Abstract
The objective of this review is to summarize the available data on the use of benzodiazepines for the treatment of behavioral and psychological symptoms of dementia (BPSD) from randomized controlled trials (RCTs). A systematic search of 5 major databases, PubMed, MEDLINE, PsychINFO, EMBASE, and Cochrane Collaboration, yielded a total of 5 RCTs. One study compared diazepam to thioridazine, 1 trial compared oxazepam to haloperidol and diphenhydramine, 1 trial compared alprazolam to lorazepam, 1 trial compared lorazepam to haloperidol, and 1 trial compared intramuscular (IM) lorazepam to IM olanzapine and placebo. The data indicates that in 4 of the 5 studies, there was no significant difference in efficacy between the active drugs to treat the symptoms of BPSD. One study indicated that thioridazine may have better efficacy than diazepam for treating symptoms of BPSD. In 1 study, the active drugs had greater efficacy in treating BPSD when compared to placebo. There was no significant difference between the active drugs in terms of tolerability. However, in 2 of the 5 studies, about a third of the patients were noted to have dropped out of the studies. Available data, although limited, do not support the routine use of benzodiazepines for the treatment of BPSD. But these drugs may be used in certain circumstances where other psychotropic medications are unsafe for use in individuals with BPSD or when there are significant medication allergies or tolerability issues with certain classes of psychotropic medications.
Keywords
Introduction
Benzodiazepines are often used in older adults for the treatment of insomnia, depression, and anxiety. 1 –4 Current data indicate that between 15% and 30% of people older than 65 years of age use benzodiazepines. Although the evidence for using benzodiazepines is limited to a few weeks for the treatment of anxiety and sleep disturbances, they are often used for significantly longer periods of time. 5 –10
Behavioral and psychological symptoms of dementia (BPSD) can be defined as a heterogeneous range of psychological reactions, psychiatric symptoms, and behaviors that are unsafe, disruptive, and impairs the care of the patient in a given environment. 11 BPSD occurs in about a third of community-dwelling people with dementia and its prevalence increases to almost 80% in skilled nursing facilities. 12,13 Available data indicate that BPSD is associated with increased caregiver burden, depression, greater cognitive and functional decline, and greater risk of institutionalization. 14 –17 The BPSD also results in lower quality of life for these individuals and leads to higher social and economic burden of caring for patients with dementia after adjusting for all other comorbidities. 18
Although treatments for BPSD are not standardized, there is some data that both nonpharmacological and pharmacological interventions have modest efficacy for the management of these symptoms and behaviors. 19,20 Pharmacotherapy is often used in combination with non-pharmacological treatments, especially for those behaviors that are refractory to management with non-pharmacological approaches. 21 Along with antipsychotic medications, benzodiazepines are often prescribed in nursing homes to treat behavioral symptoms of dementia despite lack of an appropriate indication. 22 Although benzodiazepines are often used to treat behavioral symptoms in older adults with dementia, the efficacy and tolerability of this medication class has not been systematically studied in these individuals.
The 2012 American Geriatrics Society (AGS) Beers Criteria includes 53 medications or medication classes that are divided into 3 categories: potentially inappropriate medications and classes to be avoided in older adults, potentially inappropriate medications and classes to avoid in older adults with certain diseases and syndromes that the drugs listed can exacerbate, and the medications to be used with caution in older adults. 23 The AGS-Beers criteria includes benzodiazepines in the potentially inappropriate medications class and recommends that there is strong evidence for avoiding their use in older adults, especially for the treatment of insomnia, agitation or delirium. They are also included in the potentially inappropriate medications and classes to avoid in older adults with certain diseases and syndromes that the drugs listed can exacerbate including cognitive impairment and dementia.
In this article, we systematically review and summarize the data for the use of benzodiazepines in the treatment of BPSD from randomized controlled double-blind trials.
Search Strategy
The primary aim of this review is to summarize the data from randomized controlled double-blind trials regarding the use of benzodiazepines for the treatment of BPSD.
