Abstract
Objective:
A meta-analysis was performed to better clarify the association between polymorphisms of estrogen receptor α genes rs9340799 and rs2234693 and risk of Alzheimer’s disease (AD).
Methods:
Pooled odds ratios (ORs) with 95% confidence intervals (CIs) from fixed and random effect models were calculated. Heterogeneity among studies was evaluated using the I2. Meta-regression was used to explore the potential sources of between-study heterogeneity.
Results:
A total of 23 studies about rs9340799 and 24 studies about rs2234693 were included in this meta-analysis. The combined evidence suggested that the x allele of rs9340799 had a significant protective effect on the risk of AD in codominant model (ORs = 0.893, 95%CIs = 0.822-0.970), especially for AD in Asia and sporadic AD (SAD). The p allele of rs2234693 was associated with decreased risk of AD in codominant model for patients with SAD. No publication bias was detected.
Conclusions:
This meta-analysis suggested that x allele of rs9340799 might have a protective effect on the risk of AD in Asia and in patients with SAD. In addition, the p allele of rs2234693 might decrease the risk of patients with SAD.
Introduction
As the world population is aging, Alzheimer’s disease (AD) is becoming a major public health problem. 1 Alzheimer’s disease is an age-associated neurodegenerative disease that is related to the combined interaction between environmental factors and gene variants. 2,3 Genetic variation has been postulated to influence the risk of AD. Mutations in apolipoprotein E4, PLAU, CR1, CLU, and PICALM are known to contribute to AD risk. 4 –6 As a complex disorder, the genes mentioned earlier cannot explain the overall genetic susceptibility, and it is supposed that some other genes may participate in the development of AD. The estrogen receptor α (ESR-α) gene or estrogen receptor 1 (ESR1) gene islocated throughout the brain, especially in regions involved in learning and memory, such as the hippocampus and amygdale. 7 The mutation of ESR genes may influence the effects of estrogen on AD. 8 Estrogen’s neuroprotective action could also be more direct in response to degenerative disease or injury. Because estrogen exerts its action through nuclear receptors located in the brain regions also involved in cognitive functions, 9 its neuroprotective effect is probably at least in part mediated by ESRs. 8
The gene for the ESR-α has several single-nucleotide polymorphisms, the rs9340799 (XbaI) and rs2234693 (PvuII), which may be associated with expression and function of receptor, 10,11 and most of the studies are focused on these 2 polymorphisms with AD. Four studies 12 –15 suggested that x allele of rs9340799 polymorphism might have a protective effect on risk of AD, while no associations were found in 16 articles. 1,11,16 –29 Five studies 12 –14,17,18 showed that p allele of rs2234693 polymorphism might decrease the risk of AD, and no associations were found in 16 articles. 1,11,15,16,19 –24,26 –31 Thus, these results are inconsistent, and individual studies have relatively small power to confirm this association. Therefore, we performed a meta-analysis to test the hypothesis that the mutant of the 2 polymorphisms in ESR-α may be a protective effect on AD susceptibility.
Materials and Methods
Literature Search
The available articles published in English or Chinese (up to December, 2013) were identified by extended computer-based searches from the following databases: PubMed, Web of Science, and Wan Fang Med Online. The following key words were used: “estrogen receptor alpha” or “estrogen receptor 1” or “ESR-α” or “ESR1” or “rs9340799” or “rs2234693” or “XbaI” or “PvuII” and “polymorphism” or “mutation” and “Alzheimer’s disease” or “AD”. We also reviewed the references cited in the studies and reviewed articles to identify additional studies not captured by our database searches.
Inclusion Criteria
The inclusion criteria were as follows: (1) case–control, cohort, or cross-sectional study published as original study to evaluate the association between (rs9340799 and rs2234693) polymorphisms in ESR-α gene and risk of AD; (2) numbers of each genotype were reported in case and control groups for case–control studies, or exposed and unexposed groups for cohort studies, or data provided from which numbers could be calculated; and (3) patients in each study should come from the same ethnicity and period. The most recent and complete articles were chosen if 1 data from the same population had been published more than once. Two investigators carefully reviewed all identified studies independently to determine whether an individual study was eligible for inclusion criteria in this meta-analysis.
Data Extraction
Two investigators collected the data independently and reached a consensus on all items. The following basic information was extracted from the eligible studies: first author, journal, sample size, year of publication, country, ethnicity of studied population, mean age, male sex percentage, and distributions of allele and genotype. When it came to conflicting evaluations, it was resolved by a third reviewer.
