Abstract
Currently, there are no effective treatments for Alzheimer’s disease and related disorders and age continues to be a robust risk factor. Thus, population aging in the United States may have catastrophic results if interventions are not found and implemented. This study examines possible associations between cognitive impairment and exercise, cognitive activities, and socialization. Cognitive activities, socialization, and exercise were assessed at baseline, and cognitive function was measured at baseline, 5-year, and 10-year follow-up. Controlling for baseline cognitive function, age, sex, education, diabetes, and hypertension, linear regression was performed. Engagement in cognitive activities was inversely associated with the onset of cognitive impairment at 5-year follow-up but was no longer significant at 10-year follow-up. Exercise was associated with a lower risk of cognitive impairment at 10-year follow-up but was not significant at 5-year follow-up. Associations with socialization were not statistically significant at either follow-up.
Introduction
Recent research on aging-related cognitive impairment involves potentially promising interventions that might prevent or delay the onset or progression. This research commonly involves 1 of 3 types of activities: exercise, cognitive activities, and social involvement. We test the effects of these in a head-to-head comparison using cognitive outcomes at 5 and 10 years following a baseline survey.
Research to date suggests that regular exercise might counteract aging-related cognitive declines. Although most of the studies to date are observational, the growing literature on the cognitive benefits of exercise supports the view that there is a positive association between exercise and cognitive function 1 –6 as well as an inverse relationship between regular exercise and the vulnerability to develop dementia. 7 –9 However, some studies with sample sizes <1000 did not find a statistically significant association between exercise and dementia or Alzheimer’s disease (AD). 10 –13 The number of subjects in clinical trials that examined the effects of exercise on cognitive function is limited. However, several randomized trials showed a positive effect of exercise on preservation of cognitive function. 14 –18 Animal studies add further evidence in support for exercise. 19,20
Another potential intervention involves cognitively stimulating activities such as reading or doing puzzles. Most of the research is observational but many studies have found an inverse association between engagement in cognitive activities and dementia risk 10 –12,21 as well as risk of mild cognitive impairment. 21,22 Several studies have found an association between greater engagement in cognitively stimulating activities and a decreased rate of cognitive decline. 12,21,23 A few randomized trials indicate that cognitive training can have a positive effect on cognitive function. 24 –26 Further evidence in support of cognitively stimulating activities as a possible intervention comes from basic science. 27
Social resources, including both social networks and engagement in social activities, also show potential for slowing cognitive decline. Again, this research is observational but the results are promising. 28 –31 In an autopsy study, 89 subjects were examined for AD brain pathology (amyloid load and tau tangles). Larger social networks were found to be associated with diminished negative effect of AD pathology on cognition. 32
One problem with these studies is that participation in these 3 types of activities is not unrelated. For example, individuals who exercise may have more opportunities for engaging in social activities. Also, participating in cognitively stimulating activities might make social interactions easier and more enjoyable. Few studies have simultaneously examined the associations of participation in these different types of activities on the onset of cognitive impairment. 21,33 A second problem is that most studies involved only a relatively short length of follow-up. Interventions that are successful over a period of a few years might not have long-term impact.
We used National Long Term Care Survey (NLTCS) data from 3 Waves (1994, 1999, and 2004) to examine possible associations between cognitive impairment and exercise, cognitively stimulating activities, and social resources (family, friends, and engagement).
Methods
The NLTCS is a large, nationally representative study created to assess the long-term care needs of the elderly as well as their health. It was initiated by the Health Care Financing Administration, currently the Centers for Medicare and Medicaid Services. After an initial follow-up survey in 1984, the National Institute on Aging (NIA) funded follow-up surveys in 1989, 1994, 1999, and 2004. The NLTCS sample was selected from Medicare enrollees and included both community-residing and institutionalized subjects after 1984.
For this study, 3 waves of NLTCS data were used: 1994 (baseline), 1999 (5-year follow-up), and 2004 (10-year follow-up). Exercise data were collected only on the Community Questionnaire in 1994 so only community-residing subjects were included in the sample. Those with AD, senility, or mental retardation (self- or proxy-report) were excluded at baseline, leaving a total sample size of 5280.
