Abstract
To determine the predictors of cognitive decline in a rural and remote population with Alzheimer’s disease (AD), we examined the association between cognitive change and sociodemographic, clinical, and functional data at the initial day of diagnosis. Simple linear regression analysis and multiple regression analysis were used to determine the predictors of cognitive decline as measured by the difference in the Mini-Mental State Examination over 1 year. Our sample included 72 patients with AD. Age at the clinic day appointment was 75.3 (standard deviation [SD] = 7.44). History of hypertension and decreased ability to carry out activities of daily living were statistically significant and predicted greater cognitive decline at 1 year. Many previously suggested predictors of cognitive decline were not evidenced in this study. This research helps identify clinically useful predictors of decline in a rural and remote population with AD.
Introduction
There is a considerable variation in the rate of cognitive decline in patients diagnosed with mild cognitive impairment or dementia. 1 Rapidly deteriorating scores in the Mini-Mental State Examination (MMSE) of patients with dementia have been shown to predict a poor prognosis. 2 Reliable predictors of cognitive decline would have a great clinical utility. Some have been suggested, including younger age, 3 higher education, 3 less cognitive and functional abilities at baseline, 4 family history of dementia, 4 less anxiety, 5 depression, 6 falls, 6 and higher caregiver burden. 6
Predictors of decline may vary between different populations and settings. For example, some studies have found increased duration of formal education to predict decline. 3,5,6 On the other hand, a study by Fritsch and colleagues has found increased years of education to be a predictor of less decline, 7 and a study by Suh et al found education to have no effect at all on decline. 8 There is controversy surrounding other predictors such as diabetes, gender, and body mass index. Studies often contain specific populations, and the assessments of their samples often vary. A further review of each of these potential predictors will be found in the Discussion section.
We aim to determine the predictors of cognitive decline in rural patients with Alzheimer’s disease (AD) by assessing clinical data from a rural population in the Midwestern Canadian province of Saskatchewan. Clear predictors of cognitive decline within this population could help health-care providers residing in similar communities to navigate appropriate treatment scenarios and allow for time-sensitive discussions with patients and their families.
Methods
Data Collection and Ethical Consideration
Data collection began in March 2004 at the Rural and Remote Memory Clinic (RRMC) in Saskatoon, Saskatchewan. The RRMC was created in 2004 in an attempt to alleviate gaps in dementia care for patients living in rural and remote areas of Saskatchewan. 9 Noninstitutionalized patients are referred to the clinic by their family physicians. The clinic provides an interdisciplinary approach to dementia care, shortens time to diagnosis, and reduces patient’s travel. 9 Follow-up appointments between professionals and patients separated by distance are delivered through telehealth to minimize the time, expense, and inconvenience of travel for patients and their caregivers. 10
Ethical approval was granted by the University of Saskatchewan Behavioural Research Ethics Board, and all patients and their families gave informed consent prior to participation. As the RRMC is directed toward patients with early-stage memory loss, we assumed the majority of patients were competent to provide consent. To account for instances of diminished capacity, proxy consent from all caregivers was also obtained with patients’ knowledge.
The assessment began with a preclinic telehealth appointment followed by an in-person clinic visit several weeks later. During this visit, the patient and family members met with and were jointly interviewed by a neurologist, neuropsychology team, and physical therapist. Next, these clinic team members assessed the patient individually. A standardized neuropsychological battery was administered, which included an MMSE. 9 Family members completed a questionnaire that included measures of caregiver burden, distress, health, and ratings of the patient’s behaviors and functional abilities. Patients also completed a questionnaire that included self-ratings of mood, memory, functional ability, and quality of life. The physical therapist assessed patients to determine their physical abilities and to identify the areas of concern. Unless recent imaging had been done, all patients underwent computed tomography brain scans. Sociodemographic and clinical information was collected for each patient. Following an end-of-day team conference, patients and family members met with the neurologist and neuropsychology team who provided feedback based on the day’s assessment, information about probable diagnosis, and recommendations for management and care. Patients were then seen in follow-up, generally through telehealth, at 6 weeks, 12 weeks, 6 months, 12 months, and annually thereafter or more often when clinically indicated. The MMSE was administered at each follow-up appointment. The MMSE scores recorded on the initial clinic day (diagnosis) and at the 12-month follow-up were used in this study.
