Abstract
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder, characterized by irreversible decline of mental faculties, emotional and behavioral changes, loss of motor skills, and dysfunction of autonomic nervous system and disruption of circadian rhythms (CRs). We attempted to describe the morphological findings of the hypothalamus in early cases of AD, focusing our study mostly on the suprachiasmatic nucleus (SCN), the supraoptic nucleus (SON), and the paraventricular nucleus (PVN). Samples were processed for electron microscopy and silver impregnation techniques. The hypothalamic nuclei demonstrated a substantial decrease in the neuronal population, which was particularly prominent in the SCN. Marked abbreviation of dendritic arborization, in association with spinal pathology, was also seen. The SON and PVN demonstrated a substantial number of dystrophic axons and abnormal spines. Alzheimer’s pathology, such as deposits of amyloid-β peptide and neurofibrillary degeneration, was minimal. Electron microscopy revealed mitochondrial alterations in the cell body and the dendritic branches. The morphological alterations of the hypothalamic nuclei in early cases of AD may be related to the gradual alteration of CRs and the instability of autonomic regulation.
Introduction
Alzheimer’s disease (AD) is a progressive devastating neurodegenerative disorder of the central nervous system, which has been recognized as the main cause of serious irreversible cognitive decline in elderly people. 1,2 Alzheimer’s disease has a huge social impact, starting frequently as mild cognitive impairment and resulting in dementia, as the time advances. 3,4
The pathogenesis of AD involves a substantial number of cellular and biochemical mechanisms as well as many genetic or acquired risk factors, which may contribute in the rapid decline of cognition, facilitating the clinical onset of the disease. 5
Clinical observations plead in favor of brain injury, 5 stress 6,7 or stress-related psychiatric disorders, 8,9 among the others, as potential risk factors for AD. 10,11
From the neuropathological point of view, AD is mostly characterized by the presence of amyloid-containing neuritic plaques, neurofibrillary tangles, consisting of highly phosphorylated τ proteins, dendritic and spinal pathology, synaptic alterations, and selective neuronal loss, 12 –14 which occur mostly in the limbic and neocortical areas. Chronic accumulation of amyloid-β (Aβ) peptide in brain induces neuroinflammation mediated by activated microglial cells, which offer an additional factor in the pathogenic cascade of AD. 15 It is generally thought, on the other hand, that deposits of Aβ peptide may play an important role in promoting degeneration of neurons, which are particularly vulnerable to age-related procedures, such as oxidative stress and impairments in cellular energy metabolism. 16 The blood–brain barrier disruption and the microvascular lesions also contribute substantially in plotting the neuropathological profile of AD. 17,18
The initial clinical manifestations of AD are subtle. However, as the time advances, progressive memory and learning impairment, 19 language disturbances, visuospatial disorientation, ideomotor apraxia, behavioral disturbances, depressive symptoms, 20 –22 personality changes, 23 –25 and autonomic dysfunction progressively establish as principal dominant deficits in AD. 26
Although the etiopathogenic mechanism of the sporadic cases of AD remains unknown, in spite of the constantly ongoing research, various hypotheses have been proposed, based mainly on clinical investigation and experimental data. In addition, the multiple genetic loci associated with familial AD plead in favor of the heterogeneity of the disease and support the hypothesis that the neuropathological findings in AD share a final common pathway with many other degenerative and metabolic disorders of the brain. 27,28
Oxidative stress associated with the accumulation of Aβ peptide in the cortex and the subcortical centers seems to be an important factor in the pathogenesis of AD. 29,30 In addition, morphological alterations of mitochondria in neurons and glial cells have been described in various areas of the brain including the cerebellum, 31,32 associated closely with spinal loss and dendritic pathology. In addition, morphological alterations of the Golgi apparatus have been described in early cases of AD, even in areas where the typical Alzheimer’s pathology was minimal, a fact suggesting that the mechanism of sorting and trafficking of proteins might be defective even at the initial stages of AD, given that Golgi complex is of substantial importance for the plasma membrane proteins to reach their final destination. 33,34
Autonomic dysfunction has been frequently reported in AD either as autonomic hyperactivity or as autonomic failure. It is also important that in a considerable number of cases, the autonomic responses to strong emotional or cognitive stimuli might be impaired, even in the early stages of the disease. A substantial body of evidence suggests that hypothalamus is involved in the neurodegenerative diseases including AD, 35 although its various nuclei are not involved simultaneously and in the same severity. 36 –38
In addition, stress which might be a potential risk factor is mediated by the activation of the hypothalamic–pituitary–adrenal pathway, resulting in a substantial increase in the levels of glucocorticoids. 39,40
The involvement of the hypothalamus in AD is suggested by increased volume of the third ventricle. A substantial body of evidence suggests that there are substantial molecular and cellular differences in the pathologic features of AD in the hypothalamus, 39,41 compared with those observed in hippocampal and cortical structures. 42 Unlike plaques in the cortex and hippocampus of patients with AD, hypothalamic plaques do not contain epitopes corresponding to other regions of the β-amyloid precursor proteins (β-APPs), nor do they contain τ-, neurofilament-, or microtubule-associated protein-reactive epitopes, and do not disrupt the neuropil or produce gliosis. 43 Large numbers of diffuse plaques in the AD hypothalamus labeled with an antiserum to the Aβ peptide of the β-APPs have been described, whereas Aβ peptide-immunoreactive plaques were uncommon in the hypothalamus of patients without AD. 43
In the present study, we attempted to describe the morphological alterations of the neuronal networks in the hypothalamus in early cases of AD, focusing our study mostly on the suprachiasmatic nucleus (SCN), the supraoptic nucleus (SON), and the paraventricular nucleus (PVN).
