Abstract
The objective of this review is to summarize the available data on the use of melatonin and melatonin agonist for the prevention and management of delirium in the elderly patients from randomized controlled trials (RCTs). A systematic search of 5 major databases PubMed, MEDLINE, PsychINFO, Embase, and Cochrane Library was conducted. This search yielded a total of 2 RCTs for melatonin. One study compared melatonin to midazolam, clonidine, and control groups for the prevention and management of delirium in individuals who were pre- and posthip post-hip arthroplasty. The other study compared melatonin to placebo for the prevention of delirium in older adults admitted to an inpatient internal medicine service. Data from these 2 studies indicate that melatonin may have some benefit in the prevention and management of delirium in older adults. However, there is no evidence that melatonin reduces the severity of delirium or has any effect on behaviors or functions in these individuals. Melatonin was well tolerated in these 2 studies. The search for a melatonin agonist for delirium in the elderly patients yielded 1 study of ramelteon. In this study, ramelteon was found to be beneficial in preventing delirium in medically ill individuals when compared to placebo. Ramelteon was well tolerated in this study.
Introduction
Delirium is a common neuropsychiatric syndrome seen in the elderly patients. 1 The rates of delirium vary depending upon the population that is being studied. 2 The prevalence of delirium among the elderly patients in the community is between 0.4% and 2%, whereas its incidence increases to 11% to 42% during general hospital admissions and 15% to 62% postoperatively. 2 –4 Current estimates indicate that about one-fifth of the elderly patients who are hospitalized each year in the United States experience complications because of delirium during the hospitalization. 2
Available evidence indicates that delirium contributes to poor patient outcomes irrespective of baseline patient characteristics and etiological factors. 2 Some individuals with delirium never recover to their baseline level of cognitive function following an episode of delirium. 2 The presence of delirium increases nursing time per patient, higher per-day hospital costs, and an increased length of hospital stay. 2,5 Delirium increases the risk of dementia, institutionalization, and death independent of the baseline patient characteristics or comorbidities. 6 The average cost per day in the hospital for patients with delirium is more than 2.5 times the cost among patients without delirium. 7 The cost of delirium is approximately US$6.9 billion for Medicare in-hospital expenditure, and the total the national burden of delirium on the health care system ranges from US$38 billion to US$152 billion per year. 7,8
It is estimated that about one-third of the cases of delirium are preventable and that prevention remains the most effective strategy for minimizing the occurrence of delirium and its adverse outcomes. 2 A multicomponent targeted risk factor intervention strategy that used standardized protocols for the management of 6 risk factors for delirium, cognitive impairment, sleep deprivation, immobility, visual impairment, hearing impairment, and dehydration, demonstrated that the incidence, the total number of days with delirium, and the total number of episodes of delirium could be significantly reduced by this strategy. 9 This intervention also reduced the short-term cost by US$831 per hospitalization for intermediate-risk patients and the long-term cost savings approaching US$10 000 per year from the prevention of long-term nursing home days. 10,11 One systematic review specific to the prevention of delirium in the elderly patients concluded that multicomponent interventions to prevent delirium are effective and should be implemented through synergistic cooperation between the various health care disciplines. 12
Both nonpharmacological and pharmacological strategies have demonstrated benefit in the management of individuals with delirium. 1 Nonpharmacological strategies include frequent reorientation, making eye contact, frequent touching, and using clear verbal instructions when talking to patients. 2 Other strategies include the minimizing of sensory impairments comprising vision and hearing loss by the use of assistive devices, reducing the use of physical restraints, providing treatment in a nonstimulating environment, and minimizing staff and room changes. 1 Pharmacotherapy in delirium is mainly targeted toward the treatment of its underlying causes. 3 However, it is also used to manage the patient’s behaviors when these behaviors cannot be controlled by nonpharmacological means. 2 A recent systematic review by Friedman et al indicated that pharmacological strategies show greater success in the prevention of delirium rather than in its management. 13 The investigators found evidence for delirium prevention effects with haloperidol, second-generation antipsychotics, gabapentin, and melatonin.