To assess cognitive function, study subjects were given the Short Portable Mental Status Questionnaire (SPMSQ) 34 in 1994. Only subjects with SPMSQ scores in 1994 (N = 4382) who were cognitively intact or mildly impaired were included, resulting in a baseline sample size of 3863. subjects who made <2 errors were deemed cognitively intact and those who made 3 or 4 errors were regarded as mildly impaired. Moderate impairment was defined as 5 to 7 errors and severe impairment was 8 to 10 errors; maximum number of errors possible was 10. Adjustments to scores were made for education: those with less than a high school education were permitted 1 additional error, and those with more than a high school education were permitted 1 fewer error. 34
By the 5-year follow-up, 1177 of the 3863 subjects in the sample at baseline had died, and 198 were lost to follow-up, leaving 2488 (see Figure 1) subjects. By the 10-year follow-up, 2253 had died, and 350 were lost to follow-up, leaving 1260 (see Figure 2). Mortality data include updated information from Vicki Lamb, PhD, (Personal electronic communication, April 28, 2008). For the 1994 community questionnaire, study subjects were asked several questions regarding their ability to get around inside. If subjects indicated that they were not able to get around inside or needed assistance to do this (N = 720), they were considered too disabled to exercise and were excluded from the sample. Surviving community-residing subjects were given the Mini-Mental State Examination (MMSE) 35 in 1999 to assess cognitive function. There were 1448 subjects who were cognitively intact (scores > 24); 115 were mildly impaired. There were 179 subjects with