Statistical Analysis
Departure from Hardy-Weinberg equilibrium (HWE) for the rs2234693 and rs9340799 genotype distribution of ESR-α gene in controls was tested by chi-square (χ2) analysis with exact probability (HWE: P > .05). Odds ratio (ORs) with 95% confidence intervals (CIs) were used to assess the strength of the association of the polymorphism of rs2234693 and rs9340799 with the risk of AD. We conducted analysis for 3 polymorphisms 32 considering dominant (pp + Pp vs PP; in this study, P allele mutated into p allele), (xx + Xx vs XX; in this study, X allele mutated into x allele); recessive (pp vs Pp + PP), (xx vs Xx + XX); and codominant (p vs P) and (x vs X) models for rs2234693 and rs9340799, respectively. I2 values of Higgins and Thompson 33 was used to assess heterogeneity among studies, and I2 values of 0%, 25%, 50%, and 75% represent no, low, moderate, and high heterogeneity, 34 respectively. The DerSimonian and Laird random effect model was adopted as the pooling method if substantial heterogeneity was present (I2 > 50%) 34 ; otherwise, the fixed effect model (I2 < 50%) was used as the pooling method. Subgroup analyses by types of AD and ethnicity were also carried out to explore the associations between polymorphism of rs2234693 and rs9340799 and AD risk. Meta-regression with restricted maximum likelihood estimation was performed to explore the potentially important covariates: country, age (ratio of age or mean age in case group to that in control group), and sex (ratio of male percentage in case group to that in control group) that might exert substantial impacts on between-study heterogeneity. If no significant covariates were found to be heterogeneous, the “leave-one-out” sensitive analysis 35 was carried out to evaluate the key contributor of the studies impact on between-study heterogeneity for 2 polymorphisms. Influence analysis was conducted 36 to describe how robust the pooled estimator is while we remove one of the individual studies. An individual study is suspected of excessive influence if the point estimate of its omitted analysis lies outside the 95%CI of the combined analysis. Begg’s test 37 was used to estimate publication bias.