Participants
All 72 individuals seen at the RRMC between 2004 and 2013 who were diagnosed with probable AD by National Institute of Neurological and Communicative Disorders and Stroke-Alzheimer’s Disease and Related Disorders Association (NINCDS-ADRDA) criteria 11 and had completed both a baseline clinic day MMSE and a 1-year follow-up MMSE were included in the present study. No exclusion criteria was set for this group and all 72 individuals participated.
Measures
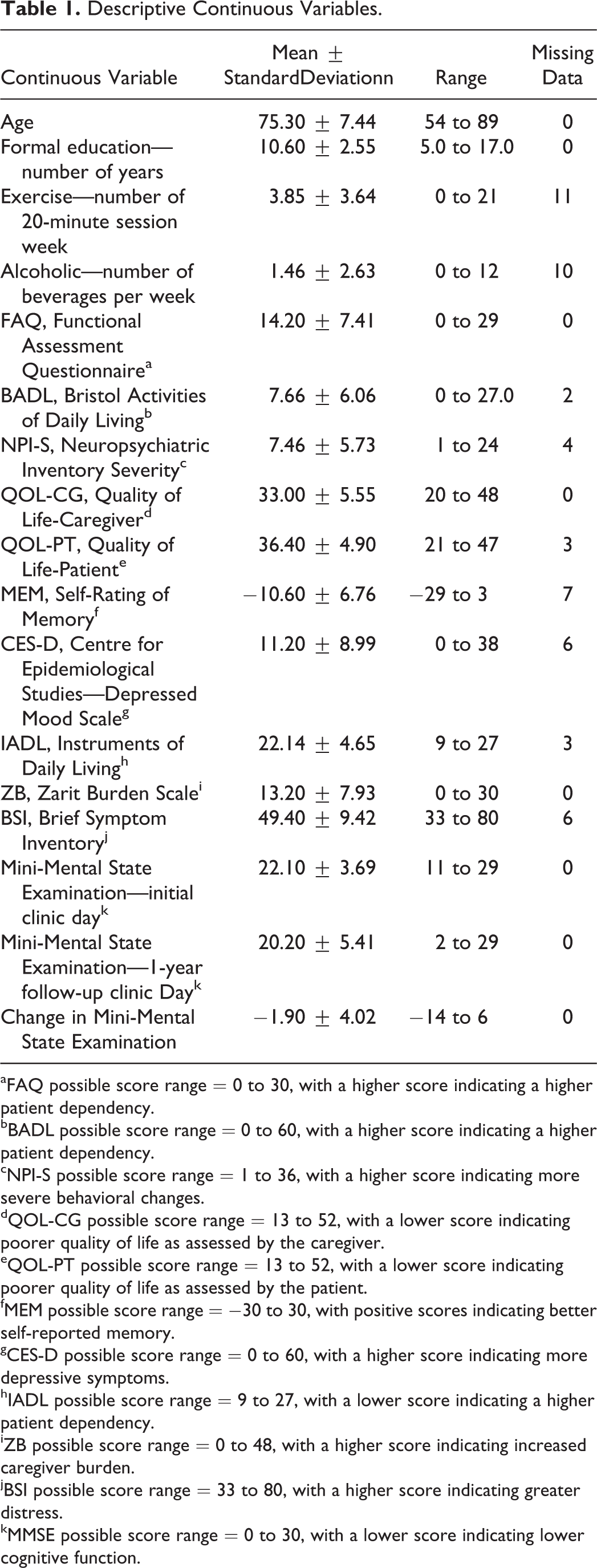
The MMSE score at 1-year follow-up visit subtracted from the MMSE score at the initial day of diagnosis (clinic day) represented a cognitive change and was the dependent variable. We have previously demonstrated that telehealth MMSE does not significantly differ from in-person MMSE. 12 Sociodemographic and clinical information, as well as functional and lifestyle status, were the independent variables in this study. These were assessed on the initial clinic day by administering patient and caregiver questionnaires. Sociodemographic variables included age, gender, years of formal education, ethnicity, and marital status. Formal education was the summed total of elementary school, high school, college, technical, or university school years completed. Ethnicity was organized into 2 groups: (1) European and (2) other. Marital status was organized into 2 groups: (1) married or common law and (2) single, divorced, separated, or widowed. Clinical variables included the family history of dementia, smoking status, number of alcoholic beverages consumed per week, exercise frequency, number of comorbidities, number of falls in the past years classified as (1) no falls, (2) 1 fall, and (3) 2 or more falls, and specific comorbidities including diabetes, heart disease, hypertension, and any psychiatric disorder. Self-rated questionnaires assessed functional and lifestyle status of the patient from his or her own perspective. These included quality of life of the patient (QOL-PT), 13 the Centre for Epidemiological Studies–Depressed Mood Scale (CES-D), 14 Instruments of Daily Living (IADL), 15 and Self-Rating of Memory (MEM). 16 Caregiver-rated questionnaires assessed the functional and lifestyle status of the patient from the caregiver’s perspective. These included Bristol Activities of Daily Living (BADL), 17 Neuropsychiatric Inventory Severity (NPI-S), 18 the Functional Assessment Questionnaire (FAQ), 19 Quality of Life of the patient (QOL-CG), 13 the Zarit Burden Scale (ZB1), 20 and the Brief Symptom Inventory. 21 A complete summary of sociodemographic and self-reported descriptive variables can be found in Tables 1 and 2.
