Abstract
Depression and cognitive impairment are frequent manifestations in Parkinson’s disease (PD). Although a few longitudinal studies have reported on depression and dementia in PD, there is a yet a lack of such studies in India. This 7-year longitudinal study is a hospital-based prospective case (n = 250)–control (n = 280) study. In all, 36.8% had PD with no cognitive impairment (PD-Normal), 27.2% of the patients with PD were affected by dementia (PDD), and 36% of the remaining patients with PD had mild cognitive impairment (PD-MCI) at baseline. After 7 years of evaluation, 32 new patients, 12 patients from the PD-MCI group and 9 patients from the PD-Normal group, were diagnosed with dementia. The 7-year prevalence rate for dementia was estimated to be 49.28%. In the Indian population, an early onset of dementia is noted among patients with PD, with the age of onset being less than 55 years. Patients with early-onset PDD showed depression symptoms that differed significantly from the controls of the same age-group. There was a major difference in verbal fluency, word list recall, constructional praxis and recall, word list recognition, abridged Boston Naming Test, word list memory with repetition, and Mini-Mental State Examination between PD-MCI and PDD groups. Hallucinations before baseline (odds ratio [OR] = 4.427, 95% confidence interval [CI] = 2.122-9.373), akinetic/tremor dominancy (OR = 0.380, 95%CI = 0.149-0.953), and asymmetrical disease onset (OR = 0.3285, 95%CI = 0.1576-0.685) can be considered as risk factors for patients with dementia. Patients with early-onset PD might be more prone to complex depression and dementia. As the disease progresses, akinetic-dominant PD, early hallucinations, and asymmetrical disease onset are the potential risk factors for the development of dementia in patients with PD.
Introduction
Parkinson’s disease (PD) is predominantly characterized as a neurodegenerative movement disorder; but over the past years, there has been an increasing awareness that the clinical spectrum of PD is much broader, also encompassing many nonmotor domains including cognition, 1 thus leading to impairments in verbal, visuospatial, executive, and memory functions. Cognitive impairment is a well-known symptom of Parkinson’s disease (PD), predominantly in elderly patients in later stages of the illness. Cognitive decline is a predictor of dementia in PD (PDD) 2 , and has important consequences for patient management. 3 The reported prevalence of PDD varies greatly (2%-81%) between studies. 4 Consistency between cognitive impairment/PDD and demographic or clinical characteristics has only been found for age and motor impairment. Dementia is a recognized sequela of PD, and its diagnostic criteria have been outlined, 5 but the cognitive phenotype is less clear. 6 In India, the prevalence rates of dementia over the past 2 decades ranged from approximately 1.4% among those 65 years and older 7 to 3.5% among those 60 years and older 8 in rural settings; in urban settings, the prevalence rates were reported as 2.44% among those 65 years and older. 9 Recently, in India, the crude prevalence rates of dementia in persons aged 65 and older have been reported to be as high as 10.6% for those living in rural areas and 7.5% for those in the urban areas. 10
Studies show that patients with PD having mild cognitive impairment (MCI; PD-MCI) are clinically characterized as intermediary between normal cognition and dementia. 11 However, controversies persist in the relationship between compromised cognition and motor aspects, age of onset of diseases, and type of evolution. 12 On the other hand, longitudinal studies are needed to understand the frequency of PDD since very little is known regarding the proportion of patients with PD who will eventually develop dementia in due course of the disease. Till date, only incidence rates of new patients with dementia have been reported. 13 –15 Caregivers, family members, and physicians will find this information useful.
The objective of the present study is to evaluate cognitive performance of patients with PD and correlate the results with clinical aspects. This study also highlights the 7-year prevalence of PDD using standardized diagnosis of PD, MCI, and dementia in a community-based cohort, thus comparing our findings with those of a non-PD population, thereby revealing the risk factors for developing dementia along with clinical characteristics of the patients.
