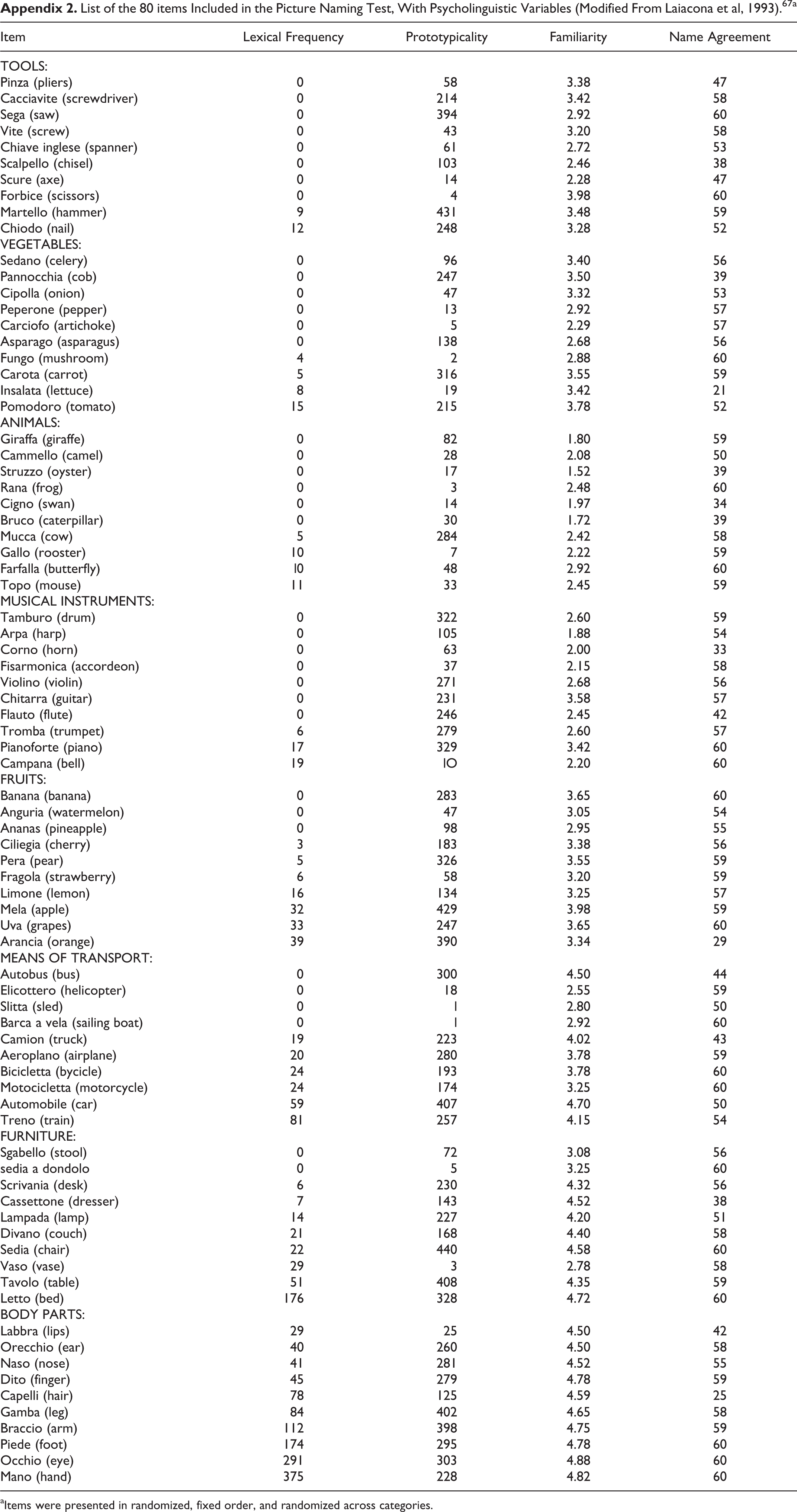
Confrontation naming was assessed as part of a neuropsychological battery tapping the main cognitive domains (attention, short- and long-term verbal memory, visuospatial and executive abilities, verbal fluency and language comprehension, mood and behavior), using a standardized test 67 composed by 80 stimuli of the Snodgrass and Vanderwart pictorial set, 68 10 for each of the following 8 categories: animals, fruits, vegetables, body parts, furniture, tools, vehicles, and musical instruments (see Appendix 2 for the list of individual items and their psycholinguistic variables). Items were presented in randomized, fixed order (and randomized across categories). Patients were asked to name aloud the item shown in the drawing displayed at the center of an A4 sheet, with no time limits. All responses were recorded by the examiner, but only the first utterance was taken into account for computing the final accuracy score (repairs were therefore scored as errors). Maximum number of correct responses is 80 and cut off for normality is an age- and education-adjusted score ≥ 68. 67
Phonemic, semantic, and formal errors were classified by consensus between 2 raters (M.G. and J.S.) following definitions reported in Table 2. Phonemic errors were neologisms or pseudowords derived from changes in the phonology of the target word. Semantic errors were real words related to the target by a category–coordinate or associate relationship, but with no phonological or purely visual relationship (like in misrecognitions, eg, box for drum, ball for orange). Formal errors were real words with phonological, but not semantic, resemblance to the target. For the sake of specificity, responses meeting the definition of semantic or formal errors were excluded from the analysis if they were double errors (eg, a response phonologically related to a semantic relative of the target, like ephelant for giraffe), or if participants perseverated a response from a previous trial. If multiple incorrect responses were provided for a same item, they were all counted as errors and included in the analysis, in an attempt to maximize the number of errors within each category.
