Abstract
We sought to investigate whether the Montreal Cognitive Assessment (MoCA) could provide a brief assessment of recall and recognition using Huntington disease (HD) and Alzheimer disease (AD) as disorders characterized by different memory deficits. This study included 80 participants with HD, 64 participants with AD, and 183 community-dwelling control participants. Random-effects hierarchical logistic regressions were performed to assess the relative performance of the normal control (NC), participants with HD, and participants with AD on verbal free recall, cued recall, and multiple-choice recognition on the MoCA. The NC participants performed significantly better than participants with AD at all the 3 levels of assessment. No difference existed between participants with HD and NC for cued recall, but NC participants performed significantly better than participants with HD on free recall and recognition. The participants with HD performed significantly better than participants with AD at all the 3 levels of assessment. The MoCA appears to be a valuable, brief cognitive assessment capable of identifying specific memory deficits consistent with known differences in memory profiles.
Memory is a complex, multidimensional construct. In order to parse it into more concrete components, neuropsychologists often refer to memory with respect to particular, constituent cognitive processes (eg, encoding vs retrieval) or the informational content to be recalled (eg, semantic vs episodic). Unfortunately, its multidimensional nature presents challenges for assessment, especially in clinical and clinical trial settings where time constraints are limiting. Brief measures that examine multiple dimensions of memory functioning are needed to facilitate assessment of patients with different forms of dementia.
Many disorders (eg, Alzheimer disease [AD], mild cognitive impairment [MCI]) have historically been characterized and diagnosed solely by changes in cognition, including memory. Other disorders, such as diseases affecting the basal ganglia (eg, Huntington disease [HD], Parkinson disease [PD]), have also been shown to produce significant changes in memory. 1 -3 For instance, in HD, overall memory, as well as visual, verbal, and episodic memory, have been shown to decline over the course of disease progression. 4 -7 Over the past few decades, the interest in and recognized importance of assessing memory in diseases such as HD and PD have increased immensely. Unfortunately, the challenge of assessing memory briefly now pervades the evaluation of these disorders as well, and many assessments of memory profiles are time consuming and difficult to administer for examiners and to complete for patients. In order to increase clinicians’ and researchers’ abilities to examine specific memory profiles, brief yet sensitive measures are needed—the Montreal Cognitive Assessment (MoCA) may be such a measure. The purpose of this study is to investigate whether the MoCA could provide a brief assessment of unique recall and recognition issues found in patients with HD and AD.
The MoCA is a relatively new, brief cognitive assessment. It was developed by Nasreddine et al as a screening measure to assess the more subtle deteriorations of memory that occur in MCI. 8 Research exploring the utility of the MoCA suggests it may have sensitivity and specificity superior to other commonly used brief cognitive assessments (eg, the Mini-Mental State Examination [MMSE]) for a variety of dementing disorders and general cognitive deficits. 2,8 -14 Researchers have demonstrated that the MoCA is effective in identifying cognitive impairment among patients with PD and HD. 15 -17 Specifically, among patients with HD, the MoCA has been shown to have high specificity and sensitivity in detecting cognitive dysfunction. 16 It has also been studied for its utility in assessing cognitive functioning among patients with AD. Cecato et al compared the total MoCA score and subtest scores among healthy control, participants with MCI, and participants with mild AD. 18 They showed that the MoCA total score effectively discriminated between the groups and that clock drawing, rhino naming, word recall, and orientation subtests differentiated between those with AD and MCI. Costa et al further demonstrated that the MoCA effectively detects changes in cognitive functioning over time among patients with AD. 19 In addition, Julayanont et al demonstrated the utility of the MoCA in predicting the risk of conversion from MCI to AD. 20 These findings suggest that the MoCA may have more diverse uses with patients with various dementia than are currently being used by researchers and clinicians. It is possible that the MoCA may be useful as a measure of recall and recognition (ie, encoding and retrieval) among patients with etiologically diverse memory disorders. Given that the MoCA examines different aspects of cognitive functioning, it should be able to identify different types of memory impairment found among patients with AD and HD. This study examined this hypothesis.