STATA version 10.0 (Stata Corporation, College Station, Texas) was used to perform statistical analyses. All reported probabilities (P values) were 2-sided, with P value less than .05 considered representative of statistically significant.
Results
Characteristics of Studies
We identified 23 articles 1,11 –31 with 25 eligible studies for this meta-analysis. A total of 20 articles that contained 23 studies about rs9340799 with 3487 cases and 8998 controls and 21 articles that contained 24 studies about rs2234693 with 4597 cases and 10 090 controls were enrolled in the meta-analysis with the risk of AD. In all, 21 articles with 24 studies were of case–control design and 1 study was of cohort design. General characteristics, genotype distributions, and the results for HWE test in control groups of the above-mentioned polymorphisms are summarized in Tables 1 and 2.
The Characteristics of rs9340799 Polymorphism Genotype Distributions in this Meta-Analysis.
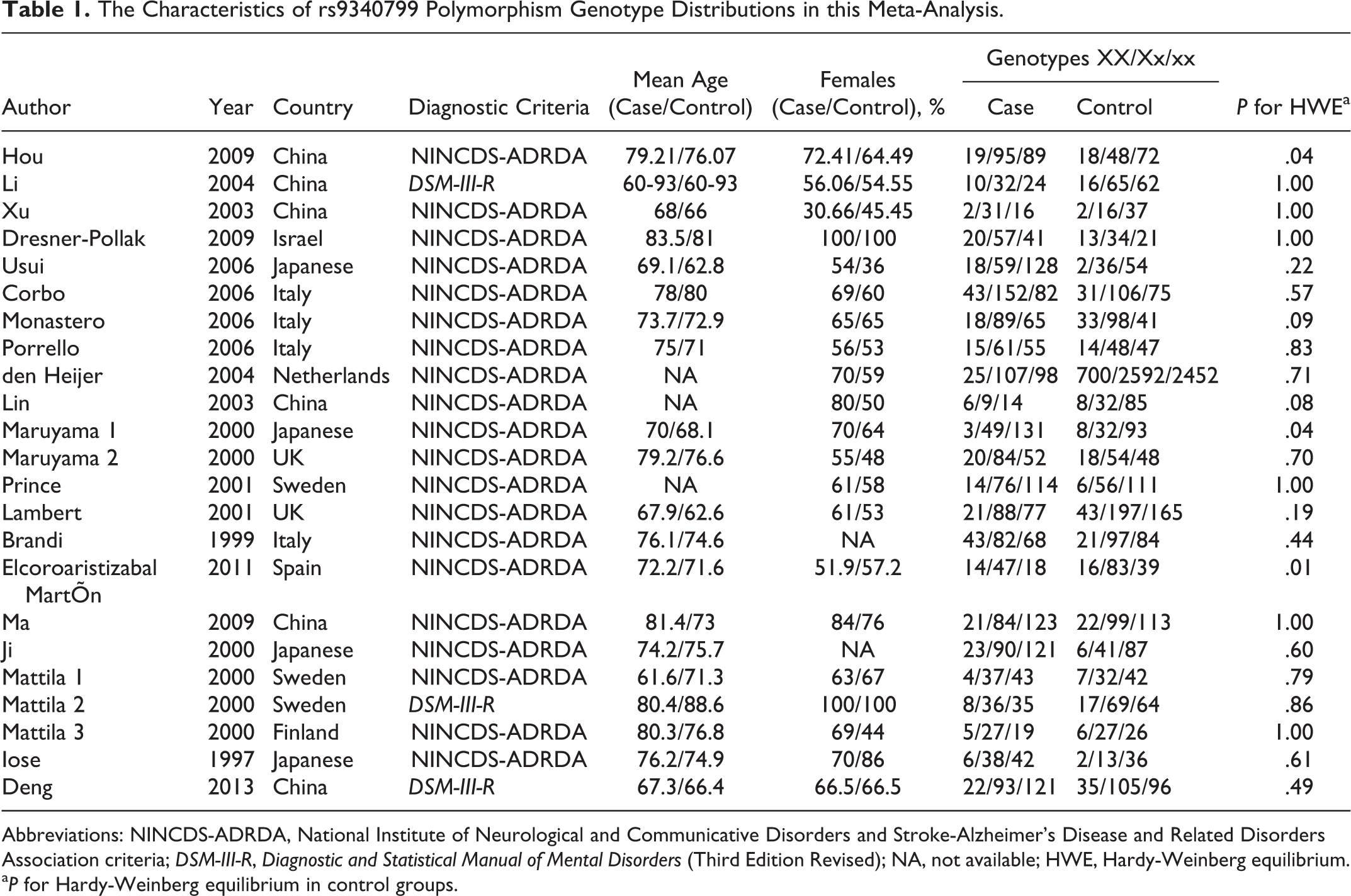
Abbreviations: NINCDS-ADRDA, National Institute of Neurological and Communicative Disorders and Stroke-Alzheimer’s Disease and Related Disorders Association criteria; DSM-III-R, Diagnostic and Statistical Manual of Mental Disorders (Third Edition Revised); NA, not available; HWE, Hardy-Weinberg equilibrium.
a P for Hardy-Weinberg equilibrium in control groups.
The Characteristics of rs2234693 Polymorphism Genotype Distributions in this Meta-Analysis.
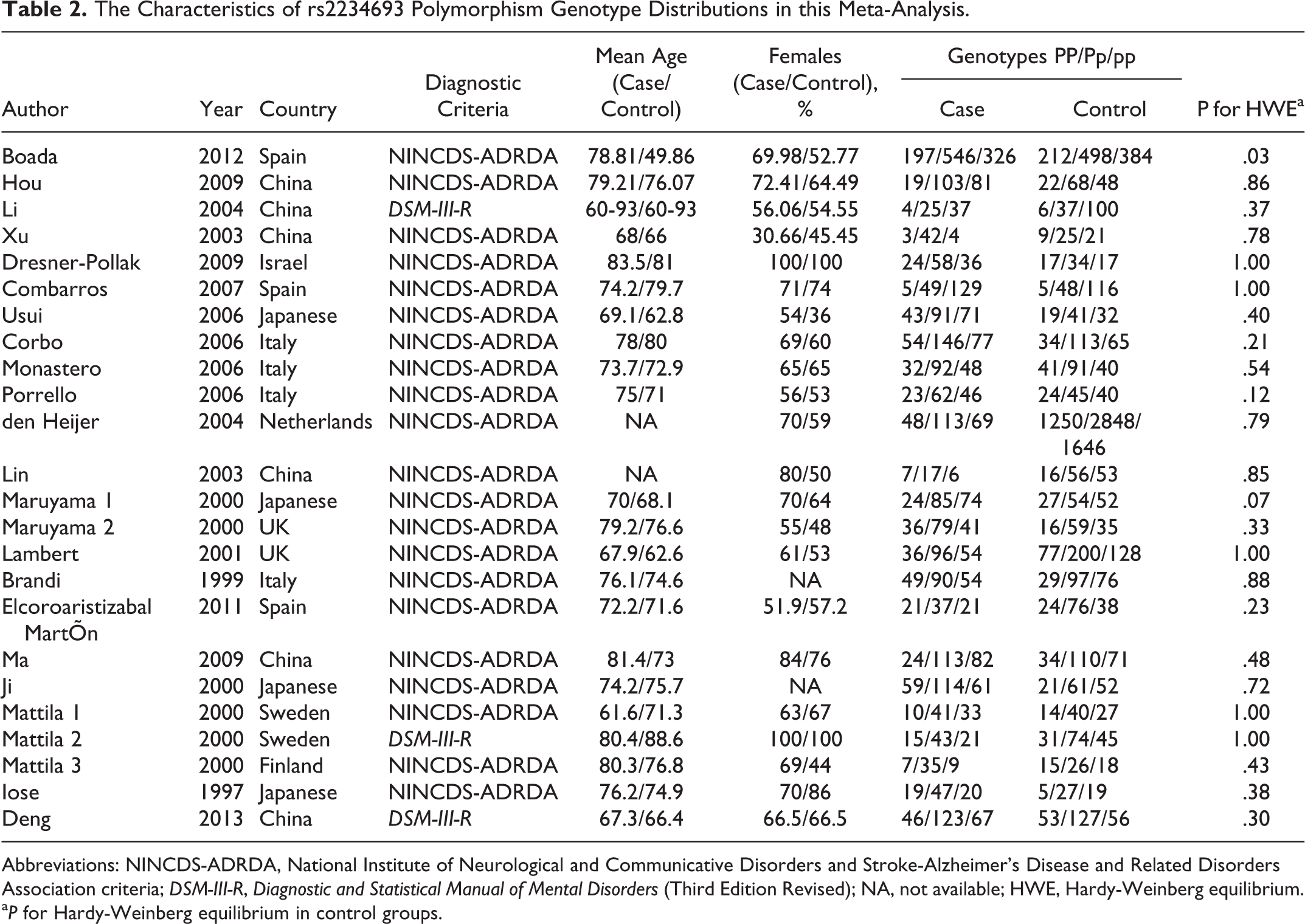