moderate/severe impairment (scores < 20). Cognitive status was unknown for 531 (23.4%) subjects. In 2004, the SPMSQ was again used to assess cognitive status. There were 862 subjects who were cognitively intact, 80 with mild impairment and 96 with moderate/severe impairment. Cognitive status was unknown for 64 (5.8%) subjects. Exercise participation, cognitive activities, and socialization were all assessed at baseline as part of the 1994 community questionnaire.
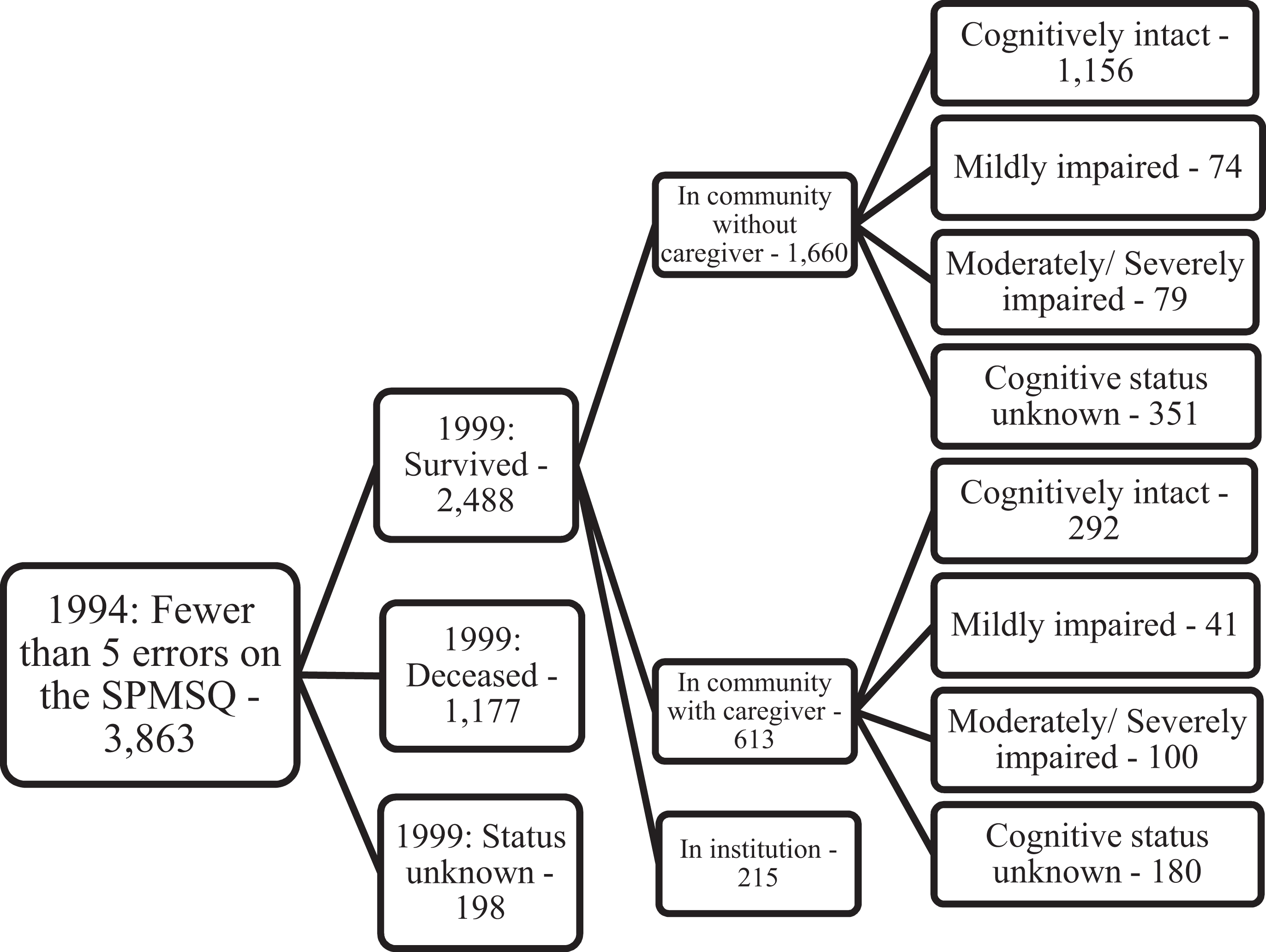
Sample from baseline (1994) to 5-year follow-up (1999). Cognitive status in 1999 was based on the Mini-Mental State Examination (MMSE) and includes all who received scores.
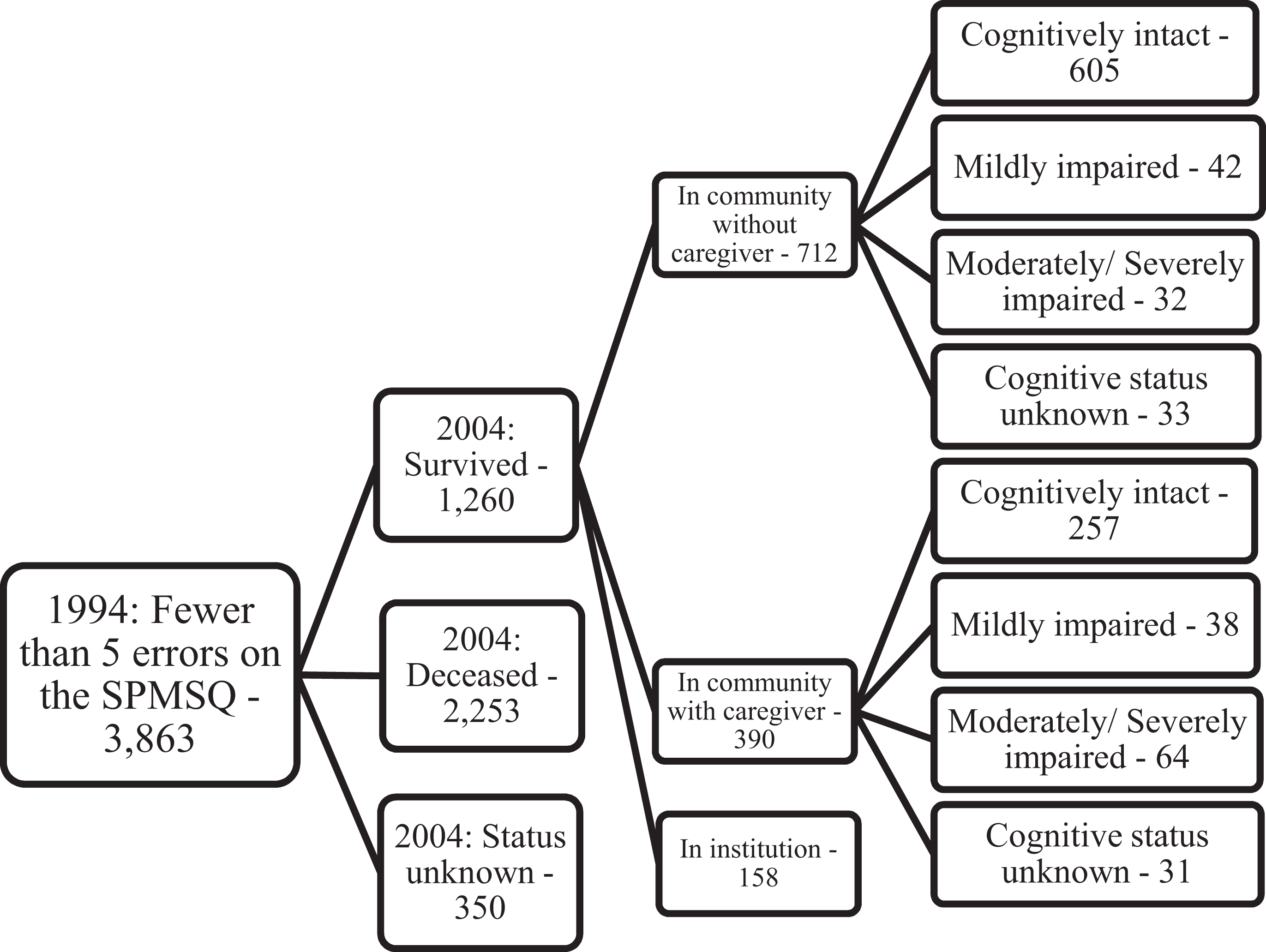
Sample from baseline (1994) to 10-year follow-up (2004). Cognitive status in 2004 was based on the Short Portable Mental Status Questionnaire (SPMSQ) in 2004 and includes all who received scores.
Data regarding 23 different physical exercise activities, including walking, jogging, hiking, biking, yoga, basketball, and so on, were collected but the 2 most common exercises, by far, were walking and gardening/yard work. Subjects were asked whether or not they participated in each activity and if so, how often during the previous 2 weeks and how long, on average, per session. Based on previous work that examined different methods of measuring exercise using data from the NLTCS and the associations of each with cognitive impairment, 2 the number of exercise sessions that were at least 20 minutes long was chosen as the optimal measure of exercise participation and was used for all analyses.