Descriptive Continuous Variables.
aFAQ possible score range = 0 to 30, with a higher score indicating a higher patient dependency.
bBADL possible score range = 0 to 60, with a higher score indicating a higher patient dependency.
cNPI-S possible score range = 1 to 36, with a higher score indicating more severe behavioral changes.
dQOL-CG possible score range = 13 to 52, with a lower score indicating poorer quality of life as assessed by the caregiver.
eQOL-PT possible score range = 13 to 52, with a lower score indicating poorer quality of life as assessed by the patient.
fMEM possible score range = −30 to 30, with positive scores indicating better self-reported memory.
gCES-D possible score range = 0 to 60, with a higher score indicating more depressive symptoms.
hIADL possible score range = 9 to 27, with a lower score indicating a higher patient dependency.
iZB possible score range = 0 to 48, with a higher score indicating increased caregiver burden.
jBSI possible score range = 33 to 80, with a higher score indicating greater distress.
kMMSE possible score range = 0 to 30, with a lower score indicating lower cognitive function.
Descriptive Categorical Variables.

Statistics
Data were analyzed using IBM SPSS version 20.0 software. Descriptive analyses were performed for all variables in order to characterize the sample. Categorical variables were described in terms of frequencies and percentages. Continuous variables were described in terms of means and standard deviations. A simple linear regression analysis was carried out in order to examine the association between each potential independent variable and the dependent variable of change in MMSE over 1 year. Based on regression analysis, independent variables associated with the dependent variable with a P < .25 became candidates for a multiple linear regression model. 22,23 All variables that were significantly correlated with our observed change in MMSE over 1 year (P < .05), as well as important individual factors (age and gender), were retained in the final multivariable model. If 2 or more variables were highly correlated (variance inflation factor greater than 10), 24 the more relevant measure referenced in the literature was included in the regression. The coefficient of multiple determination, R 2, explains the proportion of variance explained by regression with the set of independent variables used and was reported for the final multiple regression model. Several subsets of potential independent variables were fitted to identify the best fitted model using the R 2 criterion. The model with subsets of independent variables for which the R 2 value was the highest was selected as the best suitable model for these data. We tested for potentially effective modifiers, but none of the interaction terms were statistically significant. The key assumptions of linearity, independence of errors, equality of variances (homoscedasticity), and normality were verified for the final multiple regression model. 24
Results
Study Population
Our sample was comprised of 72 patients with AD. The mean patient age at the initial clinic day appointment was 75.3 (SD = 7.44, range = 54-89). Females made up 66.7% of patients, 66.7% of patients were married or common law, and 79.2% were of European ancestry. The mean MMSE score was 22.1 (SD = 3.69, range = 11-29) on initial clinic day and 20.2 (SD = 5.41, range = 2-29) at the 1-year follow-up appointment. The mean difference in the MMSE was −1.90 (SD = 4.02, range =−14-6). The mean time period between the clinic day and the 1-year follow-up appointment was 396 days (SD = 44.8; Table 1).