Abbreviations: NINCDS-ADRDA, National Institute of Neurological and Communicative Disorders and Stroke-Alzheimer’s Disease and Related Disorders Association criteria; DSM-III-R, Diagnostic and Statistical Manual of Mental Disorders (Third Edition Revised); NA, not available; HWE, Hardy-Weinberg equilibrium.
a P for Hardy-Weinberg equilibrium in control groups.
Quantitative Synthesis
The results of the pooled analysis are summarized in Table 3.
Pooled Measures on the Relation of Polymorphisms of Estrogen Receptor α Gene rs9340799 and rs2234693 to Alzheimer’s Disease.
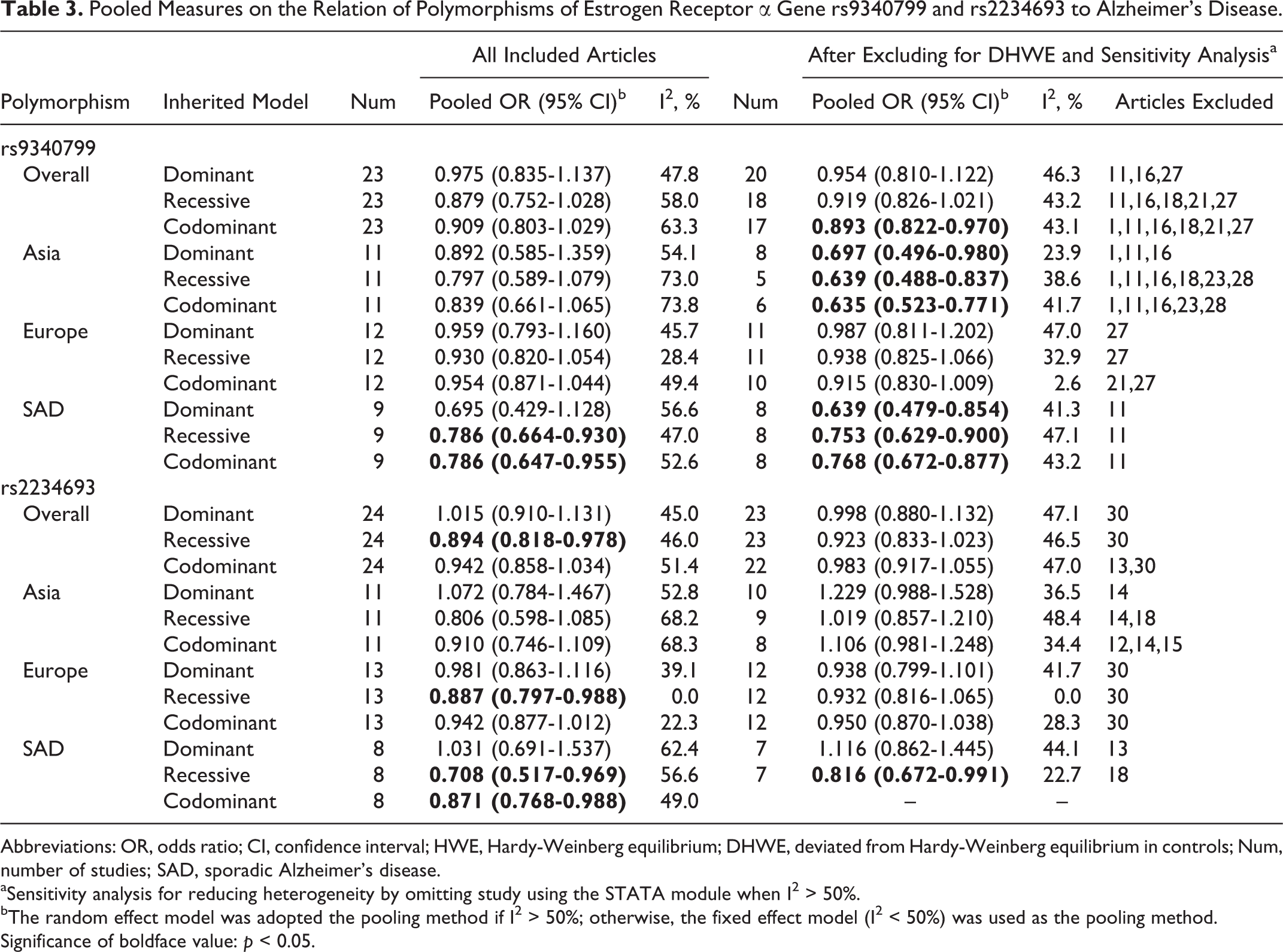
Abbreviations: OR, odds ratio; CI, confidence interval; HWE, Hardy-Weinberg equilibrium; DHWE, deviated from Hardy-Weinberg equilibrium in controls; Num, number of studies; SAD, sporadic Alzheimer's disease.
aSensitivity analysis for reducing heterogeneity by omitting study using the STATA module when I2 > 50%.
bThe random effect model was adopted the pooling method if I2 > 50%; otherwise, the fixed effect model (I2 < 50%) was used as the pooling method. Significance of boldface value: p < 0.05.
The rs9340799 polymorphism
For rs9340799 polymorphism, overall, no significant association was found between the x allele and risk of AD in dominant (ORs = 0.975, 95% CIs = 0.835-1.137), recessive (ORs = 0.879, 95%CIs = 0.752-1.028), and codominant (ORs = 0.909, 95%CIs = 0.803-1.029) models. In subgroup analysis by ethnicity, no evidence indicated the association between rs9340799 and AD susceptibility both for Asians and for Europeans in dominant model (Asian: ORs = 0.892, 95%CIs = 0.585-1.359; European: ORs = 0.959, 95%CIs = 0.793-1.160), recessive model (Asian: ORs = 0.797, 95%CIs = 0.589-1.079; European: ORs = 0.930, 95%CIs = 0.820-1.054), and codominant model (Asian: ORs = 0.839, 95%CIs = 0.661-1.065; European: ORs = 0.954, 95%CIs = 0.871-1.044). Nine studies with 1325 cases and 1173 controls were conducted for sporadic AD (SAD) solely in the overall sample. With regard to patients with SAD, the x allele was found significantly contributing to