Abstract
Purpose:
We observed that many of our helical therapy lung stereotactic body radiation therapy plans did not meet the Radiation Therapy Oncology Group (RTOG) recommended R50% (volume of 50% of the prescription dose/planning target volume), which characterizes the steepness of dose fall off. We hypothesized that despite not meeting R50%, helical therapy lung stereotactic body radiation therapy plans would confer similar local control and minimal side effects as previously reported using nonhelical treatment platforms.
Materials and Methods:
We report a retrospective review of all consecutive patients treated off-protocol with stereotactic body radiation therapy for peripheral lung lesions from 2008 to 2013 utilizing helical therapy. Seventy-four patients (81 lesions and 79 plans) were treated with doses ranging from 48 to 60 Gy in 3 to 5 fractions prescribed to the edge of the planning target volume.
Results:
Forty-eight (61%) plans had major deviation from R50%. Only 1 (<1%) plan had a major deviation from the R100%. All plans had > 95% planning target volume coverage by prescription dose, 7(8.6%) plans with 121% to 133% maximum dose, and lung V20 Gy <10% in 70 (89%) plans. With a median follow-up of 4.7 years (95% confidence interval: 4.1-5.3), local control for all patients at 1, 2, and 5 years was 94.6%, 83.4%, and 74%, respectively. For patients with primary stage I-II lung cancer (n = 46), the 1, 2, and 5-year local control: 97.2%, 94.2%, and 86.9%; RC: 97.6%, 82.5%, and 69.5%; and DM: 3%, 16%, and 33.4%, respectively. Patients treated for lung metastases (n = 26) had worse local control at 1, 2, and 5 years: 94.4%, 69.3%, and 55.5%, respectively. Side effects were rare with 2 (3%) patients reporting chest wall pain and 6 (8%) patients experiencing radiation pneumonitis, including 1 patient who had grade 5 radiation pneumonitis.
Conclusions:
Helical therapy delivers a safe and effective lung stereotactic body radiation therapy plan, despite not being able to meet RTOG’s recommended R50 conformality constraint.
Keywords
Introduction
Lung stereotactic body radiation therapy (SBRT) has been quickly adopted for treating patients with medically inoperable peripheral and central non-small cell lung cancer (NSCLC), 1,2 pulmonary metastases, 3 and even multiple lung lesions simultaneously 4 in both academic and community hospitals in the United States over the last decade. This was made possible by enhanced treatment planning software and linear accelerator hardware proving proof of principle in ongoing single- and multi-institutional phase I and II studies guiding physicians in adopting SBRT techniques. 5 More sophisticated treatment planning systems (TPSs) and algorithms were able to produce SBRT plans with tight conformality and steep dose gradients to provide the safe delivery of highly ablative radiation doses that resulted in a low toxicity profile and excellent local control (LC).
Most of the clinical data supporting lung SBRT from these early trials were derived from nonhelical platforms, such as intensity-modulated radiation therapy (IMRT), intensity-modulated arc therapy (IMAT), and 3D conformal planning with several noncoplanar beams. Helical therapy (HT) had been an option on such trials if all constraints could be met, 6 and existing technical reports had also confirmed the feasibility of using HT to deliver SBRT for lung metastases in comparison with IMAT and step-and-shoot IMRT. 7 –9
When our institution began using HT for lung SBRT in 2008, we followed the normal tissue tolerances and conformality constraints recommendations from RTOG 0915, 10 except we limited the plan maximum dose to 110% instead of the higher range of 111% to 166.7% allowed in RTOG. We noticed that while our radiation plans met the dose constraints for organs at risk (OAR), target coverage, and conformality for the prescription dose (R100%), we could not meet the measure for steepness of the dose gradient: ratio of the 50% prescription isodose volume/ planning target volume (PTV) volume (R50%).
Despite not meeting the R50%, we observed that our patients had minimal side effects and excellent LC. We thought it prudent to evaluate our entire experience of lung SBRT treated off-protocol on TomoTherapy (Accuray, Sunnyvale, California) to determine whether major deviations from R50% resulted in adverse clinical outcomes. We hypothesized that the LC and adverse effects using a helical approach to SBRT delivery would be similar to those previously reported using nonhelical treatment platforms.