Simple Linear Regression and Multiple Linear Regression Analyses
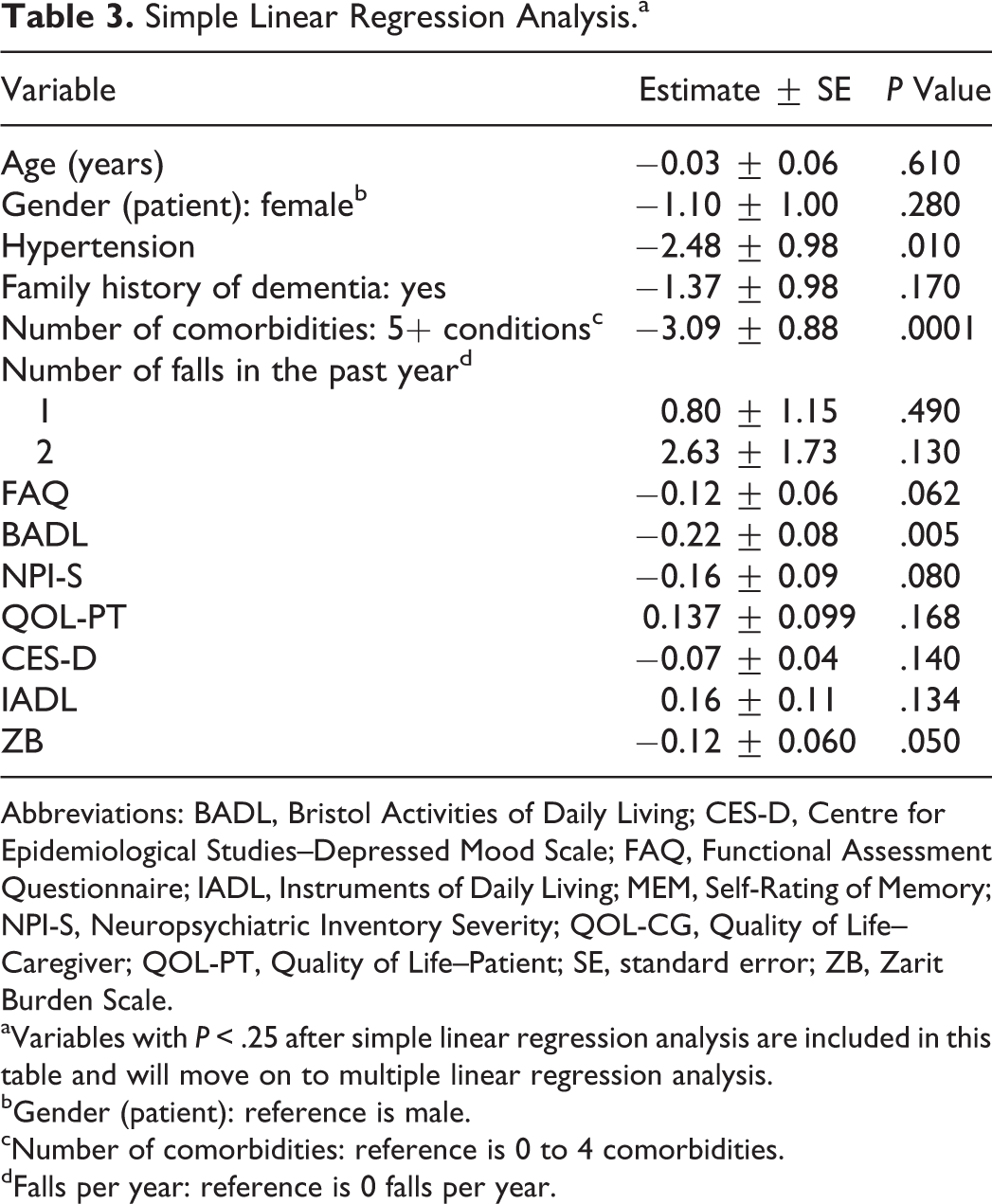
After a series of simple linear regressions, family history, hypertension, number of comorbidities (5+), number of falls, FAQ, BADL, NPI-S, QOL-PT, CES-D, IADL, and ZB scores were selected (P < .25) and included in the multiple regression analysis. These independent variables are included in Table 3. Independent variables that were not selected are not included in Table 3. In compliance with the Canadian Institutes of Health Research guidelines, gender and age were integrated into the research design by including these variables in our regression models. By including gender and age in the framework, we made certain any effects of these variables would be captured in the model.