The MoCA, like the MMSE, is a 30-point assessment of global function. 21 However, the MoCA evaluates a broader array of cognitive functions (eg, executive functioning, memory, etc) without sacrificing brevity. 10 From the perspective of examining recall and recognition memory, the MoCA provides a means of tracking free, category-cued, and multiple-choice recall—though this is an optional aspect of the instrument. Also, the MoCA appears to have decreased susceptibility to educational and cultural artifacts and is robust to major ceiling effects in normal control (NC) groups. 8,10 As a global assessment, Nasreddine et al proposed a cutoff of 26 versus 25 for identifying MCI. 8 No additional cutoff was established for dementia, as the test was not originally designed to demarcate MCI from AD. 8 Some researchers have suggested that a lower cutoff for MCI (eg, 23 vs 22) might yield greater specificity without diminishing sensitivity. 11,18 Though research has explored performance on the MoCA in AD, PD, and HD, no official cutoffs exist for these disorders as-of-yet. Nevertheless, on the whole, the MoCA appears to be a useful, brief assessment of global cognitive ability in both clinical and general populations.
Therefore, it seems that exploring the potential usefulness of particular aspects of the MoCA for the assessment of AD and HD is worthwhile as a means of enhancing cognitive assessment without increasing time demands. Given that various dementias are characterized by differing patterns of recall and encoding issues,
22
-27
it is possible that the free recall, cued recall, and recognition components of the verbal memory task on the MoCA could distinguish between types of dementia. With AD having notable encoding deficits
22,26
and HD being characterized by retrieval failures that benefit from cuing,
22,24
these 2 dementias are appropriate for this evaluation, which seeks to determine whether such nuanced aspects of memory deficits can be distinguished by assessing all levels of recall performance (ie, free recall, cued recall, and multiple-choice recognition, not just the free recall score) on the MoCA memory section. Given that AD and HD are easily distinguishable diseases, which also differ in this regard, they serve as optimal dementias upon which to perform this initial analysis to determine whether
Method
Participants
In the current, retrospective, archival study, we examined recall and recognition memory in 80 participants with well-characterized HD, 64 participants with well-characterized AD, and 183 community-dwelling, NC using the MoCA. Participants were followed at the University of California, San Diego (UCSD) Huntington Disease Center of Excellence (HD COE) and the UCSD Shiley-Marcos Alzheimer’s Disease Research Center (ADRC). The institutional review board of UCSD approved this study, and all participants provided informed consent. Participants completed the MoCA as part of a more exhaustive cognitive and medical assessment. Participants with AD were diagnosed by staff neurologists at the ADRC, and participants with HD were classified on the basis of the diagnostic confidence levels assigned by an experienced movement disorder neurologist (J.C-B.) on the Unified Huntington Disease Rating Scale. 27 Controls consisted mainly of spouses and friends of patients seen through the UCSD HD COE and ADRC. The remaining participants were recruited from the San Diego community as part of a convenience sample. Any prospective control participants with MMSE scores lower than 27 were excluded from the analyses. For demographic information and average MMSE and MoCA scores, see Table 1.
Demographic and Clinical Characteristics of HD, AD, and NC Groups.