We performed a literature search of 5 major databases: PubMed, MEDLINE, PsychINFO, EMBASE, and Cochrane Collaboration. Search terms were “Dementias” and “Benzodiazepines” with “randomized controlled trial (RCT)” as a filter in each case. The databases were searched through December 31, 2013. There was no language limit set in order to be as inclusive as possible with the search strategy.
Each author independently searched the 5 databases for studies with the search terms. Abstracts of all the studies that were noted on the initial screening were retrieved for further evaluation. Studies were selected for full-text review if they involved subjects with dementia, involved any behavioral or psychological symptom, were treatment studies, and had a randomized controlled double-blind trial design. For this review, BPSD was operationalized as any behavioral or psychological symptom that was exhibited by an individual with dementia and thought to be severe enough to need treatment with a psychopharmacological agent. Studies were excluded if they did not primarily include subjects with dementia or did not involve the treatment of BPSD. Database reviews, population-based reviews, systematic reviews, and meta-analysis were also excluded but the data available from these studies were to be used as comparison to the data available from this review. Disagreements between the 2 authors regarding study selection for this review were resolved through consensus.
Results
PubMed generated a total of 67 articles of which 3 are included in this review. MEDLINE generated 37 articles, 2 of which are included in the 3 selected from the PubMed search and 1 is an additional article. The search in PsychINFO yielded no articles. The EMBASE search found 112 articles and the Cochrane database review yielded 4 articles. None of these articles could be included in this review. One additional article was retrieved from the bibliographic search of the relevant articles.
We found a total of 5 randomized controlled double-blinded clinical trials using benzodiazepines in patients with dementia. All 5 trials were for the treatment of BPSD. Of these, 1 trial compared diazepam to thioridazine, 1 trial compared oxazepam to haloperidol and diphenhydramine, 1 trial compared alprazolam to lorazepam, 1 trial compared lorazepam to haloperidol and 1 trial compared intramuscular (IM) lorazepam to IM olanzapine and placebo.
To aid with the evaluation of data, we organized the studies in a chronological manner with the oldest study being the first to the most recent study being the last. The quality of data was evaluated using the criteria developed by the Centre for Evidence Based Medicine (CEBM) for RCT evaluation (Table 1). 24
Quality of Studies Reviewed.
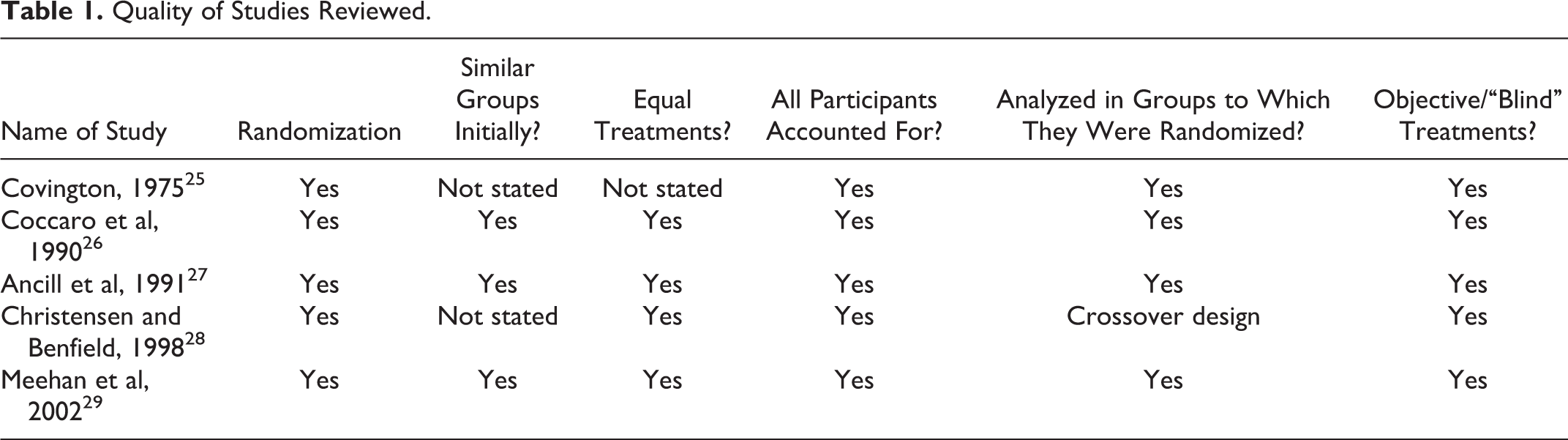
In the study by Covington, 59 older adults with dementia (diagnostic criteria not stated) and behavioral symptoms were randomized to receive either thioridizine to 200 mg a day or diazepam to 40 mg a day.