Simple Linear Regression Analysis.a
Abbreviations: BADL, Bristol Activities of Daily Living; CES-D, Centre for Epidemiological Studies–Depressed Mood Scale; FAQ, Functional Assessment Questionnaire; IADL, Instruments of Daily Living; MEM, Self-Rating of Memory; NPI-S, Neuropsychiatric Inventory Severity; QOL-CG, Quality of Life–Caregiver; QOL-PT, Quality of Life–Patient; SE, standard error; ZB, Zarit Burden Scale.
aVariables with P < .25 after simple linear regression analysis are included in this table and will move on to multiple linear regression analysis.
bGender (patient): reference is male.
cNumber of comorbidities: reference is 0 to 4 comorbidities.
dFalls per year: reference is 0 falls per year.
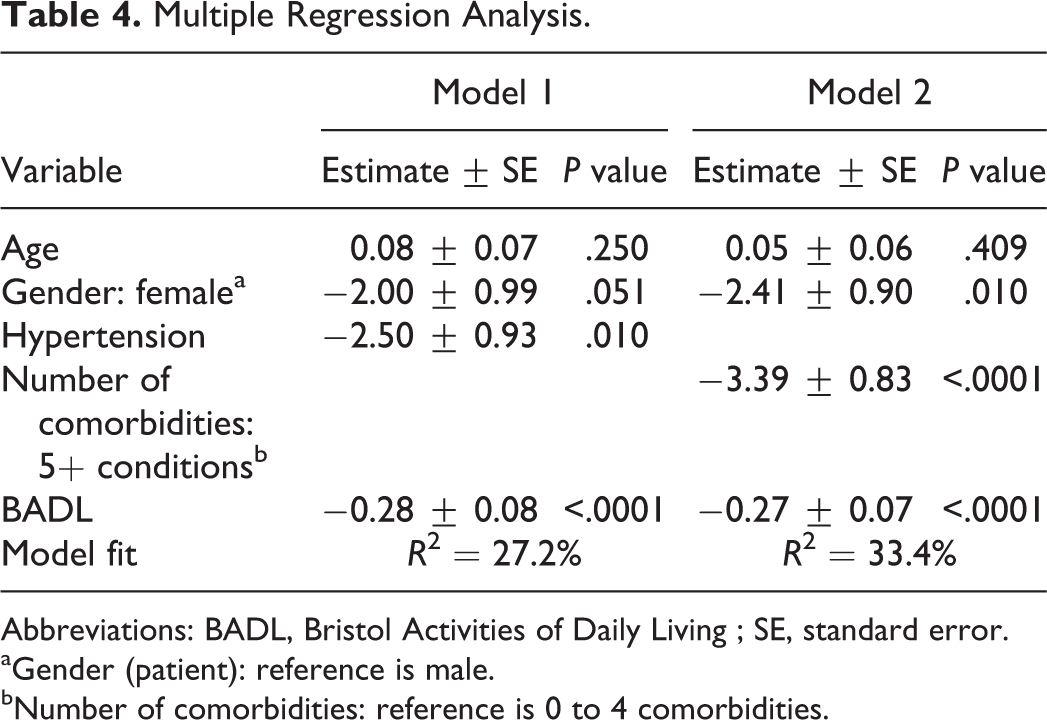
The history of hypertension and the BADL score remained significant (P < .05) in the final multivariate model (Table 4). The number of comorbidities (5+) was also significant but was highly correlated with the variable hypertension (multicollinearity exists); consequently, the number of comorbidities (5+) variable was removed from the multivariate model and the hypertension variable remained. We were unable to include the number of comorbidities (5+) in the same model with hypertension, as hypertension was included in the creation of the number of comorbidities variable. For reference, we have provided an alternate model of multivariate analysis that includes the number of comorbidities (5+) variable and removes the hypertension variable (Table 4).
Multiple Regression Analysis.
Abbreviations: BADL, Bristol Activities of Daily Living ; SE, standard error.
aGender (patient): reference is male.
bNumber of comorbidities: reference is 0 to 4 comorbidities.