Materials and Methods
This is a retrospective review of all consecutive patients treated with SBRT to peripheral lung tumors on a tomotherapy (Accuray) machine between 2008 and 2013.
All patients were simulated with a 4-dimensional (4D) computed tomography (CT) scan, and abdominal compression was used if tumor motion was > 1 cm. An internal tumor volume (ITV) was expanded 5 mm in every direction to create the PTV.
Helical therapy plans were created using tomotherapy treatment planning system (TPS; version 4.2.3.9, TomoTherapy Inc, Madison, Wisconsin), 6-MV photons, tissue heterogeneity was accounted for, a pitch of 0.143, and a modulation factor of 2. The field had a longitudinal aperture size of 2.5 cm (with an option of using 1.05 cm) and a 64-leaf binary Multileaf collimator (MLC) with a leaf width of 6.25 mm projected at isocenter. The collapsed cone convolution superposition dose calculation algorithm was used to calculate the final dose on a free breathing scan. Patients received SBRT doses ranging from 48 to 60 Gy in 3 to 5 fractions prescribed to 95% of the PTV.
Radiation treatment plans were initially limited to a maximum dose of <110%, but then in 2012, this limit was loosened. Plans were evaluated for target coverage, conformality, and normal tissue dose constraints in accordance with guidelines from RTOG 0915 10 and American Association of Physicists in Medicine Task Group 101. 11 Conformality was evaluated by 4 measures: ratio of prescription isodose volume/PTV volume (R100%) <1.2, ratio of 50% prescription isodose volume/PTV volume (R50%), maximum dose (in percentage of dose prescribed) at 2 cm from PTV in any direction (D2cm (%)), and Lung V20 <10%. The target coverage constraint of 95% of the PTV receiving prescription dose was a hard constraint and was met in all plan. Normal tissue constraints depended on the prescription dose and number of fractions, but key constraints included a total lung V20 < 10%, rib max 50 Gy, 5 cc < 40 Gy (unless the rib was within the PTV).
Data were analyzed through February 7, 2017. Patients were followed by their medical and radiation oncologists and had CT scans at 3- to 6-month intervals. Recurrence was determined via imaging in the majority of cases. Patients were discussed in a multidisciplinary tumor board and were deemed to have progressed if least 2 successive CT scans showed growth of the lesion; a PET scan was ordered in certain cases to help confirm the image represented a recurrence and not changes due to radiation. 12
The primary end point of the study was LC at the primary tumor bed (PTB). Secondary end points included regional control (RC), distant metastasis (DM), progression-free survival (PFS), overall survival (OS), and treatment-related side effects, including radiation pneumonitis (RP) and chest wall pain. Regional control was defined as control in the ipsilateral lymph nodes or ipsilateral lung other than the PTB, DM was identified as contralateral lung or extrathoracic disease. 13 The times to events were calculated from end of radiation treatment. Common Terminology Criteria for Adverse Events version 4.0 was used for grading adverse events.
This is a mainly descriptive study due to its retrospective design. Characteristics measured on a nominal or ordinal scale are described as percentages. Characteristics measured on a continuous scale are described with medians and interquartile ranges because many have skewed distributions. The inverse Kaplan-Meier method was used to calculate the median follow-up time from last day of treatment to last follow-up or death. Kaplan-Meier method was used to estimate the local, regional, and distant control rates; PFS; and OS.
Results
Between January 8, 2008, and April 10, 2013, 74 patients (81 lung lesions) were treated with 79 lung SBRT plans utilizing tomotherapy at our institution. Patient characteristics and pertinent medical history are displayed in Table 1. Interestingly, 53% of our patients had prior cancers, 19% had received previous full-course definitive radiation therapy to their thorax, and 23% had prior lung surgery. Table 2 shows the tumor characteristics of the lesions treated with SBRT.
