Abstract
Recently, a strong interest has emerged in recognizing Parkinson’s disease dementia (PDD) at a very early stage. However, the specific profile of the earliest stages of PDD is still unclear and a matter of considerable controversy. The objective of this study was to find out early neuropsychological markers for progression of dementia in this population. Fifty-eight patients with PDD were divided into 2 subgroups on the basis of the Mini-Mental State Examination: very mild and mild. The comparison with 26 normal controls shows that very mild PDD had deficits on attention/executive functions, naming, visuospatial/constructional abilities and retrieval of the episodic memory. Patients with mild PDD showed additional deficits on coding of episodic memory. Moreover, we found that in this early stage of PDD, the progression of dementia is mainly related to deterioration of attention/executive functions as well as retrieval and coding of episodic memory.
Introduction
Historically, Parkinson’s disease (PD) has been considered a motor disorder. It has been also increasingly recognized that PD is frequently associated with cognitive deficits resulting in dementia in a substantial number of patients.
Executive dysfunction is thought to be at the heart of the cognitive dysfunction in patients with PD. Besides the executive dysfunction, previous studies have reported that most patients with Parkinson’s disease dementia (PDD) also develop deficits in memory, language, and visuospatial and constructional abilities.
1
However, the lack of operational criteria to define the clinical boundaries between PD and PDD until now hindered the precise definition of different stage of cognitive impairment and dementia
2
-5
and lead to the usage of nonspecific diagnostic criteria such as
In context of current potential therapeutic approaches for PDD, it has been important to establish specific and operationalized diagnostic criteria for early and precise diagnosis of PDD.
In 2007, the Movement Disorder Society (MDS) Task Force proposed specific criteria for the diagnosis of possible and probable PDD.
1
The MDS PDD criteria have already been shown to be more sensitive for the detection of PDD, detecting up to 22% more cases than the
We suggest that the usage of MDS PDD criteria will allow us to further evaluate the early PDD by assessment of cognitive change across progression of decline from very mild PDD (vmPDD) to mild PDD (mPDD). These data may have profound clinical implications for a patient’s prognosis and physicians’ treatment strategies. For this purpose, we compared the performance of healthy controls to 2 separate groups of patients with PDD, those with mPDD and those with vmPDD (based on MDS PDD criteria) by using comprehensive neuropsychological tests.
Methods
Participants
Patients were selected from participants who sought consultation at the University Hospital “Alexandrovska,” Department of Neurology, Sofia. They presented with parkinsonian signs with or without cognitive deficits and in the advance stages required hospitalization for the treatment of parkinsonism. The research was approved by the ethics committee of Medical University-Sofia, and all participants signed an informed consent prior to enrollment.
The evaluation process comprised detailed medical history, physical and neurological examinations, cognitive evaluations, appropriate laboratory tests, and neuroimaging. All patients underwent brain computed tomography (CT) or magnetic resonance imaging (MRI). Laboratory workup included complete blood count with differential, serum electrolytes and glucose, liver, and kidney function tests, thyroid function tests, venereal disease research laboratory (VDRL) test, and serum B12 and folate levels. Information regarding past medical, neurological, and psychiatric history was obtained from patients and family members (usually patient’s spouse or children). Psychiatric evaluation included a semistructured interview and Geriatric Depression Scale (GDS), 9 using cutoff score of 4/15. This cutoff on the GDS was recently used to characterize patients having clinically significant depression. 10 We used 2 screening measures—Mini-Mental State Examination (MMSE) 11 and Dementia Rating Scale (DRS) 12 —in order to apply the MDS criteria and place participants into groups. The DRS has been shown to be a valid scale of cognitive functioning in nondemented and demented PD patients 13,14 and its subscales show strong convergent and discriminant validity. 14 The clinical dementia rating scale (CDR) 15 was also used.
All available information was evaluated by an experienced neurologist (also trained in brain imaging) and by a neuropsychologist. Diagnosis of PD was based upon the UK Parkinson’s Disease Society Brain Bank clinical diagnostic criteria for PD.
