Abstract
There are numerous measures for detecting the presence of dementia and quantifying its severity and progression. We analyzed the relations between scores on 5 commonly used measures (Mini-Mental State Examination, Montreal Cognitive Assessment, Alzheimer’s Disease Assessment Scale-Cognitive Subscale, Activities of Daily Living Scale, and Global Deterioration Scale) of 101 successive admissions to a memory clinic. Patients were included in the analysis only if they received a diagnosis of mild cognitive impairment (MCI) due to Alzheimer’s disease (AD) pathophysiological process or probable AD and if they received all measures. Regression analysis yielded 20 linear equations that allow for conversion between test scores on any 2 measures. Further, participants were grouped by MMSE scores with regard to level of disease severity, allowing for the creation of a quick reference table for estimating an approximate score range between measures. Results from this study provide a useful tool for clinicians when comparing between multiple different instruments that measure the mental status and functional ability of individuals with AD and MCI due to AD pathology.
Introduction
Alzheimer’s disease (AD) continues to be the most prevalent form of dementia, accounting for roughly 75% of all dementia diagnoses in the United States. 1 Over the past 2 decades, much progress has been made with regard to both the detection and treatment of AD, but despite this progress, it is estimated that nearly half of the roughly 5.4 million Americans who have AD remain undiagnosed. 2 Further, the prevalence of AD is projected to increase dramatically in the coming decades. 2 As the number of individuals with AD continues to grow, the use of routine and well-validated measures of mental status and functional ability in detecting and characterizing AD will continue to be of paramount importance.
Currently, there are numerous well-validated measures available to aid clinicians in the detection of dementia as well as to quantify its progression and severity. 3 To date, some of the most commonly and widely utilized measures for detecting and staging AD as well as other dementias are the Mini-Mental State Examination (MMSE) 4 and the Alzheimer’s Disease Assessment Scale-Cognitive Subscale (ADAS-Cog), 5 both of which are sensitive to AD and are highly correlated with one another. 6 With regard to functional status, both the Activities of Daily Living (ADL) scale 7 and the Global Deterioration Scale (GDS) 8 are also frequently used to determine an individual’s degree of impairment. More recently, the Montreal Cognitive Assessment (MoCA) 9 has also become increasingly used by clinicians due to its high specificity for detecting cognitive impairment as well as sensitivity to detecting early cognitive changes associated with the development of AD, such as mild cognitive impairment (MCI). 3,10 -12
Each of these instruments can be used to establish the presence of dementia as well as the degree of impairment, although clinicians' use of these scales has been shown to vary widely. 13 This article seeks to build upon and update the work of Solomon and colleagues who reported on the correlations between 5 commonly used measures of mental status/functional ability in patients with AD and provided equations to quickly convert scores on any one of these instruments to any other. 6 Although more than a decade has passed since the publication of the original article, no single measure has emerged as universal in the evaluation of dementia. Further, although measures such as the MMSE are still used widely, their sensitivity to the earliest stages of AD as addressed in the updated National Institute of Aging and Alzheimer’s Association 2011 guidelines 14 -16 has been shown to be poor relative to newer measures such as the MoCA, which has begun to gain more widespread use. 10,12,17,18
Thus, it is important to continue to establish the intercorrelation between both established and newer measures so that health care providers have a means of interpreting and converting scores from 1 commonly used instrument to another.
Methods
Retrospective analysis was performed on 101 successive admissions to a private US memory clinic between October 2012 and July 2013. Patients were screened with all 5 of the aforementioned instruments as part of their initial clinical evaluation. The MMSE (score range: 30-0, least impairment to maximal impairment), ADAS-Cog (score range: 0-70), ADLs (range: 0-100), GDS (range: 1-7), and MoCA (range: 30-0). Patients were included in the analysis if all 5 measures were administered in the same evaluation and a diagnosis of probable AD or MCI due to AD pathophysiological process was established in accordance with the updated National Institute on Aging and Alzheimer’s Association workgroup (NIA-AA) guidelines. 2 All measures were administered by the same group of clinicians with the exception of the ADL scale, which is completed by the patient’s caregiver. As data utilized for this analysis were collected during routine clinical evaluations, and analysis was performed retrospectively, institutional review board approval was not sought. Scores on individual tests were interrelated using linear regression.