Definition of Picture Naming Errors Analyzed in the Study.
Other categories of errors produced during picture naming, such as omissions, circumlocutions, superordinates, and visual errors, were recorded but not analyzed as they were not of interest for the current study.
Acquisition, Processing, and Analysis of FDG-PET Imaging
18Fluoro-deoxy-glucose positron emission tomography was performed within 3 months from completion of the naming test in the Department of Nuclear Medicine of S. Gerardo Hospital, on a General Electric Discovery LS PET/CT scanner. First, computed tomography was performed for attenuation correction, then PET scans were acquired for 15 minutes, with a thickness of 3.27 mm and a matrix of 128 × 128 pixels, and finally reconstructed following an ordered subset expectation maximization algorithm. Images were subsequently processed with Statistical Parametric Mapping (SPM) 8 (Wellcome Department of Imaging Neuroscience, London, United Kingdom; https://www.fil.ion.ucl.ac.uk/spm, running on MATLAB R2015a (MathWorks Inc, Sherborn, Massachusetts): They were reoriented along the anterior–posterior commissure, spatially normalized to the Montreal Neurological Institute reference space using an FDG-PET dementia-specific template provided on SPM, 69 and smoothed with an isotropic 3D Gaussian kernel of 16 mm FWHM.
Two types of analysis were conducted with SPM on FDG-PET images. First, distribution of hypometabolism in patients with AD was assessed, for descriptive purposes, by comparing their images with images acquired on the same PET scanner in a group of 30 neurologically healthy controls. Controls were disease-free oncologic patients undergoing PET for disease staging (14 female, mean age 66.5 years, mean education 9.1 years, mean MMSE score 28.9). The comparison was carried out with the “2-sample t test” function in SPM, including age and sex as covariates. Second, the areas of hypometabolism associated with raw number of correct naming responses as well as phonemic, semantic, and formal errors were assessed within the cohort of 70 patients. This correlation between metabolism and naming was carried out with the “regression analysis” function in SPM, including age, sex, and MMSE as covariates. For both analyses (groups comparison and correlations), significance was set at P < .05 family wise error (FWE)-corrected and only clusters with a minimum size of 100 voxels were taken into account. Anatomical labeling of loci of hypometabolism was carried out with Talairach atlas and Automatic Labeling atlas 70 u using SPM8-integrated toolbox WFU_PickAtlas.
Results
Naming Performance
Mean score on the picture-naming test indicated only mild naming impairment (Table 3), but there was great interindividual variability, as evident from Appendix 1, which shows the individual-naming scores. In particular, only one-quarter and one-third of the entire patients’ sample produced at least one phonemic or one formal error, respectively, and proportion of each of the 3 types of errors out of all the naming errors made by individual patients ranged from 2% to 20% for formal errors, to 25% for phonemic errors, and to 33% for semantic errors. The most numerous errors were semantic errors, followed by phonemic errors, while formal errors were the least common category (Table 3).
Patients’ Average Performance on the Picture Naming Test.a

a Percentages shown in brackets were calculated out of total number of stimuli (80) for total correct, and out of total number of errors produced by the entire cohort, for errors. Please note that total number of errors may not correspond to the difference between maximum test score and number of correct responses since multiple errors produced by a patient for a same item were all counted and included in the analyses.
Metabolic Correlates of Picture Naming: Number of Correct Responses and of Error Subtypes
Figure 2 shows the cluster of hypometabolism within the left ventral temporal cortex that was significantly correlated with a lower total number of correct naming responses (see Table 4 for anatomical coordinates). Figure 3 reports clusters of hypometabolism that correlated significantly with error subtypes and Table 4 lists their anatomical coordinates. Phonemic errors are associated with hypometabolism in the left supramarginal gyrus (in red), semantic errors with hypometabolism in the posterior left middle temporal gyrus and posterior inferior temporal gyrus (in blue), and formal errors with hypometabolism in the mid–anterior segment of the left middle temporal gyrus (in green).
