Abstract
The 5-hydroxytryptamine (5-HT; serotonin) neurotransmission is severely affected by the degeneration of nigrostriatal dopaminergic neurons. Here, we report the effects of the systemic administration of the 5-HT7 receptor agonist AS-19. In sham rats, the mean response of the 5-HT neurons in the dorsal raphe nucleus (DRN) to systemic AS-19 was excitatory and the mean response of the γ-aminobutyric acid (GABA) interneurons was inhibitory. In Parkinson disease (PD) rats, the same dose did not affect the 5-HT neurons and only high doses (640 μg/kg intravenous) were able to the increase GABA interneuron activity. These results indicate that DRN 5-HT neurons and GABA interneurons are regulated by the activation of 5-HT7 receptors and that the degeneration of the nigrostriatal pathway leads to decreased responses of these neurons to AS-19, which in turn suggests that the 5-HT7 receptors on 5-HT neurons and GABA interneurons in PD rats are dysfunctional and downregulated.
Introduction
The dorsal raphe nucleus (DRN) is the origin of the central serotonin (5-hydroxytryptamine [5-HT]) system and plays an important role in regulating many physiological functions, such as sleep/arousal, food intake, and mood. The DRN contains 5-HT and non-5-HT neurons, which differ in cellular morphology, electrophysiological properties, and the expression of neurotransmitters. 1 This heterogeneous organization may correspond to a variety of physiological functions that are involved in serotonergic neurotransmission and complicate the understanding of DRN functions. γ-Aminobutyric acid (GABA) interneurons are the most prevalent type of non-5-HT neurons in the DRN and well known to synapse with 5-HT neurons, 2,3 which function as interneurons in local circuits with serotonergic projection neurons and regulate their output. 4 Therefore, characterization of the properties of each cell type, especially of GABAergic cells, and their interactions may help to elucidate the various functions of the DRN. Thus, GABA interneurons contribute to the regulation of the activity of 5-HT neurons.
The 5-HT7 receptor is positively coupled to adenylate cyclase via the Gs protein. 5 The 5-HT7 receptor is present in the hypothalamus (tuberomammillary nucleus), pontine nuclei (laterodorsal and pedunculopontine tegmental nuclei and the DRN), basal forebrain (medial septal nucleus, diagonal band of Broca, and substantia innominata), and hippocampus (fields CA1 and CA3 of Ammon’s horn). 6 -8 The 5-HT7 receptor has been implicated in various functions that are related to affective disorders, 9 including mood regulation, circadian rhythmicity, and sleep disturbances. In vitro electrophysiological studies have also demonstrated that the activation of 5-HT7 receptors increases the excitability of hippocampal, thalamic, and pallidal neurons. 10
Parkinson disease (PD) is characterized by the degeneration and loss of the midbrain substantia nigra pars compacta (SNc) that produces the neurotransmitter dopamine (DA), characterized by relatively selective nigrostriatal dopaminergic degeneration. The clinical symptoms of PD are resting tremor, rigidity, bradykinesia, and postural instability. However, there is growing evidence that PD is associated with a wide variety of nonmotor features, including depression, anxiety, and cognitive decline, 11 which affect the vast majority of patients during the course of the disease and may even precede the onset of motor symptoms. 12 Experiments have proven that the 5-HT transmitter system exhibits dysfunctions in PD rats. The reduction in 5-HT is attributable to the loss of 5-HT neurons in the raphe nuclei, because an average loss of 50% of 5-HT neurons has been found in the raphe nuclei of patients with PD. 1 Neurochemical studies have observed that the 5-HT levels were decreased in cerebrospinal fluid and several brain areas in patients with PD and PD monkeys. 13 -15 The functions of several 5-HT subtypes also have changed such as the 5-HT1A, 5-HT2A, 5-HT2C, and 5-HT3 receptors. 16 -19 And some experiments also have proven that the 6-hydroxydopamine (6-OHDA) lesions of the SNc in rats decreased the firing rate of GABA interneurons in the DRN. 20 To address this issue, the present study utilized electrophysiological techniques to examine the in vivo effects of the potent 5-HT7 receptor agonist AS-19 on the firing activities in the DRNs of sham rats and the changes that occur in responses of the 5-HT neurons and GABA interneurons following 5-HT7 receptor stimulation in rats with 6-OHDA lesions of the SNc.
Materials and Methods
Animal and Drugs
The experiments were performed on male Sprague Dawley rats (230-310 g). The animals were maintained in groups of 4 in a cage at a temperature of 21°C to 22°C and were provided free access to food and water.
The animal care adhered to the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publications No. 80-23, revised 1996) and the guidelines of the Institutional Animal Care Committee of the University. Thirty-six rats were divided into 2 groups, that is, sham (n = 15) and 6-OHDA-lesioned rats (n = 21). All efforts were made to minimize the number of animals used and their suffering.