Results
Analysis of the data yielded 20 linear equations that allow for conversion between scores on any 2 measures. All relationships were linear, and correlations were generally high. Table 1 shows these regression formulas as well as their correlation coefficients and allows for precise conversations of scores between measures using the regression equations.
Linear Regressions Allowing for Conversion Between Measures.
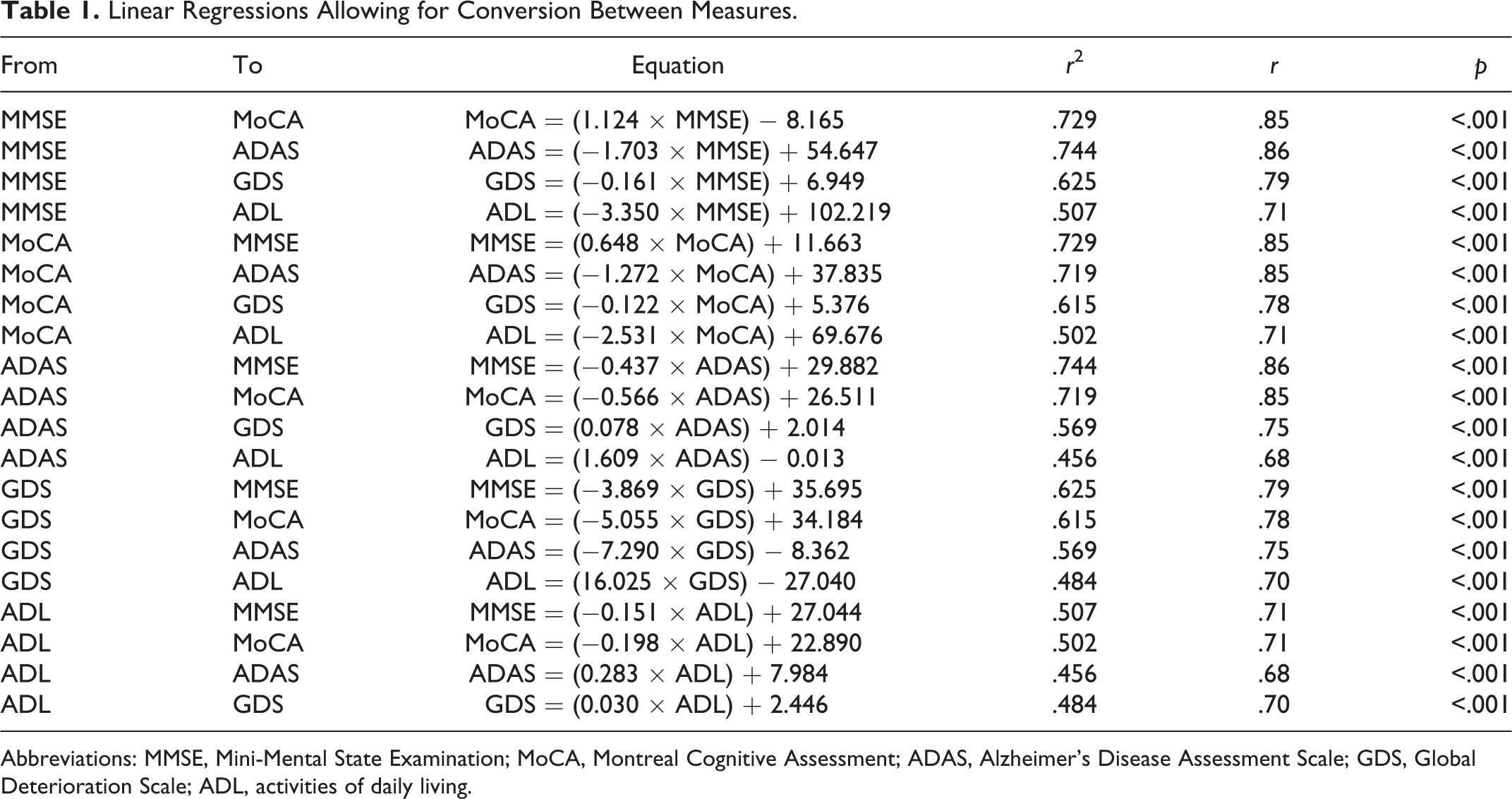
Abbreviations: MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; ADAS, Alzheimer’s Disease Assessment Scale; GDS, Global Deterioration Scale; ADL, activities of daily living.