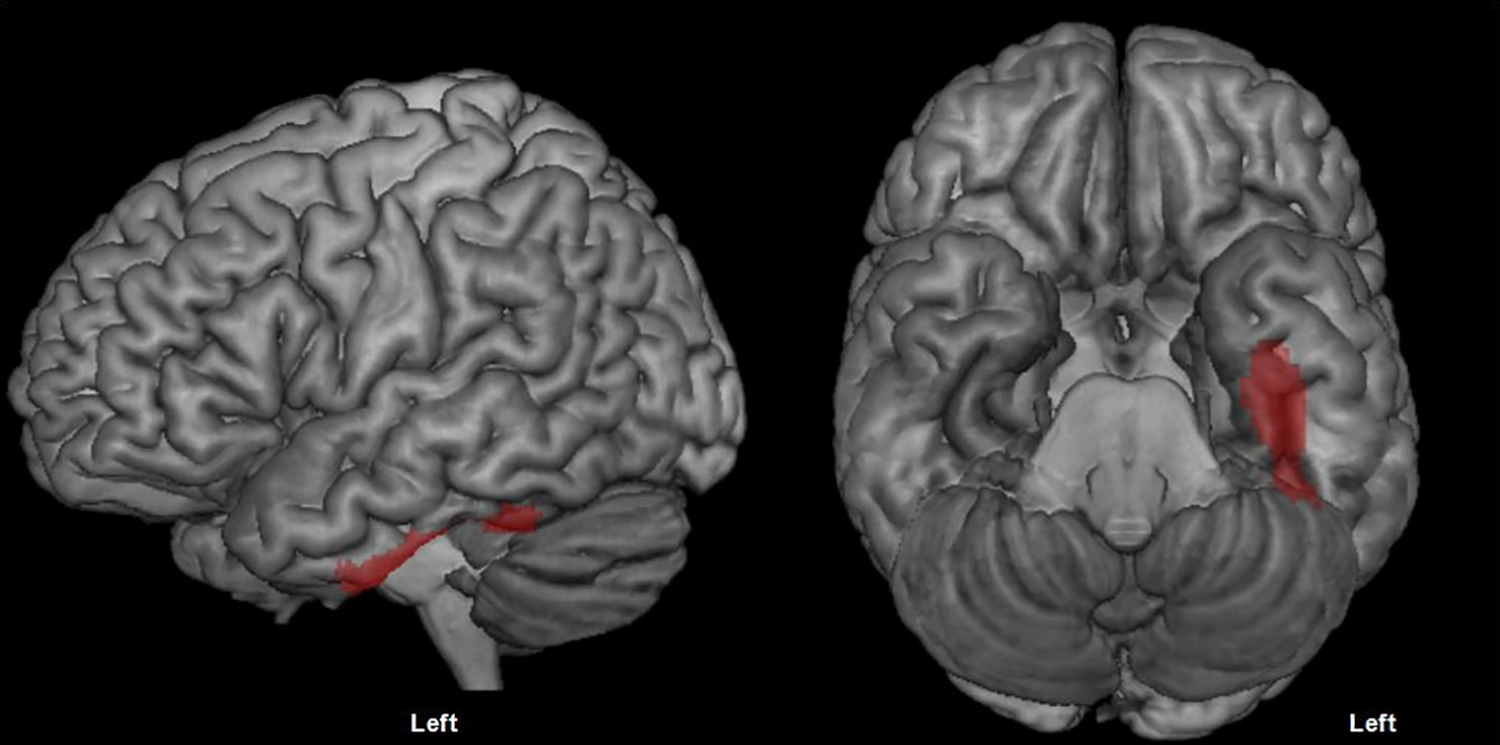
Cluster of hypometabolism correlated with a lower number of correct responses on the picture naming test (P < .05 FWE-corrected, minimum cluster size = 100 voxels).
Spatial Coordinates of Clusters Correlated With Total Number of Correct Responses on the Picture Naming Test and With Naming Error Subtypes.
Abbreviation: MNI, Montreal Neurological Institute.
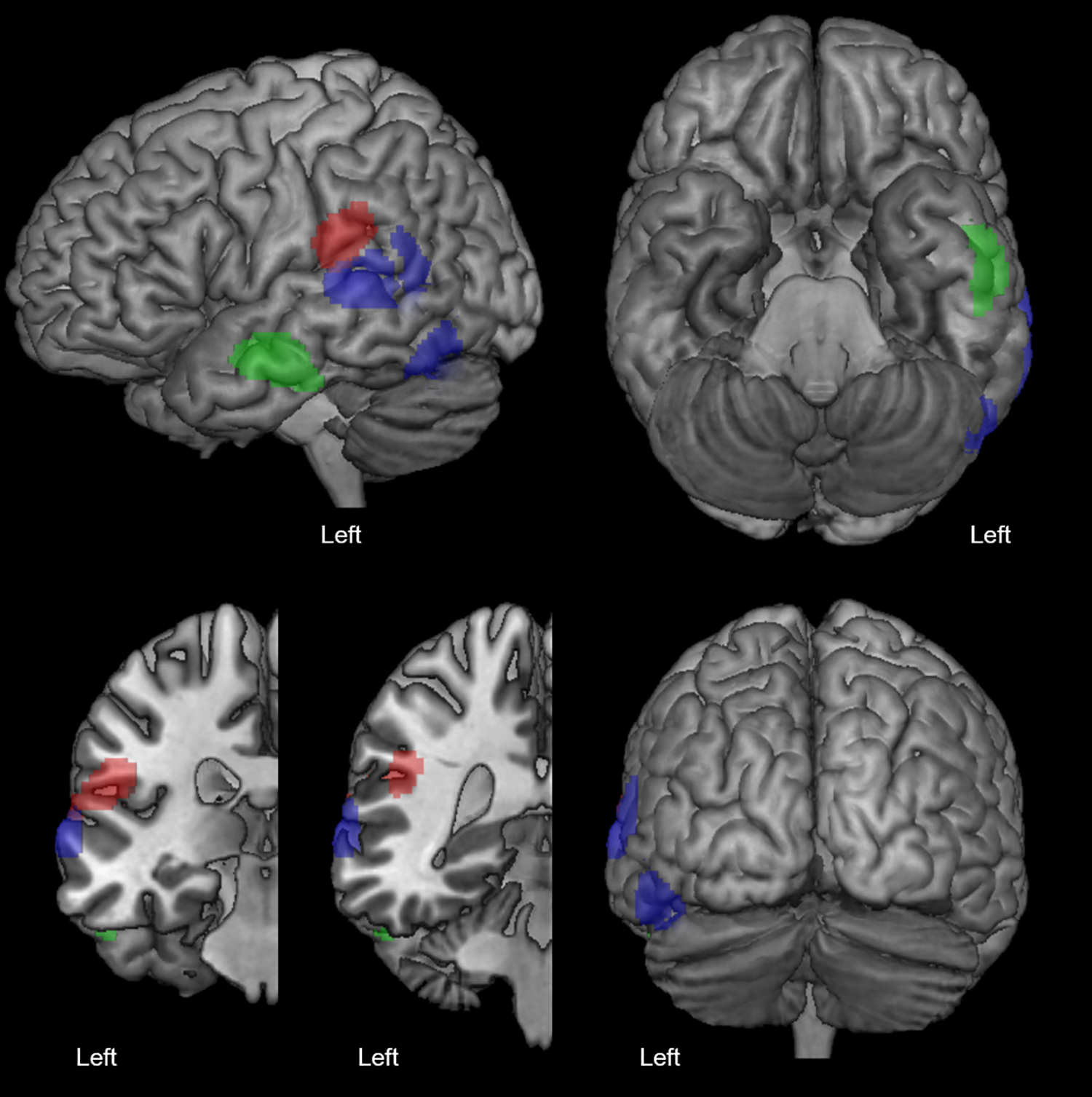
Clusters of hypometabolism correlated with a higher number of phonemic errors (in red), semantic errors (in blue) and formal errors (in green; P < .05 FWE-corrected, minimum cluster size = 100 voxels).
Discussion
In a sample of 63 patients with AD, correlation between FDG-PET brain metabolism and performance on a picture naming task identified various loci of metabolic abnormalities within the left hemisphere that are associated with naming impairment. Global performance, that is, number of accurate responses, was related to dysfunction of the ventral temporal cortex, in agreement with prior evidence that supports a crucial role of the left fusiform gyrus in semantic–lexical retrieval. 60,62,64 The 3 subtypes of naming errors considered in the study involved a specific metabolic correlate: posterior middle and posterior inferior temporal gyri for semantic errors, supramarginal gyrus, adjacent superior temporal cortex for phonemic errors, and mid–anterior segment of the middle temporal gyrus for formal errors. With the exception of semantic errors, the other 2 error categories were produced in small amounts, possibly limiting the accuracy of correlational analyses. We increased number of errors by recruiting a relatively large sample, and a mixed cohort in terms of clinical profiles (amnesic, linguistic, frontal, posterior), 66 but phenotypic heterogeneity itself might also have affected the power of the analysis since different substrates may underpin a certain type of naming errors in different disease variants. The fact that formal and phonemic errors were produced only by a minority of cases within the patients’ cohort and that the proportion of error subtypes (of all the naming errors) was highly variable across participants may also have affected the reliability and generalizability of our correlations. Nevertheless, SPM yielded clear and sizable clusters at strict significance thresholds for all 3 types of errors.
The 2 left temporal clusters emerged in association with semantic errors have been found also in previous studies. 5,8,16 -21 The posterior portion of the middle temporal gyrus, in particular, has been found to be lesioned in a focal fluent language disorder characterized by production of numerous semantic errors, that is, transcortical sensory aphasia. 70 This area lies on the ventral bank of the superior temporal sulcus 71,72 and has been described as a highly patients with region operating multimodal interactions for word comprehension and production. 20,33,73 -77 Based on its functional properties, this region has in fact been assimilated to the “semantic hub” 78 located in the left anterior ventral temporal cortex. 76 Semantic errors deriving from its dysfunction might thus be due to impaired integration of information that is necessary for activating the correct conceptual representation. 3,23,26,27,32 This mechanism is more similar to the conceptual breakdown underlying semantic errors in patients with semantic dementia 5,7,10 than to impairment of lexical retrieval. In effect, the lexical (-semantic) level of the word generation process has been localized, by Indefrey and Levelt 3,4 and by Dell, 7,14 more anteriorly along the middle temporal gyrus. A view that has received support from studies 24 -26 demonstrating that mid–anterior temporal damage generates semantic errors above and beyond impairment of comprehension or of general semantic knowledge (probably linked to more basal lesions). 