We attempted to describe the dendritic and spinal pathology in silver impregnation techniques and the morphological and morphometric alteration of the mitochondria and Golgi apparatus in association with synaptic pathology at electron microscopy.
Material and Methods
Material
This morphological study is based on examination of 12 brains of patients having AD obtained at autopsy 4 to 8 hours after death at room temperature of 4°C. All brains were derived from patients with a definite history of dementia, aged 55 to 82 years, who fulfilled the clinical, neurological, neuropsychological, and neuropsychiatric criteria of AD. All of them died 24 to 46 months following the clinical diagnosis of the disease (Table 1).
List of the AD Brains and Normal Controls Included in the Study.a
Abbreviations: AD, Alzheimer’s disease; F, female; M, male.
aThe hypothalamic nuclei were excised and studied from 1974 to 2011.
bFixation for silver impregnation techniques.
Additional 12 brains, macroscopically intact of apparently healthy individuals, who died accidentally, were used as normal controls. The definite diagnosis of AD was based on National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer's Disease and Related Disorders Association (NINCDS-ADRDA) criteria. 44
Methods
Samples from the hypothalamus were excised and processed for electron microscopy and silver impregnation techniques including rapid Golgi method, Rio Hortega and Bodian techniques.
Electron Microscopy
For electron microscopy, the specimens were immediately immersed in Sotelo fixing solution, composed of 1% paraformaldehyde, 2.5% glutaraldehyde in cacodylate buffer 0.1 mol/L, adjusted at pH 7.35. Then they were postfixed by immersion in 1% osmium tetroxide for 30 minutes at room temperature and dehydrated in graded alcohol solutions and propylene oxide. Thin sections were cut in a Reichert ultratome, contrasted with uranyl acetate and lead citrate, and studied in a Zeiss 9aS electron microscope.
Light Microscope
Silver impregnation techniques
The remaining parts of the hypothalamus were processed for silver impregnation techniques, according to rapid Golgi staining. Thus, after a 4-week fixation in formalin they were immersed in potassium dichromate (7 g potassium dichromate in 300 mL water for 10 days. Then they were immersed in 1% silver nitrate for 10 days in a dark glass jar at a temperature of 16°C. Following a rapid dehydration in graded alcohol solutions, the specimens were embedded in paraffin and cut, some of them at 100 μm and some at 25 μm, alternatively. Many sections of 25 μm were stained also with methylene blue, according to Golgi-Nissl method. 45,46 All the sections were mounted in Entellan (Merck-Millipore, Darmstadt, Germany), between 2 cover slips and studied in a Zeiss Axiolab Photomicroscope equipped with a digital camera and a computer.
We focused our study mostly on the SCN, the SON, and the PVN of the hypothalamus. The Cavalieri principle 47,48 was used in order to calculate the volume of the hypothalamic nuclei.
We estimated the dendritic arborization, the morphology and the number of the dendritic branches, and the morphology of the dendritic spines in light microscope on sections stained according to rapid Golgi method and Golgi-Nissl staining.
Morphometry
Morphometric studies were performed with an image analyzer using the Neuro J plug-in in Image J application (Java). The mean surface area of the neurons as well as the dendritic arborization were calculated in the above-mentioned nuclei of the hypothalamus in silver staining at light microscopy. 49
The neurons were estimated from the morphological and morphometric point of view, with much emphasis on the dendritic morphology, accepting the criteria posed by Jacobs et al, 50 which concern (1) the quality of staining of dendrites and (2) the sufficient contrast between neurons and background.
Dendritic arbores were quantitatively estimated in a centrifugal way for the apical dendrite and the basal dendrites according to Uylings et al. 51 We estimated the diameter of the neuronal soma, the total dendritic length, the number of dendritic bifurcations, the length and number of dendritic segments per dendritic order, as well as the spinal density per dendritic branch. Thus the dendrites, which arise from the soma of neurons up to their first symmetrical bifurcation, are considered as first-order branches. The dendritic branches which arise from the first-order branches up to their symmetrical bifurcation are considered as second-order segments or secondary dendrites, and so on.