Drugs
The following drugs were used—desipramine hydrochloride, 6-OHDA hydrochloride, and apomorphine hydrochloride from Sigma-Aldrich (St Louis, Missouri) and AS-19 [(2S)-(+)-5-(1,3,5-trimethylpyrazol-4-yl)-2-(dimethylamino) tetralin] and SB 269970 [(2R)-1-[(3-hydroxyphenyl) sulfonyl]-2-[2-(4-methyl-1-piperidinyl) ethyl] pyrrolidine] hydrochloride from Tocris (Ellisville, Missouri). The 6-OHDA and apomorphine were prepared in 0.9% saline containing 0.02% ascorbic acid. The desipramine and SB 269970 were prepared in 0.9% saline. The AS-19 was dissolved in 0.9% saline with 1% dimethyl sulfoxide. These drugs were prepared on the day of the experiment.
6-Hydroxydopamine-Induced Lesions of the Substantia Nigra DA Neurons
Unilateral 6-OHDA lesioning was carried out as previously described. 21 Briefly, animals were anesthetized with 4% chloral hydrate (400 mg/kg, intraperitoneally [IP]), pretreated with desipramine (25 mg/kg, IP), placed in a stereotaxic instrument, and injected with 6-OHDA (8 μg/4 μL) into the right SNc (Anterior Posterior (AP): −5.0 to −5.3, Medial Leteral (ML): 1.9-2.1, and Dorsal Ventral (DV): 7.1-7.3). 22 The injection was made at a rate of 0.5 μL/min using a 10-μL microsyringe (Hamilton, Switzerland). After each injection, the micropipette was left in place for an additional 5 minutes and then slowly withdrawn. In the same manner, sham rats received an injection of 4.0 μL of saline containing 0.01% ascorbic acid into the right SNc. One week after the surgery, animals were given apomorphine (0.05 mg/kg, subcutaneously) and those exhibiting more than 20 contralateral turns per 5 minutes were chosen for further investigation.
Single-Unit Recordings
Electrophysiological recordings were performed 3 weeks after the 6-OHDA lesioning of the SNc. Extracellular single-unit recordings were collected from rats that were anaesthetized with 4% chloral hydrate (400 mg/kg IP) and mounted in a stereotaxic frame as described previously. The body temperatures were maintained at 37°C ± 0.5°C, and the heart rate and pupillary diameter were monitored throughout the experiment. Glass microelectrodes (10-20 M) filled with 1% pontamine sky blue in 0.5 M sodium acetate were stereotaxically directed to the right DRN (AP: 7.8-8.0, ML: 1.6-1.9, and DV: 5.6-5.9). An oblique trajectory in the coronal plane that was angled 16° toward the DRN to the midline was chosen to avoid damaging the sagittal sinus. The neuronal firings were amplified, band-pass filtered, displayed on an oscilloscope, and stored in a computer equipped with the Spike 2 analysis system (Cambridge Electronic Design, United Kingdom) for off-line analysis.
The DRN neurons were characteristic as 5-HT-containing if they had off-line analysis—neurons with slow firing rates (0.1-4 spikes/second), long spike durations (2-4 milliseconds), single or bursting firing patterns, and action potentials with biphasic or triphasic extracellular waveforms. 23 -25
The GABA interneurons in the DRN were characterized by their shorter action potential durations (<1 millisecond), higher firing rates (5-40 spikes/second), and irregular firing patterns. The locations of all recorded neurons were histologically confirmed to be in the DRN.
Drug Administration Procedures
For the systemic administrations, the potent 5-HT7 receptor agonist AS-19 was injected intravenously (IV) in 3 doses (40, 160, and 640 μg/kg) at approximately 5-minute intervals. The potent selective 5-HT7 receptor antagonist SB 269970 was administered IV as a single dose (200 μg/kg) approximately 5 minutes following the last dose of AS-19. Working solutions (0.1 mL) were injected IV at the appropriate dosages over the course of 30 seconds.
Histology and Immunocytochemistry
At the end of each experiment, the recording site was marked by the injection of pontamine sky blue (20 A, 15 minutes). The rat was given an overdose of urethane and perfused with saline followed by 4% paraformaldehyde, and the brain was immediately removed and postfixed in the same fixative for 4 hours. The brain was then placed in phosphate-buffered saline (PBS) with 20% sucrose overnight. The brains were frozen and cut into 40-μm-thick sections using a cryostat. Cresyl violet staining of the sections mounted on gelatin-coated slides was used to determine the location of the recording sites (Figure 1A).