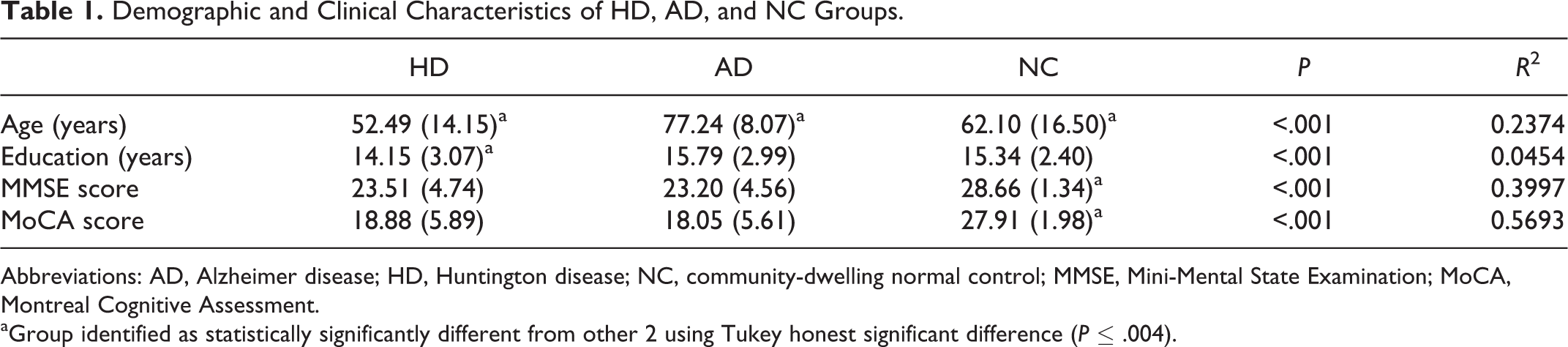
Abbreviations: AD, Alzheimer disease; HD, Huntington disease; NC, community-dwelling normal control; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment.
aGroup identified as statistically significantly different from other 2 using Tukey honest significant difference (
Procedures
Participants were given the original English version of the MoCA. During this test, a list of 5 words (FACE, VELVET, CHURCH, DAISY, RED) was read to the participants twice. After each reading, participants were asked to repeat the words back to the examiner. After the second iteration, participants were told to remember this list of words for a later portion of the test. 28 After a 5-minute delay, during which other cognitive tasks are performed, participants were asked to recall as many of the 5 words as they could, in any order. If there was a word a participant was unable to recall, they were provided with a category cue (eg, “the word you are trying to remember was a color” [for RED]). Finally, if a participant was unable to recollect the word following this prompt, they were given 3 category appropriate options (eg, “was it RED, was it BLUE, or was it GREEN” [for RED]) and asked to identify which word was the one that was on the list. The progression of this assessment lent itself to the use of binary logistic regression models, which were used to analyze the probability of success or failure on each task (ie, free recall, cued recall, recognition) as a function of diagnostic status.
Analyses
Maximum-likelihood models were used to examine differences across HD, AD, and NC groups, because of the binary nature of the outcome (ie, correct/incorrect). The analysis was a random-effects binary logistic regression panel model where words were nested within participants. Ability to recall or recognize
Results
The first analysis examined free recall outcomes (N = 1570;
The second analysis assessed category cued outcomes (N = 776,
The third analysis examined multiple-choice recognition outcomes (N = 543,
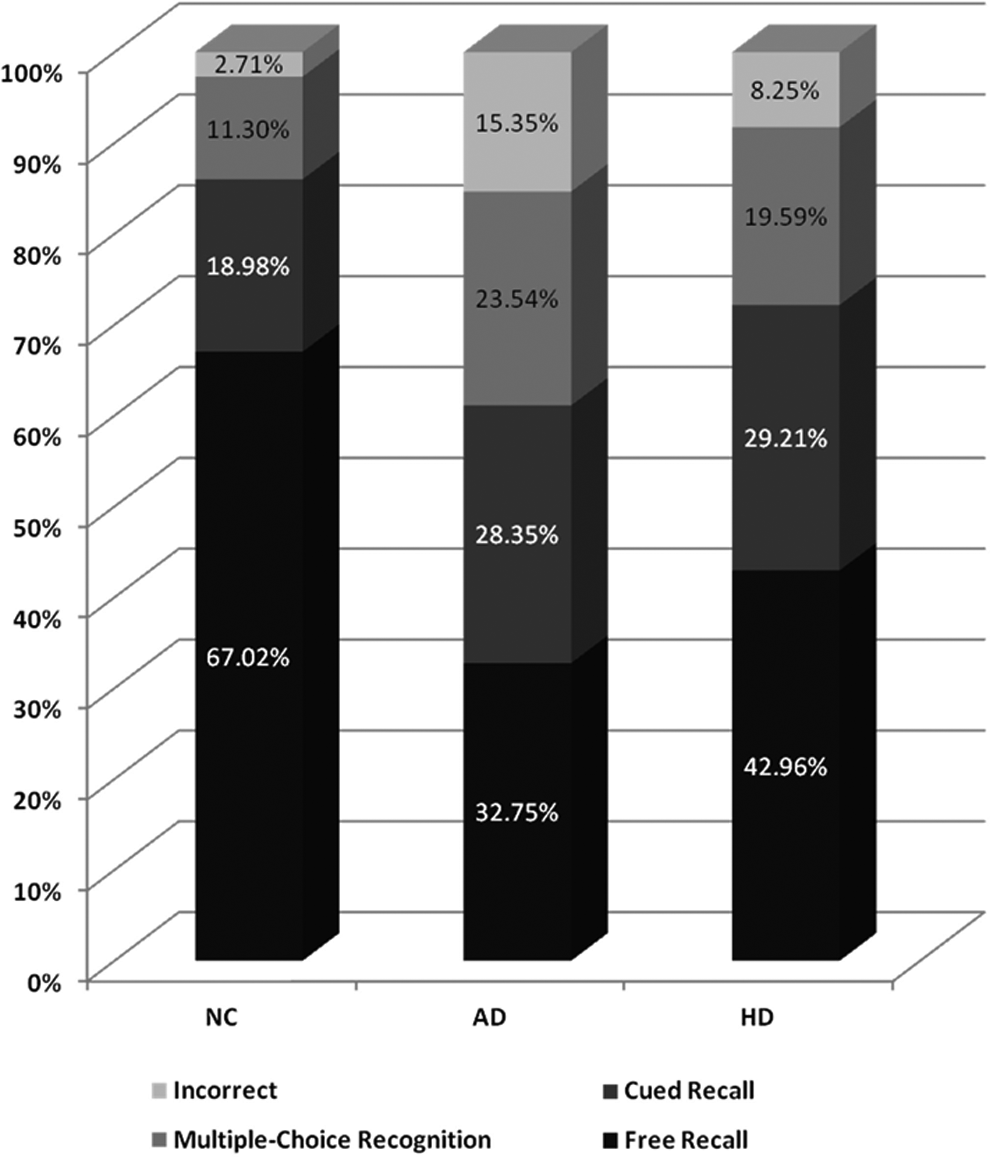