25
Patients had not been receiving any psychotropic medication for at least 3 days prior to the entry into the study. Evaluations were done weekly by the study physician and nursing staff who were blinded to the medications the patients were receiving. Evaluations included the Hamilton Anxiety Rating Scale (HARS) and the Nurses Observation Scale for Inpatient Evaluation (NOSIE). The study physician and the nurses also made weekly assessments on a subjective scale where 2 questions were answered: how mentally ill is the patient and how much have they changed from admission to the study? The anchor points for the first question were 1 = normal through 4 = moderately ill to 7 = extremely ill. On the second question, the anchor points were 1 = very much improved through 4 = no change to 7 = very much worse. Data from 19 of the original 59 patients were not included in the final analysis as the patients had been hospitalized, transferred to another institution, or died due to reasons unrelated to the drug administration. Among the 40 remaining patients, there were 20 each in the thioridazine and the diazepam groups. When compared to baseline, at the end of 4 weeks, the thioridazine group had statistically significant reduction in 4 of the 8 HARS items (
In the study by Coccaro et al, the investigators included 59 individuals who were ≥55 years of age with a
In the study by Ancill et al, 40 patients older than 65 years of age who met the
In the study by Christensen and Benfield, patients were recruited from 25 nursing homes.
28
The eligible patients were ≥65 years of age, had a single established mental disorder of organic mental syndromes (
In the study by Meehan et al, the investigators recruited clinically agitated inpatients, hospitalized, or nursing home residents who were ≥55 years and met either the National Institute of Neurological and Communicative Disorders and Stroke and Alzheimer’s Disease and Related Disorders Association (NINCDS-ADRDA) or
Summary of Studies.
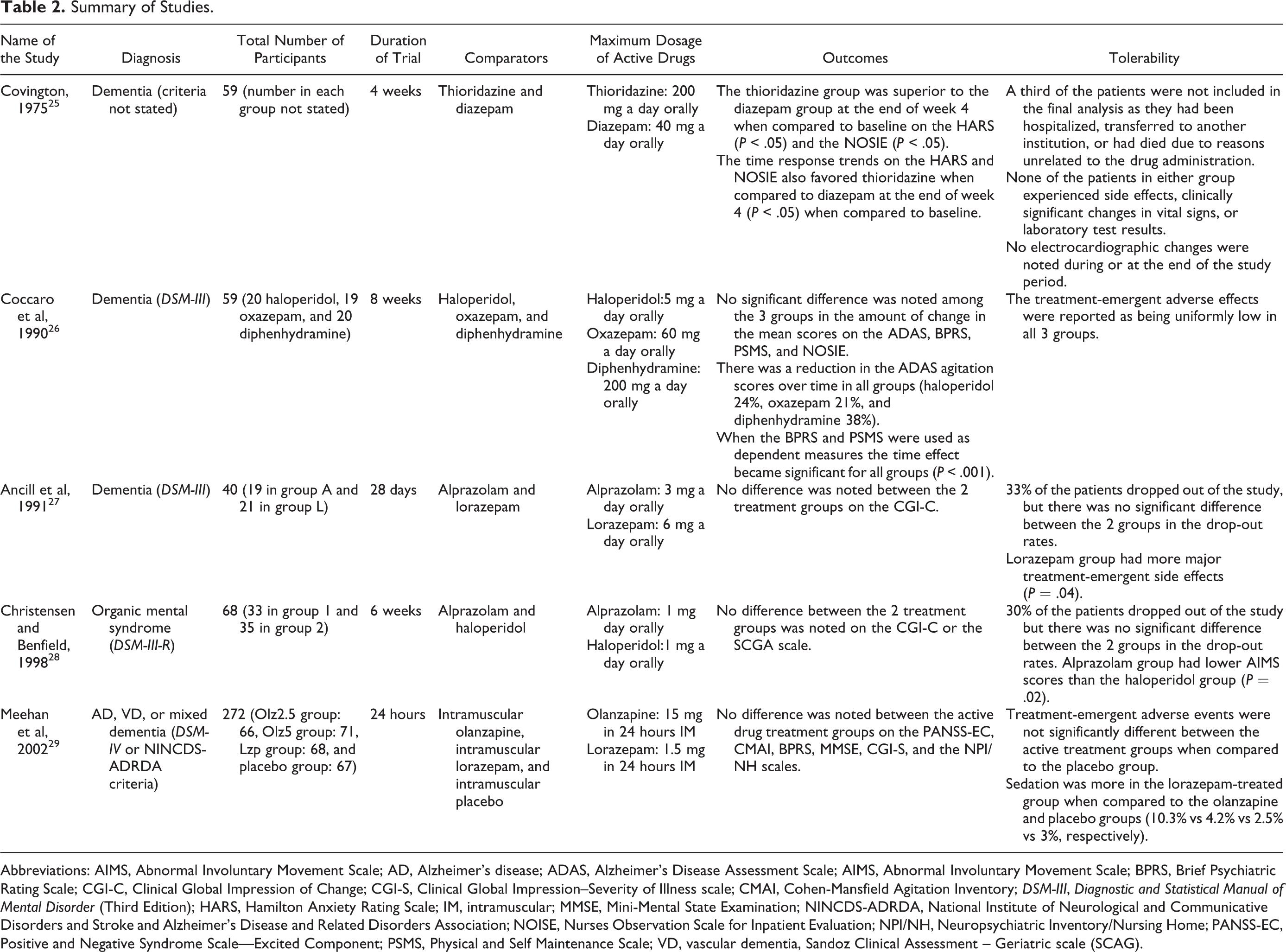
Abbreviations: AIMS, Abnormal Involuntary Movement Scale; AD, Alzheimer’s disease; ADAS, Alzheimer’s Disease Assessment Scale; AIMS, Abnormal Involuntary Movement Scale; BPRS, Brief Psychiatric Rating Scale; CGI-C, Clinical Global Impression of Change; CGI-S, Clinical Global Impression–Severity of Illness scale; CMAI, Cohen-Mansfield Agitation Inventory;
Discussion
This review indicates that there is a dearth of controlled data on the use of benzodiazepines for the treatment of BPSD. Among the 5 studies that we obtained using the search strategy, there was significant heterogeneity. The studies by Covington, Coccaro et al, Ancill et al, and Christensen and Benfield targeted BPSD over 4, 8, 4, and 6 weeks, respectively.
