Abstract
Purpose:
To evaluate the effect of autologous satellite cell and smooth muscle cell transplantation on vesicovaginal fistulas in a randomized controlled study by comparing the proportion of fistula closure and tissue composition between the 2 groups.
Materials and Methods:
Eight pigs underwent surgery to establish an experimental induced fistula. One pig was excluded due to insufficient weight gain. Seven pigs were randomized to receive either an injection of satellite cells and smooth muscle cells or no treatment. Cells were isolated from abdominal skeletal muscle and bladder smooth muscle, cultured in vitro, and marked with bromodeoxyuridine. Cell purity, growth kinetics, and differentiation potential were determined. Cells were injected around the fistula using a cystoscope. Five weeks after the injection of cells, the fistula area was examined macroscopically and histologically.
Results:
The isolated satellite cell population consisted of 76.6% neural cell adhesion molecule positive cell and the smooth muscle cell population of 88.6% α-smooth muscle actin positive cells. All pigs had a persisting fistula before treatment. One of the 4 vesicovaginal fistulas was closed 5 weeks after injection of satellite cells and smooth muscle cells compared to none in the control group. No histological differences in the tissue composition between the 2 groups were observed and no bromodeoxyuridine-labeled cells were detected around the fistula tissue in the histological examination.
Conclusion:
We established an animal model with a persisting clinical and histological confirmed vesicovaginal fistula. The study indicated that there might be an effect on vesicovaginal fistula closure using an injection of satellite cells/smooth muscle cells and alginate gel.
Keywords
Introduction
Vesicovaginal fistula (VVF) is defined as an abnormal anatomical passage between the urinary bladder and the vagina. It results in continuous urine leakage accompanied by several complications, for example, recurrent urinary tract infections, vaginal fungal infections, skin infections and irritations, and rarely pelvic pain. 1 Women affected by VVF also having psychological and social problems such as depression and isolation from society due to an unpleasant odor. 2
Although rare, VVF in the developed countries is reported as a complication to surgery of benign indication and as part of pelvic malignancy. 3 –7 It is seen as a complication to hysterectomy of malign indication, the risk being the highest in the treatment of cervical cancer. 3 The VVFs are also seen as a consequence of radiotherapy for pelvic tumors. 3,5,7
The VVFs are usually treated surgically, either with a vaginal or with an abdominal approach. 1 Success rate is the highest in primary fistula repair ranging from 71.2% to 95.7%, depending on fistula size, involvement of urethra or the bladder neck and the number of fistulas. 4,5,8 It is indicated that the success rate might be lower, when looking only at VVF in relation to malignancy. 4,6 Complications associated with surgical repair are bleeding, infection, and damage to other structures. 4 Recurrence is seen in 6.4%. 4
A cystoscopic injection of stem cells and tissue-specific cells such as smooth muscle cells (SMCs) is a potential minimal invasive treatment of VVF, hence it improves success rate of VVF closure and patient comfort, leading to faster recovery. Muscle-specific stem cells, also called satellite cells (SCs), can be isolated and cultured from a muscle sample. Satellite cells are able to survive, proliferate, and differentiate in vivo when transplanted into both human and animal tissues. 9 Skeletal myoblasts have shown a positive effect on both heart function in an animal model with heart failure, probably due to structural changes, less interstitial fibrosis, and a different composition of collagen fibers. 10 Hypothetically the combination of SCs and SMCs might be able to restore tissue with the same dynamic contractility as normal bladder wall, thus achieving fistula closure without affecting bladder function.
Injection of cells has resulted in poor outcome due to a large and rapid loss of cells and reduced migration. 11 Hydrogels are cell delivery vehicles that increase in vitro viability, proliferation, and differentiation potential of myoblast. 12 Increased survival, migration, and distribution of cells in vivo are also observed thus improving the efficacy of stem cell transplantation. 13,14
The purpose of this randomized controlled study was to establish an animal model with a histologically verified VVF and a method for cell implantation in the treatment of VVF.