The MMSE, ADAS-Cog, MoCA, and GDS were all highly correlated and reached statistical significance at the p < .001 level. Correlation coefficients for the MMSE with the aforementioned measures ranged from r = .79 to .86. The range of coefficients for the MoCA (r = .78 to .85), the ADAS-Cog (r = .75 to .86), and the GDS (r = .75 to .79) with these other measures was similarly high. The range of correlation coefficients for the ADL with the other 4 measures was somewhat lower (r = .67 to .71) but nevertheless significant (p < .001). Figure 1 shows scatter plots and linear fit for each of the regressions.
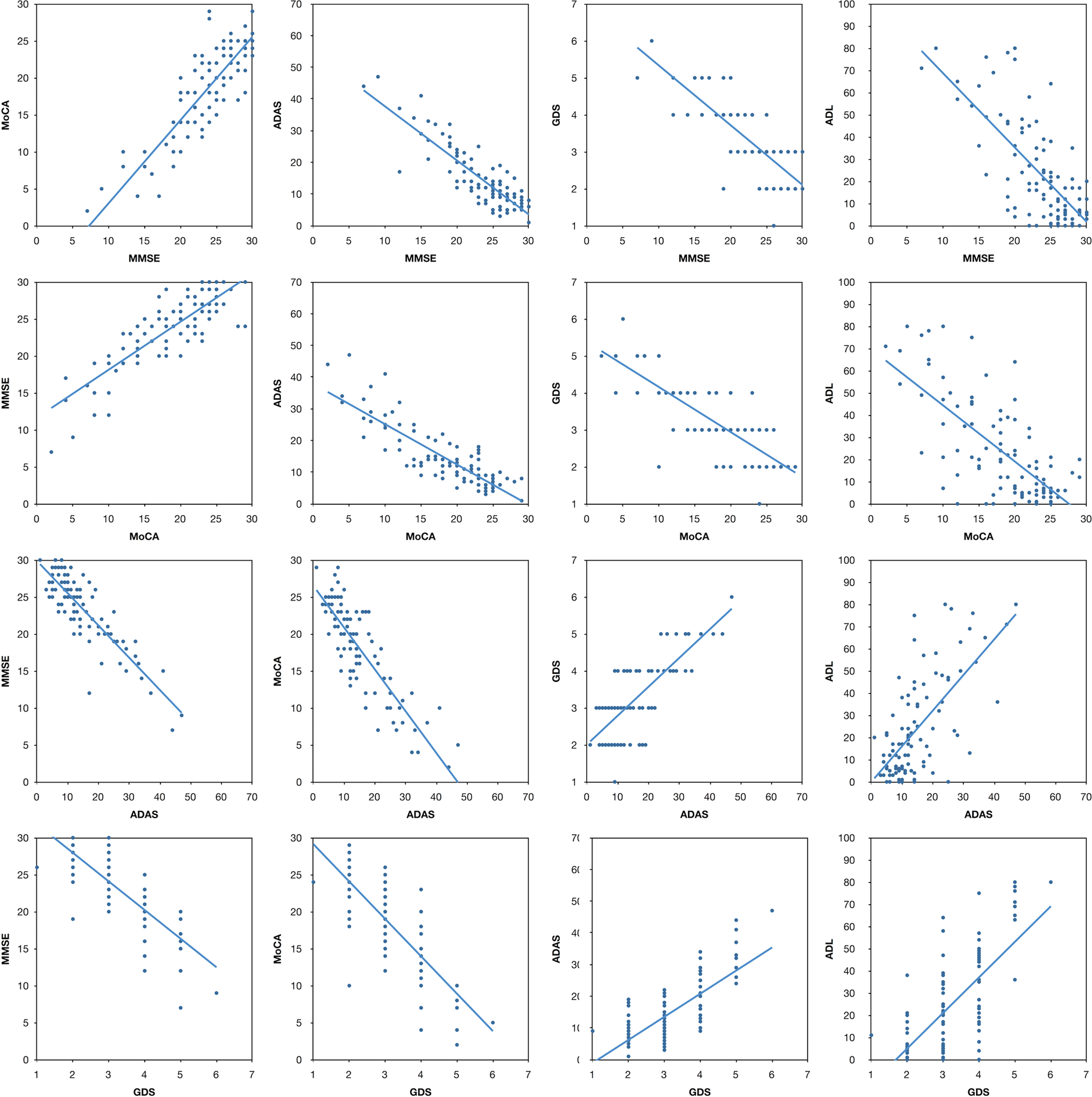