It is postulated that sundowning syndrome, the term used to describe complex behavioral symptoms in patients with dementia, is more common in the evening and at night and the alterations in the sleep–wake cycle seen in individuals with delirium have a common neurobiological substrate, that is, a disturbance in the circadian rhythm.
14
Melatonin (
A disruption to the circadian pattern of melatonin secretion has also been noted in individuals with postoperative delirium (POD).
17,18
In a recent study, the postoperative plasma concentration of melatonin at 1 hour after surgery was found to be significantly lower in individuals with delirium than in those without delirium.
20
The investigators found that after adjustment for relevant confounders, the plasma melatonin concentration was independently associated with the risk of delirium (odds ratio [OR], 0.50;
A summary of the available evidence indicates that there is a complex relationship between serum melatonin levels and the development of delirium. 15 Older adults who have an age-related suppression of their nocturnal peak levels of melatonin and a decline in the circadian amplitude of the melatonin rhythm are at a higher risk of developing delirium. 14 When these individuals encounter a physiologically stressful situation like a medical illnesses or a surgical procedure, where there is a disruption of the circadian rhythm, an episode of delirium develops. The physiological changes that develop during an episode of delirium further disrupt the individual circadian rhythm, thus perpetuating a cycle for maintaining the delirium. 23
There is some evidence that melatonin has beneficial effects on circadian rhythm disturbances in dementia. In a systematic review by de Jonghe et al, the investigators found that in 2 of the 4 randomized controlled trials (RCTs) and all 5 of the case series that used melatonin, there was an improvement in sundowning/agitated behavior in individuals with dementia. 14 As circadian rhythm disturbances are seen in both dementia and delirium, the authors postulated that melatonin may have beneficial effects in the management of delirium. A report of 2 cases indicated that melatonin at 2 mg at bedtime orally for 4 days was beneficial in the prevention and management delirium. 24 In another case report, the investigators found that melatonin given at 4 mg a day orally was beneficial in an individual with dementia and sundowning syndrome and possible delirium. 25
Interestingly, there is emerging evidence that the melatonin agonist, ramalteon, has some benefit in the management of delirium. In a case series of 3 women aged between 59 and 83 years, the use of ramelteon at 8 mg a day orally was found to be beneficial in the management of delirium. 26 A case series of 4 men and 1 woman aged between 71 and 91 years indicated that ramelteon at 8 mg at bedtime orally was beneficial in the management of delirium, and it was well tolerated. 27 In a retrospective review, the investigators found that among elderly individuals with delirium and insomnia after an acute stroke (7 individuals, mean age 76 years), the use of ramelteon was beneficial in managing their behaviors, and no adverse effects were noted with the medication. 28 In another case report, a 100-year-old Japanese man with delirium who had not responded to management with risperidone 0.5 mg once daily orally responded to a switch to ramelteon at 8 mg at bedtime orally. 29 The authors of this report indicate that the symptoms of delirium improved without any adverse effects from ramelteon (Table 1).
Case Reports and Case Series for Melatonin and Melatonin Agonist for Delirium.

It is postulated that ramelteon exerts its hypnotic action by the suppression of electrical activity via the melatonin 1 (MT1) receptor, and the circadian phase shifts due to its action on the melatonin 2 (MT2) receptor in the suprachiasmatic nucleus. 30 Ramelteon is also noted to have a 6-fold higher affinity for MT1 receptors and 3-fold affinity for MT2 receptors with a longer half-life than melatonin. 31 The higher affinity of ramelteon for the melatonin receptors is thought to have beneficial effects on delirium. 27 Ramelteon appears be fairly well tolerated, and it does not impair next-day cognitive or motor performances and is not associated with withdrawal symptoms, rebound insomnia, or abuse potential. 32
Given the growing body of evidence that melatonin and the melatonin agonist ramelteon may show some benefit in individuals with delirium, we wanted to review the data on their use in older adults with delirium from RCTs.
