Abstract
Objectives:
To investigate the association between insulin resistance (IR) and enlarged perivascular space (EPVS) in the basal gangliain nondiabetic healthy elderly population.
Methods:
A total of 235 nondiabetic healthy elderly population were recruited. A 3-level scale was used to evaluate the burden of EPVSs. The homeostasis model assessment–estimated insulin resistance index (HOMA-IR) was used for IR estimation. Correlation between IR and severity of EPVS was assessed using the regression model after adjusting demographics and cardiovascular risk factors.
Results:
The top quartile of HOMA-IR was 2.52, and 25.11% of patients showed IR. The proportion of patients with IR was higher in the moderate to severe EPVS group than in the mild group (36.51% vs 20.93%, P = .015). In multivariate logistic analysis, IR was positively correlated with the moderate to severe EPVS (adjusted odds ratio: 3.532, 95% confidence interval: 1.633-7.636, P = .001) after adjusting classical risk factors.
Conclusions:
Insulin resistance was independently correlated with EPVS in the basal ganglia in nondiabetic healthy elderly population.
Keywords
Introduction
Magnetic resonance imaging (MRI)–visible enlarged perivascular space (EPVS), a well-established neuroimaging hallmark of cerebral small vessel diseases (CSVD), is associated with other morphological features of CSVD, such as white matter hyperintensities 1 and lacunes. 2 EPVSs are fluid-filled cavities surrounding small penetrating cerebral arterioles, and they are important drainage conduit for cerebral interstitial fluid. 2 -4 The number of EPVSs increases with age, manifesting as visible round or tubular hyperintensities in the basal ganglia, centrum semiovale, and midbrain on T2-weighted brain MRI. 2,4,5 A small number of EPVSs might be normal findings, 6 but accumulating evidence has shown that an increased number of EPVSs might reflect damage to cerebral microvessels. 7 EPVS in the basal ganglia has been linked to cognitive impairment, 8 -11 depression, 12 and an increased risk of recurrent ischemic stroke. 13 Although there are some known risk factors for EPVSs, such as old age, hypertension, and atherosclerosis, 14,15 there are other unknown risk factors remaining to be explored. 16
Insulin resistance (IR) is a syndrome that correlates with a cluster of metabolic disorders, including diabetes mellitus, obesity, hypertension, and lipid abnormalities, 17,18 characterized by decreased sensitivity or responsiveness to metabolic actions of insulin. Previous studies suggest that IR is driving the increased incidence and recurrence of stroke. 19 -21 Furthermore, increasing evidence links IR to reduced cortical perfusion 22 and cognitive impairment in neurologically healthy adults in CSVD studies. 23,24 A recent study suggests that IR is related to silent lacunar infarcts that are another hallmark of CSVD. 25,26 Another study has demonstrated that type 2 diabetes mellitus may be causally associated with lacunar stroke, 16 but little is known about the unique effect of IR on EPVSs though both lacunar stroke and EPVSs are due to endothelial dysfunction as their underlying pathogenic mechanism. Therefore, the objective of this study was to investigate whether IR is associated with EPVSs independent of other clinical risk factors in nondiabetic healthy adult population.
Methods
Study Population
The present study was conducted according to the principles of the Declaration of Helsinki and was approved by the ethics committee of Shanghai Fifth People’s Hospital (approval number: 2017EC032), and written informed consent was obtained from all individual participants. We recruited nondiabetic elderly patients who were admitted to our hospital for health checkups from January 2013 to January 2017 consecutively. Those who met any of the following criteria were excluded: history of stroke, cognitive dysfunction, heart diseases, malignancies, autoimmune diseases, and hepatic or renal diseases.