Materials and Methods
This randomized study was conducted at a fully licensed Danish animal research laboratory and performed in agreement with The Danish Animal Research law. Approval was obtained from the Danish Animal Experiments Inspectorate (ref. no. 2015-15-0201-00470). Since this is a pilot project, it was only necessary with a minimum number of pigs. Based on the results from the underlying project by Lindberg et al, 15 where 50% of the pigs developed persistent fistulas, it was decided to use 4 pigs in each group to ensure pigs with fistulas in each group.
Eight female 12-week-old Landrace/Yorkshire pigs with an initial mean weight of 42.8 ± 0.71 kg were housed at The Biomedical Laboratory (University of Southern Denmark, Denmark).
They were placed 2 and 2 in 2 × 2.8 m pens on a safe vinyl floor with JELUXYl Premium Bedding (JELU-WERK, Germany) and straw. The room temperature was 21°C ± 1°C, dark/light cycle was 12 h/12 h, and the air humidity was 30% to 50%. The pigs had free access to clean tab water and were fed with Svin Enhed Classic (DLG, Denmark). Before the beginning of each procedure, animals were sedated with intramuscular (IM) metetomidin (0.05 mg/kg), midazolam (0.25 mg/kg), and atropine (0.05 mg/kg). After sedation, the animals received intravenous (IV) propofol (2.5-3.75 mg/kg), IV buprenorphine (0.03 mg/kg), and IM ampicillin (15 mg/kg). They were endotracheally intubated and connected to a respirator. During the procedures, anesthesia was maintained with either isoflurane (2.2%) or continuous IV propofol (7.7-9.2 mg/kg/h). After the procedure, the animals received percutaneous fentanyl (1.2 mg/24 h) for 3 days and IM ampicillin (16.8 mg/kg) for 5 days.
The VVF was created according to Lindberg et al. 15 A vertical laparotomy was performed from below the umbilicus to the symphysis including a peritoneal opening and through the peritoneum to reach the bladder surface. A vertical incision was made in the bladder from the apex toward the neck on the ventral and lower surface with a length of proximal 7 cm. A cuffed tracheal tube (size 6.0, Teleflex Medical, Ireland) was placed in the vagina and palpated through the bladder and vaginal wall. The tube was fixed with Babcock forceps, and an incision was made at the tip of the tracheal tube. An absorbable continuous Monocryl 3/0 was placed around the incision, thereby creating the fistula. The cuff was filled with sterile saline, and the tube was secured to the bladder wall using 2 absorbable Vicryl 3.0 sutures. The tube was cut to a length of 16 cm. The bladder was closed in 2 layers, and the peritoneum, abdominal muscle, and cutis were closed according to normal practice with Vicryl 2.0 sutures. Samples for cell isolation were taken from the bladder and the abdominal skeletal muscle. The surgical procedure was performed by 2 urologists.
Four weeks postoperatively, a cystoscopy using a flexible cystoscope (CYF-4; Olympus, Ballerup, Denmark) was performed to examine the fistula in vivo.
SCs and SMCs were isolated from the abdominal skeletal muscles and bladder tissue, respectively. Cells were handled as described by Sellathurai et al. 16 Samples were trimmed of visible connective tissue and adipose tissue, minced and digested in 3.5% collagenase type II (Worthington Biochemical Corporation, New Jersey) for 60 minutes at 37°C. Subsequently, the suspension was centrifuged at 1000 rpm for 10 minutes. The pellet was suspended in growth medium (GM) containing Dulbecco’s Modified Eagle Medium (DMEM) with 10% fetal bovine serum (FBS) and 1% penicillin, streptomycin, and Fungizone antimycotic (PSA) and filtered through 100 µm and 40 µm nylon mesh. The suspension was centrifuged at 1000 rpm for 10 minutes. The remaining cell pellet was suspended in GM, transferred to culture flasks (225 cm2), and cultured in GM at 37°C and 5% CO2. Satellite cells were cultured in flasks coated with extracellular matrix gel (ECM; Sigma-Aldrich, Denmark). The upscaled cells (15-25 × 106) were stored at −80°C until further use.
SCs and SMCs were characterized in terms of purity and growth kinetics. In addition, SCs differentiation potential was evaluated.