decreased AD risk in recessive (ORs = 0.786, 95%CIs = 0.664-0.930) and codominant (ORs = 0.786, 95%CIs = 0.647-0.955) models but not in dominant model (ORs = 0.695, 95%CIs = 0.429-1.128). After excluding 3 studies 11,16,27 that deviated from HWE (DHWE) in controls, significant associations were also found between x allele and decreased AD risk in SAD subgroup (dominant, ORs = 0.608, 95%CIs = 0.399-0.928).
The rs2234693 polymorphism
For rs2234693 polymorphism, overall, this meta-analysis showed a significant protective effect of the p allele on AD risk in recessive model (ORs = 0.894, 95%CIs = 0.818-0.978) but not in dominant (ORs = 1.015, 95%CIs = 0.910-1.131) and codominant (ORs = 0.942, 95%CIs = 0.858-1.034) models. In the analysis stratified by ethnicity, no evidence indicated the significant association between rs2234693 and AD susceptibility for Asians in 3 inherited models. For Europeans, significant association was found between the p allele and risk of AD in recessive model (ORs = 0.887, 95%CIs = 0.797-0.988) but not in dominant (ORs = 0.981, 95%CIs = 0.863-1.116) and codominant (ORs = 0.942, 95%CIs = 0.877-1.012) models. Eight studies with 1121 cases and 987 controls were conducted for SAD solely in the overall sample. With regard to patients with SAD, the p allele was found contributing significantly to decreased AD risk in recessive (ORs = 0.708, 95%CIs = 0.517-0.969) and codominant (ORs = 0.871, 95%CIs = 0.768-0.988) models. However, no association was detected in dominant model (ORs = 1.031, 95%CIs = 0.691-1.537). After excluding the study 30 that DHWE in controls, no significant associations were found between the p allele and AD risk considering recessive comparison for overall data (ORs = 0.923, 95%CIs = 0.833-1.023) and European subgroup (recessive: ORs = 0.932, 95%CIs = 0.816-1.065). The other associations were not altered significantly after excluding articles DHWE in controls.