76 The second cluster of hypometabolism that correlated significantly with semantic errors encompasses the left posterior inferior temporal cortex. In disagreement with Indefrey and Dell assumptions, 3,4,7,14 Hickok and Poeppel 5,8 mapped to this region and the adjacent middle temporal gyrus the lexical–semantic node of naming, receiving support from evidence that damage to this region is associated with pure anomia 79,80 and with the same semantic errors + preserved comprehension behavioral pattern 16,17 reported for more anterior lesions. 24 -26 Better understanding of the exact locus of interaction between semantics and lexical entries will help establish whether semantic errors associated with damage to the posterior inferior temporal gyrus derive from dysfunction at the interface between conceptual knowledge and lexical retrieval. An alternative account is suggested by studies that have highlighted a special property of this region: its vocation for visual semantics. Two recent functional MRI studies analyzed activations during picture and written 76 or auditory 81 word semantic tasks and clearly showed larger activation of the posterior inferotemporal cortex for the visual than the verbal condition. Some semantic errors, especially coordinate paraphasias (eg, orange for lemon, bus for truck), emerge for items that are visually similar to the target. The occipital–inferior temporal cluster may thus reflect the semantic plus visual character of a subset of errors produced by our patients, configuring this region as a visual semantic locus. The same account has been proposed for anomia in confrontation naming following damage to this region. In their commentary to a study correlating surgical lesions of the left inferotemporal region with anomia, 79 Hope and Price claimed that “rather than identifying the posterior ITG [inferior temporal gyrus] and posterior ILF [inferior longitudinal fasciculus] with lexical retrieval itself, we are proposing a more perceptual semantic account.” 82
Of all areas that were previously reported as lesional substrate of phonological errors, the left supramarginal gyrus, and more marginally, area Spt and the posterior superior temporal cortex emerged as specific correlates of phonemic errors in our patients with AD. Damage to the left temporoparietal junction is typical of conduction aphasia and more pertinent to the current study, typical of the language variant of AD, logopenic progressive aphasia, 83 which are characterized by repetition deficits and phonological errors. A sensory-motor account of conduction aphasia 30 purports that damage to Spt causes phonological paraphasias by interfering with the translation of phonological codes (syllable units activated in the posterior superior temporal cortex, according to Hickok) 8 into an articulatory program. Another theory 84,85 indicates the supramarginal gyrus as the critical lesion site 7,26 and impairment of the short-term memory phonological store located in this area 86 -88 as the cause of errors. Moreover, great relevance has recently been attributed to other processes that are suggested to take place in the supramarginal gyrus: sensorimotor feedback over phonological retrieval 8 and activation of prearticulatory representations (phoneme units, according to Hickok) 8 to be transferred to the articulatory programming apparatus. 20,89 Based on our findings, the cause of phonemic errors in AD is the impairment of one or more of these post-lexical–phonological operations within the supramarginal gyrus, rather than a sensory-motor interface deficit at the level of Spt since the hypometabolic cluster reached such area but was predominantly centered around the inferior parietal cortex.