25
–28
The study by Covington included patients with dementia but the diagnostic classification system that was used to make the diagnosis of dementia was not stated.
25
The Coccaro et al and Ancill et al studies used the
Although it is difficult to draw any definitive conclusions from the data obtained from this review, given the limited number of controlled studies, and the significant heterogeneity between the studies, there is some information that will be useful to clinicians taking care of individuals with BPSD. Available data indicate that in the majority of the studies, there was no significant difference in efficacy between the active drugs used in these studies to treat the symptoms of BPSD. The study by Covington indicated that thioridazine was superior to diazepam in patients with BPSD. 25 In the study by Meehan et al, the active drugs had greater efficacy in treating behavioral symptoms when compared to placebo. 29 Overall, there was also no significant difference between the active drugs in terms of treatment-emergent adverse events. In the study by Covington, there were no treatment-emergent adverse effects reported but the data on about a third of the patients were not considered in the final analysis. 25 The investigator indicated that the data from the patients were not included in the final analysis as they had either been hospitalized, transferred to another institution, or had died due to reasons unrelated to the drug administration. In the study by Coccaro et al, the treatment-emergent adverse were reported as being uniformly low and did not differ between the 3 drugs under study. 26 The overall drop-out rate was about 13% in this study. About a third of the patients dropped out in the studies by Ancill et al and Christensen and Benfield. 27,28 In the Ancill et al’s study, lorazepam group had a greater number of major treatment-emergent side effects when compared to alprazolam and this difference was statistically significant. 27 In the study by Christensen and Benfield, the haloperidol-treated group had greater AIMS overall severity scores when compared to the alprazolam group, and this result was statistically significant. 28 In the Meehan et al’s study, the lorazepam group had greater sedation when compared to the olanzapine and placebo groups but it was not statistically significant. 29
The quality of data evaluated using the criteria developed by the CEBM for RCT evaluation indicated that the majority of the trials included in this review were well conducted.