Relative frequencies of free recall, cued recall, multiple-choice recognition, and incorrect responses for normal control, participants with AD, and participants with HD. AD indicates Alzheimer disease; HD, Huntington disease; NC, normal control.
Discussion
In the current study, we examined the utility of the MoCA as a means of identifying specific memory functions (ie, recall and recognition) in AD and HD. To our knowledge, this is the first study to investigate this directly. We found that the MoCA was capable not only of differentiating NC from participants with AD and HD at each level of memory performance (ie, free recall, cued recall, and multiple-choice recognition) but also of differentiating participants with AD and HD.
Our examination of recall and recognition memory differences between participants with HD, participants with AD, and NC provides valuable insight into the prospective utility of the MoCA as a brief, clinical assessment to analyze memory profiles in dementia. It suggests that the MoCA, already recognized as a useful cognitive measure, has utility beyond that which is often exploited currently by researchers and clinicians. Clearly, examining specific aspects of recall and recognition in HD and AD populations provides detail that may be lost in analyses that simply average across these levels (eg, using the total score for memory on the MoCA).
The patterns of differentiation among HD, AD, and NC samples based upon free recall, cued recall, and multiple-choice recognition are consistent with previously reported findings using other measures of memory. 22 -25 First, the analyses demonstrate what would be expected for the differences between NC and participants with AD; specifically, that no statistically significant benefits were conferred via any form of cuing for the participants with AD. This would be anticipated, as AD is a disorder associated with encoding deficits, especially as the disorder progresses, but which may be present early in the disease course as well. 26,29 Given that other evidence has suggested that consolidation deficits may be the early indicators of AD, with encoding failure manifesting later in the disease course, 30 future research would benefit from examining clinical populations with this form of analysis controlling for disease duration or severity.