Materials and Methods
Diagnosis and Motor Evaluation
This is a hospital-based longitudinal study whose study participants belonged to various age and socioeconomic groups, who were the outpatients of the neuromedicine clinic of National Neurosciences Centre, Kolkata, from December 2004 till date. After an initial screening procedure, 309 patients were interviewed and examined by eminent neurologists. In all, 24 patients did not respond to the initial invitation, thus 285 patients remained for the study. During the follow-up, 7 patients were rediagnosed as not having PD, 8 refused to participate in mental evaluation, 4 died, and 16 misdiagnosed; hence, the remaining 250 patients with PD were eligible for the final baseline study. Potential patients were sent a recruitment letter inviting them to participate in the study. Phone contact by a trained interviewer was initiated within 2 weeks after the recruitment letter was sent. Participants were given an open-ended semistructured interviewer-administrative questionnaire that collected information on disease history, from disease onset to the baseline evaluation from patients and controls in a face-to-face interview, and a neurologist examined each patient and control face-to-face clinically after obtaining written informed consent. Each interview and evaluation took about 60 to 90 minutes. All the 530 participants (250 patients with PD and 280 healthy controls) actively took part in this study, and their good response enabled a 100% participation rate. Both participants and the accompanying family members/caregivers were allowed to answer. A meet on patients with PD was organized and the participating individuals along with their family members attended the camp, and detailed follow-up visits at the outpatient department/home took place after every 3 years, thereby accumulating total of 7 years’ study from the baseline. Before the questionnaire was formally administered, a panel consisting of neurologists, epidemiologists, and a biostatistician examined content validity. Patients were classified, based on their history and baseline evaluation, as having an akinetic-dominant, tremor-dominant, or mixed tremor/akinetic clinical subtype and symmetrical or asymmetrical symptom onset and progression. In addition, information regarding drug therapy, response to levodopa, and demographic variables was collected, including whether patients had previously experienced hallucinations while taking anti-PD medication. The diagnostic evaluation was based on disease development, clinical information, and response to levodopa as elicited at the baseline interview and from the hospital records database. Patients using antipsychotic or benzodiazepine medications were not included in the study.
In order to achieve high diagnostic specificity as well as high sensitivity, patients with PD were diagnosed according to the criteria of UK Parkinson’s Disease Society Brain Bank Research,
16
and all the cases had to meet the following criteria of clinical classification of definite, probable, and possible PD at the time of diagnosis and throughout the study period: The presence of at least 3 of the following signs: resting tremor, cogwheel rigidity, bradykinesia, and postural reflex impairment, at least one of which must be either rest tremor or bradykinesia. The disease has a unilateral onset and asymmetrical development, and the response to a dopaminergic agent should be good to excellent. No suggestion of secondary parkinsonism due to drugs (such as dopamine-blocking or dopamine-depleting agents), trauma, brain tumor, or treatment within the last 12 months. No atypical features such as prominent oculomotor palsy, cerebellar signs, vocal cord paresis, severe orthostatic hypotension, pyramidal signs, amyotrophy, or limb apraxia.
During a follow-up evaluation after 3 years, a diagnostic reevaluation was conducted by the same team who had initiated the study. To avoid the inclusion of patients with other diagnoses such as dementia with Lewy bodies, only those who fulfilled the above-mentioned criteria for PD at all examinations and were without cognitive impairment at disease onset were included in our study cohort.
The control group consisted of 280 healthy community-based, age and sex-matched volunteers. None of the controls had any diagnosable neurological disorders, cognitive impairment, or neuropsychiatric disability in their family history with similar educational levels. The experiments were conducted in accordance with the Declaration of Helsinki. Ethical approval of the research project using human subject was issued from the Institutional Ethical Committee of collaborating hospitals.
Neuropsychiatric Evaluation and Clinical Diagnosis of Dementia at Baseline
The Unified Parkinson’s Disease Scale (UPDRS), 17 Hoehn and Yahr (H&Y) staging, 18 activities of daily living, abridged Boston naming test (15 items), Mini Mental State Examination (MMSE), Scale for outcome of PD-Cognition (SCOPA-COG), 19 Clinical Dementia Rating Scale (CDR), 20 word list memory with repetition, recall and recognition, and constructional praxis with copy and recall were performed for each patient to estimate the motor symptoms and magnitude of disease severity. At baseline, dementia was diagnosed by the same neurologist based on an interview with the patient and a caregiver using the Diagnostic and Statistical Manual of Mental Disorder (Third Edition, Revised; DSM-III-R) of psychiatric disease as a guide as well as the administration of cognitive rating scales such as the MMSE 21 and CDR. To be diagnosed with dementia, the patient had to meet the DSM-III-R 22 criteria for dementia at the interview and also the following criteria: MMSE score lower than the corresponding quartile based on age and education 23 and a score of 2 or higher on the UPDRS intellectual impairment item. Presence and severity of hallucinations during the last 2 weeks before the baseline evaluation were assessed using the thought disorder item of the UPDRS. Severity of depression was detected using Montgomery Asberg Depression Rating Score (MADRS). 24 Psychometrically sound PD-specific scale SCOPA-COG which has 10 items (4 items: replicating the order in which cubes were pointed out, digit span backward, immediate, and delayed word recall), attention (2 items: counting down by threes and months backward), executive functioning (3 items: successive repetitions of fist-edge-palm movements, set shifting with dices, and fluency animals) and visuospatial functioning (1 item: mental reconstruction of figures), was used to test the specific cognitive deficits in the following domains: memory and learning, attention, executive functions, visuospatial function, and memory.