Two variables emerged as significant predictors of worsening. Patients with a history of hypertension and patients with less independence in activities of daily living (high BADL scores) experienced greater cognitive decline at 1 year. The coefficient of determination or total variance explained by the regression (R 2 value) was 27.2%. The multiple linear regression assumptions were examined and verified. The normal P–P plot of regression standardized residual showed that expected and observed values yielded excellent linearity. With regard to the independence of errors, the Durban-Watson test statistic yielded a value very close to 2, which showed that there was no serial correlation of errors. As for homoscedasticity, the plot of standardized residuals with standardized predicted values showed constant variance of the errors. Finally, a histogram of the standardized residual with a bell curve confirmed that the assumption of normality was met, and test of normality Shapiro-Wilk test showed that errors are normally distributed (P = .098). 25
Discussion
The objective of our research was to identify the predictors of cognitive decline in rural patients diagnosed with AD. History of hypertension and less independence in daily functioning were identified. Noteworthy factors that were not significant predictors after multiple regression analysis included age, years of formal education, marital status, ethnicity, family history of dementia, number of alcoholic beverages per week, exercise frequency, diabetes, heart disease, baseline level of cognitive impairment, functional abilities, quality of life, MEM, depression, self-rating of independence in activities of daily living, and caregiver burden.
Cognitive decline over 1 year was greater in patients with a diagnosis of hypertension. Patients with a history of hypertension lost an average of 2.50 points more on the MMSE within a year compared to those without. Some past studies have also reported that hypertension predicts decline, 26 –28 whereas others have found that it does not. 3,4,29 Chaves and colleagues claimed that hypertension was a predictor of cognitive decline in AD and vascular dementia. 27,30 They hypothesized that cognitive decline in dementia is affected by hypertension through the same mechanisms that cause hypertension to increase the prevalence of dementia. The present study does not address the level of blood pressure control. It may be that controlling hypertension may lessen the rate of cognitive decline but we cannot directly address this question. In conclusion, the importance of identifying blood pressure measures as a predictor of decline in AD is substantial. Although there is disparity in results between studies, the relevance of hypertension affecting cognitive decline in dementia in our rural population is apparent. This outlines the need for increased awareness of the link between hypertension and dementia in predominantly rural clinics similar to our own.
Our finding that reduced capacity to complete activities of daily living is a predictor of greater cognitive decline is both expected and consistent with other results in the literature. For every 1 point increase in the BADL score (range = 0-60), there is a predicted average decrease of 0.281 points on the MMSE over 1 year. Impaired ability to carry out activities of daily living at the time of diagnosis suggests that cognitive decline will progress more quickly. Interestingly, the IADL measure, which was completed by the patient and which assesses somewhat similar functional activities of daily living as the BADL, was not predictive of the same decline. This finding might suggest that patients who are destined to decline quickly may not understand their difficulties as much as their caregivers do, suggesting problems with awareness. Awareness of functional deficits appears to require different insight than awareness of memory deficits or difficulties with balance. 31 The importance of patients’ awareness of function in the prediction of their decline illustrates the importance of the caregiver presence at the clinic day appointment. Due to the potential burden of travel on the initial clinic day, this is of particular relevance for clinics such as our own that focus on far reaching rural populations.
Females lost a mean of 2.00 more MMSE points within a year than their male counterparts, but gender was not a statistically significant predictor in our primary multivariate model. Previous studies report that gender has no significant predictive value of rate of decline in the MMSE. 3,6,8 However, a report by Roselli and colleagues indicates that male gender is a predictor of fast cognitive decline. 21 They suggested that males have an increased ability to buffer pathological changes, which functions to ward off AD symptoms. When they are finally diagnosed, they are within a more severe disease state.
An important result in this study was that many previously suggested predictors of cognitive decline were not found to be significant. For example, younger age and more years of formal education have been found to predict decline in patients with AD in other studies. Younger age is hypothesized to predict greater decline due to the fact that AD is related to aging, and therefore, a more severe form could be manifesting in those who acquire the disease at a young age. 26,32 The faster cognitive decline seen in patients with increased years of education is thought to be due to cognitive reserve. 33,34 This reserve may act as protection from the symptoms of AD in the early stages of the disease. Symptoms do not emerge until the disease has reached a severe level and is thus capable of causing increased decline following diagnosis. Our study is suggesting that this cognitive reserve principal is not supported within a rural and remote population of Saskatchewan. This may be due to the past differences in the educational systems within primarily agricultural backgrounds. The lack of predictive value within our rural cohort shows the importance of exploring rural populations for accurate predictors of decline in order to shed light on possible limitations of broad predictions within the literature.