Scatter plot, correlation coefficients and linear fit for all tests. MMSE indicates Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; ADAS, Alzheimer’s Disease Assessment Scale; GDS, Global Deterioration Scale; ADL, activities of daily living.
Similar to Solomon’s original article, 19 patients were grouped by MMSE score into 5 categories, which have been shown to overlap with level of disease severity. 20 Table 2 provides a rapid reference guide for clinicians to convert a score on the MMSE to an approximate score on any of the 4 other measures.
Rapid Conversion Chart for Estimating Scores on Any Other Instrument Given the MMSE Score.
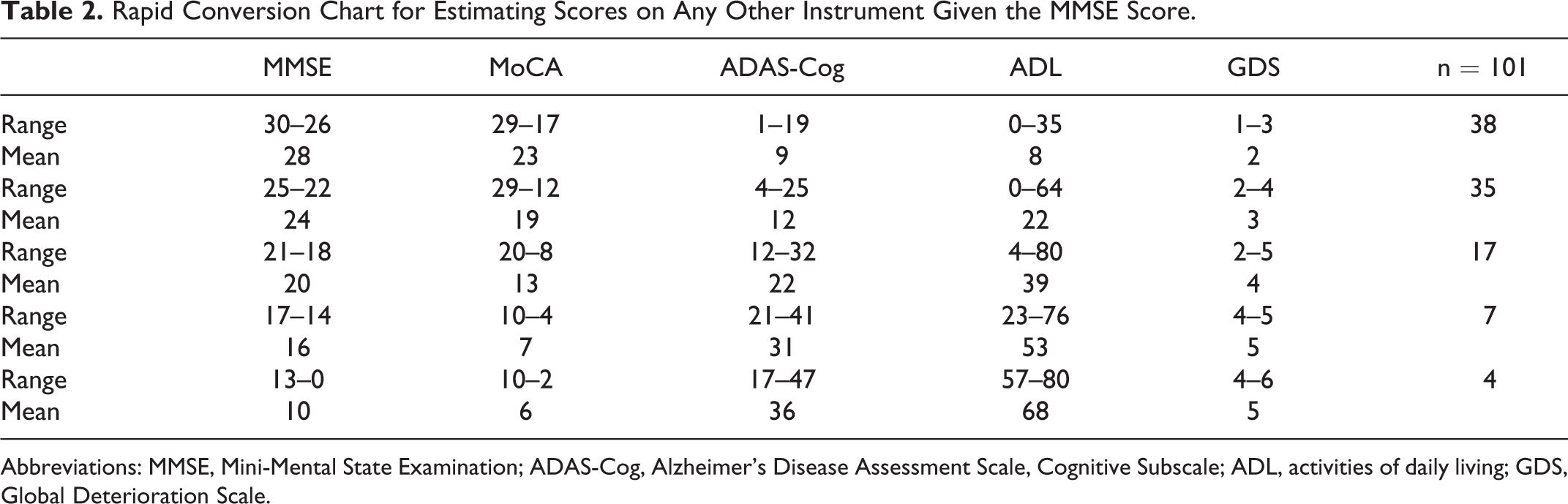
Abbreviations: MMSE, Mini-Mental State Examination; ADAS-Cog, Alzheimer’s Disease Assessment Scale, Cognitive Subscale; ADL, activities of daily living; GDS, Global Deterioration Scale.