Our results about formal errors are probably the most novel finding of the current study, for 2 reasons. First, prior data about the neural substrate of this type of lexical–phonological errors are extremely scarce. Second, the neural substrate of the node of the naming network whose dysfunction may be considered the primary source of this type of errors, that is, the lexical–phonological node has undergone revision in recent years and is no more associated with the posterior superior temporal gyrus and sulcus 3 -5 (see, for instance, Gow overview of evidence against the role of the posterior superior temporal cortex as the anatomical site of the phonological output lexical representations). 20 In a computational implementation and extension of Hickok’s and Poeppel’s model, these areas have been ascribed post-lexical–phonological functions, namely representing phonology in syllable units. 8 This view has received some support from results of 2 vascular lesion parameter mapping studies of Dell’s computational model. 7,27 One of these studies, performed by Tochadse and collaborators, 27 has gone 1 step further, identifying a possible “new” locus for the phonological lexical level. One of the computational factors they analyzed comprises formal and phonological errors and shows a correlation with a relatively vast area of vascular damage to the left fronto–temporo–parietal cortex. Within this area, the authors tentatively mapped a phonological lexical component to the anterior middle–superior temporal cortex, and a post-lexical–phonological component to the superior temporal, fronto-parietal, and fronto-insular cortex. Crucially, the peak coordinates of the anatomical correlate of formal errors emerged from the present study correspond to Tochadse mid–anterior cluster. More precisely, in their study, this cluster was not only associated with the phonological–formal factor but was also part of a large area correlated with a semantic computational factor (in accord with speech models that consider the lateral surface of the left anterior middle and superior temporal gyri as part of the language ventral pathway with a role in semantics and in lexical–semantic interface). 3,4,90,91,8,22 -26,31,72 The lexicon represents the functional link between formal and semantic errors. The overlap of lesional correlates of these 2 error categories in the more anterior segments of the left middle and superior temporal gyri suggests that this area might be the substrate of lexical representations. Our results align with this hypothesis demonstrating a one-to-one mapping between neurodegeneration of this region and formal errors, in patients with AD.
In conclusion, our findings outline a neuroanatomical map of errors produced by patients with AD in confrontation naming that associates semantic errors with neurodegeneration in 2 areas of the left posterior temporal cortex supposed to subserve visual and more general semantic representations, formal errors with hypometabolism in the left anterior middle temporal gyrus, which might then represent the locus of phonological lexical processes, and phonemic errors with degeneration in the left supramarginal gyrus underpinning phonological short-term memory or pre-articulatory representations or words.
Footnotes
The table shows individual socio-demographic and clinical features of study participants, and performance on the picture naming test. Percentages were calculated out of total number of stimuli (80) for total correct, and out of total number of errors produced by the entire cohort for error subtypes. Please note that total number of errors may not correspond to the difference between maximum test score and number of correct responses, since multiple errors produced by a patient for a same item were all counted and included in the analyses.