Patient Characteristics.a
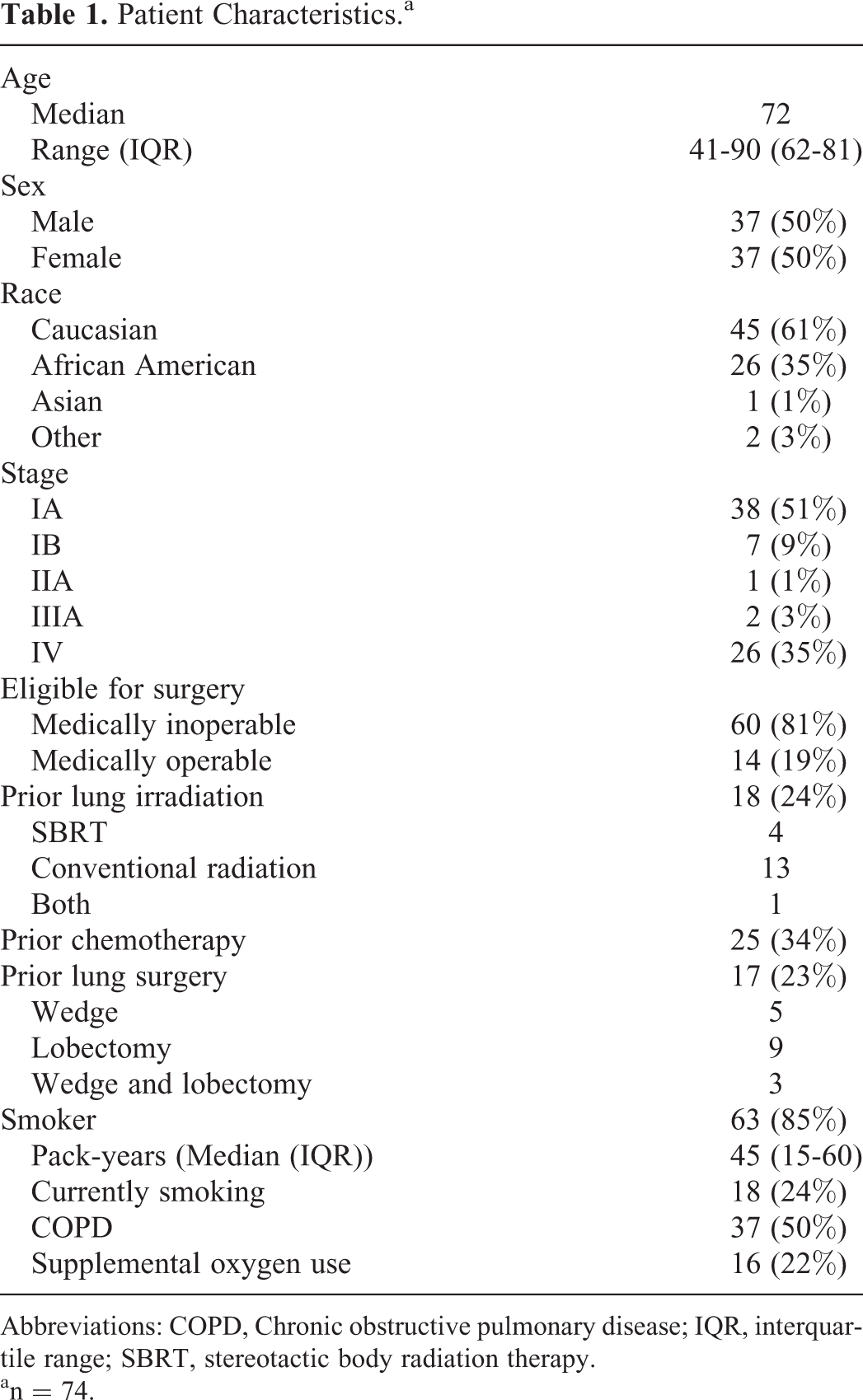
Abbreviations: COPD, Chronic obstructive pulmonary disease; IQR, interquartile range; SBRT, stereotactic body radiation therapy.
an = 74.
Tumor Characteristics.a

Abbreviations: NOS, Not otherwise specified; NSCLC, non-small cell lung cancer; SBRT, stereotactic body radiation therapy.
an = 81.
