Abstract
Characteristic frontotemporal abnormalities on structural or functional neuroimaging are mandatory for a diagnosis of probable behavioral variant of frontotemporal dementia (bvFTD) according to the new criteria. 18-Fluorodeoxyglucose-positron emission tomography (18F-FDG-PET) imaging is commonly reserved for patients with suspected bvFTD without characteristic structural neuroimaging results. We studied the diagnostic value of 18F-FDG-PET in these patients.
Methods:
The 18F-FDG-PET was performed in 52 patients with suspected bvFTD but lacking characteristic structural neuroimaging results. The clinical diagnosis of bvFTD in the presence of functional decline (bvFTD/fd+) after a follow-up period of 2 years was used as a golden standard.
Results:
The sensitivity of 18F-FDG-PET for bvFTD/fd+ was 47% at a specificity of 92%. The differential diagnosis comprised alternative neurodegenerative and psychiatric disorders and a benign phenocopy of bvFTD.
Conclusions:
The 18F-FDG-PET is able to identify nearly half of the patients with bvFTD who remain undetected by magnetic resonance imaging. In our selected group, high specificity enables exclusion of psychiatric and other neurodegenerative disorders.
Keywords
Introduction
The diagnosis of the behavioral variant of frontotemporal dementia (bvFTD) remains challenging. The until recently used international consensus diagnostic criteria for bvFTD included 5 clinical core criteria and a number of exclusion criteria. 1 Their sensitivity ranged from 36.5% to 85%. 2 –6 On the other hand, as frontotemporal abnormalities on neuroimaging were not required for a bvFTD diagnosis, psychiatric disorders could be easily misdiagnosed as bvFTD according to these criteria. 7 –10 Alzheimer’s disease (AD) is another common misdiagnosis for bvFTD. 11 –13
In view of these diagnostic problems, an international consortium, Frontotemporal Dementia Consensus Criteria (FTDC) recently revised the diagnostic criteria for bvFTD.
14
The presence of at least 3 of the 6 clinical criteria allows for a diagnosis of possible bvFTD, whereas probable bvFTD is diagnosed in the presence of possible bvFTD together with both functional decline and frontal and/or temporal abnormalities on magnetic resonance imaging (MRI), fluorodeoxyglucose-positron emission tomography (FDG-PET), or
Although the new criteria promote a more accurate diagnosis of bvFTD, a matter of concern remains that a substantial proportion of patients with bvFTD display no abnormalities in structural neuroimaging. 2,15 –17 In these cases, functional neuroimaging is generally performed as its reported general sensitivity is around 90%. 9,18 The specificity of functional neuroimaging for bvFTD is questionable as frontal abnormalities have been described in patients with both depression and schizophrenia. 19 –21 Only 1 study examined the direct relationship between structural imaging and 18F-FDG-PET in bvFTD and found that 18F-FDG-PET was abnormal in only 2 (22%) of the 9 patients with normal structural neuroimaging. 22 We retrospectively investigated the diagnostic value of 18F-FDG-PET in a larger cohort of patients with suspected bvFTD but lacking distinctive structural neuroimaging features.
Methods
Patients were recruited from the VU University Medical Center Alzheimer Center, Amsterdam, the Netherlands, which is a tertiary memory and behavior clinic with high interest and expertise in early-onset dementia.
All patients received a 1-day program of investigations including history taking, a physical, neurological, and neurobehavioral examination including the Clinical Dementia Rating Scale (CDR), Mini-Mental State Examination (MMSE), Frontal Assessment Battery (FAB), neuropsychological assessment, MRI or computed tomography (CT), cerebrospinal fluid investigation, and electroencephalogram. Results were discussed in a multidisciplinary meeting in the presence of a behavioral neurologist, psychiatrist, and neuroradiologist where a clinical diagnosis was established according to their respective diagnostic criteria. 1,23 –26 When results of standard investigations were insufficient to reach a clinical diagnosis, additional diagnostics, for example, psychiatric evaluation or 18F-FDG-PET were performed when appropriate. All patients were kept in regular follow-up.