24
However, these trials had has some limitations. They were short-term trials that lasted between 24 hours and 8 weeks. The study by Christensen and Benfield may have also included some individuals who had delirium as the diagnostic inclusion criteria was that of Organic Mental Syndrome according to the
The maximum doses of the drugs used orally in these studies were alprazolam 1 mg and 3 mg a day, diazepam 40 mg a day, lorazepam 6 mg a day, oxazepam 60 mg a day, diphenhydramine 200 mg a day, haloperidol 1 mg and 5 mg a day, and thioridazine 200 mg a day. 25 –28 These drugs were prescribed in 1 to 3 times a day dosing. The IM doses of lorazepam and olanzapine were 1.5 and 15 mg a day divided in 3 injections. 29 The APA practice guideline for AD and other dementias indicates lorazepam doses to be from 0.5 to 1.0 mg every 4 to 6 hours. 30 The standing oral doses range from 0.5 to 1.0 mg and may be given from 1 to 4 times per day. Standing doses of oxazepam vary from 7.5 to 15.0 mg and may be given 1 to 4 times per day. The dosages of haloperidol, diazepam, and thioridazine used in these studies and the prescribing patterns are typical of what would be used in older adults with dementia. 21,31 Thioridazine is not currently used in clinical practice, especially for older adults given the risk of QTc prolongation and Torsades. 32 Also, diphenhydramine is not recommended for use in older adults given its anticholinergic properties and adverse effect profile. 23,33
Our searches did not yield any database reviews, population-based reviews, systematic reviews, or meta-analysis for the use of benzodiazepines for the treatment of BPSD. However, we did find a meta-analysis for the use of benzodiazepines for delirium, which indicated that there are no adequately controlled trials that was found to support the use of benzodiazepines in the treatment of nonalcohol withdrawal-related delirium among hospitalized individuals and that at this time benzodiazepines cannot be recommended for the control of delirium. 34 A meta-analysis of benzodiazepines for the treatment of insomnia, which included data from 45 RCTs of which 15 studies included patients ≥65 years and 4 studies involved exclusively older patients, the investigators found that the use of benzodiazepines results in a decrease in sleep latency and a significant increase in total sleep time when compared to placebo. However, there was also significantly increased side effects, especially daytime sedation, dizziness, and lightheadedness, but this did not result in increased discontinuation rates. There was no dose–response relationship for the beneficial effect seen with the use of benzodiazepines. 35 The data from the current review could be positioned between the results of these 2 meta-analyses. The controlled data evaluating the use of benzodiazepines for the treatment of BPSD is greater than what is noted for the treatment of delirium but significantly less than what is available for the use of benzodiazepines for the treatment of insomnia.
As noted earlier in this review, the AGS-Beers criteria caution the use of benzodiazepines in older adults, especially among those with cognitive impairment for the treatment of insomnia, agitation, or delirium. 23 The APA practice guideline indicates that benzodiazepines may have a higher likelihood of side effects and a lower likelihood of benefit than antipsychotics but they are occasionally useful in treating agitation in certain patients with dementia particularly those in whom anxiety is prominent . 30 The guideline also cautions the long-term use of these drugs and states that they may be useful on an occasional as-needed basis for individuals who have only rare episodes of agitation or those who need to be sedated. The guideline also recommends using caution when dosing these drugs and keeping the doses to a minimum. The data from this review do not provide additional information so as to contradict the recommendations of the AGS-Beers criteria or the APA guideline.