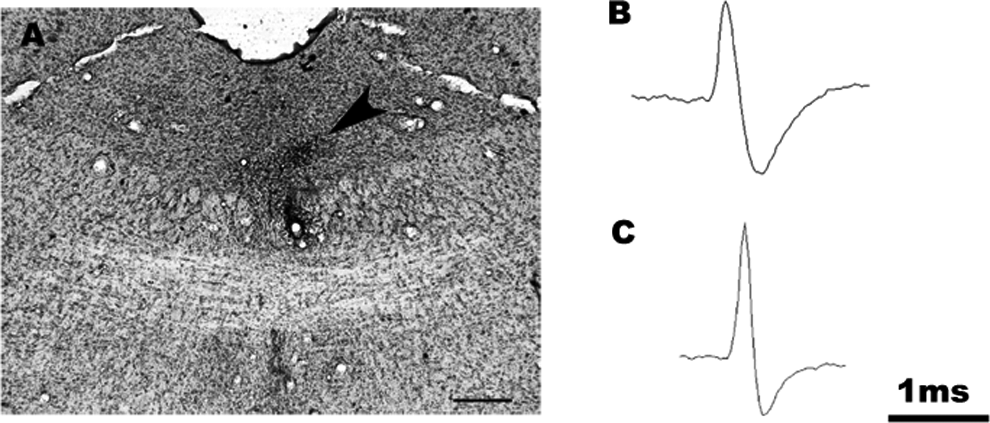
Histological photomicrograph showing the recording site (arrow) in a dorsal raphe nucleus (DRN) marked with iontophoretically injected pontamine sky blue in a sham-operated rat (A). Representative spikes of 5-hydroxytryptamine (5-HT; B) and γ-aminobutyric acid (GABA; C) neurons in the DRN. Scale bar = 0.5 mm.
To determine the extent of nigral dopaminergic degeneration, sections of the SNc from the rats that received 6-OHDA injections were examined with immunocytochemical staining for tyrosine hydroxylase (TH). Briefly, sections were preincubated with 3% bovine serum albumin in PBS containing 0.3% Triton X-100 for 30 minutes at room temperature and then incubated at 4°C for 48 hours with anti-TH polyclonal antibodies (1:1000; Chemicon, Buena Park, California). Next, the sections were incubated for 2 hours with biotinylated anti-mouse immunoglobulin G (1:200; Chemicon) and then incubated for 2 hours with the avidin–biotin–peroxidase complex (1:200; Vector Laboratories, Burlingame, California) at room temperature. Finally, the section was exposed at room temperature for 10 to 15 minutes to a solution of 0.05% 3,3′-diaminobenzidine (Sigma-Aldrich) containing 0.01% H2O2, which served as a chromogen for subsequent visualization. A rinsing procedure was applied to the sections between each step, excluding the blocking solution step and the addition of the primary antibody. The sections were rinsed, mounted on gelatin-coated slides, dehydrated, and cleared in xylene and coverslipped. Only rats with complete or nearly complete losses of TH immunoreactivity in the SNc were used in the analysis of the electrophysiological recordings (Figure 2A and B).
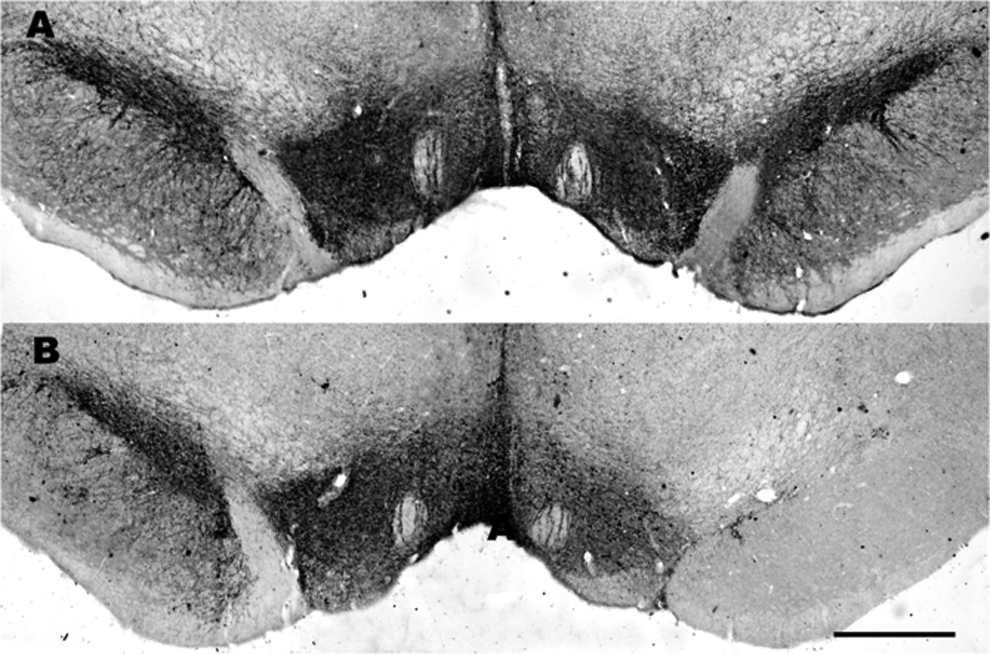
Photomicrographs of the tyrosine hydroxylase (TH) immunocytochemical staining of the substantia nigra pars compacta (SNc) and ventral tegmental area (VTA) after sham (A; n = 15) or 6-OHDA lesion surgeries (B; n = 21). The SNc dopaminergic neurons on the injected side (right) are compared to those on the uninjected side (left). Note the complete and partial degeneration of the TH-immunoreactive (IR) neurons in the SNc and VTA, respectively, on the lesioned side. Scale bar = 0.5 mm (A).