Clinical and Laboratory Variables
Clinical information, including age, sex, smoking status, comorbid conditions, hypertension, dyslipidemia, and intake of antiplatelet or statin medications, was obtained within 8 hours after admission through face-to-face interviews by neurological residents. Hypertension was defined as a high systolic (≥140 mm Hg) or diastolic (≥90 mm Hg) blood pressure, 27 or patients were currently taking antihypertensive medications. Hypercholesterolemia was defined as total cholesterol level ≥240 mg/dL 28 or currently taking lipid-lowering agents. Intracranial arterial stenosis was defined as more than 50% stenosis in intracranial arteries as shown on magnetic resonance angiography. 29 Carotid artery stenosis was defined as more than 50% stenosis in the carotid artery as shown on ultrasound. Fasting venous blood samples were drawn to measure levels of glucose, insulin, creatinine, and total cholesterol. The body mass index (BMI) was calculated as weight (kg) divided by the height squared (m 2 ). Overweight (BMI, 23.0-24.9) and obesity (BMI ≥25.0) were classified according to the World Health Organization criteria for the Asia-Pacific region. 30
Brain MRI and EPVS
All MRI scans were performed using a 3.0 T MRI system (Magnetom Skyra, Siemens, Germany). Axial T2-weighted sequences, fluid-attenuated inversion recovery (FLAIR), T1-weighted sequences, and diffusion-weighted imaging were used for this study. EPVS defined as round, oval, or linear-shaped lesions with a diameter smaller than 3 mm along the course of penetrating arteries, with signal intensity similar to that of cerebrospinal fluid on all sequence spaces and without a T2-hyperintense rim on FLAIR.
5
To count the total burden of EPVSs, 2 neuroradiologists who were blind to clinical details of the patients rated EPVSs in the basal ganglia and stratified their scores of EPVSs as follows: 1 = 0 to 10 EPVSs, 2 = 11 to 25 EPVSs, and 3 = >25 EPVSs. 31 If asymmetrical EPVSs were observed on both hemisphere, the hemisphere with more EPVSs was selected. Interobserver agreement values for the EPVS score was 0.888. Any disagreement about the number of EPVSs was resolved by consulting with a neuroimaging expert.
Insulin Resistance Evaluation
The homeostasis model assessment–estimated insulin resistance index (HOMA-IR)was used to identify patients with IR. HOMA-IR was calculated as fasting insulin (μU/mL) × fasting glucose (mmol/L)/22.5, 32 and its top quartile was used to define IR as reported in a previous study. 19
Statistical Analyses
Statistical analyses were performed using the SPSS 21.0 software (IBM Corp, Armonk, New York). We dichotomized basal ganglia EPVSs into degree 1 (score 1) and degrees 2 + 3 (score 2, score 3), reflecting mild versus moderate/severe EPVS, respectively, as previously reported. 1,31,33 For demographic and clinical features, continuous variables with a normal distribution were presented as mean ± standard deviation and compared using the independent sample t test, continuous variables with a non-normal distribution were presented as median (P25, P75) and compared using the nonparametric test. Categorical variables were expressed as frequency (percentage) and compared using the χ2 test. Logistic regression was used to assess the relationship between IR and the severity of EPVSs. The following risk factors were included as reported previously 26 : age, sex, hypertension, hypercholesterolemia, smoking, and BMI. Ordered logistic regression was used to assess the association between IR and the total EPVS scores. All probability values were 2-tailed, and P ≤ .05 were considered to be statistically significant.
Results
Characteristics of the Study Population
A total of 235 patients were included in the analysis. The mean age of the moderate to severe EPVS group and the mild group were 74.98 ± 8.74 years and 66.00 ± 8.60 years, respectively. No significant difference was observed in the proportion of males and females. The top quartile of HOMA-IR was 2.52, and 25.11% of patients showed IR (HOMA-IR ≥2.52). The proportion of patients with IR was larger in the moderate to severe EPVS group than that in the mild group (36.51% vs 20.93%, P = .015). The proportion of BMI ≥25 tended to be larger in the moderate to severe EPVS group than in the mild group (P = .061). No significant difference was observed in the comparison of smoking status, hypertension, and other cardiovascular risk factors between the 2 groups (Table 1).
Characteristics of Subgroups Based on severity of EPVS.