| Patient | Sex | Age | Education |
Symptoms Duration |
MMSE | Picture Naming | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total Correct | Semantic Errors | Formal Errors | Phonemic Errors | ||||||||||
| n. | % | n. | % | n. | % | n. | % | ||||||
| CASE 1 | female | 73 | 5 | 42 | 21 | 54 | 67.5 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 2 | female | 78 | 13 | 42 | 18 | 49 | 61.3 | 5 | 11.4 | 1 | 2.3 | 2 | 4.5 |
| CASE 3 | male | 84 | 5 | 18 | 25 | 52 | 65.0 | 3 | 10.0 | 0 | .0 | 0 | .0 |
| CASE 4 | female | 71 | 8 | 48 | 19 | 55 | 68.8 | 2 | 10.5 | 0 | .0 | 1 | 5.3 |
| CASE 5 | female | 77 | 11 | 12 | 28 | 71 | 88.8 | 1 | 11.1 | 0 | .0 | 1 | 11.1 |
| CASE 6 | male | 77 | 5 | 36 | 19 | 68 | 85.0 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 7 | female | 80 | 5 | 36 | 23 | 63 | 78.8 | 2 | 15.4 | 0 | .0 | 1 | 7.7 |
| CASE 8 | female | 74 | 8 | 12 | 26 | 55 | 68.8 | 3 | 9.4 | 0 | .0 | 0 | .0 |
| CASE 9 | female | 78 | 5 | 36 | 19 | 65 | 81.3 | 0 | .0 | 1 | 5.0 | 0 | .0 |
| CASE 10 | male | 75 | 8 | 24 | 26 | 33 | 41.3 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 11 | female | 70 | 5 | 12 | 28 | 62 | 77.5 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 12 | female | 83 | 5 | 36 | 22 | 65 | 81.3 | 0 | .0 | 1 | 8.3 | 0 | .0 |
| CASE 13 | female | 76 | 5 | 18 | 28 | 59 | 73.8 | 2 | 12.5 | 0 | .0 | 0 | .0 |
| CASE 14 | female | 63 | 5 | 36 | 18 | 73 | 91.3 | 0 | .0 | 1 | 20.0 | 1 | 20.0 |
| CASE 15 | male | 57 | 8 | 30 | 27 | 72 | 90.0 | 0 | .0 | 1 | 11.1 | 1 | 11.1 |
| CASE 16 | female | 61 | 8 | 24 | 21 | 73 | 91.3 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 17 | male | 75 | 8 | 24 | 19 | 53 | 66.3 | 2 | 8.7 | 0 | .0 | 0 | .0 |
| CASE 18 | female | 57 | 11 | 24 | 28 | 80 | 100 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 19 | female | 72 | 5 | 24 | 23 | 71 | 88.8 | 2 | 20.0 | 0 | .0 | 0 | .0 |
| CASE 20 | male | 69 | 10 | 24 | 18 | 68 | 85.0 | 1 | 7.1 | 0 | .0 | 1 | 7.1 |
| CASE 21 | male | 72 | 13 | 30 | 20 | 62 | 77.5 | 3 | 18.8 | 2 | 12.5 | 4 | 25.0 |
| CASE 22 | female | 53 | 13 | 30 | 22 | 73 | 91.3 | 1 | 8.3 | 0 | .0 | 0 | .0 |
| CASE 23 | male | 78 | 5 | 36 | 23 | 65 | 81.3 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 24 | female | 81 | 12 | 18 | 24 | 75 | 93.8 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 25 | female | 70 | 6 | 24 | 24 | 70 | 87.5 | 0 | .0 | 0 | .0 | 1 | 14.3 |
| CASE 26 | female | 75 | 8 | 18 | 28 | 43 | 53.8 | 4 | 11.1 | 0 | .0 | 1 | 2.8 |
| CASE 27 | male | 84 | 5 | 12 | 24 | 75 | 93.8 | 0 | .0 | 0 | .0 | 1 | 16.7 |
| CASE 28 | female | 50 | 13 | 42 | 21 | 73 | 91.3 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 29 | female | 67 | 5 | 12 | 26 | 73 | 91.3 | 1 | 33.3 | 0 | .0 | 0 | .0 |
| CASE 30 | female | 70 | 5 | 24 | 18 | 45 | 56.3 | 3 | 11.5 | 1 | 3.8 | 1 | 3.8 |
| CASE 31 | male | 74 | 13 | 18 | 24 | 61 | 76.3 | 1 | 5.6 | 1 | 5.6 | 0 | .0 |
| CASE 32 | female | 72 | 8 | 36 | 23 | 68 | 85.0 | 2 | 18.2 | 0 | .0 | 0 | .0 |
| CASE 33 | male | 75 | 13 | 24 | 20 | 65 | 81.3 | 0 | .0 | 1 | 7.1 | 2 | 14.3 |
| CASE 34 | male | 73 | 8 | 12 | 21 | 33 | 41.3 | 1 | 2.6 | 1 | 2.6 | 1 | 2.6 |
| CASE 35 | female | 57 | 13 | 48 | 18 | 61 | 76.3 | 1 | 5.6 | 1 | 5.6 | 1 | 5.6 |