Data Treatment and Statistical Analysis
The baseline activities of the identified neurons were recorded for 5 to 10 minutes prior to any treatment. The changes in firing rate were quantified by analyzing 5-minute epochs after each AS-19 or SB 269970 injection. Comparisons of the firing rates between the different groups and the effects of drugs were assessed using 2-way analysis of variance (ANOVA). When appropriate, post hoc multiple comparisons were performed using Sidak tests. All data are expressed as the means ± standard error of the mean. The statistical analyses were performed using SPSS 15.0 for Windows, and the level of significance was set as P < .05.
Results
All of recording sites in the sham and 6-OHDA-lesioned rats were verified to be within the DRN (Figure 1A). The 6-OHDA-lesioned rats in this study consistently turned toward the side contralateral to the lesion more than 20 times within 5 minutes (41 ± 3 turns/5 minutes) following apomorphine stimulation.
The 5-HT neurons exhibited a typical long-duration action potential of 2 to 4 milliseconds that displayed a prominent positive deflection followed by a negative or negative/positive transient (Figure 1B). 23 The GABA interneurons in the DRN were characterized by their shorter action potential durations (<1 millisecond; Figure 1C). 26 -29
Effects of AS-19 and SB 269970 on DRN 5-HT Neurons
The systemic administration of the selective 5-HT7 receptor agonist AS-19 affected the DRN 5-HT neurons of the sham and PD rats differently (Figure 3A and B). Two-way ANOVA reported significant effects of both 5-HT7 receptor agonist (F 3,68 = 9.421, P < .0001) and 6-OHDA SNc lesioning (F 1,68 = 21.01, P < .0001), as well as an interaction between them (F 3,68 = 8.839, P < .0001; Figure 3C). In sham rats, Sidak multiple comparisons test revealed that the administration of AS-19 at 40 µg/kg did not affect the DRN 5-HT neurons (P = .057); however, significant effects were observed at the dose of 160 and 640 μg/kg (P = .0014 and <.0001, respectively), suggested that the administration of AS-19 (160-640 μg/kg, IV) dose dependently increased the firing rates of the DRN 5-HT neurons. In comparison to the sham rats, the administration of AS-19 in the 6-OHDA-lesioned rats did not affect the firing rates of the DRN 5-HT neurons at any of the doses tested (40-640 μg/kg, IV). To determine the effects of interaction between AS-19 and SNc lesioning on the putative DRN 5-HT neurons, we further compared the neuronal firing rates between the sham and PD model rats at each doses. Sidak multiple comparisons test reported significant differences between those 2 groups only at the baseline condition (P < .0001) and 40 μg/kg of AS-19 (P = .009). In addition, the excitation of the firing rate produced by AS-19 was completely reversed by the subsequent administration of SB 269970 at the dose of 200 μg/kg (IV) in both groups (Figure 3A and B), which indicated that this effect was mediated by 5-HT7 receptor.