Rasmusson and colleagues found that having first-degree relatives with a history of dementia predicted decline in patients with AD older than 65 years. 35,36 It was thought that patients with a family history of dementia would have a severe form and would be at risk to decline quickly. Our study found that the family history had no predictive value for the rate of decline, with other studies reporting similar results. 2,4 A possible explanation is that the experience of family and close knit rural community members who have witnessed this disease in the past could aid in recognition of the disease and influence patients to seek help sooner. Even though the patient may be having a more severe form of the disease, access to prior care could result in slower cognitive decline initially.
Limitations
There are limitations to this study that warrant discussion. First of all, the sample size for the AD group (n = 72) is somewhat small compared to similar studies that included samples of 107 to 156 participants. 3 –5,8 A study by Brodaty et al found that 6 months was too short to accurately predict rapid decline. 37 Because of this, we chose to assess a smaller sample size from a follow-up of 1 year, even though 6-month follow-up data were available and would have ensured a larger sample.
A second limitation that many of these studies face is the difficulty of controlling for the varying disease stages of participants when first assessed. Although this creates disparity among the subjects, we feel that our study reflects the real-world situation of many memory clinics. By including all potential disease stages at baseline, we are identifying predictors of progression that will be valuable to clinicians who are also unaware of the exact disease stage with which their patient will present.
A third limitation is the challenge to be consistent with other studies assessing predictors of cognitive decline. A study authored by Marra et al states that, “the methodical differences between these AD studies may account for much of the inconsistency in identifying correct predictors of progression.” 38(p216) The inconsistencies are seen within the time periods assessed, the inclusion criteria of the participants, and the definitions of rapid cognitive decline derived. The purpose of identifying increased cognitive decline is to be able to adjust treatment plans accordingly, as well as to allow appropriate discussions with patients and their family in a timely manner. Based on this, our study was designed to address some of the limitations in previous research: we have used a shorter time period in order to identify the most imperative predictors, an inclusive sampling frame that is representative of the typical clinic population, and have chosen not to determine a minimal rate of rapid cognitive decline in order to identify all of the patients within our population who require attention sooner than others. Means of identifying these patients in the future in a rural setting will help to better coordinate our health-care team.
A fourth limitation is that the variables for exercise frequency per week and alcohol consumption per week had missing data in greater than 10% of the population.
A fifth limitation is that we could not address the effect of all individual comorbidities (22 of them) on cognitive change since our sample size is not very large (n = 72). The variables hypertension and comorbidities (+5) were highly correlated as the creation of the number of comorbidities variable included the hypertension condition. We chose to include hypertension in our final model because it is statistically significant on its own and is a known risk factor of dementia. The following comorbidities were included in our data collection and were presented to the patient population for selection: memory problems/confusion, arthritis/rheumatism/bursitis, cancer, convalescence from surgery, diabetes, thyroid disease, epilepsy/convulsions/seizures, hardening of the arteries, hyperlipidemia, heart disease/heart attack, angina, heart failure, rheumatic heart disease, high blood pressure, liver trouble, kidney trouble, Parkinson’s disease, hearing loss, poor vision not relieved by corrective eyewear, chronic respiratory problems, stroke, head injury, stomach trouble/ulcer/gallbladder trouble, and psychiatric/psychological problems. Further research needs to be directed toward the effect of multiple comorbidities on cognitive change within rural populations.
Conclusion
The care of patients with AD may be better individualized by identifying predictors of cognitive decline. Our study outlines the uniqueness of our rural population when it comes to identifying predictors of cognitive decline. As a clinic primarily providing care to rural patients, these results help to refine our clinical knowledge and may allow us to improve care to our target population, as well as provide a benefit to clinics similar to our own.
Footnotes
Authors’ Note
Statistical analysis was conducted by Chandima Karunanayake and Drew Hager. This manuscript has not been published elsewhere nor is it under simultaneous consideration by another journal.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by College of Medicine, University of Saskatchewan, the Canadian Institutes of Health Research (CIHR) New Emerging Team Grant (CIA-63947; D Morgan) and the CIHR-SHRF Chair Grant (ACH-93185) and the Saskatchewan Health Services Research Foundation.