For the quantitative analysis, we applied Image J program after a calibration for the specific types of microscope (Carl Zeiss Axiolab Photomicroscope) and we estimated the dendritic arborization according to Sholl method of concentric circles. 52 Thus concentric circles were drawn, at intervals of 15 μm centered on the soma of the neuron, and the dendritic intersections within each circle were counted. The dendritic spines were counted on 3 segments of the dendritic field. The first segment, 20 to 30 μm in length, was located on primary dendrite, the second segment, 20 to 30 μm in length, on the secondary dendrite and the third one, 40 to 50 μm, on the tertiary dendrite.
In electron microscopy, we applied the stereological analysis according to Nyengaard and Gundersen 53 and West. 54,55 The quantities comprise number, length, surface area, volume, and spatial distribution for mitochondria 56 and cisternae and vesicles of the Golgi complex. 57
We estimated also the mean nuclear area, the dendritic profiles,
58
the total number of the dendritic spines per dendritic branch, the pre- and postsynaptic terminals,
59
–61
and the number of synaptic vesicles per presynaptic component.
61
The statistical analysis of the data was evaluated by Student
Results
Silver Impregnation Technique
The limits of the hypothalamus in the human brain are situated anteriorly to the level of lamina terminalis and posteriorly to a level through the posterior commissure and the posterior edge of the mammillary body.