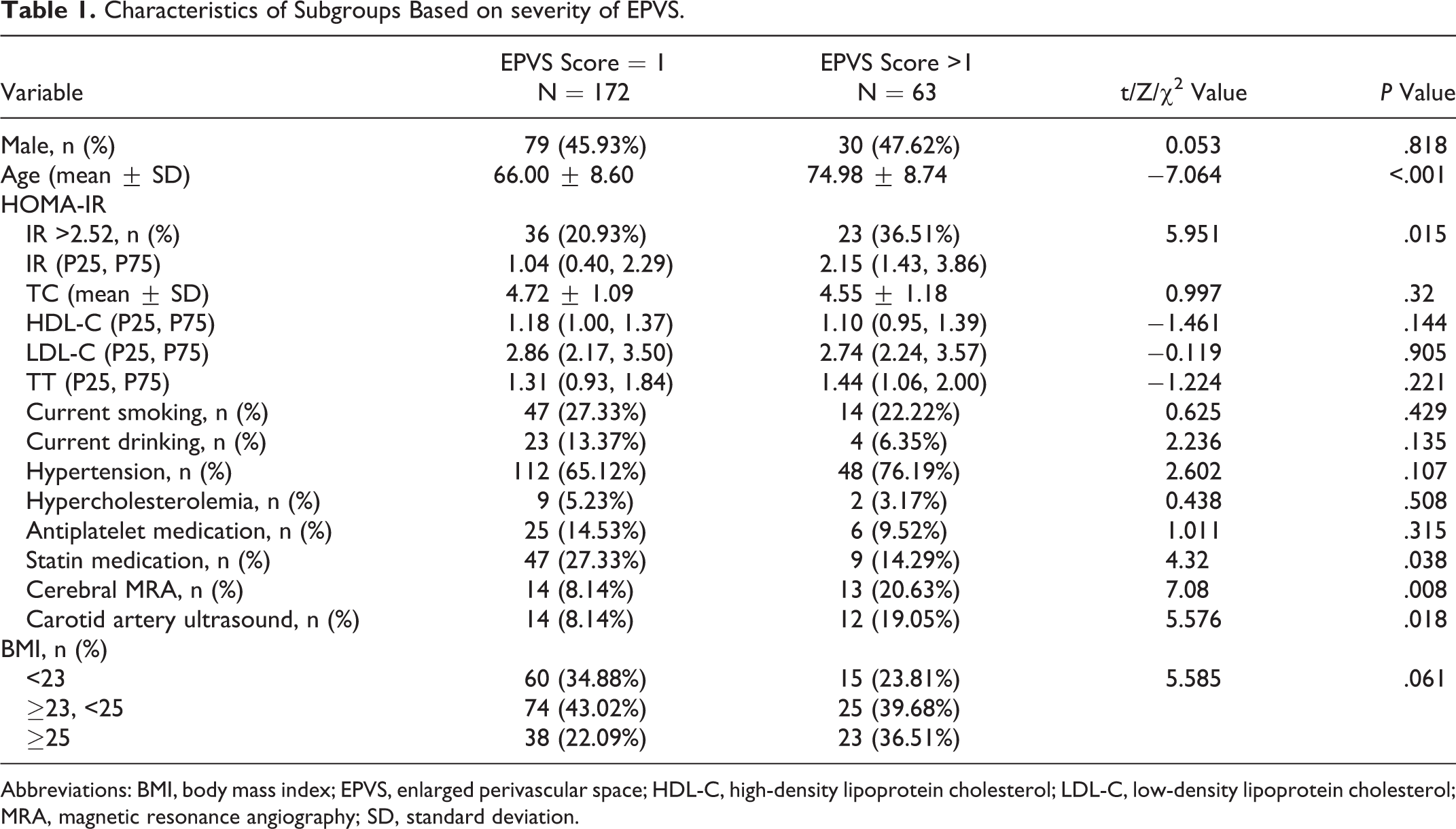
Abbreviations: BMI, body mass index; EPVS, enlarged perivascular space; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; MRA, magnetic resonance angiography; SD, standard deviation.