Cases were drawn from our database of cerebral 18F-FDG-PET scans performed between January 2005 and February 2008, based on a reported suspicion of bvFTD. We checked the absence of characteristic frontotemporal abnormalities on MRI. Moreover, an atrophy pattern typical for any neurodegenerative disorder or gross white matter abnormalities had to be absent based on visual neuroradiological assessment. 27 Finally, a minimal clinical follow-up duration of 2 years after the initial 18F-FDG-PET was required for inclusion. Clinical diagnoses at ≥2 years of follow-up were used for the analysis. A flowchart describing the inclusion process is displayed in Figure 1. Of the 228 patients who had undergone 18F-FDG-PET, 52 fulfilled the inclusion criteria. Structural neuroimaging in these cases consisted of MRI in 51 of 52 cases. There were 29 normal MRIs and 22 MRI scans with nonspecific abnormalities such as minor white matter abnormalities, mild medial temporal atrophy, or mild global atrophy. One patient had a normal CT scan of the brain.
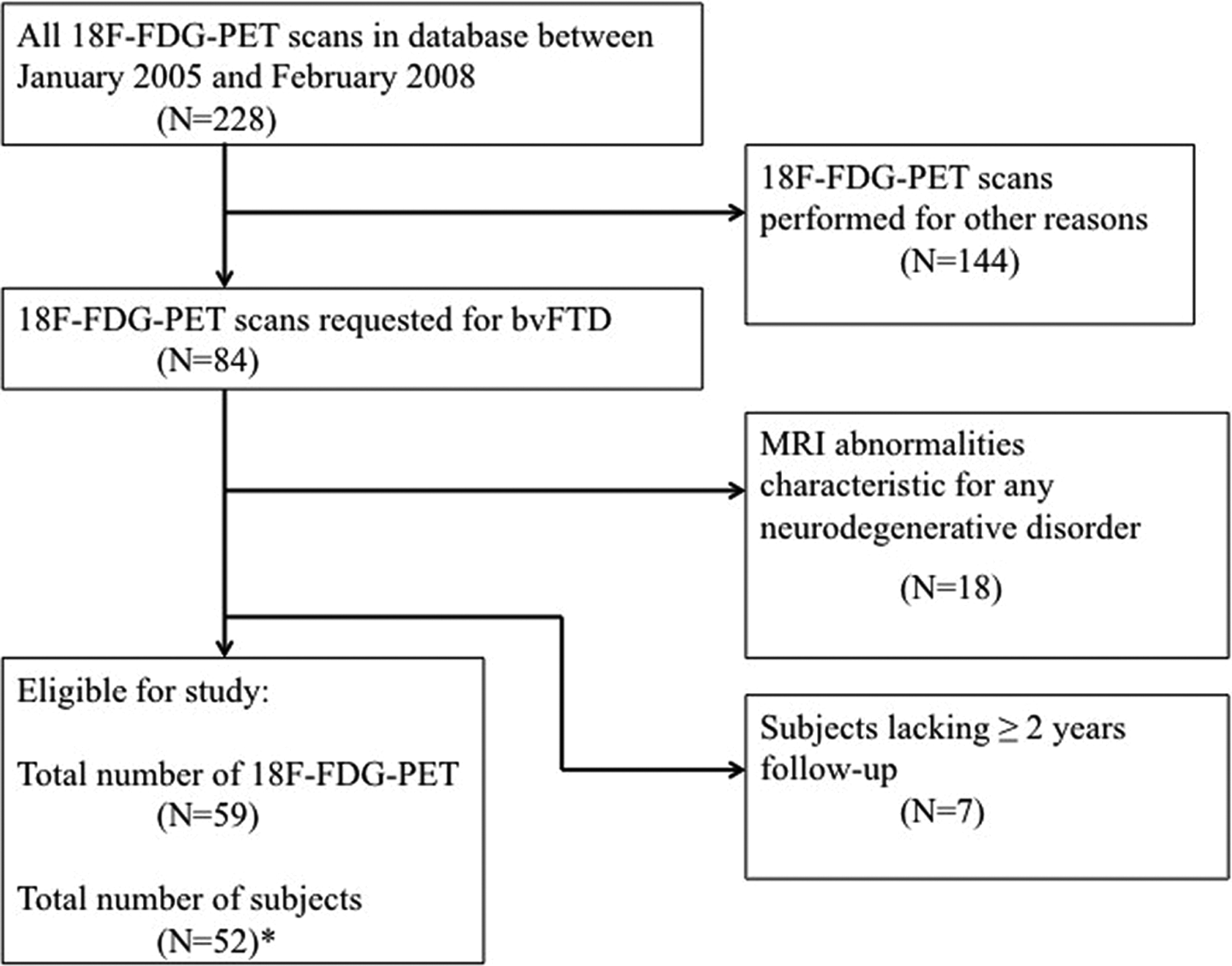
Flowchart of patient selection. *Repeated 18-fluorodeoxyglucose-positron emission tomography (18F-FDG-PET) was performed in 7 patients.