16
Patients were between 55 and 85 years, native speakers with at least 8 years of formal education. Exclusion criteria were presence of major depression (based on
Parkinson’s Disease Dementia Groups
The diagnosis of PDD was made based on MDS task force criteria for dementia 1 : abnormalities in at least 2 cognitive domains, cognitive decline, and cognitive deficiency severe enough to impair activities of daily living. In addition, the onset of PD preceded the development of dementia by at least 12 months in all patients.
The attention/executive functions were evaluated through DRS subscales for attention, initiation, and conceptualization; for memory we used memory subscale of DRS, for constructional abilities—constructional subscale of DRS and for language abilities—semantic verbal fluency (supermarket).
The lack of internationally accepted criteria for staging of dementia in PD made us, similarly to other studies in the domain, 6 adopt MMSE score equal to or higher than 20/30 to categorize mPDD group.
In addition, an upper cutoff of 24 was chosen to divide the 2 subgroups of patients with PDD: vmPDD (24-30 on MMSE) and mPDD (20-23 on MMSE).
Based on significant theoretical knowledge, in Alzheimer disease (AD) a score of 24 is often used as the split point between very mild and mild dementia. 17-18 Several authors reported MMSE score below 24 in patients with PD as an indicator for definitive dementia 7,19-20 and MMSE score higher than 24 as a sign for possible cognitive impairment or early dementia. 21
Severity of Parkinsonism in patients with PD was evaluated using the Unified Parkinson’s Disease Rating Scale (UPDRS) part III
22
and the Hoehn and Yahr Scale
23
in the patient’s best on state.
Normal Controls Group
In order to compare the neuropsychological performances of PDD groups, we included a control group comprising 26 age-matched participants without history or symptoms of psychiatric and neurological disease and with preserved integrity of cognitive functions.
Neuropsychological Assessment