SCs and SMCs were seeded at a density of 6000 cells/cm2 in 12-well plates containing coverslips coated with ECM and cultured in GM followed by harvest after 48 hours. For differentiation, SCs were grown in proliferation medium consisting of GM with 10% horse serum. Cells were changed to GM at 80% confluence, and 20 hours later to differentiation medium (DM) containing DMEM with 5% FBS, 1 µM insulin (Life Technologies, Denmark), 1 µM cytosine arabinoside (Sigma-Aldrich, Denmark), and 1% PSA. Coverslips were harvested after 17 days in DM.
Growth kinetics of SCs and SMCs were evaluated by seeding cells at a density of 3500 cells/cm2 in 6-well plates in GM. Cells were detached with trypsin/EDTA and counted every day for 6 days using a Bürker hemocytometer. The counting was made in triplicates.
Four weeks postoperatively, the pigs with persisting VVF were randomized to either injection of hydrogel encapsulated SCs/SMCs or no treatment. Randomization was blinded using GraphPad QuickCalcs. 17
Satellite cells and SMCs were thawed and cultured in GM containing 0.01 mM bromodeoxyuridine (BrdU; Sigma-Aldrich, Denmark) for 4 days before injection.
A small number of cells were seeded on coverslips and harvested 2 days later for immunocytochemistry.
Before injections of SCs/SMCs were performed, a flexible cystoscope was used to examine the fistula in vivo and place a wire guide (Roadrunner Hydrophilic PC Wire Guide 0.035 in/145 cm; Cook Medical, Bloomington, Indiana, USA) in the urethra. A cystoscopic injection needle (5 Fr × 8 mm; Cook Medical) was inserted through the operative channel of a rigid cystoscope (22.5 Fr and 12° optics, Olympus, Ballerup, Denmark), and a total of 5 mL sterile 1% sodium alginate gel (diluted in phosphate-buffered saline [PBS]; 0.1 mg/mL PBS; PRONOVA UP LVM; BioPolymer AS, Norway), containing 18 × 106 SCs and 4.5 × 106 SMCs, was injected in 4 places around the fistula. Subsequently, a new injection needle was inserted and 0.3 mL diluted calcium chloride (0.01 mg/mL PBS) was injected at the same sites to induce cross-linking in the gel and formation of a fixed gel structure. In order to immobilize the bladder by continuous bladder drainage to reduce tension of fistula area, an endotracheal tube with cuff (size 4.0 mm, Unomedical, Denmark) was inserted in the bladder, cut and secured to the vaginal wall using absorbable Vicryl 1.0.