As a gold standard, we used a diagnosis of bvFTD based on clinical diagnostic criteria in the presence of functional decline (bvFTD/fd+) after at least 2 years of follow-up. 1 Evidence of functional decline was collected via the case notes (BK, AD, YP) revealing progressive cognitive problems based on the MMSE, FAB, CDR, or neuropsychological tests. 28 –30 Moreover, admission in a day care unit or nursing home due to cognitive and/or behavioral problems was considered as evidence for functional decline. Cases with a clinical diagnosis of bvFTD without significant functional decline were labeled bvFTD/fd−. Investigators were unaware of final positron emission tomography (PET) diagnosis. Disease duration was calculated as the time difference between first symptoms as reported by the informant and PET scanning.
Fluorodeoxyglucose-Positron Emission Tomography Imaging
Fluorodeoxyglucose-positron emission tomography imaging of all patients was performed on an ECAT EXACT HR+ scanner (Siemens/CTI, Knoxville, Tennessee) equipped with a neuroinsert to reduce the contribution of scattered photons. This scanner enables the acquisition of 63 transaxial planes over a 15.5-cm axial field of view, thus allowing the whole brain to be imaged in 1 bed position. The properties of this scanner have been reported elsewhere. 31 All patients rested for 10 minutes with the eyes closed in a dimly lit room with minimal background noise, when 18F-FDG was injected intravenously. Thirty-five minutes later, the patients underwent a 10-minute transmission scan followed by a 15-minute emission scan (3 × 5 minute frames). To restrict patient movement, a head holder was used. In addition, using laser beams, head movement was checked on a regular basis. The PET sinograms were corrected for dead time, tissue attenuation using the transmission scan, decay, scatter, and randoms. Next, images were reconstructed using a standard filtered back projection algorithm and a Hanning filter with a cutoff at 0.5 times the Nyquist frequency. A zoom factor of 2 and a matrix size of 256 × 256 × 63 were applied to the data, which resulted in a voxel size of 1.2 × 1.2 × 2.4 mm with a spatial resolution of 7 mm full width at half-maximum (FWHM) at the center of field of view. Both the original reconstructed volume scan and a voxel-based image analysis tool (Alzheimer's discrimination analysis tool PALZ, PMOD Technologies Ltd., Zurich) to detect brain areas with reduced metabolism were available to the reviewers. 32
The images were reviewed by 2 independent professionals in nuclear medicine who were unaware of the clinical diagnosis or results of structural imaging (BB and KH). Imaging was interpreted as positive (FTD pattern), normal, or deviant otherwise (non-FTD pattern). A positive 18F-FDG-PET scan was defined as a typical FTD pattern of hypometabolism in the lateral frontal and/or anterior temporal regions. Images were read conservatively meaning that isolated hypometabolism in the frontal mesial cortex was not reported as abnormal as this may be associated with normal aging and with depression when combined with abnormalities in dorsolateral cortex. 32,33
In case of disagreement, consensus was sought. The interrater variability was 0.7, indicating substantial agreement. Seven patients had their FDG-PET imaging repeated with an interval of at least 1 year.