Discussion
The results of this analysis provide a clinically useful tool for converting test scores between 5 commonly used measures of mental status and functional ability in patients with both probable AD and MCI due to AD pathology as determined by the updated NIA-AA guidelines. 2
Similar to the original analysis, results from commonly used measures such as the MMSE, ADAS-COG, ADCQ, and GDS were analyzed. With the exception of ADLs, intercorrelations were high among measures (r = .70 to .86). These results corroborate the previous findings 19 and suggest that each instrument is measuring similar cognitive functions. As with the previous analysis, the relationships between the ADL and the other measures were somewhat lower (r = .67 to .71) but nevertheless significant. As suggested previously, 19,21 this may be a consequence of the ADL being caregiver rated, as opposed to clinician rated, and being a functional, rather than cognitive, measure.
Given the recent establishment of new clinical criteria to diagnose individuals with MCI due to AD pathology and the continued emphasis on screening and diagnosis at earlier stages in the disease, 22 -24 this analysis included patients who were diagnosed not only with probable AD but also with MCI due to Alzheimer’s disease pathophysiological process. Further, this analysis used scores on the MoCA, as numerous recent studies have demonstrated the MoCA’s increased sensitivity to detecting cognitive problems due to AD, particularly at early stages in the disease, compared to measures such as the MMSE. 10 -12,17,25
Yet, despite the proliferation of such studies demonstrating the MOCA’s utility as a superior cognitive screening instrument, 13,18 the MMSE continues to be the most commonly used clinical measure for detecting and monitoring cognitive impairment, despite a number of documented limitations. 26 Given these limitations and in concert with the MoCA’s increased sensitivity to subtle cognitive changes associated with MCI, as well as the NIA’s updated diagnostic criteria, providing clinicians with a useful means to convert and interpret between common measures of mental status and functional ability fills an important gap, particularly as no other published literature currently provides the means to do so. By providing a straightforward method to convert and interpret between the measures, clinicians may benefit from continuity when tracking cognitive performance over time in a clinical setting as well as within longitudinal research studies.
Unlike the previous analysis, the current analysis included patients diagnosed with MCI due to AD pathology who generally had a lower degree of overall cognitive and functional impairment due to their early stage of the disease. Further, the population utilized for this analysis were those who sought treatment or were referred to a specialized memory clinic, and therefore, the results are not necessarily generalizable to the public. Finally, given our small sample size, it will be important for these findings to be replicated in the literature so that their validity can continue to be established.
Overall, this analysis may provide a useful tool for clinicians who often need to quickly convert or interpret scores between commonly used measures of mental status and functional ability. It is important to note that no single measure should be considered adequate for establishing an accurate diagnosis. However, as the population continues to age and the incidence of dementia, specifically Alzheimer’s disease, continues to increase, it remains paramount that the relations between commonly used measures are quantified in the hope of helping health care professionals continue to provide accurate and efficacious care to those individuals affected.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