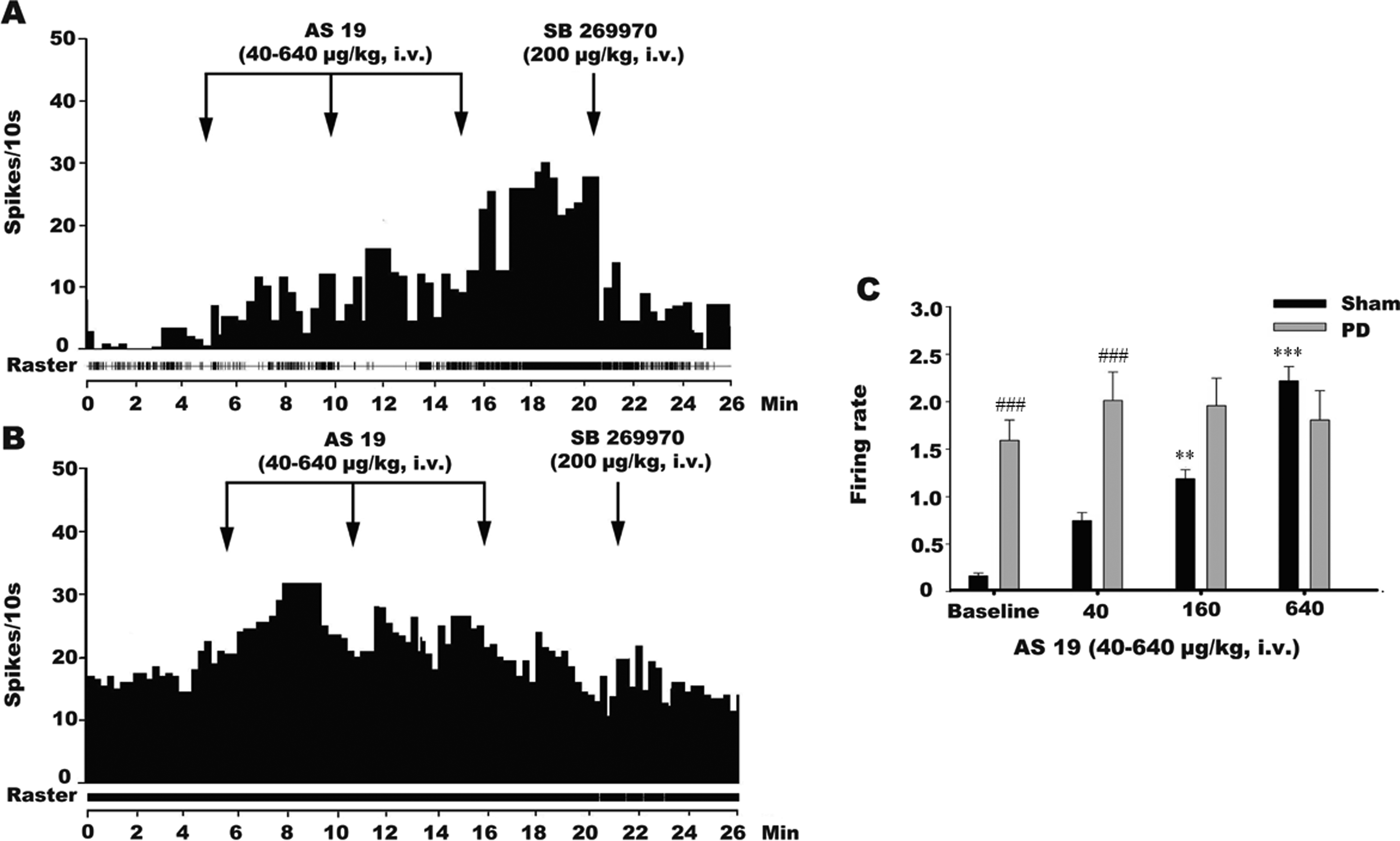
Histograms showing the effects of the systemic administration of the selective 5-hydroxytryptamine (5-HT)7 receptor agonist AS-19 on the 5-HT neurons in the dorsal raphe nucleus (DRN) of sham-operated and SNc-lesioned rats. The systemic administration of AS-19 at 40 to 640 μg/kg (intravenous [IV]) to sham-operated rats affected the firing rates of the 5-HT neurons in the DRN in an excitatory manner (A and C; n = 8). The excitatory effect of AS-19 was reversed by the administration of the selective 5-HT7 receptor antagonist SB 269970 at 200 μg/kg (IV) in the sham rats. At the same dose, AS-19 exhibited no effect on the 5-HT neurons in the DRNs of the substantia nigra pars compacta (SNc)-lesioned rats (B and C; n = 11). The arrows indicate the time of the IV administration of AS-19 or SB 269970. *P < .01 versus the baseline firing rate. Compare to the sham rats, the firing rate increased significantly at baseline, 40, and 160 dose in PD rats (C; n = 11). # P < .05 versus the firing rate of sham operation.
Effects of AS-19 and SB 269970 on DRN GABA Interneurons
We next examined the effect of AS-19 and SB 269970 on DRN GABA interneurons in the sham and PD rats (Figure 4A and B). Two-way ANOVA reported significant effects of both 5-HT7 receptor agonist (F 3,60 = 4.465, P = .0094) and 6-OHDA SNc lesioning (F 1,60 = 5.55, P = .0218), as well as an interaction between them (F3,60 = 4.498, P = .0065; Figure 4C). In the sham rats, the administration of AS-19 dose dependently inhibited the firing rates of the DRN GABA interneurons and elicited a significant effect at the high dosage of 640 μg/kg IV (post hoc, P = .042). In the 6-OHDA-lesioned rats, AS-19 increased the firing rates of the examined DRN GABA interneurons at the dosage of 640 μg/kg (post hoc, P = .048). Sidak multiple comparisons test also reported significant differences between those 2 groups at the baseline condition (P = .014), 40 μg/kg (P = .019), and 60 μg/kg (P = .039) of AS-19. In addition, the inhibition of the firing rate produced by AS-19 was completely reversed by the subsequent administration of SB 269970 at the dose of 200 μg/kg (Figure 4A and B), which indicated that this effect was 5-HT7 receptor mediated in rats.