Statistical Analysis
For statistical analysis, SPSS version 20.0 was used. Group comparisons were made using chi-square test for categorical data,
Results
Demographic data and scores on cognitive and functional scales are displayed in Table 1. Overall, the number of Neary criteria that were fulfilled at baseline varied between 2 and 5 (median 4).
Demographic Variables, MMSE, FAB, and CDR scores in the Total Cohort, bvFTD/fd+, and bvFTD/fd−.
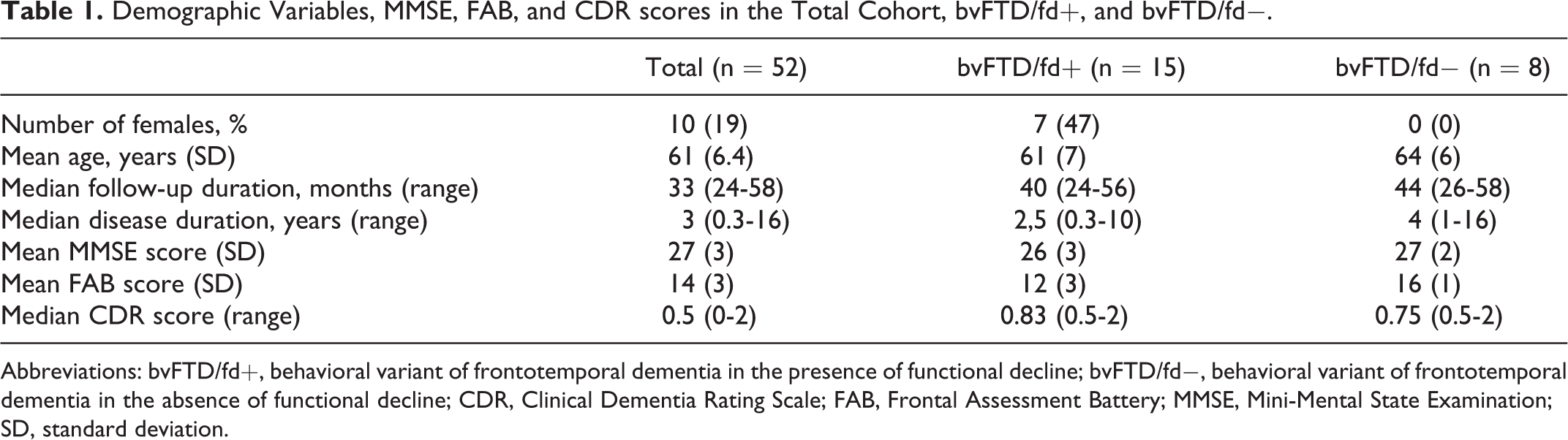
Abbreviations: bvFTD/fd+, behavioral variant of frontotemporal dementia in the presence of functional decline; bvFTD/fd−, behavioral variant of frontotemporal dementia in the absence of functional decline; CDR, Clinical Dementia Rating Scale; FAB, Frontal Assessment Battery; MMSE, Mini-Mental State Examination; SD, standard deviation.
We identified 15 cases fulfilling the clinical diagnosis of bvFTD (based on the presence of all 5 Neary criteria) in combination with functional decline at ≥ 2 years of follow-up (bvFTD/fd+). 1 Eight cases fulfilled the clinical criteria for bvFTD without displaying functional decline over time (bvFTD/fd−).
Frontal and/or anterior temporal hypometabolism on 18F-FDG-PET was present in 10 cases. In 29 cases, the 18F-FDG-PET was rated as normal and in 13 cases as abnormal according to a non-FTD pattern. The sensitivity of FDG PET for bvFTD/fd+ was 47% (95% confidence interval [CI] 22%-73%) at a specificity of 92% (95% CI 77%-98%; Table 2). Figure 2 shows examples of 18F-FDG PET scans.
Relation Between Results of 18F-FDG-PET and Diagnosis After Follow-Up.