In this study we performed neuropsychological battery consisting of subtests and modified short forms of commonly used neuropsychological measures for further comprehensive evaluation of neuropsychological profile of both PDD groups. Episodic memory was assessed with Buschke Free and Cued Selective Reminding Test (FCSRT) 24 estimating free recall (number of items retrieved over 3 learning trials), total recall (number of words recalled with free and cued procedures over 3 learning trials), number of intrusions, recognition, and delayed free and total recall. Attention and executive functions were tested by Trail Making Test 25 part A (TMT-A) and part B (TMT-B; assessing the time to correctly relay all items in each of the trials), Modified Card Sorting Test 26 (MCST; assessing the number of categories achieved and perseverative errors), Digit Span forward of the Wechsler Adult Intelligence Scale, 27 and the Stroop Test 28 (assessing, in the third part, the number of items correctly named in 45 seconds). Language abilities were determined by the 15-item subset of Boston Naming Test (BNT), 29 the semantic verbal fluency (categories animals, assessing number of animals produced in 60 seconds), and the phonemic verbal fluency (letters M, assessing the number of words produced in 60 seconds). 30 Visuospatial abilities and constructional praxis were evaluated by clock drawing test 31 and ability to copy 5 complex designs. 32
Statistical Analysis
An analysis of variance (ANOVA) on each test was performed separately. Bonferroni method was used to counteract for multiple comparisons. The difference between both PDD groups in the Hoehn and Yahr staging and UPDRS motor subscale were evaluated with nonpair
Results
Demographic characteristics of patients and controls are presented in Table 1. Analysis of variance showed no significant intergroup differences in age (
Demographic Characteristics of Controls and Patients With Very Mild and Mild PDDa
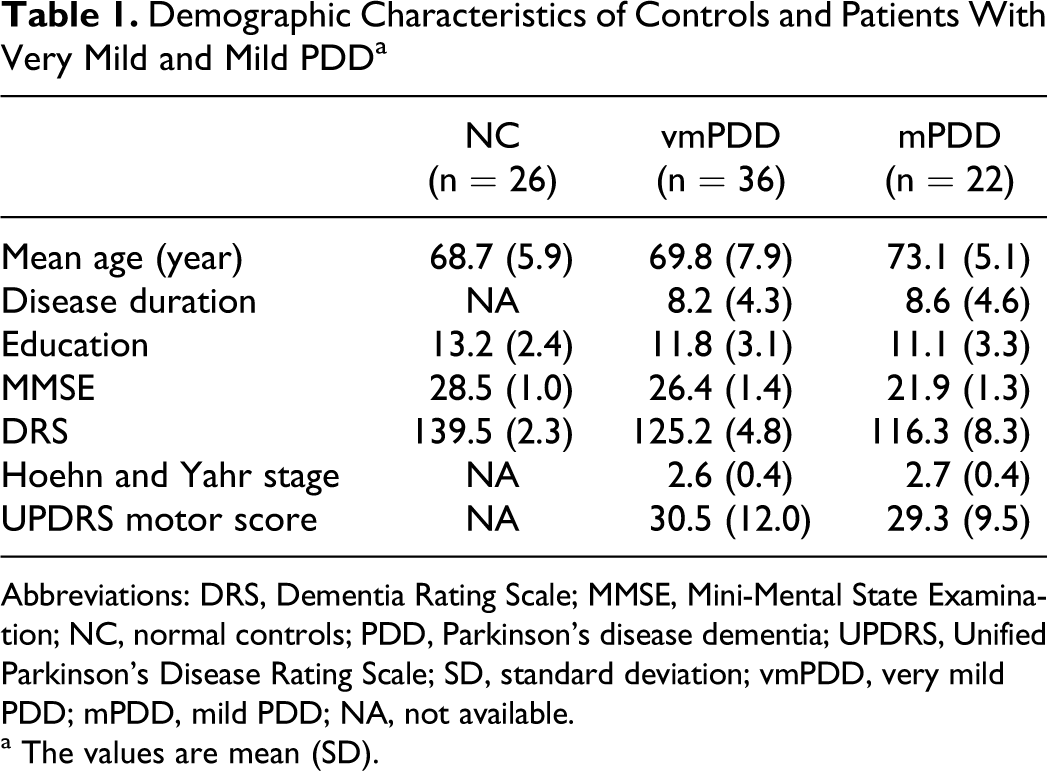
Abbreviations: DRS, Dementia Rating Scale; MMSE, Mini-Mental State Examination; NC, normal controls; PDD, Parkinson’s disease dementia; UPDRS, Unified Parkinson’s Disease Rating Scale; SD, standard deviation; vmPDD, very mild PDD; mPDD, mild PDD; NA, not available.
a The values are mean (SD).
On MMSE and DRS, the post hoc pairwise comparison using Bonferroni method (
The neuropsychological performances of the 3 groups are demonstrated in Table 2.