| CASE 36 | female | 70 | 18 | 24 | 22 | 69 | 86.3 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 37 | male | 64 | 10 | 36 | 21 | 20 | 25.0 | 1 | 1.6 | 1 | 1.6 | 1 | 1.6 |
| CASE 38 | male | 82 | 5 | 12 | 25 | 59 | 73.8 | 1 | 5.0 | 0 | .0 | 2 | 10.0 |
| CASE 39 | male | 85 | 15 | 48 | 19 | 80 | 100 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 40 | female | 62 | 5 | 24 | 26 | 46 | 57.5 | 1 | 5.0 | 1 | 5.0 | 0 | .0 |
| CASE 41 | female | 77 | 5 | 18 | 25 | 53 | 66.3 | 0 | .0 | 0 | .0 | 1 | 5.0 |
| CASE 42 | female | 77 | 5 | 12 | 27 | 62 | 77.5 | 2 | 10.0 | 0 | .0 | 0 | .0 |
| CASE 43 | male | 79 | 5 | 12 | 28 | 71 | 88.8 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 44 | male | 73 | 13 | 24 | 25 | 19 | 23.8 | 0 | .0 | 0 | .0 | 1 | 2.0 |
| CASE 45 | male | 66 | 8 | 12 | 24 | 79 | 98.8 | 1 | 20.0 | 0 | .0 | 0 | .0 |
| CASE 46 | male | 55 | 19 | 42 | 18 | 69 | 86.3 | 4 | 26.7 | 1 | 6.7 | 1 | 6.7 |
| CASE 47 | male | 76 | 5 | 24 | 25 | 72 | 90.0 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 48 | male | 74 | 8 | 18 | 28 | 58 | 72.5 | 1 | 4.8 | 0 | .0 | 0 | .0 |
| CASE 49 | female | 69 | 9 | 36 | 18 | 52 | 65.0 | 4 | 11.8 | 0 | .0 | 0 | .0 |
| CASE 50 | male | 67 | 9 | 18 | 27 | 78 | 97.5 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 51 | male | 66 | 8 | 24 | 28 | 77 | 96.3 | 1 | 33.3 | 0 | .0 | 0 | .0 |
| CASE 52 | male | 72 | 13 | 12 | 27 | 77 | 96.3 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 53 | male | 78 | 16 | 48 | 22 | 73 | 91.3 | 1 | 16.7 | 0 | .0 | 1 | 16.7 |
| CASE 54 | female | 68 | 11 | 36 | 27 | 71 | 88.8 | 2 | 13.3 | 0 | .0 | 0 | .0 |
| CASE 55 | female | 64 | 5 | 36 | 22 | 69 | 86.3 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 56 | male | 57 | 18 | 24 | 27 | 77 | 96.3 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 57 | male | 81 | 5 | 36 | 21 | 64 | 80.0 | 1 | 6.3 | 0 | .0 | 0 | .0 |
| CASE 58 | male | 74 | 8 | 30 | 21 | 70 | 87.5 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 59 | female | 72 | 5 | 12 | 28 | 48 | 60.0 | 1 | 2.9 | 0 | .0 | 0 | .0 |
| CASE 60 | male | 75 | 18 | 24 | 22 | 45 | 56.3 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 61 | female | 73 | 5 | 12 | 27 | 68 | 85.0 | 0 | .0 | 0 | .0 | 0 | .0 |
| CASE 62 | female | 80 | 5 | 36 | 21 | 42 | 52.5 | 1 | 2.3 | 0 | .0 | 0 | .0 |
| CASE 63 | female | 77 | 5 | 24 | 23 | 54 | 67.5 | 1 | 5.0 | 0 | .0 | 0 | .0 |
List of the 80 items Included in the Picture Naming Test, With Psycholinguistic Variables (Modified From Laiacona et al, 1993).67a
| Item | Lexical Frequency | Prototypicality | Familiarity | Name Agreement |
|---|---|---|---|---|
| TOOLS: | ||||
| Pinza (pliers) | 0 | 58 | 3.38 | 47 |
| Cacciavite (screwdriver) | 0 | 214 | 3.42 | 58 |
| Sega (saw) | 0 | 394 | 2.92 | 60 |
| Vite (screw) | 0 | 43 | 3.20 | 58 |
| Chiave inglese (spanner) | 0 | 61 | 2.72 | 53 |
| Scalpello (chisel) | 0 | 103 | 2.46 | 38 |
| Scure (axe) | 0 | 14 | 2.28 | 47 |
| Forbice (scissors) | 0 | 4 | 3.98 | 60 |
| Martello (hammer) | 9 | 431 | 3.48 | 59 |
| Chiodo (nail) | 12 | 248 | 3.28 | 52 |
| VEGETABLES: | ||||
| Sedano (celery) | 0 | 96 | 3.40 | 56 |
| Pannocchia (cob) | 0 | 247 | 3.50 | 39 |
| Cipolla (onion) | 0 | 47 | 3.32 | 53 |