Follow-Up Evaluation of Patients With PD
All the patients with PD and control participants who initially participated in the study were reinvited by letter or telephonic calls to participate in follow-up evaluations at every 3-year span from the baseline. The final evaluation was conducted from February 2013 to August 2013. Both MCI and dementia were diagnosed by a renowned neuromedicine doctor (TKB) along with his team of trained neurologists based on the clinical interview for cognitive screening and neuropsychological tests at National Neurosciences Centre, Kolkata. The results of the interview, rating scales, and neuropsychological tests had to be compatible with a diagnosis of dementia. In cases of inconsistencies between these measures, all of the available material were reviewed, and both raters made an independent diagnosis of dementia or no dementia according to DSM-III-R criteria. In cases of disagreement, a consensus diagnosis was achieved.
Data Analysis
In order to detect cognitive impairment, the group of patients with PD was divided into 3 subgroups based on the clinical neurological assessment and standardized instruments. (1) Parkinson’s disease with normal cognition (PD-NLCog), that is, patients with no apparent cognitive impairment. (2) Parkinson’s disease with mild cognitive impairment (PD-MCI), that is, corresponding to cognitive complaints coming from the patients or their families; the reporting of a relative decline in cognitive functioning during the past year by a patient or an informant; cognitive disorders as evidenced by a clinical evaluation; absence of major repercussions on daily life; and absence of dementia (≥2 standard deviations [SDs] below control groups). (3) Parkinson’s disease with dementia (PDD)—dementia according to the DSM-III-R, starting at least 1 year after the initialization of PD and without the characteristics of dementia with Lewy bodies.
To analyze the differences between patients with normal and controls, the Student’s t test for independent samples and the χ2 tests were used without adjusting for other variables. The relationship of the cognitive aspects with age, age of onset, duration, motor aspects of the PD and occurrence of depression was studied in the 3 subgroups using analysis of variance. Parametric and nonparametric statistical analyses were used according to the situation under study at a significance level of P < .05 with Yates’ correction using SPSS version 11.
Results
Baseline, clinical, and demographic data of patients with PD and control participants are summarized in Table 1. A total of 530 study participants were included. A total of 250 patients with PD with a mean age of onset of 53.1 ± 11.2 years (age range, 37–86 years; 162 males and 88 females), UPDRS score of 31.2 ± 5.20, and H&Y staging scale of 2.4 ± 1.1 and 280 controls were recruited in this study. Mean (±SD) age of the patients was 57.89 (±12.06) years and was 56.42 (±9.68) years for controls, ranging from 35 to 89 years. Most of the patients reported an increase in tremor and imbalance during periods of stress. The age (t = 1.55, P = .12) and sex (P = .96) distribution of patients and controls was similar. Family history for PD was found to be positive in 8.2% cases, in the present study. All except 11 patients with PD were treated with antiparkinsonian medication. In all, 36.8% of the patients had PD with no cognitive impairment, 27.2% had PDD and 36% of the remaining patients had PD-MCI. Patients with PD-MCI were excluded from the study on dementia, thus the remaining 160 patients with PD were recruited for the analysis of prevalence of dementia.
Baseline Demographic and Clinical Characteristics of Patients With PD and Control Participants, MADRS to Determine Depression in Different Age-Groups, and MMSE Scores to Assess Dementia.