By application of the silver impregnation technique in association with Golgi Nissl method, we could visualize the hypothalamic nuclei, focusing our study particularly on the SCN, the SON, and the PVN. The morphological and morphometric estimation of the neurons, which have been impregnated by silver nitrate, revealed a substantial decrease in the neuronal population and loss of dendritic branches in the patients who had AD (Figure 1, 1A, 1B, and 1C), as compared to normal controls (Figure 1, 2A, 2B, and 2C). Abbreviation of the dendritic arborization was prominent mostly in the neurons of SCN which was associated with marked decrease in the number of dendritic spines (Figure 1, 1A and 1B) in comparison with the normal control brains (Figure 1, 2A and 2B). The same morphological alterations concerning the dendritic branches and the spines were also observed in the SON and PVN of the hypothalamus in AD (Figure 1, 1C) in comparison with normal controls (Figure 1, 2C).
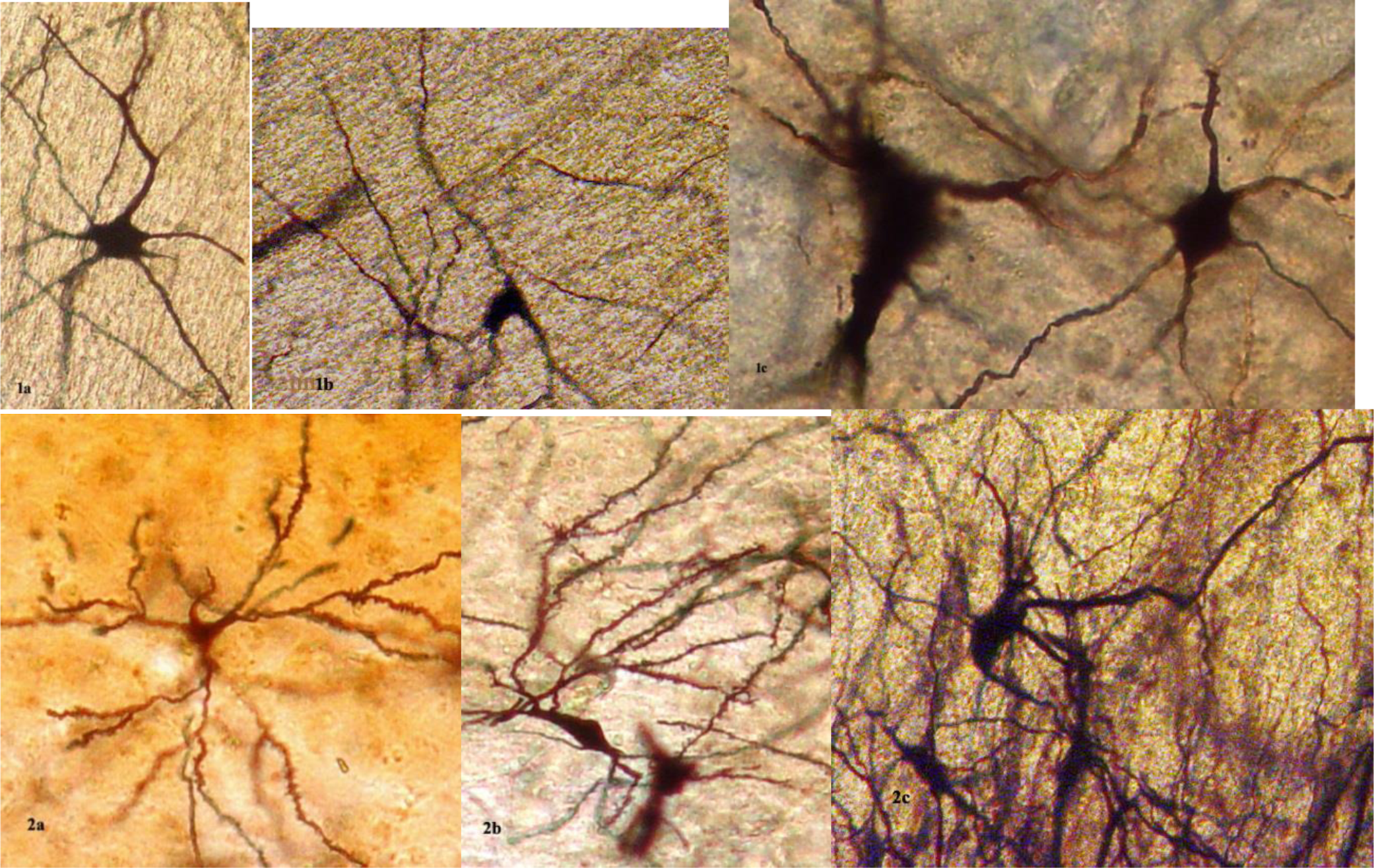
Neurons of the SCN (1A, F 75 years), supraoptic nucleus (SON; 1B, M 63 years), and PVN (1C, F 66 years) of the hypothalamus of Alzheimer’s disease (AD) brains. The loss of dendritic branches and spines is obvious in comparison with SCN (2A M 76 years, 2B F 68 years) and PVN (2C M 76 years) of normal control brains. Golgi silver impregnation technique (magn ×1200: 1A, 1B, 2A, 2B, 2C; ×2400: 1C). F indicates female; M, male; magn, magnification; PVN, paraventricular nucleus; SCN, suprachiasmatic nucleus.
The morphometric estimation of the dendritic spines of the neurons of the SCN and SON revealed a dramatic decrease of their number in AD brains in comparison with normal controls (Table 2).
Average Dendritic Spines per Dendritic Arbor in SCN and SO Neurons Based on Measurements of 100 Neurons (
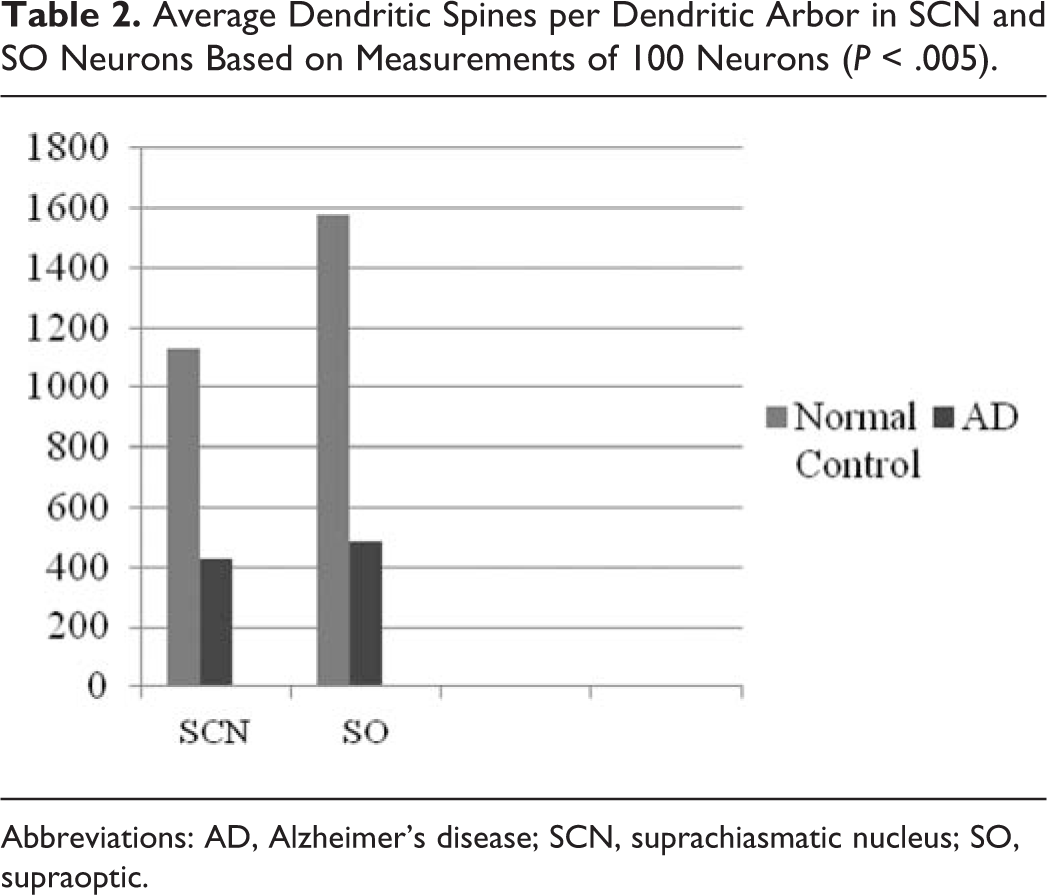
Abbreviations: AD, Alzheimer’s disease; SCN, suprachiasmatic nucleus; SO, supraoptic.