| Peperone (pepper) | 0 | 13 | 2.92 | 57 |
| Carciofo (artichoke) | 0 | 5 | 2.29 | 57 |
| Asparago (asparagus) | 0 | 138 | 2.68 | 56 |
| Fungo (mushroom) | 4 | 2 | 2.88 | 60 |
| Carota (carrot) | 5 | 316 | 3.55 | 59 |
| Insalata (lettuce) | 8 | 19 | 3.42 | 21 |
| Pomodoro (tomato) | 15 | 215 | 3.78 | 52 |
| ANIMALS: | ||||
| Giraffa (giraffe) | 0 | 82 | 1.80 | 59 |
| Cammello (camel) | 0 | 28 | 2.08 | 50 |
| Struzzo (oyster) | 0 | 17 | 1.52 | 39 |
| Rana (frog) | 0 | 3 | 2.48 | 60 |
| Cigno (swan) | 0 | 14 | 1.97 | 34 |
| Bruco (caterpillar) | 0 | 30 | 1.72 | 39 |
| Mucca (cow) | 5 | 284 | 2.42 | 58 |
| Gallo (rooster) | 10 | 7 | 2.22 | 59 |
| Farfalla (butterfly) | l0 | 48 | 2.92 | 60 |
| Topo (mouse) | 11 | 33 | 2.45 | 59 |
| MUSICAL INSTRUMENTS: | ||||
| Tamburo (drum) | 0 | 322 | 2.60 | 59 |
| Arpa (harp) | 0 | 105 | 1.88 | 54 |
| Corno (horn) | 0 | 63 | 2.00 | 33 |
| Fisarmonica (accordeon) | 0 | 37 | 2.15 | 58 |
| Violino (violin) | 0 | 271 | 2.68 | 56 |
| Chitarra (guitar) | 0 | 231 | 3.58 | 57 |
| Flauto (flute) | 0 | 246 | 2.45 | 42 |
| Tromba (trumpet) | 6 | 279 | 2.60 | 57 |
| Pianoforte (piano) | 17 | 329 | 3.42 | 60 |
| Campana (bell) | 19 | lO | 2.20 | 60 |
| FRUITS: | ||||
| Banana (banana) | 0 | 283 | 3.65 | 60 |
| Anguria (watermelon) | 0 | 47 | 3.05 | 54 |
| Ananas (pineapple) | 0 | 98 | 2.95 | 55 |
| Ciliegia (cherry) | 3 | 183 | 3.38 | 56 |
| Pera (pear) | 5 | 326 | 3.55 | 59 |
| Fragola (strawberry) | 6 | 58 | 3.20 | 59 |
| Limone (lemon) | 16 | 134 | 3.25 | 57 |
| Mela (apple) | 32 | 429 | 3.98 | 59 |
| Uva (grapes) | 33 | 247 | 3.65 | 60 |
| Arancia (orange) | 39 | 390 | 3.34 | 29 |
| MEANS OF TRANSPORT: | ||||
| Autobus (bus) | 0 | 300 | 4.50 | 44 |
| Elicottero (helicopter) | 0 | 18 | 2.55 | 59 |
| Slitta (sled) | 0 | l | 2.80 | 50 |
| Barca a vela (sailing boat) | 0 | l | 2.92 | 60 |
| Camion (truck) | 19 | 223 | 4.02 | 43 |
| Aeroplano (airplane) | 20 | 280 | 3.78 | 59 |
| Bicicletta (bycicle) | 24 | 193 | 3.78 | 60 |
| Motocicletta (motorcycle) | 24 | 174 | 3.25 | 60 |
| Automobile (car) | 59 | 407 | 4.70 | 50 |
| Treno (train) | 81 | 257 | 4.15 | 54 |
| FURNITURE: | ||||
| Sgabello (stool) | 0 | 72 | 3.08 | 56 |
| sedia a dondolo | 0 | 5 | 3.25 | 60 |
| Scrivania (desk) | 6 | 230 | 4.32 | 56 |
| Cassettone (dresser) | 7 | 143 | 4.52 | 38 |
| Lampada (lamp) | 14 | 227 | 4.20 | 51 |
| Divano (couch) | 21 | 168 | 4.40 | 58 |
| Sedia (chair) | 22 | 440 | 4.58 | 60 |
| Vaso (vase) | 29 | 3 | 2.78 | 58 |
| Tavolo (table) | 51 | 408 | 4.35 | 59 |
| Letto (bed) | 176 | 328 | 4.72 | 60 |
| BODY PARTS: | ||||
| Labbra (lips) | 29 | 25 | 4.50 | 42 |
| Orecchio (ear) | 40 | 260 | 4.50 | 58 |
| Naso (nose) | 41 | 281 | 4.52 | 55 |
| Dito (finger) | 45 | 279 | 4.78 | 59 |
| Capelli (hair) | 78 | 125 | 4.59 | 25 |
| Gamba (leg) | 84 | 402 | 4.65 | 58 |
| Braccio (arm) | 112 | 398 | 4.75 | 59 |
| Piede (foot) | 174 | 295 | 4.78 | 60 |
| Occhio (eye) | 291 | 303 | 4.88 | 60 |
| Mano (hand) | 375 | 228 | 4.82 | 60 |
aItems were presented in randomized, fixed order, and randomized across categories.
Acknowledgments
The authors would like to thank Dr Cristina Mapelli for her help in patients’ assessment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