The radiation prescription, target coverage, and normal tissue dose constraints to the lung and rib are reported in Table 3. All lesions had acceptable PTV and ITV coverage with only 7(9%) of plans with max dose >120%. Table 4 summarizes the deviations from RTOG 0915 conformality constraints. The R50% conformality constraint was not met in any of the plans; 31 (39%) had minor deviations and 48 (61%) of the plans had major deviations. Figure 1 shows the variance in the actual R50% compared to the RTOG 0915 goals. There is more variance for the smaller PTV volumes since the smallest field size is 1.05 cm, and therefore more surrounding tissue would be targeted. Two radiation plans treated 2 lesions simultaneously; however, these plans were able to meet R100% and did not have the largest deviations from R50%. The outlier R50% deviation and the only plan to have a major deviation from R100% was for a plan to treat the smallest tumor in the study that had a PTV of 3.7 cc.
Radiation Treatment Details and Dose Volume Histogram (DVH).

Conformality Constraints in 79 Tomotherapy Plans for Lung SBRT.a

Abbreviations: PTV, planning target volume; SBRT, stereotactic body radiation therapy.a The RTOG 0915 constraints were used. R50% and D2 cc were based on PTV volume. The R100% goal was <1.2, minor deviation >1.2 but <1.5, and major deviation >1.5.

The area under the line labeled “Recommended R50%” is acceptable, the area between the lines is with minor deviation, and the area above the lines would not have been an acceptable R50% per the RTOG 0915 requirements. The variation in R50% was greater for small lesions. Variance in R50% for the plans with single lesions per isocenter (79 plans).
Four of the 74 patients were lost to follow up. All other patients were evaluated for response with a CT scan of the chest at least once after the completion of SBRT. Median follow-up for surviving patients was 4.7 years (range: 0.5-89.5 months). Local control for all patients at 1, 2, and 5 years were 94.6%, 83.4%, and 74%, respectively. For patients with primary stage I to II lung cancer (n = 46), the 1, 2, and 5-year LC: 97.2%, 94.2%, and 86.9%; RC: 97.6%, 82.5%, and 69.5%; and DM: 3%, 16%, and 33.4%, respectively. Patients treated for lung metastases (n = 26) had worse LC at 1, 2, and 5 years: 94.4%, 69.3%, and 55.5%, respectively. Overall survival at 1, 2, and 5 years for the stage I to II primary lung cancers and for patients with metastatic cancer were 76.1%, 55.8%, 33.3% and 68.7%, 59.8%, 35.5%, respectively. Progression-free survival at 1, 2, and 5 years for the stage I to II primary lung cancers and for patients with metastatic cancer were 92.3%, 72.9%, 51.5% and 60%, 38.6%, and 20.6%, respectively.
Side effects were minimal with only 2 (3%) patients reporting chest wall pain and 6 (8%) patients recorded as having RP: 4 patients had grade 1-2 RP, 1 patient had grade 3 RP, and 1 patient had grade 5 RP. Since the lung and rib dose constraints were met, it is unlikely related to the larger R50%.
Discussion
This is the largest published report with almost 5 years follow-up on dosimetric and clinical outcomes of 74 patients (81 lung lesions) who were treated with lung SBRT using a helical delivery platform from 2008 to 2013. In this report, we assess the clinical validity of the R50% dose gradient constraints put forth by RTOG 0915 in patients treated with HT. In our study population, we did not find that major deviations from R50% increased side effects. The 1, 2, and 5-year LC in stages I to II primary lung cancer of 97.2%, 94.2%, and 86.9%, respectively, and in patients with metastatic cancer of 94.4%, 69.3% and 55.5%, respectively, were similar to previously reported outcomes. 1,3,14 –17
The R50% is the ratio of the 50% IDL/PTV volume that describes the steepness of the dose gradient for radiation plans. This measure is used to evaluate the conformality of lung SBRT plans, yet it has never been clinically validated. One would strive for a sharp dose gradient to limit dose to OARs, yet if all of the dose constraints are met, it is unlikely the patient would have increased side effects even if the R50% doesn’t meet the recommended values. The R50% constraint would more likely be critical when treating medial tumors near critical central OARs and when treating multiple lung lesions or reirradiation. One theoretical benefit of a gradual dose gradient is that the higher doses may contribute to increased microscopic tumor killing in surrounding lung tissue.