Abbreviations: bvFTD, behavioral variant of frontotemporal dementia; bvFTD/fd+, behavioral variant of frontotemporal dementia in the presence of functional decline; bvFTD/fd−, behavioral variant of frontotemporal dementia in the absence of functional decline; 18F-FDG-PET, 18-fluorodeoxyglucose-positron emission tomography; N, number.
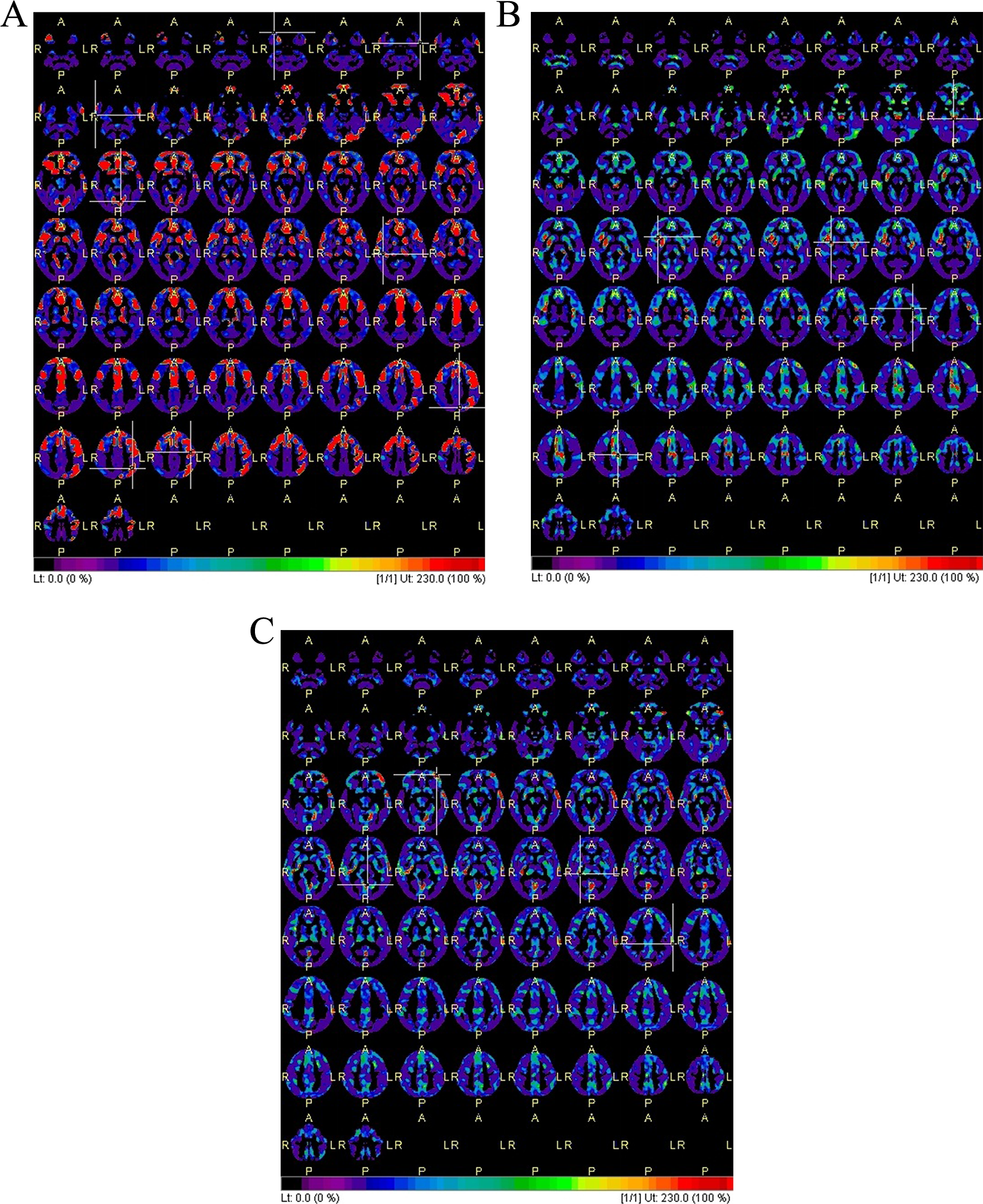
Representative images of 18-fluorodeoxyglucose-positron emission tomography (18F-FDG-PET) scans of true-positive, true-negative, and false-negative patients. The color red means significantly decreased uptake compared to the 18F-FDG-PET scan of age-matched control participants. A, Male, 66 years old, with a true-positive 18F-FDG-PET scan with an eventual diagnosis of behavioral variant of frontotemporal dementia in the presence of functional decline (bvFTD/fd+). The scan shows significantly decreased uptake in frontal and left more than right parietotemporal regions. B, Male, 65 years old, with a true-negative 18F-FDG-PET scan with an eventual diagnosis of depression. C, Male, 57 years old, with a false-negative 18F-FDG-PET scan with an eventual diagnosis of bvFTD/fd+.