Search Strategy
The purpose of this review is to summarize the data from RCTs on the use of melatonin and melatonin agonist ramelteon for delirium in older adults. We performed a literature search of 5 major databases: PubMed, MEDLINE, PsychINFO, Embase, and Cochrane Library. Search terms were “delirium,” “melatonin,” “melatonin agonist,” and “ramelteon.” The databases were searched through March 31, 2014. There was no language limit set in order to be as inclusive as possible with the search strategy.
Each author independently searched the 5 databases for studies with the search terms. Abstracts of all of the studies that were noted on the initial screening were retrieved for further evaluation. Studies were selected for full-text review if they involved patients who were ≥60 years of age, were diagnosed as having delirium by any diagnostic criteria, were prevention or management studies, used melatonin or a melatonin agonist, and had an RCT design. Case reports, case series, database reviews, population-based reviews, systematic reviews, and meta-analysis were also excluded, but the data available from these studies were to be used as a comparison to the data available from this review. Disagreements between the authors regarding the study selection for this review were resolved through consensus.
We operationalized prevention as a strategy to avoid the development of a new episode of delirium or to avoid the recurrence of another episode of delirium. Management was operationalized as a strategy to control the symptoms of delirium once they occurred.
Results
For melatonin, the search strategy yielded a total of 250 possible articles. This included 66 articles in PubMed, 22 articles in MEDLINE, 6 articles in PsychINFO, 148 articles in Embase, and 8 articles in Cochrane Library. After a review of the abstracts, 8 articles were obtained for a full-text review. Of these, 6 articles were excluded as they were case reports, editorial, or reviews. Only 2 articles met the inclusion criteria. No additional articles were retrieved from the bibliographic search of the relevant articles.