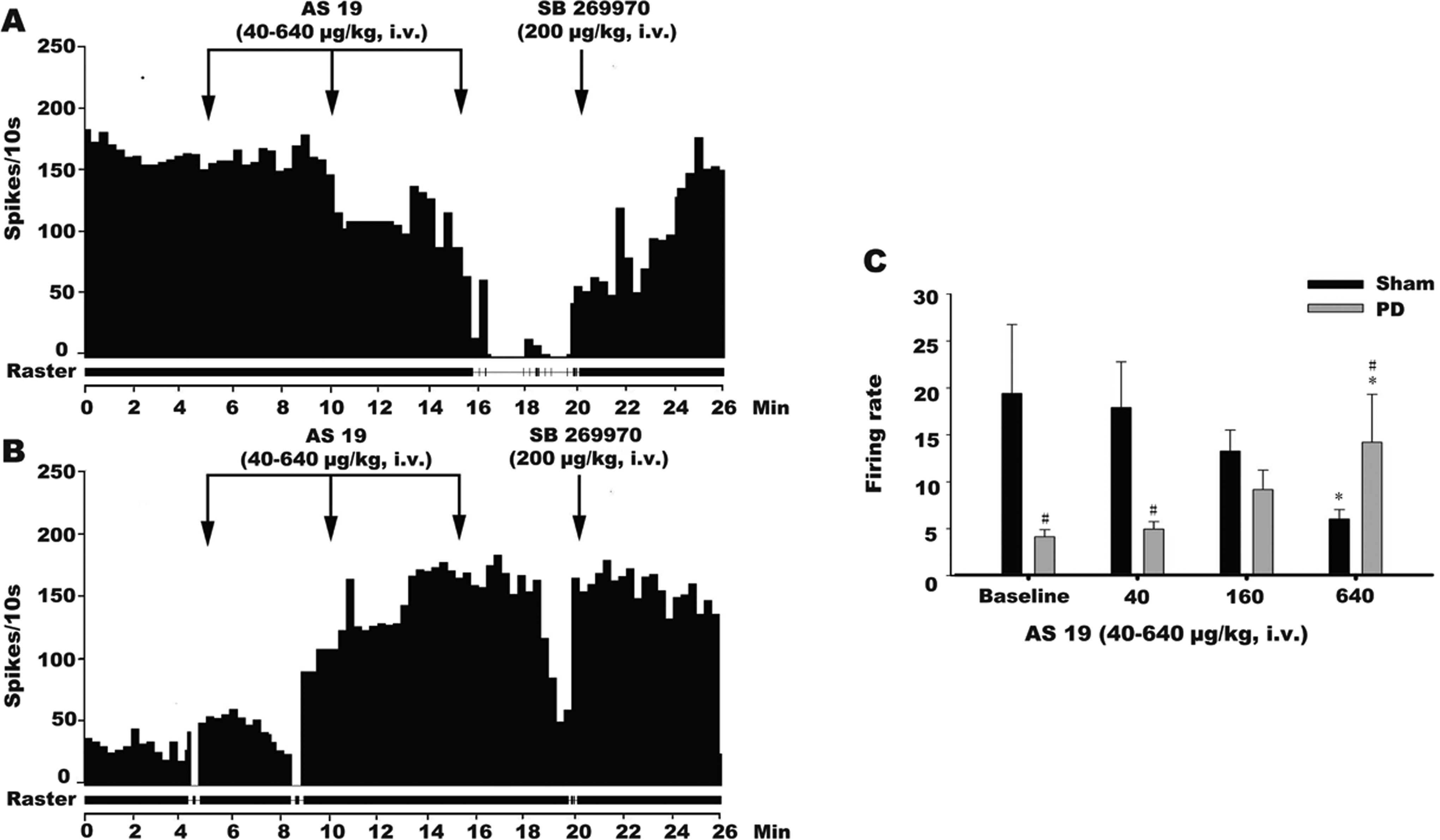
Histograms showing the effects of the systemic administration of the selective 5-hydroxytryptamine (5-HT)7 receptor agonist AS-19 on the γ-aminobutyric acid (GABA) neurons in the dorsal raphe nuclei (DRNs) of sham-operated and substantia nigra pars compacta (SNc)-lesioned rats. The systemic administration of AS-19 at 640 μg/kg (intravenous [IV]) in the sham-operated rats affected the firing rates of the GABA neurons in DRN in an inhibitory manner (A and C; n = 7). The inhibitory effect of AS-19 was reversed by the administration of the selective 5-HT7 receptor antagonist SB 269970 at 200 μg/kg (IV) in the sham-operated rats. AS-19 increased the firing rates of the GABA neurons in the DRNs of the SNc-lesioned rats, and this excitation was significant only at the high dose of 640 μg/kg (B and C; n = 10). The excitatory effect of AS-19 was reversed by the administration of the selective 5-HT7 receptor antagonist SB 269970 at 200 μg/kg (IV) in the SNc-lesioned rats. The arrows indicate the time of the IV administration of AS-19 or SB 269970. *P < .01 versus the baseline firing rate. Compare to the sham rats, the firing rate decreased significantly at baseline and 40 dose and increased at 640 dose in PD rats (C; n = 10). # P < .05 versus the firing rate of sham operation.