Sources of Heterogeneity and Sensitivity Analysis
Before and after excluding studies DHWE in controls, strong evidence of heterogeneity (I2 > 50%) was found in some inherited models considering the association of 2 polymorphisms with AD. Univariate meta-regression with the covariates of country, age (ratio of age or mean age in case group to that in control group), and sex (ratio of male percentage in case group to that in control group) showed that no covariates had a significant impact on between-study heterogeneity.
The key contributor of the study to between-study heterogeneity was assessed by the “leave-one-out” sensitive analysis. Low and moderate heterogeneities (I2 < 50%) were found in any inherited models following the exclusion of certain studies. After excluding for DHWE and sensitivity analysis, the association of ESR-α gene rs9340799 polymorphism with AD risk was found significant in the codominant model for overall data (ORs = 0.893, 95%CIs = 0.822-0.970) and 3 inherited models in Asia (detailed data are shown in Table 3). However, no significant association was found between the ESR-α gene rs2234693 p allele and AD risk in recessive model for overall data (ORs = 0.923, 95%CIs = 0.833-1.023) and European subgroup (ORs = 0.932, 95%CIs = 0.816-1.065). Figure 1 presented the forest plot of ORs for AD in codominant model of rs9340799 polymorphism and AD after excluding articles DHWE in controls and sensitivity analysis. Figure 2 showed the forest plot of ORs in recessive model for rs2234693 polymorphism and the risk of AD after excluding articles DHWE in controls and sensitivity analysis.
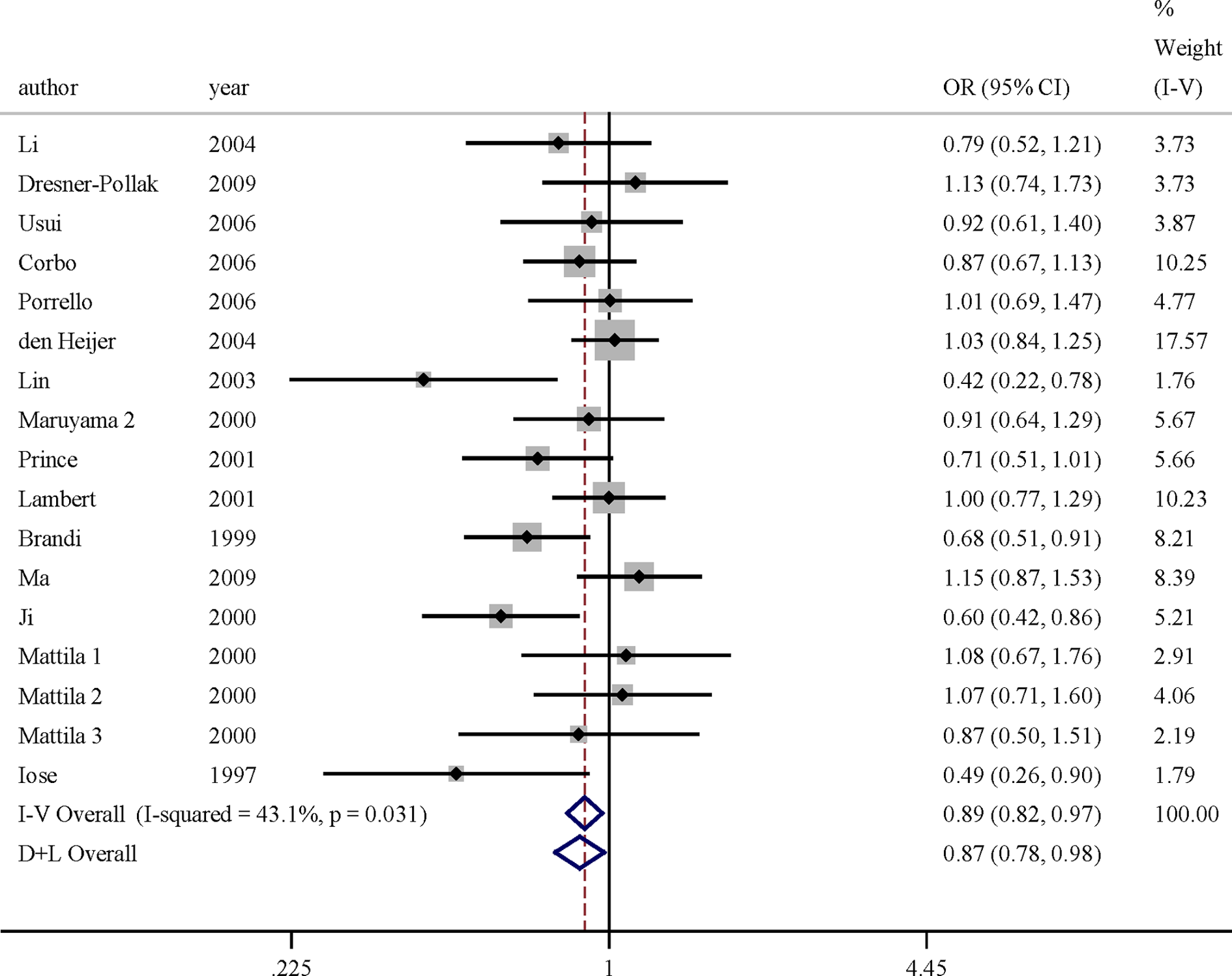
Meta-analysis for AD risk depending on the rs9340799 polymorphism in the ESR-α gene. Forest plots of relationship between polymorphism of ESR-α gene rs9340799 and AD risk in codominant model after excluding articles that deviated from Hardy-Weinberg equilibrium (HWE) in controls and sensitivity analysis. White diamond denotes the pooled OR. Black squares indicate the OR in each study, with square sizes inversely proportional to the standard error of the OR. Horizontal lines represent 95% confidence intervals (CIs). AD indicates Alzheimer’s disease; ESR-α, estrogen receptor α; OR, odds ratio.