The search strategy yielded a total of 7 articles for the melatonin agonist ramelteon. Of the 7 articles, 6 were obtained from PubMed. No articles were obtained from the search of MEDLINE, PsychINFO, or Embase databases. One article was obtained from the search of Cochrane Library. After a review of the abstracts, 5 articles were obtained for a full-text review. Of these, only 1 article met the inclusion criteria. The other articles were 2 case series and 2 case reports. No additional articles were retrieved from the bibliographic search of the relevant articles.
To aid with the evaluation of data, we organized the studies in a chronological manner with the older study being the first to the most recent study being the last. The quality of data was evaluated using the criteria developed by the Centre for Evidence Based Medicine (CEBM) for RCT evaluation (Table 2). 33
Quality of Studies Reviewed.

Melatonin
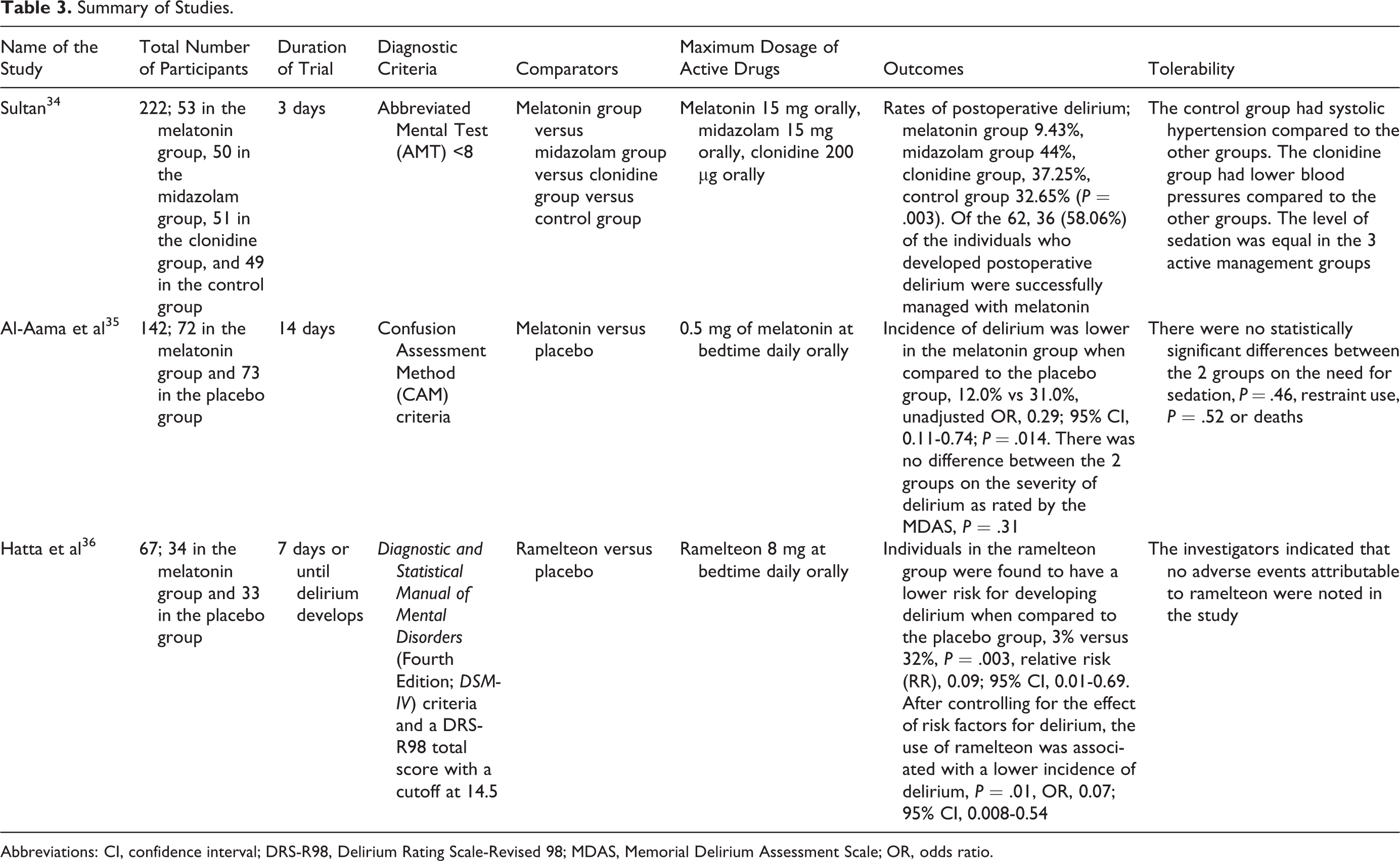
In the study by Sultan, 300 individuals ≥65 years of age who were scheduled for hip arthroplasty were screened for inclusion into this study.
34
Abbreviated Mental Test (AMT), a test approved by the Royal College of Physicians and the British Geriatric Society for routine assessment of cognitive function in the elderly patients, was conducted in all individuals. Those individuals with scores <8 indicating cognitive dysfunction were excluded from the study. Subsequently, individuals were randomized to 1 of the 4 groups. Group 1 (control) received no premedication. Group 2 received melatonin 1 capsule of 5 mg melatonin at sleep time and another 5 mg 90 minutes before the operative time. Group 3 received midazolam, 1 tablet of 7.5 mg at sleep time and another 7.5 mg, 90 minutes before the operative time. Group 4 received clonidine, 1 tablet of 100 μg at sleep time and another 100 μg 90 minutes before the operative time. The doses of midazolam and clonidine used for preoperative sedation were standard doses. All individuals received spinal anesthesia according to the standard techniques. Abbreviated Mental Test was repeated on the same day of operation (POD-0) and for 3 consecutive PODs. Individuals who scored <8 on the AMT were considered to have developed POD. These individuals received melatonin 5 mg at 9
In the study by Al-Aama et al, 145 individuals who were ≥65 years of age who were admitted to the internal medicine inpatient service of a hospital were randomly assigned to 1 of the 2 arms of the study using a computer–generated, blocked-randomization sequence to receive either melatonin or placebo.