Electron Microscopy
Electron microscopy revealed morphological alterations of the dendritic profiles concerning mostly the secondary dendritic branches in a substantial number of neurons of the SCN, the SON, and the PVN of the hypothalamus in Alzheimer’s brains, as compared to normal controls. Considerable decrease in spine density was observed in the secondary and tertiary dendritic branches of neurons in hypothalamic nuclei, which were particularly marked in the SCN. Reduction in the size of the spines or giant spines were also observed in a considerable number of neurons of the SCN (Figure 2, 3A and 3B). In a substantial number of dendritic spines, large multivesicular bodies were seen.
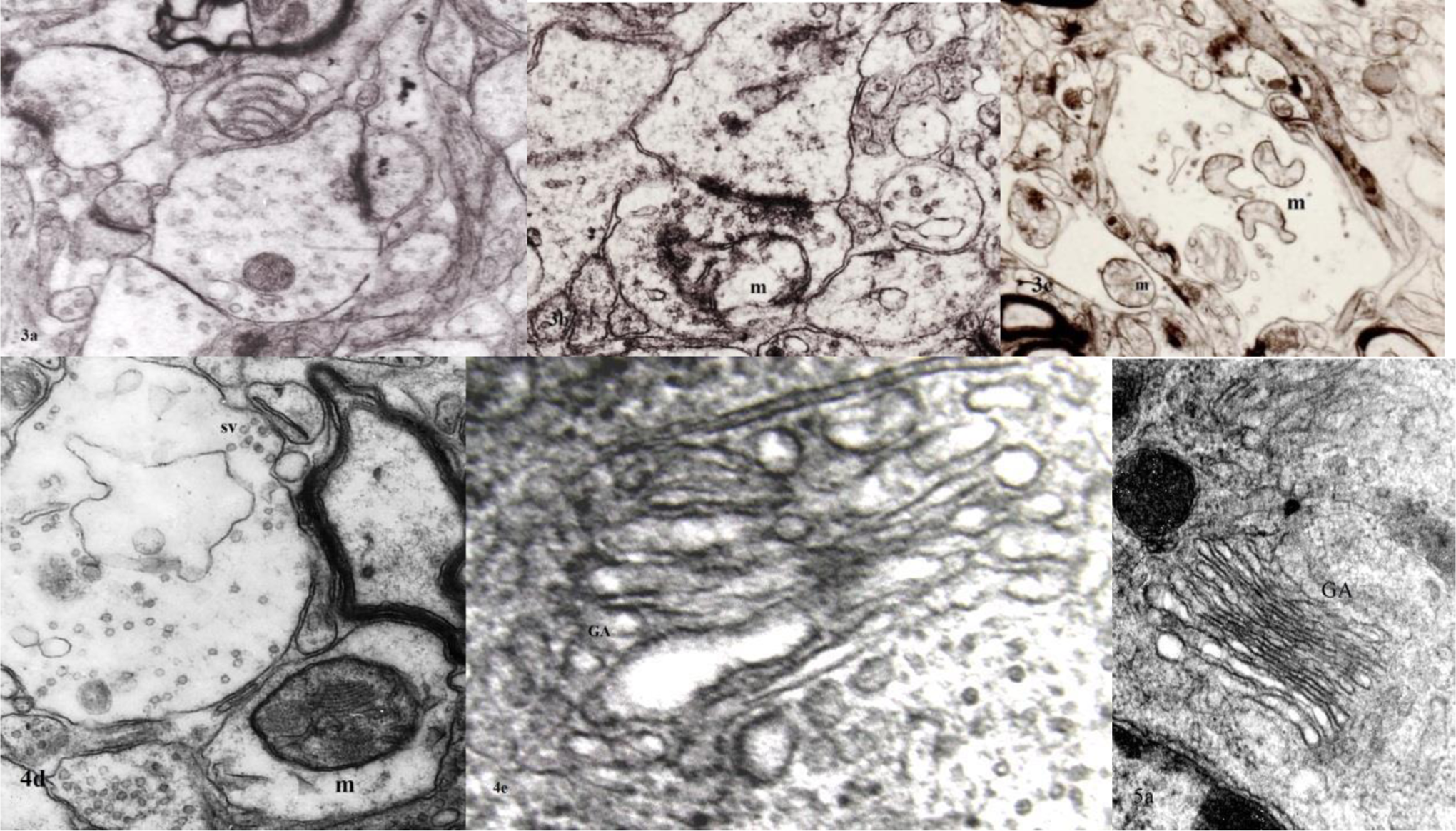
Reduction in the size of the dendritic spines (3A) in synaptic profiles and giant spine (3B) in suprachiasmatic nucleus (SCN) in AD (M 78 years). Marked alterations of mitochondria are also observed in paraventricular nucleus (PVN; 3C) and supraoptic nucleus (SON; 4D) in AD (M 72 years). The alteration of Golgi apparatus (4C, M 72 years) is obvious in comparison with normal control (M 75 years) (5A). Electron micrographs magn ×128 000: 3A, 3B, 4D; ×110 000: 3C; ×136 000: 4C; ×67 000: 5A). AD indicates Alzheimer’s disease; F, female; M, male; magn, magnification.