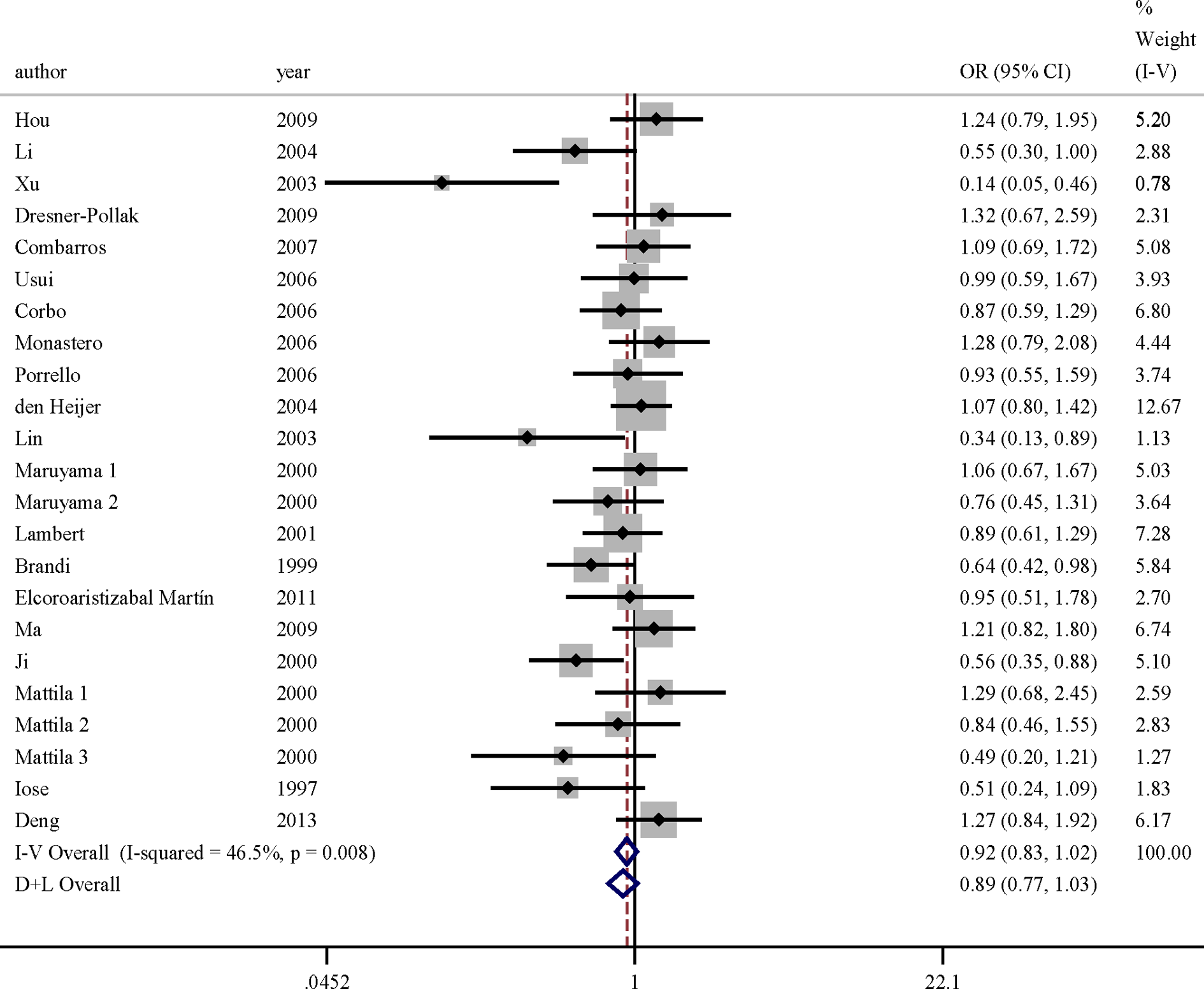
Meta-analysis for AD risk depending on the rs2234693 polymorphism in the ESR-α gene. Forest plots of relationship between polymorphism of ESR-α gene rs2234693 and AD risk in recessive model after excluding articles that deviated from Hardy-Weinberg equilibrium (HWE) in controls and sensitivity analysis. White diamond denotes the pooled OR. Black squares indicate the OR in each study, with square sizes inversely proportional to the standard error of the OR. Horizontal lines represent 95% confidence intervals (CIs). AD indicates Alzheimer’s disease; ESR-α, estrogen receptor α; OR, odds ratio.