Of the eight 18-FDG-PETscans of patients with bvFTD/fd+ having a non-FTD pattern of hypometabolism, 5 scans were rated as normal, 1 scan displayed an AD pattern of hypometabolism, and 2 scans displayed an atypical pattern of hypometabolism. Repeated 18F-FDG-PET was performed in 7 patients. In none of these cases 18F-FDG-PET evolved to a typical bvFTD pattern. A survey of all clinical diagnoses is given in Table 3.
Survey of Clinical Diagnosis After Follow-Up in 52 Patients With Initially Suspected bvFTD.
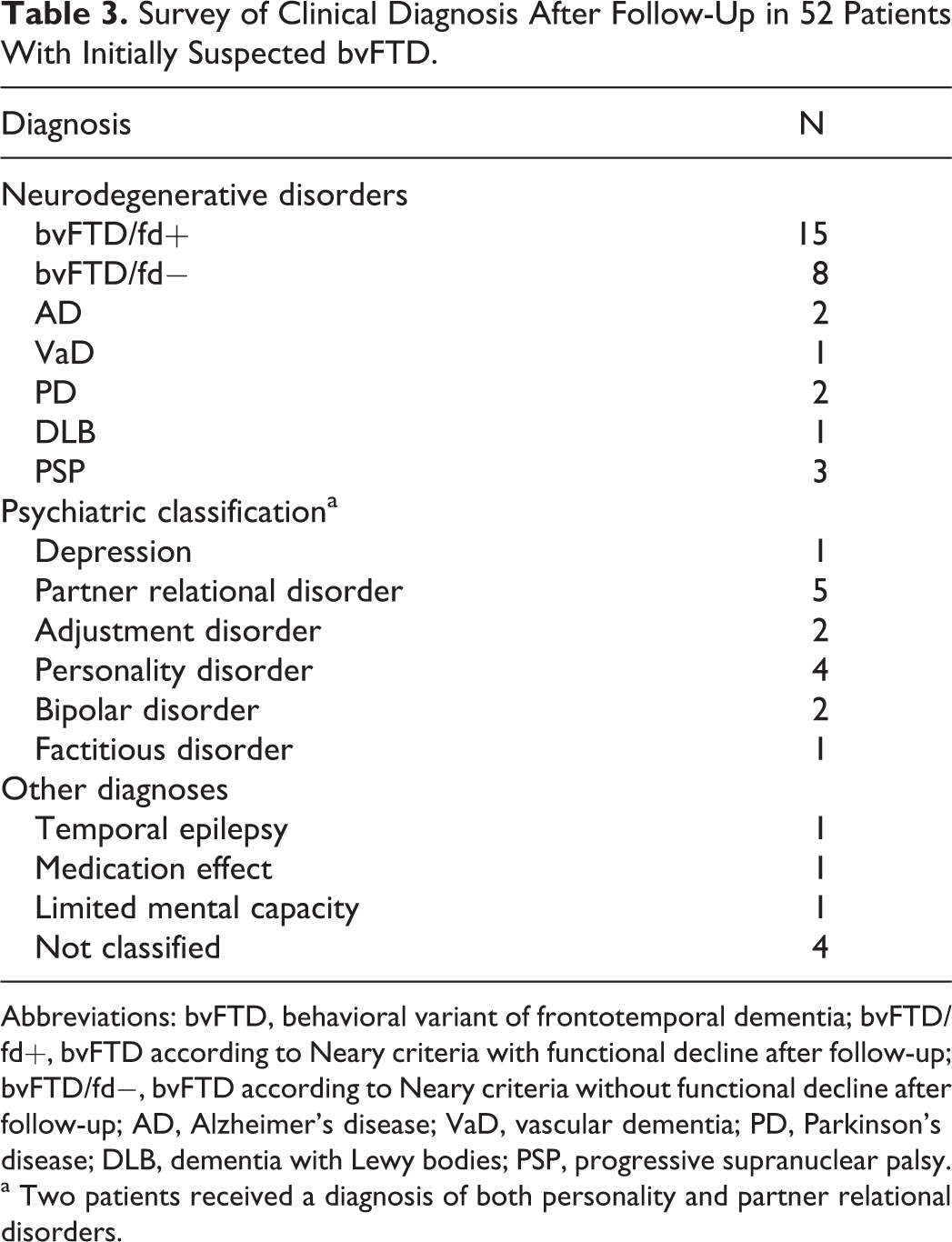
Abbreviations: bvFTD, behavioral variant of frontotemporal dementia; bvFTD/fd+, bvFTD according to Neary criteria with functional decline after follow-up; bvFTD/fd−, bvFTD according to Neary criteria without functional decline after follow-up; AD, Alzheimer’s disease; VaD, vascular dementia; PD, Parkinson’s disease; DLB, dementia with Lewy bodies; PSP, progressive supranuclear palsy.
a Two patients received a diagnosis of both personality and partner relational disorders.
Three patients with bvFTD/fd+ came to autopsy and a postmortem diagnosis of FTD-TDP43 was established. The 18F-FDG-PET scans of all 3 patients were rated as typical for bvFTD. One of these patients was carrier of a C9orf hexanucleotide repeat expansion.
Discussion
We investigated the additional diagnostic value of 18F-FDG-PET in the diagnosis of bvFTD in a selected cohort with noncharacteristic structural neuroimaging results.