At the free recall level, perhaps as would be expected, NC participants were significantly more likely to correctly recall the target word than participants with HD. However, at the cued recall level, NC and participants with HD performed statistically equivalently. Thus, among NC and participants with HD who were unable to recall freely a target word, their probability of recalling correctly after a category cue was statistically equivalent. Yet, for those of the participants with HD whose memory had been impacted severely (ie, those who could not recall the word even when cued), it seems that both retrieval and encoding deficits manifest, as even recognizing the correct word became a task with statistically significant proportions of failure compared to NC participants. This is consistent with past research that has shown that patients with HD in more advanced stages of the disease show recognition deficits comparable to patients with AD. 31 However, we cannot conclude definitively that encoding deficits, not retrieval deficits, were the mechanisms underlying the subpar recognition performance observed in our study. These findings are consistent with past research and provide statistical, face, and clinical validity to the MoCA with respect to its ability to examine memory encoding and retrieval processes specifically.
Another finding in support of the MoCA’s usefulness was that it identified a relative increase in performance for participants with HD in the category cued recall condition. This is consistent with past research that has demonstrated that individuals with HD benefit from cuing.
22
-25
However, it is important to remember that these analyses utilized the standard progression of clinical assessment using the MoCA, meaning that
The greatest clinical implications of this finding appear to be that the MoCA can be used in a way that is beyond its typical scoring to provide greater detail regarding memory deficits. The fact that such a brief test can distinguish these 2 differing dementias in a way that is consistent with what is known about them is meaningful. Although this is not meant to insinuate that the MoCA would be a diagnostic tool per se, it does provide evidence for the MoCA’s ability to provide greater clinical insights than often gleaned from it presently. This is particularly true given the fact that at the free recall level (which is all that is scored on the MoCA), participants with HD and AD did not differ significantly from one another. This finding is consistent with data from Table 1, which shows that neither MoCA nor MMSE scores alone differentiated these 2 groups, but these analyses of memory subtyping were able to do so. Thus, although it may not be meaningful clinically per se to differentiate patients with HD and AD in this fashion—as there are many well-established means to do this—it is meaningful to find that specific types of memory differences can be identified by the memory portion of the MoCA beyond its present scoring of providing a point for every freely recalled word. This type of additional analysis may be useful in distinguishing other, less differentiable memory disorders (eg, vascular dementia and AD) by assessing particular memory profiles. Patients with HD and AD merely provided a worthwhile opportunity to explore this possibility among 2 distinguishable diseases to confirm that the known patterns of memory deficits were, in fact, identified by such a brief—and previously untapped—resource.
There are a few cautions that should be borne in mind when interpreting these findings. First, we used convenience, although well characterized, samples from 1 academic center. Second, the nature of the MoCA is such that the possibility of differential carryover effects poses a realistic concern. Although our use of panel data analysis helps to control for the within-person differences in recall typologies, it cannot replace methodological protections for such prospective problems (eg, randomization of participants in free recall only, cued recall only, and multiple-choice recognition only conditions for analysis). Also, it is important to bear in mind the sequential nature of our analyses; specifically, entering individuals who performed perfectly at the free recall level would have affected
Conclusion
Our research shows that the MoCA is a test with utility beyond what is acknowledged widely or considered in its scoring. It suggests that the MoCA is a rapidly administered measure capable of identifying specific memory deficits in both AD and HD. These findings hold implications for clinical practice. The MoCA offers a brief and sensitive measure to assess patients with AD and HD and differentially identify areas of memory deficit. This knowledge will provide practitioners with necessary information to tailor interventions that address the specific presentation of memory impairment. Consistent with previous research, this supports the value of the MoCA as a brief cognitive assessment for many forms of dementia. It seems that future research should further explore the full breadth of the usefulness of the MoCA, exploring correlates of differential recall on this test and considering including this aspect in its scoring.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the UCSD Huntington Disease Society of America Center of Excellence and UCSD Shiley-Marcos Alzheimer Disease Research Center NIH P50 AG 005131.