The use of benzodiazepines is associated with significant adverse effects, especially in older individuals. 36 Common adverse effects include drowsiness, lethargy, and fatigue. 37 These medications are also associated with impaired motor coordination, dizziness, vertigo, slurred speech, blurry vision, mood swings, and hostile or erratic behaviors. 37 They are associated with increased risk of falls and fracture. 38,39 Benzodiazepines are also implicated in the development of cognitive dysfunction and increasing the risk of dementia. 40,41 Benzodiazepines may cause severe adverse effects when coadministered with certain medications like the opioids. 37 This combination results in cardiovascular and hemodynamic instability. Respiratory depressant effects of opioids become exaggerated in individuals with chronic obstructive pulmonary disease. 37 Benzodiazepines are also associated with the development of tolerance, dependence, and withdrawal symptoms causing significant care issues in older adults. 37 A recent Canadian study found that there was a significant association between benzodiazepine-related drug interactions and health care costs among elderly adults. 42 One study indicated that the use of benzodiazepines was associated with an increased risk of death when compared to users of atypical antipsychotics (rate ratio 1.28, 95% confidence interval 1.04-1.58) in nursing homes. 43
Comparing the data available from this systematic review with controlled data for other psychotropic agents for treating BPSD indicates that none of these medication classes have sustained benefit for the treatment of BPSD and they have significant adverse effects. Antipsychotic medications have the largest body of data for the treatment of BPSD but they have limited efficacy and significant adverse effects including the risk of cerebrovascular events and death. 44 The use of anticonvulsants is limited by their lack of benefit and significant adverse effects. 45,46 The use of antidepressants for the treatment of BPSD indicates limited benefit although they have a fairly benign adverse effect profile. 47,48 The role of cholinesterase inhibitors and memantine for the treatment of BPSD still remains unclear. 49,50
Given this limited data, is there any place for the use of benzodiazepines in the treatment of BPSD? Although available data do not support the routine use of benzodiazepines for the treatment of BPSD, these medications may be used in certain circumstances. These include situations where other psychotropic medications are unsafe to use in individuals with BPSD, for example, hepatic failure, severe Parkinson’s disease, Dementia with Lewy bodies, status postmyocardial infarction, severe cardiac arrhythmias, or when there are co-occurring disorders, that is, alcohol or benzodiazepine withdrawal. 51 –56 Benzodiazepine use may also be justified when individuals with BPSD have significant medication allergies or tolerability issues with certain classes of psychotropic medications. 57 –59
The APA practice guideline indicates that when using benzodiazepines in individuals with cognitive impairment , agents such as oxazepam and lorazepam that do not require oxidative metabolism in the liver and have no active metabolites are the preferred agents. 30 Oral lorazepam or IM doses in emergencies may be given on an as-needed basis. The guideline also states that agents with long half-lives, that is, clonazepam and those with long-lived metabolites, that is, diazepam, chlordiazepoxide, clorazepate, and flurazepam, which can take weeks to reach steady state levels, especially in elderly patients, should not generally be used in these individuals.
When prescribing benzodiazepines to individuals with BPSD, the goal should be to optimize their use whereby the clinical benefit is maximized while minimizing the adverse effects. A good formulation of the patient’s behaviors, the use of appropriate and standardized scales to qualify and quantify these behaviors and combining non-pharmacological and pharmacological treatments methods will increase the chance of success in treating these behaviors. 60
Conclusions
This systematic review indicates that the evidence for using benzodiazepines for the treatment of BPSD is limited. Available data are hindered by a few controlled studies with significant heterogeneity in their study designs and a small number of participants and being of short duration. Although fairly well tolerated in these studies, benzodiazepines have the potential to cause significant serious adverse effects, especially in older individuals. Given the available information, there is inadequate data to recommend the routine use of benzodiazepines for the treatment of BPSD. These medications should only be used in individuals with BPSD in exceptional circumstances with close monitoring of their doses and adverse effects.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