To assess the extent of engagement in cognitively stimulating activities, responses to 4 questions were used to develop a cognitive activity score. The questions asked subjects if within the last week they: (1) did any reading including books, newspapers, or magazines, (2) participated in any hobbies, (3) played games or worked on puzzles, or (4) listened to compact discs, records, or tapes. One point was given for each “yes” response to each of the 4 questions resulting in a 4-point scale with a potential range of 0 to 4.
A 4-point scale was also created to score the level of socialization. Subjects were asked whether they kept in touch with relatives either in person or by talking on the telephone and if so, how often in the past month. They were asked the same questions regarding keeping in touch with friends. If subjects saw or talked with relatives on the telephone at least 6 times over the past month, they received a point; if less than 6 times (including never), they received no point. Similarly, subjects received a point if they saw or talked on the telephone with friends at least 6 times in the past month and no points if less than 6 times. One point was awarded if they attended at least 1 meeting of a club or other organization in the past month. One point was also awarded if they attended religious services at least once in the past month. Thus, the range of possible scores for socialization is 0 to 4.
Data Analysis
The analysis compared the cognitive score at each follow-up with the measures of exercise and social and cognitive activities after controlling for the baseline SPMSQ. Because the NLTCS changed the measure of cognitive impairment between rounds, the analysis for 1999 was based on the MMSE, and the 2004 follow-up was based on the SPMSQ.
