Abstract
Over the last decade, a large number of experimental observations have suggested a relationship between alterations in cholesterol homeostasis and Alzheimer’s disease (AD). Moreover, epidemiological studies have pointed an association between statin treatment and a decrease in the risk of having AD. For these reasons, a large number of clinical trials have been carried out to determine whether the statins can prevent the progression of AD. However, these studies did not provide clear evidence for the therapeutic efficacy in AD. We consider that there are a number of explanations for this failure that may provide guidance for selecting and clinically developing statins with therapeutic efficacy in AD.
Over more than 10 years, a relationship between alterations in cholesterol homeostasis and Alzheimer’s disease (AD) has been reported. 1 Long-term prospective population-based studies have indicated that elevated cholesterol levels in midlife are associated with an increase in the risk to suffer AD in later life. 2,3 Moreover, polymorphisms in apolipoprotein E (apoE) and other proteins involved in cholesterol metabolism are considered as risk factors for AD. 4 Experimental support for the neuroprotective activity of statins has been provided by a large number of studies, reporting that statins, in particular simvastatin, have a direct protective effect on the central nervous system mediated by the changes in the brain cholesterol homeostasis or other mechanisms such as the control of neuroinflammation. 5 These results have been confirmed by several epidemiological studies 6 –8 , wherein it has been described that a previous treatment with statins is associated with a decrease in the risk of having AD. 9 However, not all epidemiological studies conclude that statins are beneficial in the setting of AD, 6 and rare cognitive problems have been associated with statin therapy in a limited number of cases. 10 Major reasons for these discrepancies are the statin selected for each study, the time of initiation of statin therapy with respect to age and the time of AD onset, and the selected population for the studies. For these reasons, we conclude that an unbiased analysis of the main well-regulated clinical studies is a need to define the main master lines in the use of statins for AD.
To determine the potential use of statins for AD, we analyzed the ongoing or completed clinical trials (CTs; http://www.clinicaltrials.gov/) using statins for the AD prevention or treatment. We decide to select these CTs because they are well-controlled and developed studies. These large CTs have been carried out to determine whether the statins already in the market as antihypercholesterolemic drugs may prevent the progression of AD (Table 1). However, these studies did not provide clear evidence for the therapeutic efficacy in AD. Taking into account these considerations, we hypothesized that changes in the design of CTs and in the selection of statins for AD could increase the possibility of success. We propose the use of statins specifically designed for neuroprotection (with high brain penetration and/or high neuroprotective profile) to be used early in the time course of the disease, selecting patients with hypercholesterolemia, and using a set of biomarkers that includes cholesterol homeostasis and inflammatory biomarkers. We also consider that there are a number of explanations for this failure that may provide guidance for selecting and clinically developing statins with therapeutic efficacy in AD.
Clinical Trials for Alzheimer’s Disease Using Statins a
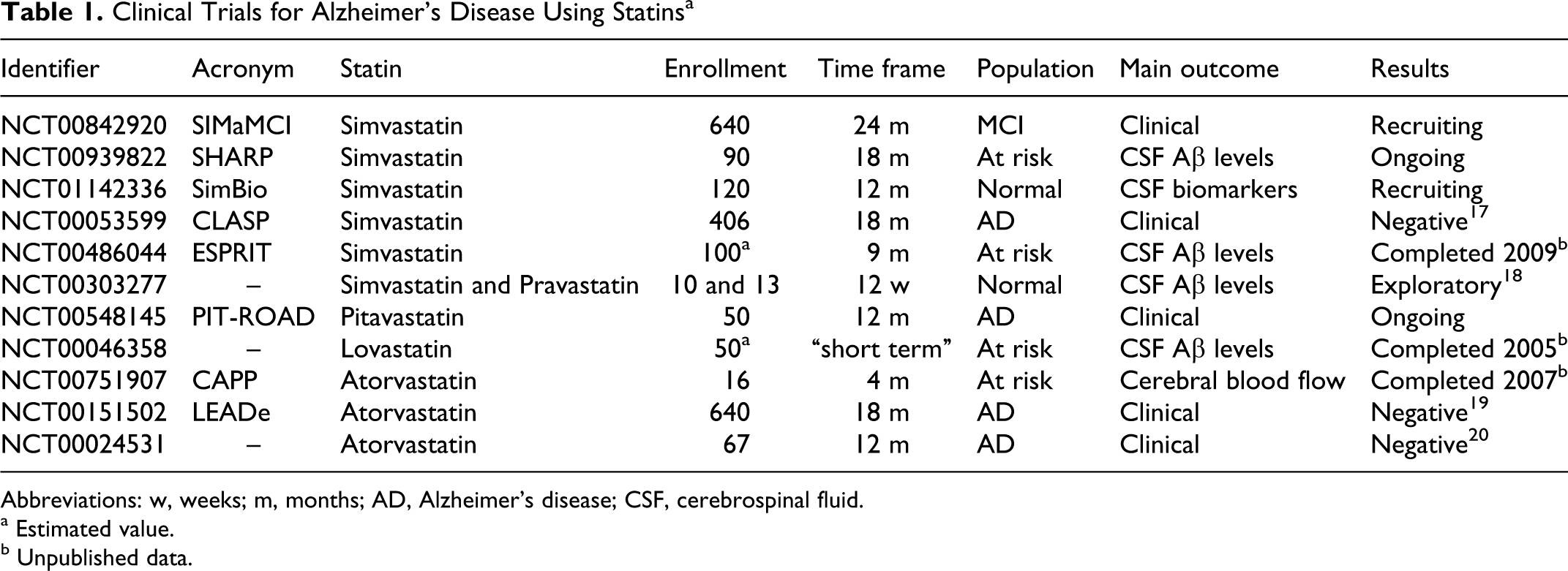
Abbreviations: w, weeks; m, months; AD, Alzheimer’s disease; CSF, cerebrospinal fluid.