We found that 18F-FDG-PET had a relatively low sensitivity of 47% for bvFTD at a high specificity of 92%. Yet, assuming that structural neuroimaging has a sensitivity of 50% to 70% for bvFTD, the overall sensitivity of neuroimaging increases substantially by identifying nearly half of the undiagnosed patients using 18F-FDG-PET. 2,9 However, our results show that a diagnosis of bvFTD cannot reliably be excluded using 18F-FDG-PET. This is supported by findings in a subgroup of patients with bvFTD carrying a C9orf hexanucleotide repeat expansion. 34 As pathological verification is missing in the majority of cases, it cannot be excluded that some of our patients with bvFTD/fd+ did not have neurodegenerative bvFTD. However, the presence of an alternative neurodegenerative disorder seems unlikely, given the fact that the majority of the 18F-FDG-PET scans without bvFTD patterns was normal. Also, we tried to avoid misdiagnosis using objective measures of functional decline instead of, for example, the informant-based history. On the other hand, 18F-FDG-PET appears to be highly accurate in differentiating bvFTD from clinical bvFTD mimics such as psychiatric disorders. Although former studies have described frontal abnormalities on functional neuroimaging in a variety of psychiatric disorders such as depression and schizophrenia, 19 –21 18-F-FDG-PET was normal in the patients with depression and bipolar disorder included in our study. As our cohort included a relative low number of patients with mood disorders and no patients with schizophrenia, the high specificity of 18-F-FDG-PET for bvFTD/fd+ might be an overestimation.