The 1999 data were analyzed using a linear regression of MMSE scores (dependent variable) on SPMSQ scores at baseline, controlling for age, sex, education, and presence of 2 medical conditions selected based on their associations with dementia (diabetes 36 or hypertension 37 ), as well as measures for level of participation in exercise, engagement in cognitively stimulating activities, and level of socialization. An “errors-in-variables” model that controls for the reliability of the SPMSQ and self-reports for exercise was used. A reliability of 0.82 was used for the SPMSQ. 34 For exercise, reliability was set at 0.75. The Women’s Health Initiative 38 reported a value of 0.67 and a study examining risk of breast cancer and bicycle riding among German women reported 0.43. 39
Analysis of the 2004 data was based on a linear regression on change between 1994 and 2004 of the SPMSQ score (dependent variable), with controls for baseline SPMSQ score, age, sex, education, presence of applicable medical conditions (diabetes or hypertension), exercise participation, cognitive activities, and socialization. As described previously, errors-in-variables adjustments were made for the reliability of the exercise measurement as well as the baseline SPMSQ. 40 We also tested a model in which the regression was based on the difference between the 2004 scores and the predicted score from a linear regression of the 2004 score on the 1994 score. The results from the 2 methods were virtually identical. All analyses were performed using STATA version 10.
In our previous analysis of the effects of exercise using this sample, we controlled for reports of diabetes and hypertension. There was no association of cognitive scores with these diseases. We also tested a simple count of the number of 9 other conditions. It was also not significant and did not affect the results. Removing subjects who reported Parkinson’s disease or having had a stroke also did not change the results. 2
Results
Baseline (1994) characteristics of the total sample are presented in Table 1. Characteristics for the total sample, those with SPMSQ scores in 1994 and subjects who were cognitively intact or mildly impaired in 1994, are displayed. Subjects with SPMSQ scores are similar to the total sample. Approximately one-third were between the age of 75 to 79 at baseline and about one-third were male. The educational level of those who were cognitively intact/mildly impaired at baseline was slightly higher with 70.2% having at least some high school. Table 2 presents the baseline cognitive status of subjects who had SPMSQ scores in 1994.