35
A total of 72 individuals were assigned to the melatonin group and 73 to the placebo group. Exclusion criteria for the study were expected hospital stay or life expectancy of <48 hours, inability to communicate in English, inability to take oral medications, history of an intracranial bleed or seizures, history of a nontherapeutic international normalized ratio ≤1 or ≥4 while on warfarin, or has a known allergy to the study compounds. Once admitted, the study participants received either the 0.5 mg of oral melatonin or the placebo prior to sleep in a double-blinded fashion. This medication regimen was continued until discharge from the hospital, death, or up to 14 days. The primary study outcome measure was delirium, defined according to the Confusion Assessment Method (CAM) criteria. The secondary outcome measure was the severity of delirium measured with the Memorial Delirium Assessment Scale (MDAS). The participants were tested every 24 to 48 hours using the CAM and the MDAS. Of the 149 people who were initially enrolled in the study, 122 completed the study with 61 participants in either arm of the study. A smaller proportion of participants in the melatonin group developed delirium when compared to the placebo group (12.0% vs 31.0%; unadjusted OR, 0.29; 95% confidence intervals [CIs], 0.11-0.74;
Melatonin Agonist
In a study by Hatta et al, 67 individuals aged between 65 and 89 years who were admitted to ICUs or to regular acute wards for medical problems were randomized to receive either ramelteon 8 mg at bedtime orally or placebo.
36
Individuals who could not take medications orally or had a life expectancy of less than 48 hours were excluded from the study. Individuals were randomized using a sealed envelope method in a rater-blind manner to receive either ramelteon or placebo. The randomization was done using a random number table with sequentially numbered, opaque, and sealed envelopes used to conceal the allocation sequence. The study medication was managed by nurses and administered daily at 9
Summary of Studies.
Abbreviations: CI, confidence interval; DRS-R98, Delirium Rating Scale-Revised 98; MDAS, Memorial Delirium Assessment Scale; OR, odds ratio.
Discussion
Data available from 4 case reports and 3 case series indicate that melatonin and the melatonin agonist ramelteon have shown some benefit in the prevention and management of delirium. 24 –29 Both melatonin and ramelteon were well tolerated in these reports.
Evidence from this systematic review indicates that there is limited but emerging data on the use of melatonin and ramelteon for delirium in the elderly patients from controlled study. The 2 melatonin studies obtained through a systematic search of the literature point to the fact that melatonin shows beneficial effect for both the prevention and the management of delirium in late life. The only ramelteon study obtained from the systematic search indicates that ramelteon shows beneficial effect in the prevention of delirium in late life.
The study by Sultan included pre- and post-hip arthroplasty patients who were prescribed melatonin for the prevention and treatment of delirium. Melatonin was compared to midazolam, clonidine, and control groups.
34
The incidence of POD was lower in the melatonin group compared to the other groups,
Although not available as a research article, data noted in the form of an abstract from the European Geriatric Medicine Conference, 2013, indicate that de Jonghe et al completed a multicenter, double-blind RCT of 3 mg of melatonin versus placebo for preventing delirium in individuals >65 years of age with hip fracture.
37
Primary outcome was the incidence of delirium according to the
The quality of data evaluated using the criteria developed by the CEBM for RCT evaluation indicated that the studies included in this review were well designed. However, there are some limitations to these studies. In the study by Sultan, AMT <8 was used as the diagnostic criteria for delirium.
34
Although used in some studies to detect delirium, the AMT was initially designed to detect cognitive impairment and not specifically delirium.