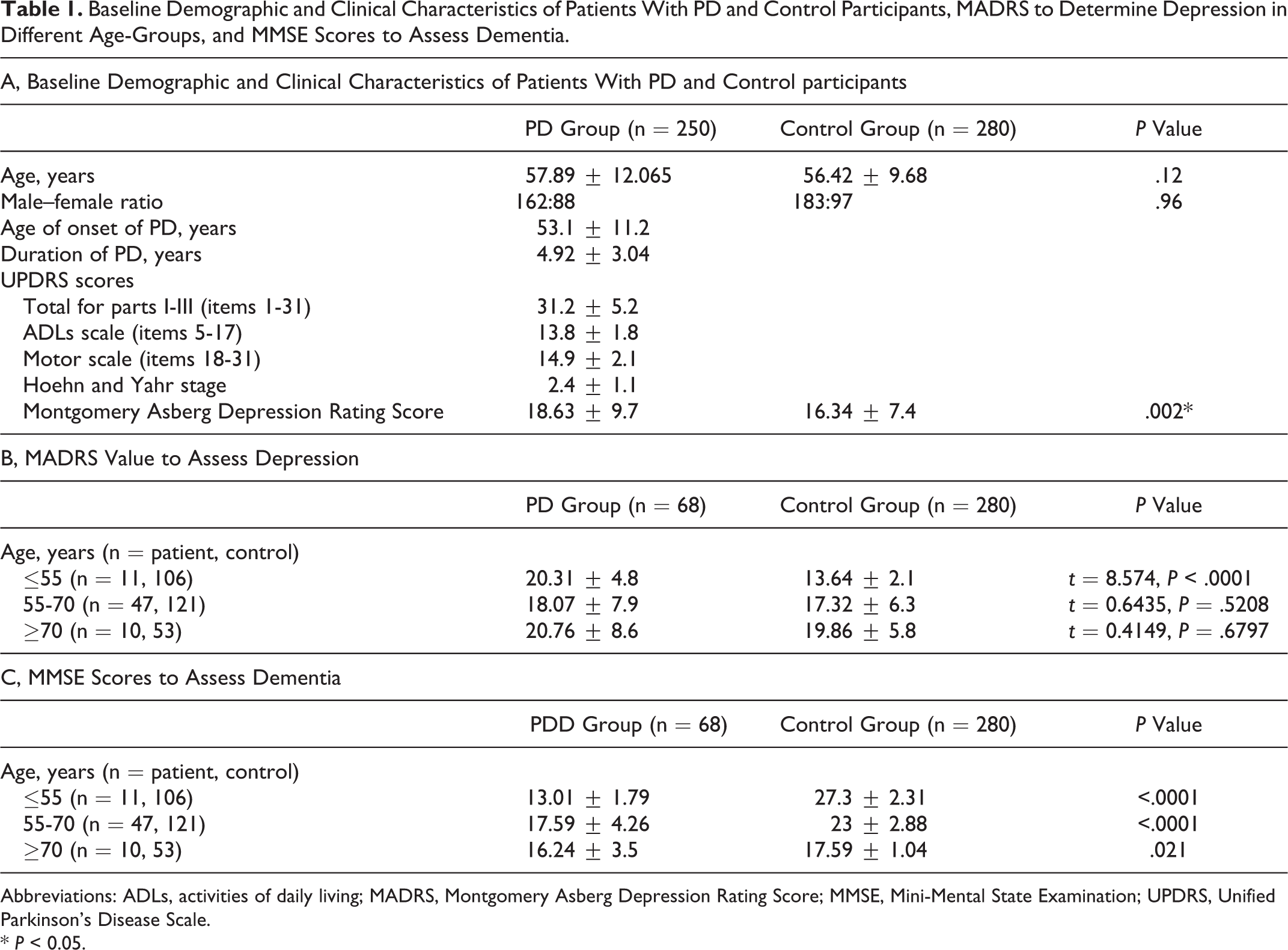


Abbreviations: ADLs, activities of daily living; MADRS, Montgomery Asberg Depression Rating Score; MMSE, Mini-Mental State Examination; UPDRS, Unified Parkinson’s Disease Scale. * P < 0.05.