Baseline Characteristics of Sample by Cognitive Status, 1994.
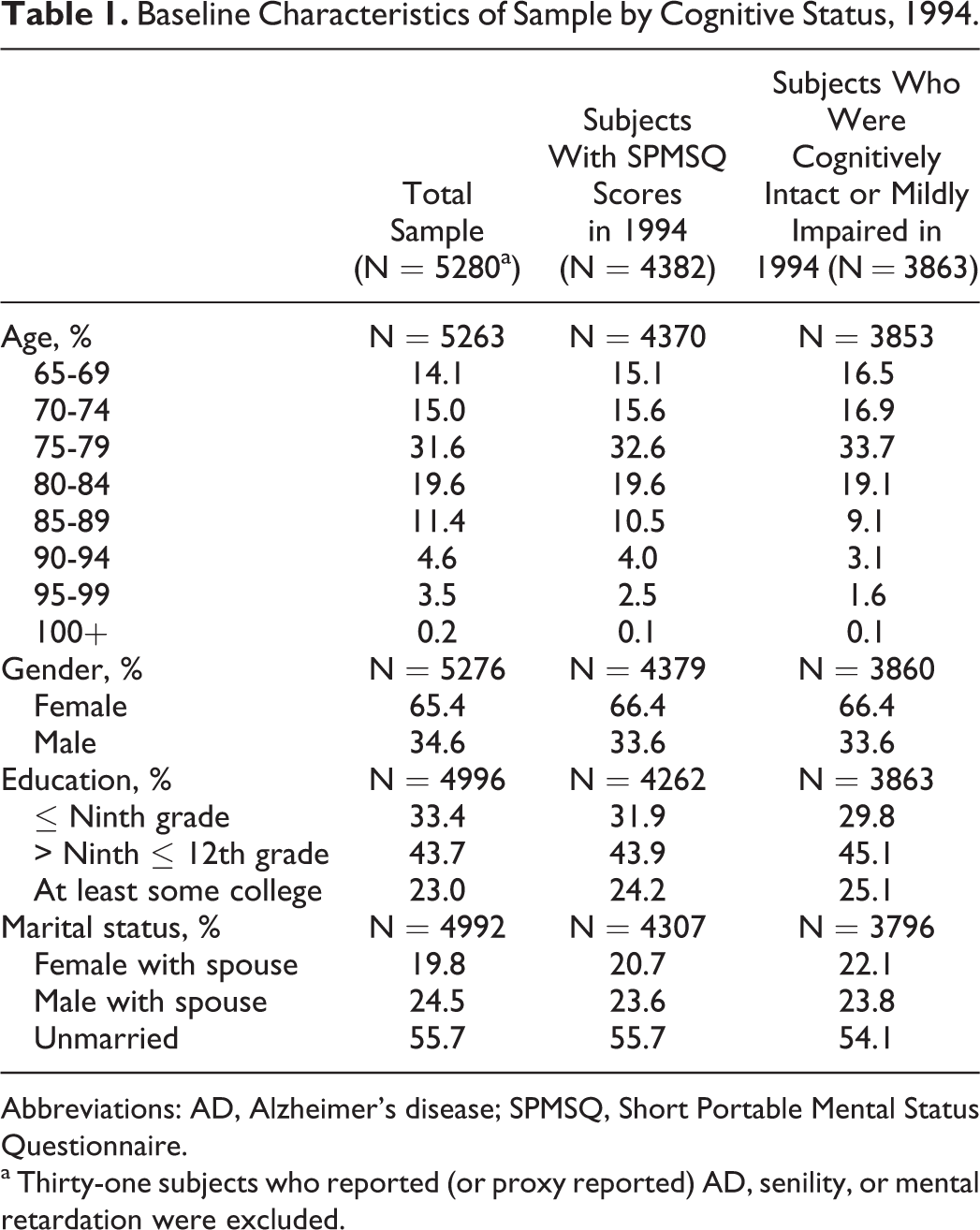
Abbreviations: AD, Alzheimer’s disease; SPMSQ, Short Portable Mental Status Questionnaire.
a Thirty-one subjects who reported (or proxy reported) AD, senility, or mental retardation were excluded.
Baseline Cognitive Status of Subjects With SPMSQ Scores in 1994.a

Abbreviation: SPMSQ, Short Portable Mental Status Questionnaire.
a N = 4382.
Table 3 shows the results from the linear regressions on the MMSE scores in 1999 and the change between 1994 and 2004 on the SPMSQ, 5-year, and 10-year follow-up, respectively, controlling for exercise participation, engagement in cognitive activities, and socialization. Subjects who were living in the community and were cognitively intact or mildly impaired in 1994 and were subsequently tested in later waves of data collection were included in the analysis. Engagement in cognitively stimulating activities was associated with less cognitive impairment at the 5-year follow-up (P < .001). However, by the 10-year follow-up, although the association was in the right direction, results were not statistically significant. For exercise, there was no significant association between exercise and risk of cognitive impairment at the 5-year follow-up. However, at the 10-year follow-up, exercise showed a significant association with less cognitive impairment (P = .018). Finally, for socialization, the associations at both time points were in the right direction but not significant.
The Results of a Single Linear Regression Model Estimating the Effects of Cognitive Activities, Exercise, and Socialization Reported in 1994 on the 1999 MMSE Scores and on the Change in the SPMSQ Between 1994 and 2004.a
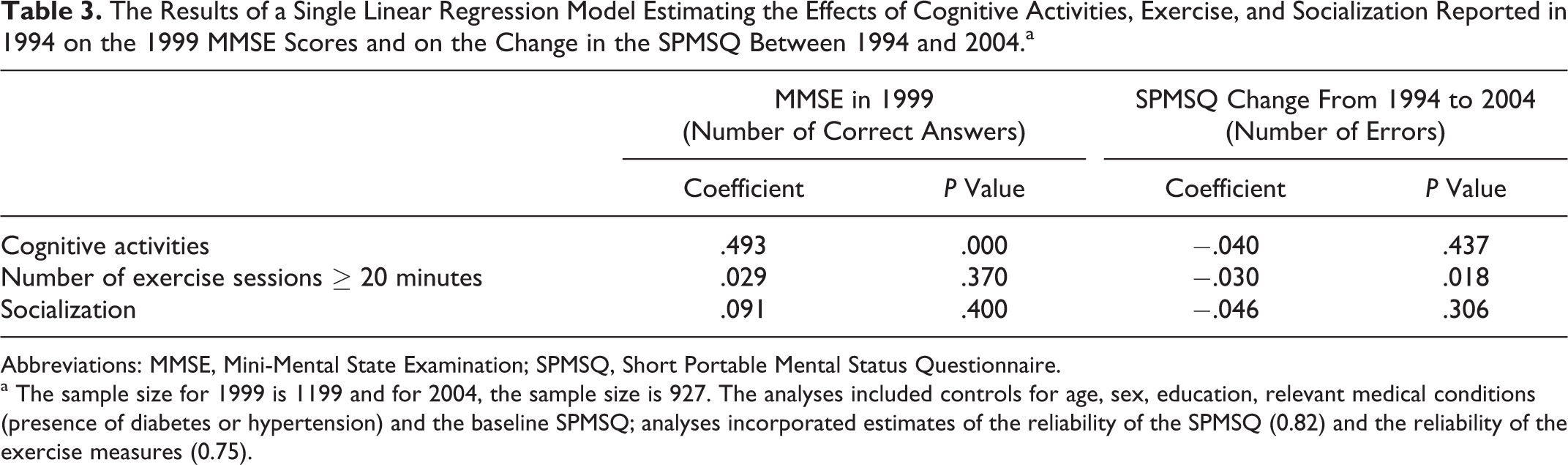
Abbreviations: MMSE, Mini-Mental State Examination; SPMSQ, Short Portable Mental Status Questionnaire.
a The sample size for 1999 is 1199 and for 2004, the sample size is 927. The analyses included controls for age, sex, education, relevant medical conditions (presence of diabetes or hypertension) and the baseline SPMSQ; analyses incorporated estimates of the reliability of the SPMSQ (0.82) and the reliability of the exercise measures (0.75).
Linear regressions were also performed controlling for, one at a time, cognitive activities, exercise, and socialization; results were similar to those described previously (data not shown).
Discussion