38
–41
Not using another more validated diagnostic criteria like the
Did the wide range in the dosing strategy for melatonin have any effect on the outcome of the 2 studies? The Sultan study used melatonin at a dose of 15 mg in total, and the Al Aama et al study used melatonin at a dose of 0.5 mg a day. This variability of dosing for melatonin is due to the fact that melatonin is categorized by the US Food and Drug Administration as a dietary supplement and not as a drug. 42 The melatonin supplements are associated with large variations in effective doses and serum concentrations partly due to the differences in their purity and integrity. This variability in dosing may have contributed to the different outcomes noted in the 2 studies. The Sultan study when compared to the Al Aama study used a higher dose of melatonin. The ramelteon study used the 8 mg at bedtime orally dosing strategy. The approved adult dose for ramelteon in the United States is 8 mg at bedtime orally, although other studies of ramelteon have used the drug in the dosage range from 4 to 32 mg daily orally. 43
Despite their limitations, what do these studies indicate? A conservative assessment informs us that melatonin may have some benefit in preventing and managing symptoms of delirium in older adults. However, there is no evidence that melatonin reduces the severity of delirium or has any effect on behaviors or functions in these individuals. Melatonin was well tolerated in the 2 studies. Although difficult to make a definitive conclusion based on data from just 1 study, current evidence indicates ramelteon may prevent the onset of delirium in older adults with acute medical illness. Ramelteon was well tolerated in this study.
How does the evidence for using melatonin and ramelteon in older adults with delirium compare to the data on the other pharmacological agents for delirium? The Cochrane review by Lonergan et al indicates that low-dose haloperidol has similar efficacy and side-effect profile when compared to olanzapine and risperidone in the management of delirium. 44 High-dose haloperidol was associated with a greater incidence parkinsonism than the atypical antipsychotics. Low-dose haloperidol was reported to be effective in decreasing the degree and duration of delirium in postoperative patients when compared to placebo. A meta-analysis by Zhang et al indicated that dexmedetomidine sedation was associated with less delirium when compared to sedation produced by other drugs (pooled RR, 0.39; 95% CI, 0.16-0.95). 45 Both typical (RR, 0.71; 95% CI, 0.54-0.93) and atypical antipsychotics (RR, 0.36; 95% CI, 0.26-0.50) decreased delirium occurrence when compared to placebo. Multicomponent interventions (RR, 0.71; 95% CI, 0.58-0.86) were effective in preventing delirium. No difference in the incidences of delirium was found between acetylcholinesterase inhibitors and placebo (RR, 0.95; 95% CI, 0.63-1.44). Prevention of POD did not shorten the length of hospital stay (pooled standard mean difference = −0.06; 95% CI, −0.16 to 0.04). The meta-analysis by Teslyar et al, which included treatment with haloperidol (3 studies), risperidone (1 study), and olanzapine (1 study), indicated that perioperative use of prophylactic antipsychotics to be effective in reducing the overall risk of POD in elderly individuals, pooled RR (RR = 0.51, 95% CI, 0.33-0.79). 46 A meta-analysis on the efficacy of perioperative interventions to decrease the incidence of POD indicates that perioperative geriatric consultation (OR, 0.46; 95% CI, 0.32-0.67) and lighter anesthesia (OR, 2.66; 95% CI, 1.27-5.56) were associated with a decreased incidence of POD. 47 There is some evidence to suggest the efficacy of prophylactic haloperidol (OR, 0.62; 95% CI, 0.36-1.05), bright light therapy (OR, 0.20; 95% CI, 0.03-1.19), and general as opposed to regional anesthesia (OR, 0.76; 95% CI, 0.47-1.23). A systematic review of 3 RCTs indicates that intervention strategies to prevent delirium are most efficacious in reducing its incidence in both surgical and medical patients. 12 They may also have benefit on the duration, severity of delirium, and the functional status. Strategies to manage delirium are ineffective in older individuals admitted to medical services. But, they show some benefit in older adults admitted for surgery where there is a shorter duration of symptoms and a diminished severity of delirium. There is no effect of intervention strategies on the length of stay or mortality. The systematic review by Friedman et al indicates that pharmacological strategies show greater benefit in preventing delirium rather managing it. 13 The investigators found evidence for haloperidol, second-generation antipsychotics, gabapentin, and melatonin for the prevention of delirium.