Five weeks after the pigs received an injection of SCs/SMCs, the animals were euthanized with pentobarbital (34 mg/kg), and the fistula area was removed and examined macroscopically.
The fistula area was fixed in 4% formaldehyde and embedded in paraffin. Sections of 3 µm were cut and stained with hematoxylin and erosin and Sirius red according to standard procedures.
Cells cultured on coverslips were stained immunohistochemically for neural cell adhesion molecule (NCAM), α-smooth muscle actin (ASMA), desmin, and BrdU, and sections from fistula area were stained for BrdU according to Table 1.
Overview of the Used Antibodies for Immunocytochemistry and Immunohistochemistry.a
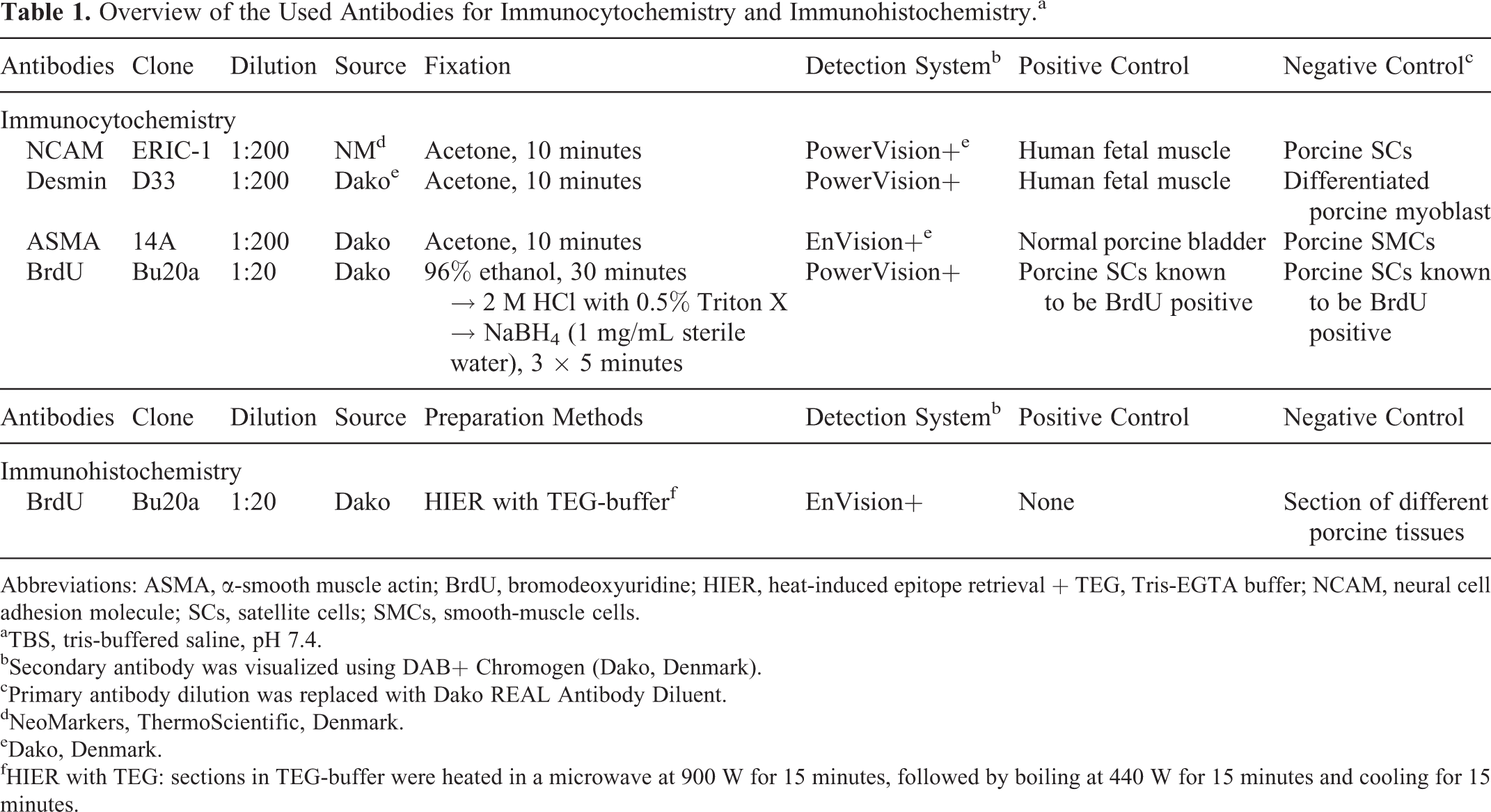
Abbreviations: ASMA, α-smooth muscle actin; BrdU, bromodeoxyuridine; HIER, heat-induced epitope retrieval + TEG, Tris-EGTA buffer; NCAM, neural cell adhesion molecule; SCs, satellite cells; SMCs, smooth-muscle cells.
aTBS, tris-buffered saline, pH 7.4.
bSecondary antibody was visualized using DAB+ Chromogen (Dako, Denmark).
cPrimary antibody dilution was replaced with Dako REAL Antibody Diluent.
dNeoMarkers, ThermoScientific, Denmark.
eDako, Denmark.
fHIER with TEG: sections in TEG-buffer were heated in a microwave at 900 W for 15 minutes, followed by boiling at 440 W for 15 minutes and cooling for 15 minutes.
The ratios of NCAM-positive SCs and ASMA-positive SMCs were determined to assess the purity of the cell cultures. The ratio of BrdU positive cells were determined to assess the labeling efficiency in cells prior to transplantation.
Cells were counted using CAST 2.1.6.0 (Olympus) by inserting systematic random counting frames in 5% of the coverslip area.
Calculation and graphs were made in GraphPad Prism, version 5.01. All estimates were expressed as mean ± standard error of the mean.