Discussion
The main results of the present study revealed the following: The systemic administration of the potent 5-HT7 receptor agonist AS-19 increased the mean firing rates of the 5-HT neurons in the DRN at the cumulative dose of 640 μg/kg (IV) in the sham rats. The systemic administration of AS-19 did not affect the mean firing rate of the 5-HT neurons of the rats with 6-OHDA lesions in the SNc. The systemic administration of the putative 5-HT7 receptor agonist AS-19 inhibited the mean firing rate of the GABA interneurons in the DRN at the cumulative dose of 640 μg/kg (IV) in the sham rats. The systemic administration of the potent 5-HT7 receptor agonist AS-19 increased the mean firing rate of the GABA interneurons at the cumulative dose of 640 μg/kg (IV) in rats with 6-OHDA lesions of the SNc.
Production of the Rat PD Model
In the present study, the PD model was achieved by unilateral 6-OHDA-induced lesions of the SNc DA neurons. 6-Hydroxydopamine was the first agent used to produce an in vivo animal model of PD. 21 The most common rodent PD model involves a unilateral injection of 6-OHDA into the SNc or the medial forebrain bundle (MFB), leading to rapid cell death within 1 to 3 days. After injection of 6-OHDA into the SNc, the drug is selectively toxic to DA neurons by the generation of free radicals. 6-Hydroxydopamine is an effective toxin in rats, mice, and primates and has been used predominantly to produce unilateral lesions. In rodents, the extent of DA depletion can be assessed by examining behavioral responses to amphetamine and apomorphine. 30 In this study, the 6-OHDA-lesioned rats consistently turned toward the side contralateral to the lesion more than 20 times within 5 minutes (41 ± 3 turns/5 minutes) following apomorphine stimulation. Thus, those data prove that the PD model was successfully produced.
The Firing Activity of the 5-HT Neurons in the DRN After SNc Lesioning
In rats with 6-OHDA lesions of the SNc, the firing rate of DRN 5-HT neurons markedly increased. A more recent study has also found both increased the firing rate of DRN 5-HT neurons and increased cytochrome oxidase activity in DA-deleted rats. 31 However, Guiard et al 32 have reported that the selective lesion of DAergic neurons induced by 6-OHDA decreases the spontaneous firing activity of DRN 5-HT neurons. This discrepancy may be explained by the following 3 reasons. First, it is possible that the rats with variable degrees of nigrostriatal lesion display different electrophysiological characteristics. In our previous study, we found a total loss of TH immunoreactivity in the SNc of the lesioned rats. 17 These results are in good agreement with those reported in rats with 6-OHDA lesions of the SNc showing a total or subtotal loss of TH immunoreactivity in the SNc and reduction of more than 95% in DA levels in the striatum. 33,34 Second, the intracerebroventricular injection of 6-OHDA may provoke alterations in other intracerebral structures that influence the firing activity of 5-HT neurons. Third, changes in the firing activity of 5-HT neurons may also relate to electrophysiological recordings at different time intervals after injection of 6-OHDA, since several studies have shown that the firing activity of thalamic parafascicular and subthalamic neurons has a time course of changes after 6-OHDA lesions of the SNc in rats. 35 -37 These changes at different time intervals may represent a compensating mechanism for the damaged DAergic system.
Effects of AS-19 on the 5-HT and GABA Interneurons in the DRN of the Sham Rats
The 5-HT7 receptor is a described member of the large family of 5-HT receptors, of which 14 members have already been described. 38,39 Several studies have demonstrated that the 5-HT7 receptor is not only present in the cortex but also in the thalamus, hippocampus, hypothalamus, amygdala, and dorsal raphe in a variety of species. 40,41 This receptor has been demonstrated to be positively coupled to adenylate cyclase via Gs proteins and to stimulate cAMP synthesis following activation by 5-HT and 5-HT7 receptor agonists. 42 Thus, activation of the 5-HT7 receptor results in an increase in the excitability of 5-HT neurons.
Regarding the GABA interneurons, the systemic administration of the potent 5-HT7 receptor agonist AS-19 inhibited the mean firing rate of these neurons in the DRN at the cumulative dose of 640 μg/kg (IV) in the sham rats. AS-19 increased the firing rates of the GABA interneurons through the activation of the 5-HT7 receptor. However, the 5-HT1A receptor can inhibit the firing rates of GABA interneurons. AS-19 could excite 5-HT neurons to release 5-HT, and the subsequent stimulation of 5-HT1A receptors on the GABA interneurons would result in the inhibition of the firing rate. It is possible that the effects of AS-19 included antagonism at low doses. However, the 5-HT1A receptor is more strongly affected than the 5-HT7 receptor at high doses; thus, the systemic administration of the potent 5-HT7 receptor agonist AS-19 at the high dose of 640 μg/kg inhibited the mean firing rate of the GABA interneurons in the DRNs of the sham rats.