When compared to controlled studies of melatonin in individuals with dementia, how does this particular review matchup? A meta-analysis of 3 studies indicated that melatonin had non-significant effects on cognition when compared to placebo on the Mini-Mental State Examination (MMSE) and the Alzheimer's Disease Assessment Scale (ADAS) cognitive scale. 48 In 2 of these studies, improvements in psychopathological behaviors (decreased depression, anxiety, apathy, hallucinations, delusions, agitation, irritability, and appetite disturbances) were noted for the change scores from the Neuropsychiatric Inventory (NPI) at 7 weeks using 2.5 mg a day of melatonin and on the ADAS non-cognitive scales at 4 weeks using 3 mg a day of melatonin. There was no benefit noted for melatonin on activities of daily living when compared to placebo. There were no reported adverse effects associated with melatonin use. In a systematic review by de Jonghe et al, the investigators found a total of 9 reports including 4 RCTs and 5 case series for the use of melatonin in older adults with dementia and sundowning/agitated behaviors. 14 In all, 2 of the 4 RCTs and all 5 of the case series found an improvement in sundowning/agitated behaviors with melatonin, but the data on its efficacy on sleep quality and daytime functioning were not conclusive. The data from the current review have a similar theme with a limited number of controlled studies indicating that melatonin may have some benefit in the prevention and management of delirium in older adults and has good tolerability.
The data on the use of ramelteon in individuals with dementia are limited to 1 case report and 1 case series. In a report by Asano and colleagues, the authors describe the case of a 79-year-old man with severe Alzheimer’s disease who had failed trials of donepezil, memantine, risperidone, and quetiapine but responded well to a trial of ramelton at 8 mg at bed time orally. 49 There was an improvement in the individual’s scores on the Japanese version of the Neuropsychiatric Inventory (NPI) and on the frequency-weighted score to the Behavioral Pathology in Alzheimer’s Disease Rating Scale from 56 and 89, respectively, prior to the ramelteon trial to 20 and 30 points, respectively, on these scales after 1 week. There was a significant improvement noted on the individual’s sleep along with the behavioral improvements. The effect of the medication was sustained 3 months post discharge from the hospital and the medication was well tolerated. In a case series of 4 individuals, 2 men and 2 women aged between 71 and 80 years having a diagnosis of dementia with Lewy bodies, a trial of ramelteon at 8 mg a day orally over 8 weeks resulted in an improvement in the visual hallucinations, excessive daytime sleepiness, and rapid eye movement sleep behavioral disorder. 50 In these individuals, the cognitive and daily living scores did not change with the ramelteon trial. There was also no change in the extrapyramidal symptoms with the use of ramelteon.
The data for using melatonin as a sleep agent were reviewed in 2 meta-analyses. In the meta-analysis by Brzezinski et al, the investigators used data from 17 different studies. 51 This meta-analysis indicated that use of melatonin reduced sleep onset latency by 4.0 minutes, increased sleep efficiency by 2.2%, and increased the total sleep duration by 12.8 minutes. Hangover effect that was noted with hypnotic agents was not noted with melatonin. A meta-analysis by Buscemi et al found that melatonin decreases sleep onset latency (weighted mean difference ([WMD]: −11.7 minutes). 52 The investigators also found that the sleep latency was decreased to a greater extent in individuals with delayed sleep phase syndrome (WMD: −38.8 minutes) when compared to individuals with insomnia (WMD: −7.2 minutes). There was no evidence of adverse effects from melatonin in this meta-analysis. The effect of melatonin in individuals with secondary sleep disorders and sleep disorders accompanying sleep restriction was noted in a meta-analysis by Buscemi et al. 53 The data from this meta-analysis indicated that in 6 RCTs, melatonin was found to have no effect on sleep onset latency in individuals with secondary sleep disorders. In 9 RCTs, melatonin had no effect on sleep onset latency in people who had sleep disorders accompanying sleep restriction. In 17 RCTs, melatonin use was not associated with any adverse effects.