Results
All pigs had a persistent fistula 4 weeks postoperative and at the time of cell injection. One pig was excluded from the study at the time of randomization due to insufficient weight gain, hence 4 pigs were assigned for injection of SCs/SMCs and 3 pigs included in the control group. Sufficient weight gain was seen in the remaining pigs, both in the treatment and control groups (70.1 ± 4.6 kg and 78.4 ± 8.3 kg) with a small plateau in the treatment group at the time of SCs/SMCs injection (Figure 1). In the treatment group, One pig developed an incisional hernia due to fast and extensive growth and was surgically treated. A second pig developed a fistula between the bowel and the VVF, and a third pig had a VVF located in the same level as cervix and urethra. No animals died during the study period.

Weight gain in pigs in treatment group and control group during the study period. Data shown as mean weight ± standard error of the mean.
The isolated SC population consisted of 76.7% ± 4.0% NCAM-positive cells and SMC population of 88.6% ± 4.1% ASMA-positive cells (Figure 2A and B). However, SCs were not able to differentiate in vitro (Figure 2D). Doubling times for SCs and SMCs were 24.6 ± 1.4 hours and 28.3 ± 4.1 hours, respectively (Figure 3). With 98.8% ± 0.6% BrdU-positive cells prior to transplantation, nearly all cells were BrdU labeled (Figure 2C).

Immunocytochemical staining of satellite cells and smooth muscle cells. Cells were positive for α-smooth muscle actin (A), neural cell adhesion molecule (B), and bromodeoxyuridine (C) in 76.7%, 88.6%, and 98.8%, respectively. No desmin-positive myotubes with more than 2 nuclei were observed after exposure of satellite cells to proliferation medium for 7 days and differentiation medium for 17 days. A few myotube-like cells containing 2 to 3 nuclei were observed (D; original magnification—A, B, D: ×29, and C: ×10).
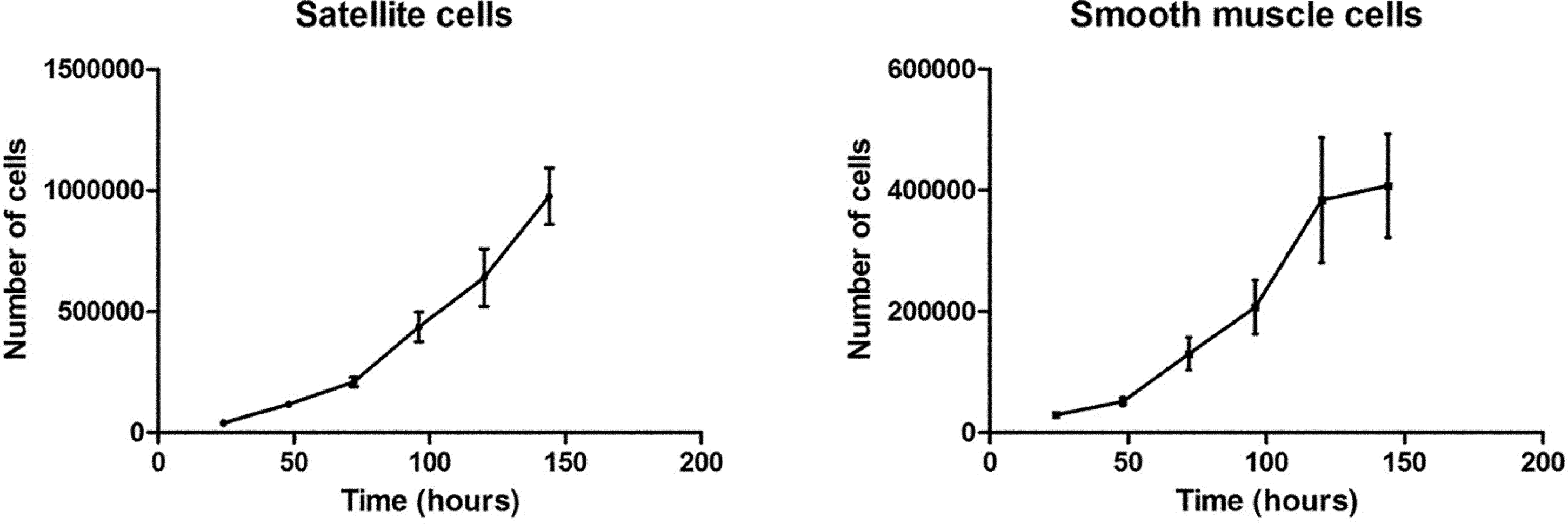
Growth curve of satellite cells (SCs) and smooth muscle cells (SMCs). Cells were counted as triplicates after 24, 48, 72, 96, 120, and 144 hours. Doubling time for SCs and SMCs was 24.6 hours and 28.3 hours, respectively. Data shown as mean ± standard error of the mean.