A total of 241 patients had completed the 7-year longitudinal study (4 patients had address and phone numbers changed hence could not be tracked by the study team, 2 patients did not visit the clinic, and 3 patients died approximately at the end of the sixth year). Thus, a total of 9 patients with PD (3.6%) dropped out without dementia being diagnosed. In all, 68 (27.2%) patients with PD were diagnosed as having dementia at baseline; and 32 new cases, 12 patients from PD-MCI group and 9 patients from the PD-Normal group, were diagnosed with dementia after 7 years of evaluation. Thus, a total of 121 patients with dementia were identified at the end of 7 years. In no cases were the patients with dementia later diagnosed as not having dementia. Reversible dementia was not observed. The average population in the middle of the observation period was (250 + 241)/2, so the 7-year prevalence rate was calculated to be 49.28%. Hallucinations before baseline (odds ratio [OR] = 4.427, 95% confidence interval [CI] = 2.122-9.373), akinetic/tremor dominancy (OR = 0.380, 95% CI = 0.149-0.953), and side of disease onset (symmetrical/asymmetrical; OR = 0.328, 95%CI = 0.156-0.685) were determinant risk factors for patients with dementia.
Direct multiple comparisons between the PD subgroups and the control groups (CGs) were subsequently made for the data showing significant differences (Table 2). Chi-square and Mann-Whitney U test show no statistically significant difference between the PD-NLCoG subgroup and CG, with respect to word list recall, constructional praxis and recall, and word list recognition (Table 2). A comparison was made between patients with PDD and without dementia, and higher values in H&Y scale were observed in the PDD group (Table 2) having the disease for a greater duration.
Sociodemographic, Clinical, and Cognitive Aspects at Baseline in the Total Patients With Parkinson’s Disease (PD) and in the Parkinson Subgroups With no Cognitive Alterations (PD-NLCog), With Mild Cognitive Impairment (PD-MCI), With Dementia (PD-Dem), and Control in Means (SD), as Also the Results of Comparisons (PD × CG) and Post Hoc Pairwise Multiple Comparisons.
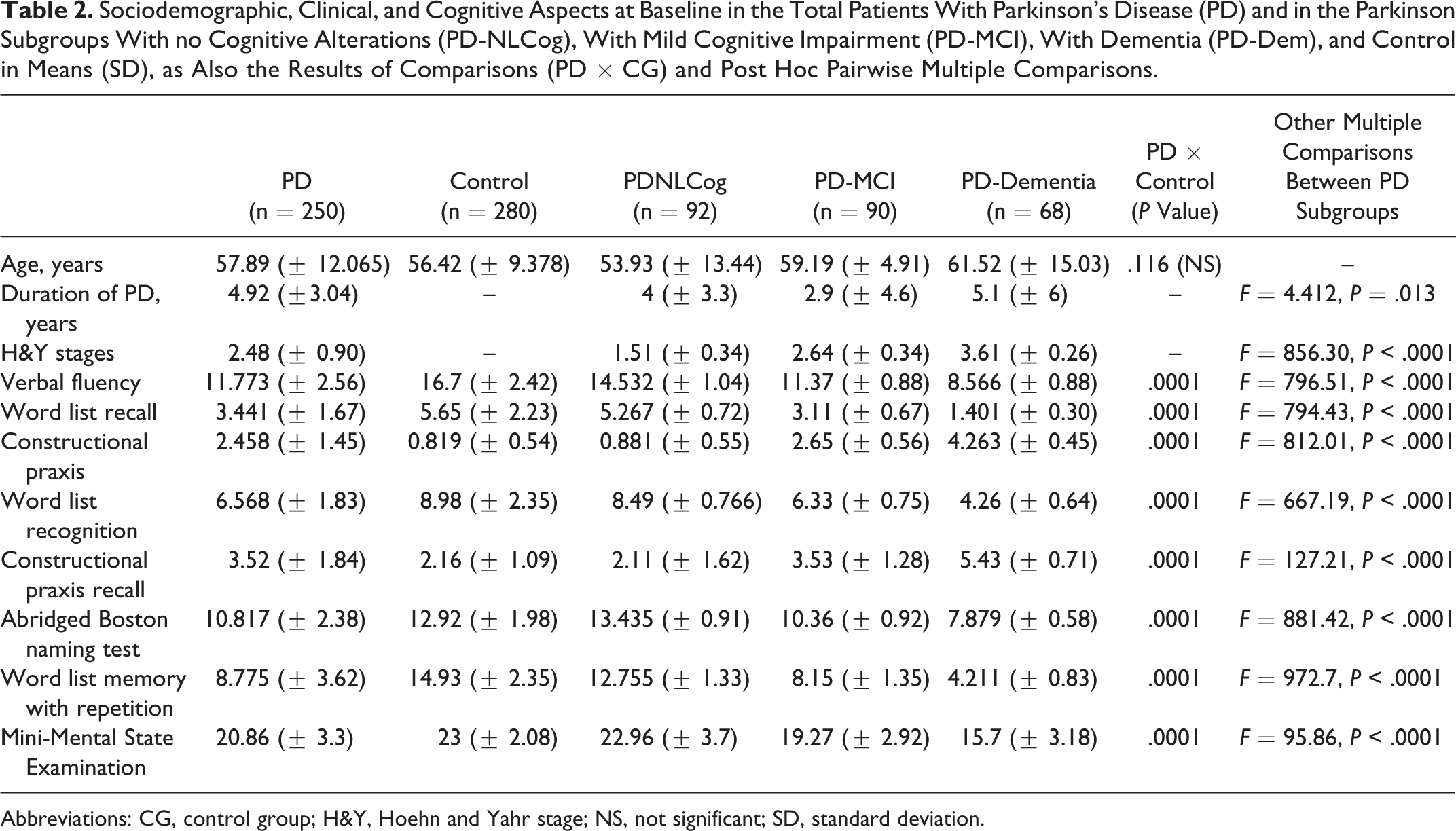
Abbreviations: CG, control group; H&Y, Hoehn and Yahr stage; NS, not significant; SD, standard deviation.