Influence Analysis and Publication Bias Evaluation
For both of the polymorphisms, after exclusion of articles DHWE in controls and sensitivity analysis, no individual study was found having excessive influence on the pooled effect in the above-mentioned inherited models with regard to AD. Begg’s test was used to assess the publication bias. No individual study was detected to have publication bias.
Discussion
Alzheimer’s disease is a progressive neurodegenerative disorder that begins with mild memory deficit and cognitive decline. Alzheimer’s disease could ultimately lead to severe dementia in a few years. 30 Finding from this meta-analysis indicated that x allele of rs9340799 polymorphism might have a protective effect on AD in Asia and in patients with SAD. In addition, the p allele of rs2234693 polymorphism might decrease the risk of patients with SAD. Since the gender is a relevant risk factor for developing the disease, we conducted a subgroup analysis by gender, and no associations were found both in male and in female for polymorphisms of rs9340799 and rs2234693, respectively (Supplementary Table S1).
In our meta-analysis, some analyses present moderate or high heterogeneity after excluding the studies departure from HWE. Between-study heterogeneity is common in meta-analysis for genetic association studies, 38 and exploring the potential sources of between-study heterogeneity is the essential component of meta-analysis. 39 The between-study heterogeneity might arise from an indeterminate number of characteristics that vary among studies. The possibilities related to the disease-effect diversity, such as study quality, genotyping, clinical heterogeneity (diagnosis for AD patients, allelic or locus), lifestyle factors, and so on, could not be ruled out, although no significant individual study influence on the pooled effects was observed with influence analysis. Thus, for polymorphisms of rs9340799 and rs2234693, we used meta-regression to explore the causes of heterogeneity for covariates. However, country, mean age, and sex were not found to be important sources of disease-effect heterogeneity in this meta-analysis (the details of the results are listed in Supplementary Table S2). Furthermore, the between-study heterogeneity persisted in some genetic models, especially in Asian and SAD subgroups, suggesting the presence of other unknown confounding factors. Alzheimer’s disease is a complex etiology and pathophysiology generated by the combined effects of genes and environment factors. As no covariates had a significant impact on between-study heterogeneity, we used STATA comments for “leave-one-out” sensitive analysis to reduce the between-study heterogeneity to I2 < 50%. The key contributor of the studies to this low between-study heterogeneity assessed by the “leave-one-out” sensitive analysis was also excluded, and the results are present in Table 3.
Meta-analysis that can summarize and review previously published quantitative research has been recognized as an effective method to solve a wide variety of clinical questions. To our knowledge, this is the first meta-analysis conducted to assess the association between polymorphisms of rs9340799 and rs2234693 and AD risk. Our meta-analysis also showed some advantages. First, substantial number of cases and controls were pooled from different studies, which significantly increased the statistical power of the analysis. Second, no publication biases were detected, indicating that the pooled results may be unbiased. However, there were some limitations in this meta-analysis. First, between-study heterogeneity was found in some subgroup and overall analyses in this meta-analysis, and the results were not consistent before and after sensitivity analysis for polymorphisms of rs9340799 and rs2234693 with the risk of AD. This suggested that more studies need to be performed to investigate the association between polymorphisms of rs9340799 and rs2234693 and AD risk. Second, some studies were reported appear to be specific to SAD, while most of the studies were reported overall AD. Further studies should be conducted by subgroups of AD types, such as SAD, late-onset AD, and so on.
Conclusions
This meta-analysis suggested that x allele of rs9340799 might have a protective effect on risk of AD in Asia and in patients with SAD. In addition, the p allele of rs2234693 might decrease the risk of AD for patients with SAD. Further studies are needed to confirm these results.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