In the SCN and the PVN, in a considerable number of dendritic profiles, the mitochondria were characterized by marked morphological alterations, such as disruption of the cristae and accumulation of fibrillary material (Figure 2, 3C and 4D). Many of them were very small or giant.
In a morphometric estimation of the mitochondria in dendrites, dendritic spines, and cell body of neurons of the SCN in normal control brains, we noticed that the ellipsoid mitochondria of the dendritic spines appear to have an average diameter of 650 ± 250 nm and a mean axial ratio of 1.9 ± 0.2. In addition, the round mitochondria appeared to have a mean radius of 350 nm. In AD brains, mitochondria in the neurons of the SCN appear to have an average diameter of 440 ± 250 nm and a mean axial ratio of 1.7 ± 0.2. The round mitochondria appear to have a mean radius of 235 nm (Table 3). The alteration of the cristae was also considerable in the mitochondria of hypothalamic neurons in AD in comparison with normal controls (Table 4).
The Mean Diameter of Mitochondria in AD and NC Brains in SCN Neurons, Based on Estimation of 500 Mitochondria (
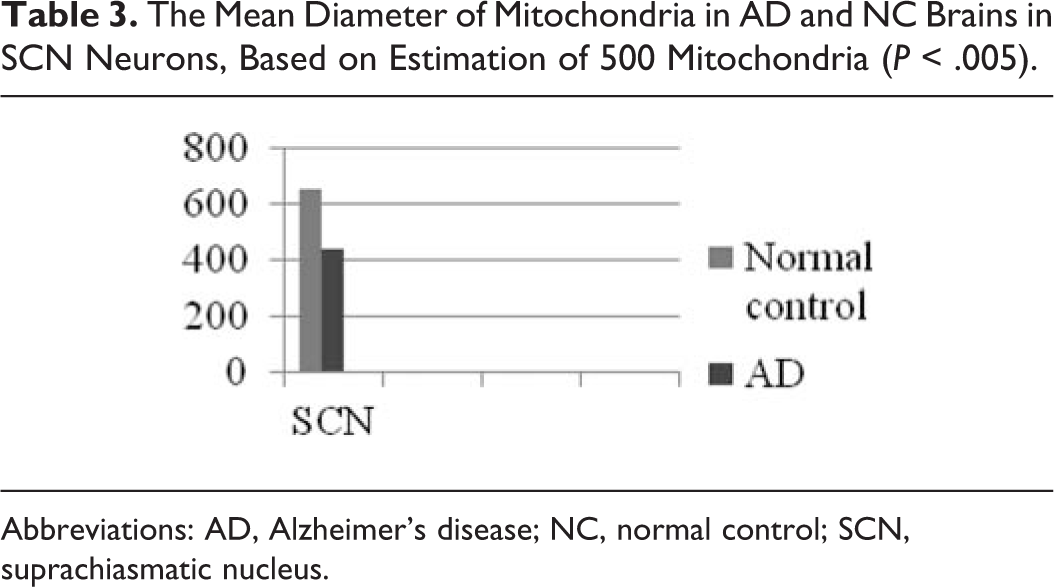
Abbreviations: AD, Alzheimer’s disease; NC, normal control; SCN, suprachiasmatic nucleus.