Association Between IR and Severity of EPVS
Correlation analysis was performed between demographic as well as clinical variables and the severity of EPVSs. It was found that IR along with age, statin medication, intracranial arterial stenosis, and carotid artery stenosis was linked to moderate to severe EPVSs (P < .05, respectively; Table 1). After normalizing these classical risk factors, IR was found to be positively associated with the moderate to severe EPVS in the binary logistic regression model (adjusted odds ratio: 3.532, 95% confidence interval [CI]: 1.633-7.636, P = .001). Meanwhile, aging (adjusted odds ratio: 1.145, 95% CI: 1.095-1.198, P < .001) and intracranial arterial stenosis (adjusted odds ratio: 2.920, 95% CI: 1.074-7.942, P = .036) increased the likelihood of developing moderate to severe EPVS, whereas statin medication decreased the likelihood of developing moderate to severe EPVS (adjusted odds ratio: 0.361, 95% CI: 0.147-0.887, P = .026; Table 2).
Association of Insulin Resistance and Other Cardiovascular Risk Factors With Moderate to Severe EPVS.a

Abbreviations: CI, confidence interval; EPVS, enlarged perivascular space; OR, odds ratio; MRA, magnetic resonance angiography.
a Model 1: binary logistic regression (LR) based on the severity of EPVS (EPVS score =1 vs EPVS score = 2 + 3); the enter and delete of variables were forward LR.
The association between IR and other cardiovascular risk factors with increasing severity of EPVS was assessed using the ordered logistic regression (model 2). In this statistical model, the odds ratio was presumed for each level (parallel line examination = 0.337), and the tendency still remained significant, only the odds ratio changed mildly, implying that IR, aging, and intracranial arterial stenosis increase the likelihood of increased numbers of EPVSs, whereas the statin medication decreased the tendency (Table 3).
Association of Insulin Resistance and Other Cardiovascular Risk Factors With Increasing Severity of EPVS Occurrence.a
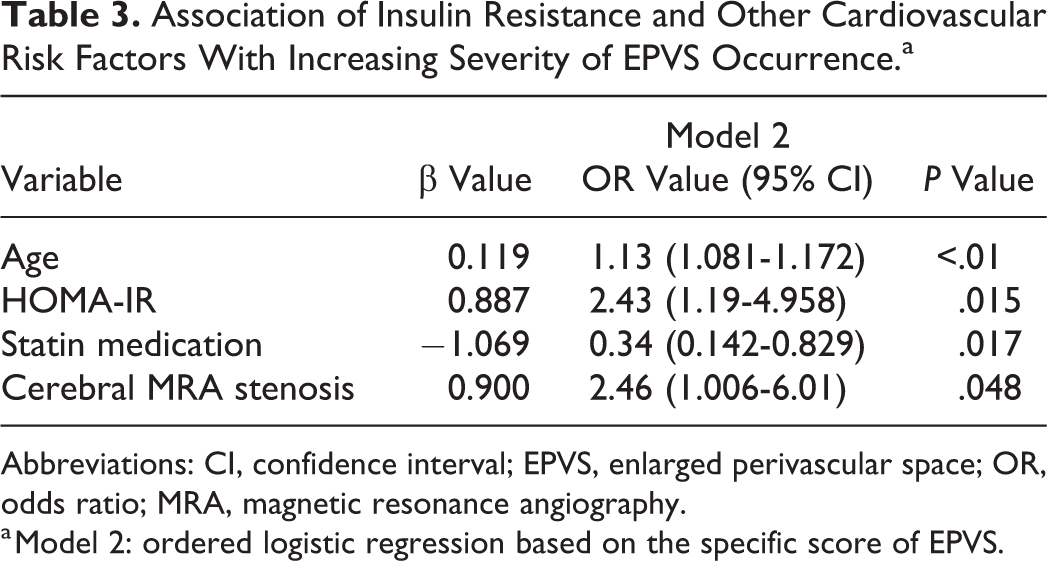
Abbreviations: CI, confidence interval; EPVS, enlarged perivascular space; OR, odds ratio; MRA, magnetic resonance angiography.
a Model 2: ordered logistic regression based on the specific score of EPVS.
Discussion
In the present study, the association between IR and EPVSs in a nondiabetic elderly population was investigated. It was found that IR along with aging and intracranial arterial stenosis was related to increased severity of EPVSs, independent of other clinical risk factors.