This is one of only a few studies that have simultaneously examined the associations of cognitive activities, exercise, and socialization with changes in cognitive abilities. The results add to the growing body of research indicating that engaging in cognitively stimulating activities lowers the risk of cognitive impairment, although this effect waned with time in our study and that exercising, independently, lowers the risk of impairment as well. There are several findings in our study that merit further discussion.
We anticipated that greater socialization would lower the risk of cognitive impairment. Although the associations between increased socialization and a lower risk of cognitive impairment at both the 5- and 10-year follow-up time points were in the right direction, results were far from significant (P values were .400 and .306, respectively). This contrasts with many, but not all, 41 previous studies examining the association between socialization and cognitive impairment. Inconsistent results from these studies may reflect differences in study design.
We were surprised that cognitive activities showed such a strong association at 5 years (P < .001) but no significant association at 10 years (P = .437). This result is in contrast with the finding that exercise was associated with risk of cognitive impairment at the 10-year follow-up (P = .018) but was not significant at the 5-year follow-up (P = .370). These findings for exercise are similar to those from an earlier, more detailed study performed using the NLTCS. 2
There are several possible explanations for these unexpected results. One hypothesis links to the theory of cognitive reserve. 42 Investigators have speculated that through various activities (ie, education), individuals can build up their cognitive reserve that will protect them against insults from disease pathology (ie, plaques and tangles of Aβ and tau) that would otherwise affect their cognitive functioning. This theory provides an explanation for individuals who, upon autopsy, are found to have abundant plaques and tangles in their brains; however, prior to death, they were assessed as having unimpaired cognitive function. Both cognitively stimulating activities and exercise have been suggested as interventions to build cognitive reserve. Our results may suggest that cognitively stimulating activities build cognitive reserve that lasts over a limited time period (ie, 5 years) and diminishes with time, whereas physical exercise increases cognitive reserve that is maintained over a longer time span (ie, 10 years).