a Estimated value.
b Unpublished data.
Commercially Available Statins Were Not Selected for Neuroprotective Activity
Commercially available statins were selected and developed for their plasma cholesterol-lowering effects. Indeed, atorvastatin that has been used in 3 CTs for AD (NCT00751907, NCT00151502, and NCT00024531) does not get into the brain, although simvastatin that has an acceptable blood-brain barrier (BBB) penetration has been also evaluated. It appears necessary to identify novel statins with differential structural features that result in a stronger intrinsic neuroprotective activity and physicochemical properties that result in a greater brain penetration. These novel compounds are expected to have a better dissociation between central neuroprotective effects and dose-limiting peripheral unwanted effects and thus display neuroprotective effects at well-tolerated doses in the clinical setting.
Statins Should be Studied in Predementia AD or Even in Presymptomatic AD
Epidemiological studies have shown that hypercholesterolemia is an early risk factor associated with AD and that elevated plasma concentrations of low density lipoprotein (LDL) cholesterol (LDL-C) in the midlife are associated with risk of late-life AD. 11,12 However, the majority of CTs are designed to evaluate the outcome in patients diagnosed for full-blown AD (NCT00024531, NCT00151502, NCT00548145, and NCT00053599) although 2 ongoing studies using simvastatin are being performed in cognitive normal participants (NCT01142336 and NCT00303277) and in mild cognitive impairment (MCI) (NCT00842920). Moreover, since some studies suggest that the statins may have a beneficial effect after long-term administration, 13,14 a prolonged follow-up period of treatment could increase the possibility of success. A similar treatment period that the VITACOG study 15 could be applied for statin treatments in predementia AD.
Hypercholesterolemic Patients Should be Selected for Proof-of-Concept Studies
Because hypercholesterolemia in humans 11,12 and in animals 16 is a risk factor associated with the cognitive decline, high total cholesterol (TC) or LDL-C should be recruiting patient selection criteria. However, in the past, only 1 CT (NCT00303277) used this criterion to select their participants (TC > 200 mg/dL and/or LDL-C > 130 mg/dL), while in the NCT01142336, the inclusion threshold for LDL was only of 70 mg/dL. On the contrary, 2 studies have used hypercholesterolemia as exclusion criteria: NCT00842920 and NCT00053599. Additionally, the main risk factor for AD (apoE ∊4 allele) exacerbates hypercholesterolemia, 4 also supporting the view that hypercholesterolemia should be an inclusion criterion. If cholesterol-independent effects of statins are crucial, hypercholesterolemia could complicate the interpretation of outcomes, but if statins protect by cholesterol-dependent effects, hypercholesterolemia could be a critical parameter.
Biomarker Strategy
A translational research strategy should be defined in order to optimize the development process. Biomarkers of the effects on plasma and brain cholesterol should be used to provide a proof-of-mechanism and to select the doses to be used in proof-of-concept studies. Other than standard AD progression biomarkers (Aβ species and P-tau, brain imaging), additional strategies should be considered such as monitoring neuroinflammation (by positron emission tomography) or brain atrophy (by magnetic resonance imaging). Additionally, brain blood flow measurement could also be a good indicator for statin effects on the function of cerebral vasculature.
In conclusion, although CTs using statins have not provided clear evidence for therapeutic efficacy in AD, we consider that a successful strategy should include identifying novel statins with optimized brain penetration and/or stronger intrinsic neuroprotective activity. Second, learning from the completed or ongoing CTs must be useful to design new trials with a greater probability of success. The CTs with these novel statins must be designed to prevent the progression in very early rather than dementia stage of AD and selecting participants with proved hypercholesterolemia. It is expected that such a strategy leads to the long sought demonstration of the therapeutic efficacy of statins in AD.
Footnotes
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JSB and JV are Neuron Bio employees.
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: funded by Neuron Bio and supported by grants from the Agencia de Innovación y Desarrollo de Andalucía and from the CDTI of the Ministerio de Ciencia e Innovación, and by funds from the Torres Quevedo program (JSB).