Average Number of Cristae per Mitochondrion Based on Measurements of 500 Mitochondria of SCN (
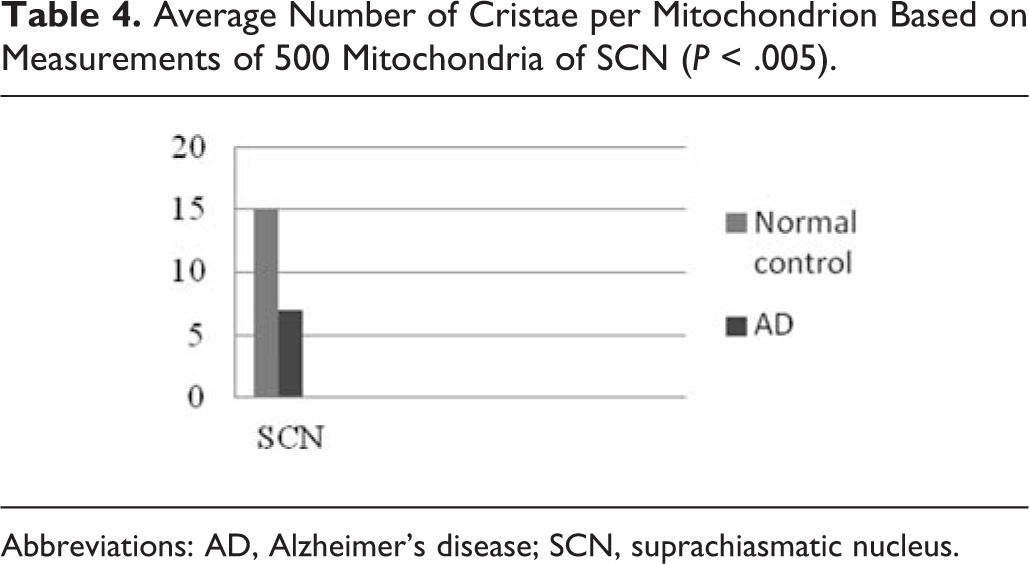
Abbreviations: AD, Alzheimer’s disease; SCN, suprachiasmatic nucleus.
Morphological alterations of the mitochondria were also seen in a considerable number of astrocytes in AD brains, in comparison with normal controls.
In a substantial number of neurons of the SCN and PVN of the hypothalamus, the Golgi apparatus appeared to be fragmented and atrophic (Figure 2, 4E) in comparison with normal controls (Figure 2, 5A). It was noticed that the atrophy or the fragmentation of Golgi apparatus (Table 5) and the mitochondrial alterations coexisted with dendritic and spinal pathology in the majority of neurons.
The Volume of Golgi Apparatus in nm3, Based on Measurements of 100 Neurons of SCN (
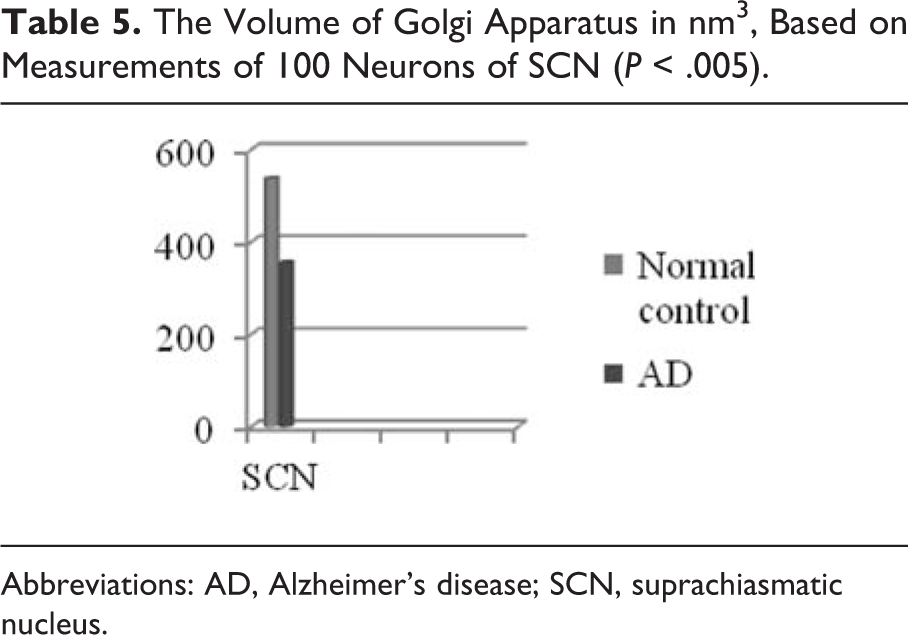
Abbreviations: AD, Alzheimer’s disease; SCN, suprachiasmatic nucleus.