Another possible explanation for the association between a higher cognitively stimulating activity score and a lower risk of cognitive impairment at 5 years but not at 10 years could be that study subjects continued to undertake cognitive activities during the first 5 years of the study, thus still benefiting if a continuous “dose” of cognitive activities is required to have an effect, but gave up these activities by the 10-year follow-up. To take this a step further, perhaps giving up the pursuit of cognitive activities is a marker of cognitive decline. 43,44 In contrast, subjects who were exercising at baseline may have continued to exercise through the 10-year follow-up or the beneficial effects of exercise earlier in life may have lasted, even if subjects ended their exercise routines, until the 10-year follow-up.
Finally, cognitive activities and exercise are both associated with a lower risk of cognitive impairment, but they may work through different pathways. Cognitively stimulating activities have been shown to increase neuronal density 45 and therefore may directly affect cognitive function through brain plasticity. It is possible that despite numerous Aβ plaques and tau tangles, individuals who engage in cognitively stimulating activities have increased the number of neurons in their brains, providing them with enough reserve to perform cognitive tasks without impairment. Exercise also impacts on brain plasticity in that it affects brain density 46 and brain volume. 47 Colcombe et al found that older subjects who were more physically fit experienced a diminished loss of brain density when compared to older subjects who were less physically fit. 46 In a randomized trial comparing aerobic and nonaerobic exercise for older adults (and with a nonexercising, younger-age control group), subjects assigned to the aerobic intervention increased their brain volume, both gray and white matter. Neither the nonaerobic group nor the younger control group experienced these increases. 47
The pathways through which cognitive activities and physical exercise impact brain plasticity may very likely overlap; however, exercise may work through additional pathways. For example, AD and other dementias are associated with cardiovascular risk factors such as high blood pressure and high levels of cholesterol 37,48 as well as diabetes. 36 It is well understood that exercise lowers the risk of cardiovascular risk factors, cardiovascular disease, and diabetes. 49 It is therefore possible that exercise lowers the risk of cognitive impairment indirectly through lowering cardiovascular risk and risk of diabetes. This indirect pathway may have a longer-lasting impact on cognitive impairment and may continue to the 10-year follow-up.
We must note a limitation of this study that may impact our results. The SPMSQ was used to measure cognitive function at baseline and at the 10-year follow-up. The MMSE was used for the 5-year follow-up, and many subjects were not tested, perhaps because they appeared cognitively intact during earlier sections of the interview.
In addition relying on the SPMSQ and MMSE for our outcome measures lacks precision as these instruments were designed to provide a gross measure of cognitive status and they both have well known limitations as outcome measures of cognitive change. Notwithstanding this limitation, our results indicate associations between cognitive impairment and engagement in cognitively stimulating activities, as well as physical exercise and we are therefore encouraged that an association does in fact exist. More precise outcome measures might reveal a more robust association.
We must note that our study results show association and not causation. We speculate that both exercise and cognitive activities independently lower the risk of cognitive impairment but this needs to be confirmed in randomized trials. Individuals with better cognitive function may engage in exercise and cognitive activities more compared to individuals with cognitive impairment. However, we consider this unlikely since subjects with moderate or severe cognitive impairment at baseline were excluded from the study.
Finally, once subjects in the NLTCS became institutionalized, data on their cognitive status were no longer collected. Therefore, our results cannot be generalized to the entire US population, only to the noninstitutionalized US population who would have been assessed as cognitively intact or mildly impaired on the SPMSQ in 1994. In summary, the results of this study add to the growing body of research indicating that exercise and engagement in cognitive activities may have potential as interventions to lower the risk of cognitive impairment and dementia.
Footnotes
Acknowledgments
We thank the National Archive of Computerized Data on Aging, Ann Arbor, for providing the National Long Term Care Survey (NLTCS) data and Vicki Lamb for her invaluable assistance with using the NLTCS.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the National Institutes of Health-NIA, Grant number P30 AG12836, the Boettner Center for Pensions and Retirement Security at the University of Pennsylvania, and National Institutes of Health-National Institute of Child Health and Development Population Research Infrastructure Program R24 HD-044964, all at the University of Pennsylvania; the University of Pennsylvania Alzheimer’s Disease Core Center (National Institutes of Health - NIA), Grant number P30 AG10124 and the Marian S. Ware Alzheimer Program. JQT is the William Maul Measey-Truman G. Schnabel Jr, MD, Professor of Geriatric Medicine and Gerontology.