It was initially surprising to observe that HT plans could not meet the R50% constraints since tomotherapy had been shown to be ideal for SBRT and can accurately localize dose to within 2 to 2.4 mm. 18 Prior studies have shown that HT was not found to increase integral dose and HT had better conformality compared to step-and-shoot IMRT (coplanar plans only) as well as provided a more uniform target dose and improved critical structure sparing compared to IMAT. 9,7,8 In 2006, a feasibility report was published based on 9 medically inoperable T1/2 N0 M0 patients with NSCLC who underwent treatment planning with 4D-CT in a double vacuum based immobilization, received 60 Gy in 5 fractions within 10 days, had daily pretreatment MVCT image guidance, and an average fraction delivery time of 22 minutes. 19 Clinically, none of the patients experienced ≥ grade 2 pulmonary toxicity and the authors concluded that SBRT using HT proved to be safe and technically feasible. 18,19 However, none of these reports had reported on the dose gradient, or R50%, so it is possible this limitation was not identified.
Treatment planning optimization parameters in tomotherapy TPS include a target coverage constraint that is fixed relative to the other target or OAR constraints. This is beneficial in a sense that the prescribed target coverage is always met, but there is no ability to normalize the dose (ie, prescribe to an isodose line). By adhering to clinical practice of keeping the maximum dose to <110%, we achieved relatively uniform dose throughout the target at the expense of a shallow dose gradient outside the PTV and resulting in high values of R50%. Due to the helical nature of tomotherapy, the selected field width will have an effect on dose “spillage” adjacent to the PTV in the superior–inferior direction. Our HT unit is not equipped with dynamic jaws to minimize this effect. Most of the treatment plans utilized 2.5 cm field width to maintain reasonable beam-on time. It is also possible that the HT plans were not able to meet R50% due to the fact that HT is limited to coplanar delivery so that the “dose to nontarget regions is higher than that achieved using noncoplanar arcs with circular collimators, especially for small isolated spherical targets.”(pp. 147) 18 We also noticed that plans with smaller lesions resulted in worse conformality and larger R50% values. It may be possible that using a smaller field size of 1 cm and permitting a greater than 110% max hotspot would result in more HT plans achieving the desired R50%. 20
Our patients were still treated using HT with the plans that didn’t meet R50% because the lung and rib dose constraints and tumor coverage goals were met. We found that the LC and low rate of RP and chest wall pain were similar to previously reported studies. 1,21 Side effects of chest wall pain and RP after SBRT using HT were minimal; however, there was 1 patient who had grade 5 RP. This patient was an unusual case with multiple prior lung surgeries and full-course thoracic radiation treatment for her prior history of lung cancer. Stereotactic body radiation therapy was her only ablative option due to her poor performance status. A previous report from Japan on 30 patients treated with SBRT using HT had 2 patients with grade 5 RP which the authors primarily attributed to the size of the Gross tumor volume (GTV), 22 however in our patients case, her GTV was only 1.2 cm, so her increased risks of RP were most likely from prior lung surgeries and underlying poor lung function.
This is a retrospective study whose sample size was limited by the number of patients seen at this institution. Failures were not routinely biopsied since oftentimes the risk of complications from a biopsy was high in these medically inoperable patients. Local, regional, and distant controls were evaluated based on CT or PET scans. Timing of follow-up scans could also affect the time when a patient was deemed a failure and thus their PFS. 23,24 The set-up, planning, and delivery techniques for these patients were consistent, and follow-up was adequate for a majority of patients. Our results are similar to other studies that used different treatment platforms to deliver SBRT. This report provides clinical evidence to support the use of HT for lung SBRT.
Conclusion
This single-institutional experience is the largest published report using HT to treat lung lesions with SBRT with almost 5-year follow-up data. We found that HT delivered safe and effective lung SBRT plans despite not meeting the R50% constraints suggested by RTOG. The R50% constraint may become more clinically significant when treating multiple lesions or repeating SBRT near a previously irradiated site.
The next step is to design and undertake a comparative, multi-institutional study that has sufficient statistical power to identify differences in dosimetry from different machines and practitioners, as well as to further explore potential risk factors for RP and prognostic factors for LF, PFS, and OS. Future dosimetric and clinical comparisons between various linear accelerator machines used to deliver SBRT could help to further refine lung SBRT and the R50% recommendations.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