As an explanation for the relative low sensitivity of 18F-FDG-PET, we might have performed PET scanning too early to detect any abnormalities. Indeed as structural imaging was largely normal, we may have selected a subgroup that is too early in the disease course for detection of hypometabolism. It is generally acknowledged, however, that hypometabolism on PET predates abnormalities on structural neuroimaging. 14 At more detailed inspection of our cohort, patients with bvFTD/fd+ having frontotemporal hypometabolism had shorter disease duration than patients with bvFTD/fd+ having normal or inconclusive FDG-PET. Moreover, repeated functional imaging in 2 patients with bvFTD/fd+ having normal and inconclusive results of baseline functional imaging after on average 2 years showed no meaningful changes.
Our study is the first to address the additional diagnostic value of 18F-FDG-PET in patients with suspected bvFTD having negative structural neuroimaging. These results are particularly important for the interpretation of the FTDC clinical diagnostic criteria. The current sensitivity of 76% of probable bvFTD according to these criteria clearly reflects the overall sensitivity of neuroimaging as neuroimaging findings are incorporated within these criteria.
Another feature of probable bvFTD according to the new criteria is the presence of functional decline. Thus, it is prevented that patients with a clinical picture similar to bvFTD but lacking functional decline, the so-called “benign bvFTD phenocopy syndrome,” are being labeled as cases with a neurodegenerative disorder. In our cohort, we identified 8 cases with this syndrome (bvFTD/fd−) that fit the characteristic male predominance. 20,35 The 18F-FDG-PET was abnormal in only 1 of these patients. Although this cannot exclude an underlying neurodegenerative process, it is more likely that these patients have an alternative explanation for their behavioral change, such as personality traits or an undetermined psychiatric disorder. There are sporadic reports of absence of neurodegeneration on autopsy in possible “benign phenocopy” patients with bvFTD but this needs to be confirmed in larger series. 36,37 Limitations of our study are its retrospective nature that might have influenced the systematic inclusion of patients and prevented us from collecting a standard set of measures for functional decline in all cases. Also, circular argument may have been of influence as results of PET imaging were used in the original diagnostic process. Although measuring the impact of 18F-FDG-PET in this setting seems unfit, we have tried to preempt this limitation by blinded review of all relevant scans and by maximizing the length of clinical follow-up. Moreover, as limited postmortem data were available, we used the second best condition (clinical outcome after long-term follow-up) as “silver” standard. Finally, a relative small sample size may have influenced the power of the present study. This is inherent to the low general prevalence of bvFTD and the selection of MRI-negative cases.
Conclusion
18-Fluorodeoxyglucose-positron emission tomography adds to the clinical diagnosis of probable bvFTD when structural imaging is normal although its sensitivity in this situation is modest. Its relatively higher specificity enables to rule out bvFTD mimicks. A prospective study on the diagnostic value of 18F-FDG-PET in patients presenting with a frontal lobe syndrome is currently ongoing. 38
Our results underscore that the ideal biomarker for bvFTD is still missing. This warrants further research in multiple modalities to identify underlying neurodegeneration in clinical bvFTD.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. B. J. Kerklaan reports no disclosures. B. N. M. Van Berckel. K. Herholz reports having been a consultant for GE Healthcare, Lilly and Elan, and having received a research grant from Avid Radiopharmaceuticals. Dols A reports no disclosures. W. M. Van der Flier reports no disclosures. P. Scheltens serves/has served on the advisory boards of: Genentech, Novartis, Pfizer, Roche, Danone, Nutricia, Jansen AI, Baxter and Lundbeck. He has been a speaker at symposia organized by Lundbeck, Lilly, Merz, Pfizer, Jansen AI, Danone, Novartis, Roche, and Genentech. He serves on the editorial board of Alzheimer’s Research & Therapy and Alzheimers Disease and Associated Disorders and is a member of the scientific advisory board of the EU Joint Programming Initiative and the French National Plan Alzheimer. The Alzheimer Center receives unrestricted funding from various sources through the VUmc Fonds. Dr Scheltens receives no personal compensation for the activities mentioned earlier. Y. A. L. Pijnenburg reports no disclosures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