Neuropsychological Performance of 3 Groups
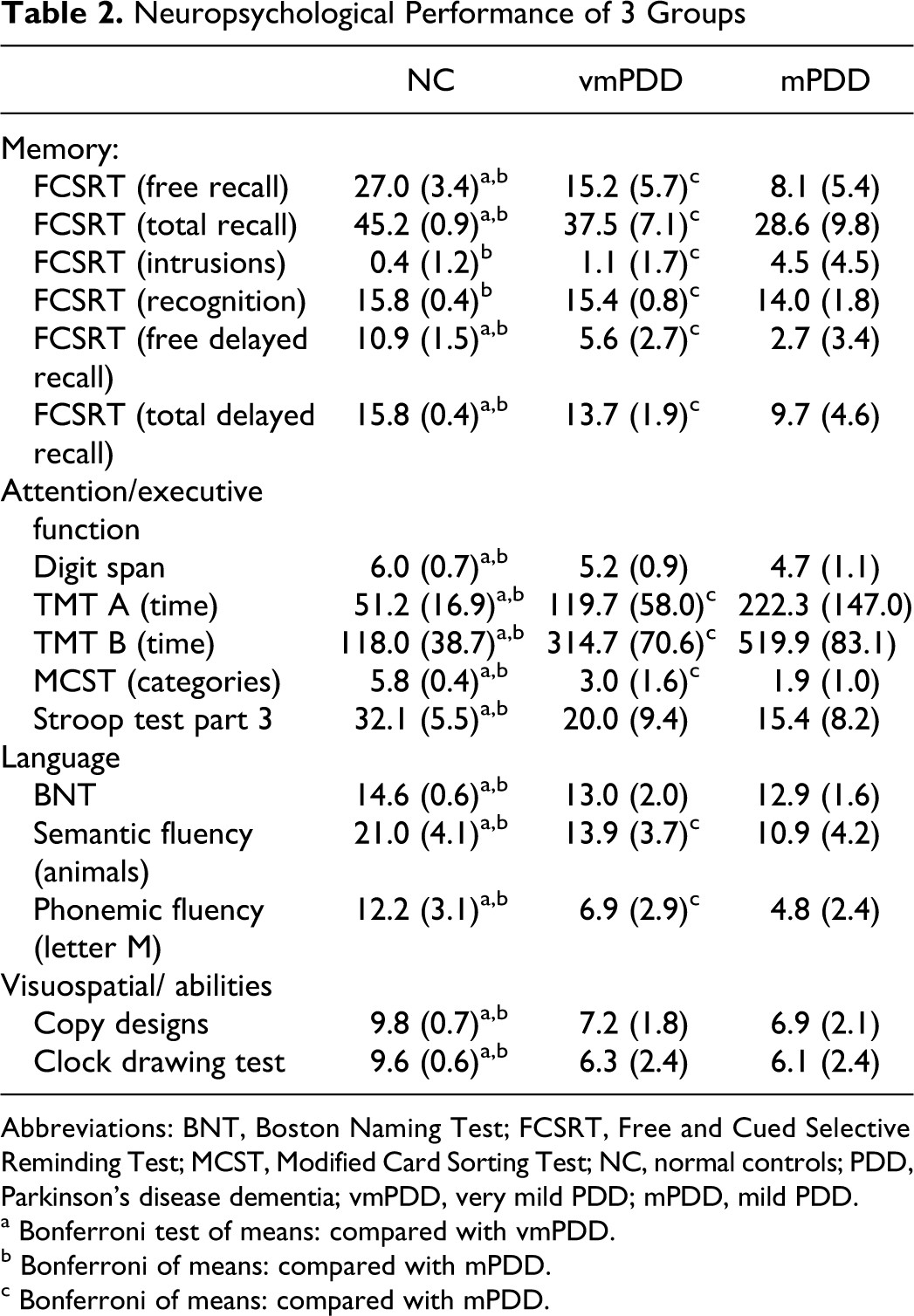
Abbreviations: BNT, Boston Naming Test; FCSRT, Free and Cued Selective Reminding Test; MCST, Modified Card Sorting Test; NC, normal controls; PDD, Parkinson’s disease dementia; vmPDD, very mild PDD; mPDD, mild PDD.
a Bonferroni test of means: compared with vmPDD.
b Bonferroni of means: compared with mPDD.
c Bonferroni of means: compared with mPDD.
On FCSRT statistical analysis indicated that both PDD groups performed significantly worse than controls on immediate and delayed free recall and immediate and delayed total recall, but more pronounced changes were noted in mPDD. However, only mPDD group revealed significantly lower score on the recognition compared to controls (
In terms of tasks encompassing various aspects of attention and executive functions, post hoc analysis also revealed marked difference between both PDD groups and controls. The post hoc analysis indicated that the mPDD group has more significant impairment than vmPDD on TMT-A (
Post hoc comparisons on language tasks indicated that both PDD groups were significantly impaired on BNT in comparison to controls (controls/vmPDD
The comparison between groups on clock drawing test and copy complex designs indicated significantly lower scores for both PDD groups as compared to controls. However, no difference was found between patients with vmPDD and mPDD on these tests (clock drawing test
Discussion
The cognitive changes in nondemented PD are typically characterized by a frontal–subcortical impairment with decreased attention and executive function leading to progressive impairment in prefrontal tasks, visuospatial skills, and memory. 33 The appearance of significant functional impairment resulting from cognitive impairment is considered as the primary feature that differentiates mild cognitive impairment (MCI) from PDD. 1,34 In this study, we found that the vmPDD stage is related to deficits on attention/executive functions, naming, visuospatial/constructional abilities, and retrieval of the episodic memory. Moreover, we found that the progression of dementia to the mPDD stage is mainly related to deterioration of attention/executive functions as well as retrieval and coding of episodic memory.