There was a significant difference between PD-MCI and PDD groups in verbal fluency, word list recall, constructional praxis and recall, word list recognition, abridged Boston Naming Test, word list memory with repetition, and MMSE (Table 2). Patients with PDD demonstrated high depression when compared with the controls (Table 1B). In the Indian population, early dementia among patients with PD is significantly noted, with age of onset being less than 55 years (Table 1, panel C). Patients with early-onset PDD showed depression symptoms, which differed significantly from the controls of the same age-group (Table 1, panel B).
Discussion
The present study participants comprised patients with idiopathic PD, whose mean age is 57 years. We found that 49.28% of a population-based and representative cohort of patients with PD developed dementia during the 7-year of the study period. Patients depicting hallucinations prior to the baseline evaluation were at higher risk of developing dementia than those with tremor-dominant parkinsonism. Thus, our finding is consistent with those of the previous longitudinal studies 3,15 ; although considerably lower results have been reported 13,14,25 in studies using less rigorous methods. The pathophysiology of dementia in PD appears to be of a multifactorial nature, encompassing subcortical and cortical neuronal populations. Our observation reveals that the patients with dementia having PD who had an early onset of symptoms (age < 55 years) showed higher MADRS scores than that of the controls (t = 8.574, P < .0001). There has been a significant difference in the scores among patients with PD having dementia when depression sets in much early and disease duration is greater (Table 1, panel B). In the present study, patients with PD having dementia presented a functional decline and worse clinical condition in the H&Yscale, which is more marked than those without dementia (Table 2).
A strong association was found between the early occurrence of dementia and greater gravity of motor compromise in patients with PD compared to controls (Table 1, panel C), as assessed by the H&Y stages, and this finding is similar to the studies previously reported from the Brazilian population. 26 Thus, compared to cognitively preserved individuals, the MMSE ratings differed significantly among patients who had PD much early in their life, thereby dementia setting in among individuals having PD for a longer duration. These individuals were found to have a more advanced clinical stage of dementia and had greater functional impairment. Interestingly, of the 13 patients with PD, 9 with dementia had early-onset PD with greater severity of the disease (Table 3). Of 96 patients with PD, 72 (75%; age range: 55-65 years) had dementia, whereas only 12 (32%) individuals belonged to this same age-group of 37 in the “no dementia” subsection; thus, it can be concluded that dementia sets in comparatively early in patients with PD within the same age-group with advanced disease stages.
Characteristics of Patients With PD Who Did and Did Not Develop Dementia.

Abbreviations: F, female; M, male; PD, Parkinson’s disease; χ2, chi-square. * P < 0.05.
The following background, however, should be taken into account during the interpretation of the findings of this study. First, this is a hospital clinic-based study with a selection procedure based on age of onset and disease duration. Therefore, the results of this study cannot be generalized to the population with PD in general. The percentage of patients with impaired cognition in this study cannot be interpreted as a prevalence estimate, which limits the possibility to compare our findings with the prevalence rates in other studies.