Discussion
Several hypothalamic nuclei are affected in AD and other neurodegenerative disorders. 62 It seems that the hypothalamic nuclei are not involved simultaneously at the early stages of AD. The SCN seems to be more seriously affected than the others in aging, 63 given that in previous studies, it was clearly revealed that the total cell population in the SCN is decreased in aging and in a dramatic way in AD. 64
However, the correlation of the morphological and morphometric alterations of the dendrites in the hypothalamic nuclei with those of the neocortex and the cerebellum reveals clearly that the alterations in the hypothalamus are rather modest in comparison with those of the acoustic, visual, prefrontal areas of the brain hemispheres, and the cerebellar cortex. 65 –68 Since hypothalamus is the principal subcortical center of the homeostatic and autonomic-related processes, it is reasonable that some nuclei such as the SON and the PVN ones reserve substantial synaptic density in comparison with the neocortex, even in the advanced stages of AD.
The fact that the SCN demonstrated more severe dendritic alterations and synaptic loss than the SON and PVN might explain the phenomenon of desynchronization of circadian rhythms (CRs) in the majority of the patients who have AD 69 and cognitive decline, 70 given that the SCN is of crucial importance for the generation and the synchronization of CRs in man. 71,72 It is reported that changes in the CR of arterial blood pressure and circadian temperature may occur in patients with AD 73 especially during the nighttime. 74 –76 Also changes in the melatonin levels is not an unusual finding in advanced senility and AD. 77 –79 Sundown syndrome, on the other hand, frequently associated with increased motor activity, has been a rather common phenomenon in advanced cases of AD. 80
In the majority of neurons of hypothalamic nuclei, mitochondrial alterations were prominent in the cell body and the dendritic profiles. It is well known that mitochondria play a fundamental role in the energy supply of the cell as adenosine triphosphate-generating organelles and their role is crucial in the alteration of reduction-oxidation potential of cells, in the free radical scavenging, in the intracellular calcium regulation, and in the activation of apoptotic process. 81,82 Normally, the number of mitochondria is very high in dendrites and especially in pre- and postsynaptic terminals, given that they are the major energy generators for the synaptic activity. Mitochondrial dysfunction might be a substantial factor for Aβ peptide neurotoxicity and at the same time is also associated with oxidative stress, which may play an important role in activating the pathogenic cascade of AD. 83 –86 It is reasonable that impaired mitochondrial biogenesis contributes to mitochondrial dysfunction in AD. 87
Mitochondrial motility and accumulation are related to the functional state of the neuron, because mitochondria are transported to regions where the necessity for energy is particularly high, as it occurs in the dendritic and axonal profiles and the synaptic components. 88 The shape and size of mitochondria are not stable because they undergo continual fission and fusion, leading to their fragmentation or elongation accordingly. Recent studies reported increased mitochondrial fission and decreased fusion, due to increased Aβ peptide interaction with the mitochondrial fission protein Drp 1, inducing increased mitochondrial fragmentation, impaired axonal transport of mitochondria, and synaptic degeneration in AD. 85 Nevertheless, a prominent decrease in the size of the mitochondria is observed in aging-related neurodegenerative diseases 82,83 and particularly in the early stages of AD presumably prior to the onset of the obvious cognitive dysfunction. 89 Normally, a limited number of dendritic spines contain mitochondria, which are mostly small and round, which are increased in number inside the dendritic branches during the synaptogenesis and hormonal instability. 90 It is important to emphasize that mitochondrial alterations are associated with synaptic loss in patients with AD, due to blockage of mitochondrial energy production 91 even before neuritic plaques and τ pathology are detected. 92,93
The morphological alteration of the mitochondria, which is observed in hypothalamic nuclei, pleads in favor of a generalized mitochondrial dysfunction in AD, which may be associated with aged neurons, neuronal loss, and synaptic alterations.
Dendritic spine density was decreased in the dendritic arborization in a considerable number of neurons in the SCN of the hypothalamus. The loss of dendritic spines correlates with impairment in neuronal communication and is an important reason for the dysfunction of the neuronal circuits in AD. In previous studies, it was realized that the loss of dendritic spines coincides with the morphological alterations of the mitochondria and the Golgi apparatus. 89,91 –93 In experimental level of a mouse model of Aβ peptide deposition, it was revealed that nonfibrillar Aβ peptide may exert spine toxicity and contribute among other factors in the dramatic decrease of spine density. 94
The role of the hypothalamus in the regulation of CRs, its critical role in the maintenance of energy homeostasis, 95 in feeding behavior, 96,97 and in thermoregulation of the body starts becoming unstable during the clinical course of AD 98,99 as well as in experimental models of AD. 100,101 In other types of dementia, such as in behavioral variant of frontotemporal dementia, degeneration of the hypothalamic nuclei relates to significant feeding disturbances. 102
In conclusion, neuropathological alterations of the hypothalamus may play an important role in the autonomic dysfunction and homeostatic changes, phenomena which frequently occur in the advanced stages of AD.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