The data for ramelteon as a sleep agent have been reviewed in 2 recent meta-analyses. In the meta-analysis by Lui and Wang, the investigators found that data from 8 studies indicated that ramelteon use resulted in significant improvements in subjective and polysomnographic sleep latency, total sleep time, and Rapid Eye Movement (REM) latency. 54 Subjective sleep latency was reduced in individuals in the 18 to 64 years age group and not those 65 years or older. Ramelteon use was not associated with a greater risk for any frequent adverse events when compared to controls. In a meta-analysis by Kuriyama et al, the investigators included data from 13 trials. 55 This meta-analysis indicated that ramelteon was associated with reduced subjective sleep latency and improved sleep quality, but not with subjective total sleep time. Ramelteon use was associated with improvements in latency to persistent sleep, total sleep time and sleep efficiency. The only significant adverse event noted with ramelteon use was somnolence when compared to the control group (RR, 1.97, 95% CI, 1.21–3.20).
As inappropriate polypharmacy leads to greater cognitive and functional decline in older adults, in these individuals, lesser the number of medications prescribed, better will be the outcome. 56,57 If that is the case, would the prescription of melatonin or ramelteon add to the burden of care for elderly with delirium? In a conference presentation of a double-blind RCT of placebo and melatonin in medically ill individuals receiving conscious sedation, Mistraletti et al found that the prescription of melatonin daily from the third ICU day until discharge from the hospital, reduced the use of hydroxyzine, benzodiazepine, haloperidol, propofol, and opiates. 58 It also resulted in a decrease of pain events, anxiety, agitation, and the use of physical restraints. The information from this study indicates that the prescription of melatonin may reduce the use of other medication and improve outcomes in older adults with delirium. The data for ramelteon having such an effect on older adults with delirium is not available at the present time.
Where does the current information place melatonin and ramelteon in the algorithm for the prevention and management of delirium in older adults? Based on the evidence, it appears that melatonin and ramelteon can be prescribed to vulnerable elderly patients who are at high risk of developing delirium, especially those admitted to acute medical and pre- and postoperative services. Melatonin may also have some benefit in the management of delirium that has already developed in older adults. Melatonin and ramelteon should be prescribed as once-a-day dosing prior to sleep time. The exact dose of melatonin to be used in these individuals is unclear with the dosage ranging between 0.5 and 10 mg. For ramelteon, 8 mg dosing orally appears to be well tolerated in older adults with delirium. The duration of prescription for both melatonin and ramelteon in these individuals remains unclear at this time. Given the strong data for multicomponent interventions to prevent delirium, any prescription of melatonin or ramelteon should be combined with this management modality so-as-to achieve optimal outcomes in these individuals.
Although the available data are promising, larger well-controlled trials for both melatonin and ramelteon with cognitive, functional, and behavioral assessment tools completed at different times of the day, using multiple fixed doses of these 2 agents and for longer duration, will need to be completed before we can have a definitive answer on their role in care of older adults with delirium.
Conclusions
Available data from this current review indicate that there is a dearth of controlled studies on the use of melatonin and ramelteon for the prevention and management of delirium in older adults. Available data from 2 studies indicate that melatonin may have some benefit in prevention and management of delirium in older adults. Data from 1 study indicate that ramelteon may help prevent delirium in older adults with medical illness. Melatonin and ramelteon were noted to be well tolerated in these studies. Given this information, melatonin and ramelteon could be prescribed to older adults at high risk of developing delirium especially those who are medically ill. Melatonin may also have a role in the management of delirium in the elderly.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