The patient’s ability to perform his or her daily activities independently is a predicting factor for quality of life in PD, where the limitations associated with PD may be further aggravated by a cognitive impairment. 4 This combination is likely to increase the family burden, which may result in early institutionalization of the patient. 2 Literature survey reports very few studies of MCI among PD patients. Tedrus et al. 26 too have done similar study and found that 31% of their PD patients had MCI which is quite close to our data (36% PD patients in our cohort are having MCI). Few longitudinal studies have been carried out, but according to Janvin et al, 11 the presence of MCI in PD, as also in the population at large, is associated with a greater risk of developing dementia. In our study, 12 patients who had PD-MCI eventually developed dementia at the end of the seventh year. Thus, clinical identification and evaluation of MCI among patients with PD are necessary since such patients are prone to dementia in the long run.
Inferior results in the semantic verbal fluency in patients in the PDD and PD-MCI subgroups as compared to those in the PD-NLCog or CG groups suggest involvement of specific cognitive domains such as executive and memory functions similar to other reports found in the literature. 27,28
Motor disorder (failure in the planning and sequential control of their movements), a major factor for patients with PD, has been depicted by the inferior performance in the praxis trials, which is similar to the study conducted among Brazilian patients. 26 This finding could suggest that the visual–constructive abilities of the patients assessed in this study could have been affected to a certain extent by an executive disorder. 29 The memory difficulties expressed in their recall deficit of the word list in this study could reflect the involvement of multiple cortical and subcortical neural pathways and not temporal–limbic involvement 4 caused by a slowing up of the information process or by impairment of evocative strategies more than a mnemic deficit as such. These mechanisms also contribute to the difficulties shown in the word list memory with repetition task, and this finding is similar to that of Tedrus et al. 26
The major methodological limitation of our study was the extended period of assessments, and thus a degree of attrition in this group of elderly individuals was anticipated. Dementia might reduce the survival in patients with PD, and in some cases, patients developed dementia between the last evaluation and death, indicating that the prevalence observed is a conservative estimate. The primary population comprised a cross-sectional small sample of patients with PD, with a mean duration of disease baseline of nearly 6 years, where the patient selection bias is hospital based. An optimal design would be to include successive recently diagnosed patients with PD and those suffering for long period and assess them annually.
The study is part of the contour PD study for the past 7 years, with a progressive longitudinal cohort study of patients with PD who are extensively profiled on phenotype, disability, and global outcomes of health with different assessment instruments/scales that have been found to be valid and reliable in PD. This 7-year study was divided into 3 interstudies: data taken from the baseline, follow-up study after 3 years, and the final evaluation after 3 years of the second follow-up; the total data have been summarized as a 7-year continuous study. The strength of this study includes moderate representative individuals from the same community-based PD population, standardized diagnostic criteria for the diagnosis of PD, dementia and cognitive impairment, comprehensive evaluation battery, independent testing of motor and mental symptoms, inclusion of large control groups, a detailed follow-up period, and comparatively low attrition rate. Other previous studies have used smaller and lesser representative samples and a shorter follow-up time. 12,13 We were particularly careful, however, to classify meticulously and include in our analyses patients who were cognitively impaired and demented, making it less likely that another disease with parkinsonian features explained the associations.
All the 530 individuals were residents of India and were assessed by the same team of physicians belonging to the core research PD group; therefore, diagnostic procedures to identify dementia and cognition were consistent, whereas in contrast a study by Aarsland et al 3 had patients recruited from different countries, thus there are chances of misdiagnosis in some cases. The control group (non-PD individuals) was not based on self-report; the data were reported by trained neuroresearch associates. Thus, we can eradicate the fact that there was no underestimation or overestimation of the risk of dementia in this group. This is one of the first studies from India stretched over such a period of time to follow-up patients with PD and report the dementia rates.
In summary, it is found in the present study that cognitive compromise that appeared in our patients with PD is not only expressed in dementia but also in MCI. The characterization of cognitive involvement and its gravity in PD is of great clinical importance, since it can be of use in better orientation of the patients and the introduction of therapeutic strategies. Indian patients with PD with early age of onset might be more prone to develop depression and dementia. Akinetic-dominant PD, early hallucinations, and asymmetrical disease onset are potential risk factors for the development of dementia in patients with PD as the disease progresses.
Footnotes
Acknowledgment
This research was supported by a grant from Anthropological Survey of India, Ministry of Culture, Government of India. We are thankful to the team of doctors, patients with PD and control subjects for voluntarily taking part in this research work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