After cell injection, 1 of the 4 VVFs was closed compared to none in the control group (Figure 4). No histological differences in tissue composition between the 2 groups were detected (Figure 5). Both groups showed signs of regeneration with equal formation of collagen fibers and muscular tissue, although distribution patterns appeared different (Figure 5). We were able to detect the transition between vaginal stratified squamous epithelium and bladder urothelium in all pigs in the control group, indicating persistence of the VVF in all pigs (Figure 6A). Presence of localized aggregates of multinucleated giant cells was seen in the treatment group (Figure 6B). The BrdU labeled, implanted cells could not be recovered.

Macroscopic illustration of a closed fistula (A) and a persisting fistula (B). One closed fistula was seen in the treatment group, while 3 persisted despite an injection of satellite cells and smooth muscle cells.

Histological sections of fistula areas in the treatment group and the control group. HE staining (A-B) and Sirius red staining (C-D) showing no differences in tissue structure between the 2 groups (original magnification: ×0.5).

HE staining of fistula epithelia showing formation of a vesicovaginal fistula (A) and multinucleated giant cells at the site of injection, indicated by arrows (B; original magnification—A: ×10 and B: ×40).
Discussion
We have successfully created a reliable animal model with a persisting VVF and proposed a method for cell implantation as a possible treatment of VVF. One of the 4 VVFs was closed after SCs/SMCs injection. No differences were seen in the overall tissue composition between the 2 groups 5 weeks after injection. No studies have evaluated the effect of stem cell transplantation on VVF closure. The effect of stem cell transplantation on fistula closure has primarily been examined in small-scaled studies in patients with perianal fistulas due to Crohn disease. 18,19 We found an effect of SCs/SMCs in 1 of the 4 treated pigs. However, our success rate is lower than the effect of autologous adipose-derived stem cells (ASCs) or bone marrow-derived mesenchymal stromal cells (MSCs) on perianal fistula closure. Lee et al found an effect of autologous ASCs in 82% (27 of 33) of patients 18 and Ciccocioppo et al found an effect of autologous MSCs in 58% (7 of 12) of patients. 19 These studies indicate an effect of stem cells on fistula closure in settings with complex fistulas. The lower success rate of SCs/SMCs in this study may be due to differences in fistula type and stem cell source. The fistula between the bowel and the VVF, which we discovered in 1 pig, could have affected the healing process due to continuous leakage of intestinal fluid through the VVF, hence maintaining the fistula. The location of 1 VVF at the same level as cervix and urethra in another pig may also have affected the possibility of fistula closure. The size of the persisting fistulas might also explain the low number of closed fistulas. All remaining fistulas had a size over 10 mm (data not shown), which is indicated to predict difficult fistula closure. 8 It would have been desirable to include more animals in our study, given that the small number of animals could be the reason why no effect was found.
Histological examinations showed that the injection of SCs/SMCs and alginate gel did not induce formation of additional connective tissue or muscular tissue. The role of SCs and SMCs in fistula closure in the treatment group is therefore unknown. It could be due to cell transplantation, bulking effect of the gel, and induction of a reaction in the tissue or a combination of all. One study found a bulking effect of a urethral submucosal injection of hydrogel on stress urinary incontinence in a multicenter study. 20 To the extent of our knowledge no studies have examined the effect of hydrogel or hydrogel in combination with cells as a bulking agent on fistula closure. Spontaneous closure cannot be excluded; however, this explanation is less feasible since the remaining pigs all had persisting VVFs.