In the domain of memory, both PDD groups showed significant difference in the long-term episodic memory profile but not in the short-term memory. The memory profile typically described in PD includes poor free recall, improved total recall (enhanced performance when given retrieval cues), few intrusions, and relative sparing of recognition performance suggesting a retrieval deficit. 35 -38 In fact this was the memory profile that we found in our vmPDD group. In contrast, patients with AD typically exhibit a memory profile with poor free and total recall, high number of intrusions, and impaired recognition indicating an encoding deficit. 39,40 The marked difference in memory profile between PD and AD probably reflects the difference in the involved brain regions. 41 With the progression of dementia syndrome and the development of mild dementia, patients demonstrated a memory profile that partially resembles typical PD memory profile and partly the one typically observed in AD. Patients with mPDD showed impaired free recall with improved cued recall similar to typical PD profile. On other hand, our patients with mPDD also showed increased number of intrusions in immediate recall and impaired recognition that is typically observed in patients with AD. 39 These data suggest the episodic memory impairment in mPDD is probably due to deficit of retrieval as well as encoding.
Cognitive deficit in the domain of attention/executive functions was found in both PDD groups. With the progression of dementia, it could be noticed that a deterioration in some neuropsychological tests largely accepted as measurements of attention and executive functions, such as selective attention, psychomotor speed, concept formation, set formation and set shifting, and cognitive flexibility.
In the domain of language, PD patients with very mild dementia showed marked deficit in verbal fluency tasks and naming. With the progression of global cognitive deficit, a deterioration in verbal fluency tasks was found but not in naming. In comparison to verbal fluency, naming is considered to be very sensitive to the integrity of the semantic system and imposing fewer demands on effortful self-initiated retrieval. 42 In the domain of visuospatial and constructive abilities, our vmPDD group demonstrated significant deficit without marked progression in the transition to mPDD.
One other published study has also addressed the profile of mPDD.
6
However, unlike our study, Song and colleagues evaluated the progression from PD-MCI to mPDD based on
Our study has some limitations that need to be addressed. First, an important concern in studies involving patients with PD is the accuracy of clinical diagnosis. Although we cannot completely exclude misdiagnosis in our patients, this possibility was minimized by using detailed clinical, neuropsychologic, and neuroimaging assessment, and trained research team consisting of neuropsychologist and neurologist (trained in brain imaging). Furthermore, we used the UKPDS Brain Bank criteria, estimated in a clinicopathological study to have a diagnostic accuracy of 90%. 43 Second, the impaired motor performance may have affected patients’ scores on cognitive tests. However, the neuropsychological examination was performed in the patient’s on state and we did not find significant differences in the UPDRS motor subscale between both PDD groups. Thus, it is less likely that motor function significantly contributed to this difference.
In conclusion, we found several significant differences in the cognitive profile of PD in the transition between very mild and mild dementia. First, the progression of dementia is related predominantly with the progression of attention/executive function deficit. This suggests that in this stage of dementia, the deterioration in frontostriatal dysfunction is still predominant as it is in PD-MCI stage. These data were supported by several studies investigating the profile of cognitive impairment in patients with PDD. 3,44,45 Second, with the development of mild dementia PD patients demonstrate additional deterioration in memory process that was considered related to frontostriatal dysfunction, as well as to dysfunction in hippocampus, similar to AD. Recently, several neuropathological and functional studies showed that hippocampus is seriously impaired in PD, 41,46,47 and this involvement increases with the progression of cognitive deficit. 48 -50 Therefore, we found that the transition from very mild to mild dementia is primarily related to deterioration of attention/executive function and memory.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by a grant from the Medical University-Sofia, Bulgaria.