Effects of AS-19 on 5-HT and GABA Interneurons in the DRNs of PD Rats
In the PD rats with 6-OHDA lesions in the SNc, the systemic administration of AS-19 did not affect the mean firing rate of the 5-HT neurons. The potential reasons for this observation include the following. First, AS-19 could increase the firing rates of 5-HT neurons through the activation of the 5-HT7 receptor; however, the 6-OHDA lesioning of the SNc induced an increase in the firing rates of the 5-HT neurons in the DRN, and the firing pattern was altered toward a burst-firing pattern in the PD rats. This explanation agrees well with our recent reports. 17 The firing of 5-HT neurons in the DRN is primarily under the control of the 5-HT receptor. 43 Neuroanatomical and neurophysiological studies have established that 5-HT1A receptors are coupled to a K+ channel through a G protein and are also coupled to adenylate cyclase. 44 The 5-HT1A receptors are localized to the soma and dendrites of 5-HT neurons in the DRN where they function as autoreceptors. Thus, their activation leads to the hyperpolarization of the membranes of 5-HT neurons, which inhibits spontaneous firing and reduces 5-HT release in the area of the cell body and the target structures of the 5-HT projections. 45 Therefore, it seems that the physiological role of the somatic and dendritic 5-HT1A autoreceptors is to function as sensors that respond by influencing the firing of 5-HT neurons and 5-HT release when the concentration of endogenous transmitter in the extracellular space is changed. The 6-OHDA lesions of the SNc induced an increase in the firing rates of the 5-HT neurons in the DRN, and the firing patterns shifted toward burst firing in the PD rats. This shift might have induced an increase in 5-HT release in the DRN, which might have inhibited the firing rates of the 5-HT neurons in the DRN through the activation of the 5-HT1A receptor. These 2 effects were nearly equal; thus, the systemic administration of AS-19 did not affect the mean firing rates of the 5-HT neurons in the rats with 6-OHDA lesions of the SNc. Additionally, the effect of AS-19 may have been elicited by a decline in the sensitivity of the 5-HT7 receptor; a similar effect has been observed in the 5-HT1A receptor. 17 It is also worth noting that in the present study 6-OHDA lesion induces an increment in the firing rate of DRN. However, other studies have reported a decrease 32 and not a change 46,47 in the DRN neurons from 6-OHDA-lesioned rats. This inconsistency observed in the firing rate of DRN may be due to the different brain areas lesioned by 6-OHDA. Previous studies observed firing rate of DRN after intraventral tegmental area or right MFB injection of 6-OHDA, whereas in our model, the SNc was injected with 6-OHDA.
The systemic administration of the potent 5-HT7 receptor agonist AS-19 at the cumulative dose of 640 μg/kg (IV) increased the mean firing rate of the GABA interneurons of the rats with 6-OHDA lesions of the SNc. The GABA interneurons are regulated by the 5-HT1A and 5-HT7 receptors. The AS 19-mediated increase in the firing rates of the GABA interneurons involved the 5-HT7 receptor. However, similar to the 5-HT neurons, 6-OHDA lesions of the SNc can also induce increases in the firing rates of 5-HT neurons in the DRN, and the firing patterns shifted toward burst firing in the rats, which might have induced an increase in 5-HT release in the DRN. This increase in 5-HT could have inhibited the firing rates of the GABA interneurons in the DRN via the activation of the 5-HT1A receptor, and this receptor could have exhibited a decline in sensitivity in the PD rats. Therefore, the systemic administration of the potent 5-HT7 receptor agonist AS-19 increased the mean firing rate of the GABA interneurons at the high dose of 640 μg/kg (IV) in the PD rats.
About SB 269970
Additionally, SB 269970 was able to reverse the excitatory and inhibitory effects of AS-19 in 5-HT and GABA interneurons, respectively, which indicates that these were 5-HT7 receptor-mediated effects. AS-19 is not a highly selective 5-HT7 receptor agonist; several studies have demonstrated that the selectivity of AS-19 for this receptor is moderate compared to its selectivity of 5-HT1A and 2a-adrenergic receptors. Additionally, AS-19 has affinities for the 5-HT2A, D1a, histamine H1, and muscarinic M1 receptors. 48 However, the 5-HT7 receptor antagonist SB 269970 has been reported to have a high affinity for the 5-HT7 receptor and excellent selectivity for this receptor over other receptors (250-fold greater selectivity for the 5-HT7 receptor relative to the 5-HT1A, 5-HT2, 5-HT4, 5-HT6,1, D2, and D3 receptors), with the exception of the 5-HT5A receptor (50-fold greater selectivity). 49,50 Several studies have also found that SB 269970 penetrates the central nervous system well and exhibits the features of a 5-HT7 receptor antagonist. 39 In sum, of the examined neurons, the excitatory and inhibitory effects produced by the systemic administration of AS-19 were both reversed by SB 269970 in both sham and SNc-lesioned rats, which indicates that the these effects were 5-HT7 receptor mediated.
Footnotes
Authors’ Note
Wang Shuang and Zhao Yan contributed equally to this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No. 81501175), the Science and Technological Project (2013K21-22-02, 2016JQ8043), and the Foundation of Education Department (15JK1616) of Shaanxi Province, China.