It is clear that some reaction against injected material happened. We found localized multinucleated giant cells in pigs treated with injections containing SCs/SMCs and calcium cross-linked alginate gel, indicating a chronic inflammatory reaction. This is consistent with Chan et al who observed increased activation of dendritic cells in vivo after an injection of calcium cross-linked alginate gel compared to agarose gel, collagen gel, and barium cross-linked alginate gel. 21 Since we used autologous cells, these results indicate that the formation of multinucleated giant cells is related to the use of calcium as a cross-linker mechanism. In future clinical studies one might choose a less immunogenic material for cell delivery such as human fibrin.
Cell Characteristic, Survival, and Migration
We characterized cells according to purity, growth kinetic, and differentiation potential. Results indicate good purity levels. Growth of fibroblast-like cells was seen in both the SC and the SMC cultures (data not shown). Fibroblast in the cell culture might have induced formation of connective tissue, which would have induced fistula closure. With respect to the differentiation potential of SCs Perruchot et al observed that SCs from 6-week-old piglets were able to form myotube in vitro with a fusion index of 65.3% after 4 days in DM. 22 However, we did not succeed in inducing differentiation of SCs using the same protocol. Satellite cells in our study were isolated from muscle tissue of 12-week-old pigs and the relatively older age could be an explanation for why our SCs did not differentiate in vitro. Zhu et al indicated that differentiation potential declined with age, reporting a poor differentiation of SCs from 6- to 7-month-old pigs compared to 5- to 6-week-old piglets. 23
Surviving transplanted cells were not found in the tissue sections. We were not able to detect any BrdU-positive cells at the time of evaluation. The lack of BrdU-positive cells may be due to dilution of BrdU during cell division or low survival rate of injected cells. One study reported detection of BrdU-labeled ASCs 2 and 4 weeks after transplantation subcutaneously but not after 8 and 12 weeks. 24 This could indicate that long-term BrdU-detection after in vivo transplantation is difficult. Since BrdU is diluted at each cell division, the possibility of detection depends on the cell growth kinetics. They did not report the growth rate of their ASCs. We did not evaluate the tissue structure and BrdU-labeling at an earlier stage after injection. Therefore, we can neither exclude that stem cells may have had an effect at earlier stages of regeneration nor determine whether the lack of BrdU-positive cells is due to cell death or dilution. Detection of transplanted cells could be improved by transfecting the cells with a green fluorescent protein (GFP) vector, which has been tried in mice. 25 However, there are no reports on stable GFP-transfected porcine SCs.
The cell injection method may have affected cell survival and migration as well as healing potential. The use of 2 separate injection needles for stem cell suspension and calcium chloride, respectively, could have affected cell survival. An imprecise injection of calcium chloride increases the risk of incomplete formation of hydrogel surrounding the cells, thereby affecting cell survival. Cell survival and migration may be increased by the use of a different material, for example, 3-dimensional (3D) hydrogel scaffolds. Hill et al showed an improved cell survival and migration with increasing pore size in a 3D alginate scaffold. 26 A scaffold, which is not liquid, may have improved cell survival and migration, but may also be more difficult to use for cystoscopic injections. Alternatively, the injections could have been made both around and in the fistula tract and in combination with scraping of the epithelium to promote the healing process. The cell survival and migration might also have been affected by the dynamic movement of the bladder, which was tried to avoid by using continuous bladder drainage after cell injection. This might not be as effective in immobilizing the bladder as a catheter in humans.
Conclusion
We have established a reliable animal model with a persisting clinical and histologically confirmed VVF in a simple surgical procedure. We suggest a method for cell transplantation and bulging, which can be used in future studies. Cystoscopic guided injection of stem cells could be a possible treatment of VVF in an outpatient setting with minimal patient discomfort, but has to be optimized further according to, for example, formation of supporting gel, cell survival, and cell detection methods.
Footnotes
Abbreviations
Acknowledgments
The authors thank Olympus Denmark for providing us with equipment for cystoscopy. The authors would also like to thank, Lone Christensen at The Department of Pathology, University Hospital Odense, for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. The project was funded by The Family Hede Nielsen Foundation, The Research Board Odense University Hospital, The Ingemann O. Bucks Foundation, The Director Jacob Madsen & Wife Olga Madsen Foundation, The Memorial Fund of Market Gardener Ove William Buhl Olesen and Wife Edith Buhl Olesen, and Department of Clinical Research, University of Southern Denmark.