It proposed that endothelial dysfunction and atherosclerosis were the main pathologic mechanisms of CSVDs, 34,35 including EPVSs. Endothelial dysfunction contributed to the increased permeability of the blood–brain barrier, which led to deposition of plasma and leakage of inflammatory cells into the vascular wall and the adjacent tissue space, initiating or accelerating the formation of EPVSs. Our study demonstrated that IR, aging, and intracranial arterial stenosis increased the likelihood of developing EPVSs, whereas the statin medication decreased this tendency. The abovementioned factors were all related to the function of endothelial cells, 36 -39 suggesting that endothelial dysfunction might play a pivotal role in the pathogenesis of EPVS.
IR is not an exclusive feature of diabetes mellitus, it is also a fundamental mechanism of the metabolic syndrome and its components, namely, hypertension and hypercholesterolemia, which were also known as risk factors for EPVSs. 14,15 Previous studies suggested that IR was responsible for the increased incidence and recurrence of stroke. 19 -21 In CSVD regions, increasing evidence has linked IR to reduced cortical perfusion 22 and cognitive impairment in neurologically healthy adults. 22 Recently, a small number of studies demonstrated a possible causal relationship between IR and lacunar infarction, 26,25 but little is known about the unique effect of IR on EPVSs though they share the common pathogenic mechanisms. In our study, the correlation between IR and EPVSs in nondiabetic elderly population was analyzed and IR was found to be related to the increased severity of EPVSs, which suggests that IR is a risk factor of EPVSs, irrespective of diabetes mellitus and other cardiovascular risk factors. Previous studies have demonstrated that IR was definitely correlated with endothelial dysfunction in many cardiovascular diseases, 37,38 which might be confirmed by molecular and cellular mechanistic studies. For example, IR increased lipolysis and induced production of pro-inflammatory cytokines, which in turn contributed to endothelial dysfunction. 37,38 Therefore, we hypothesized that IR was involved in initiating or accelerating the formation of EPVSs through compromising the function of endothelial cells.
Atherosclerosis has been observed as a pathological feature of EPVSs,34 many typical vascular risk factors that are known to be associated with atherosclerosis contribute to the development of EPVSs. Association between EPVSs in the basal ganglia and atherosclerosis has been explored, and EPVSs were related to not only extracranial atherosclerosis 40 -42 but also intracranial atherosclerosis. 15 Our research also demonstrated that intracranial arterial stenosis was associated with increased numbers of EPVSs in the basal ganglia, demonstrating that a common pathogenic mechanism, such as endothelial dysfunction, might be responsible for the occurrence of both EPVSs and intra- and extra-atherosclerosis.
The present study had a couple of main strengths. Firstly, the association between IR and EPVSs was investigated. Secondly, the present study was conducted in a nondiabetic elderly population, which excluded the effect of confounding factors such as diabetes mellitus, coronary heart diseases, chronic kidney diseases, and stroke.
However, the present study also has a number of limitations. Firstly, the homeostasis model assessment–estimated insulin resistance index (HOMA-IR) method was used to measure IR rather than the euglycemic hyperinsulinemic clamp method which is the gold standard. The clamp method is unsuitable for clinical and epidemiological studies, so HOMA-IR is a commonly used alternative in many studies. 19,26,22,43,44 Secondly, we did not collect white matter hyperintensities, lacunes, and microbleeds in cerebral MRI in the current study. These structural changes may be found simultaneously in one patient, so the relationship between IR and EPVS may be interfered. But in the previous study, the same research method was applied. 26 Thirdly, health-check patients are not completely healthy. An American study showed that of 348 individuals who were subject to in-person history and physical examinations, only 51.7% passed the screening procedures and were regarded as healthy. 45 We developed a set of exclusion criteria and performed comprehensive physical examination of the nervous system to ensure all patients were nondiabetic healthy elderly population as much as possible.
In conclusion, IR was positively related to increased severity of EPVSs, together with aging and intracranial arterial stenosis, independent of other clinical risk factors in a nondiabetic healthy adult population. Longitudinal studies are needed to confirm the causal relationship between IR and EPVSs.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Shanghai Municipal Committee of Science and Technology (17411967700 and 19401935700), Shanghai Minhang District Health and Family Planning Commission for constructing big disciplines (2017MWDXK02), and Shanghai Municipal Commission of Health and Family Planning (Number 20184Y0178).
